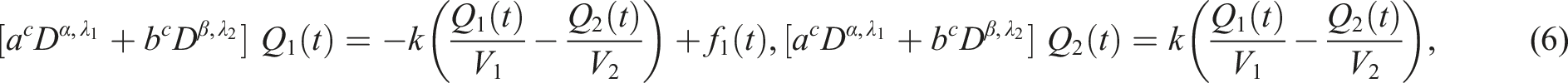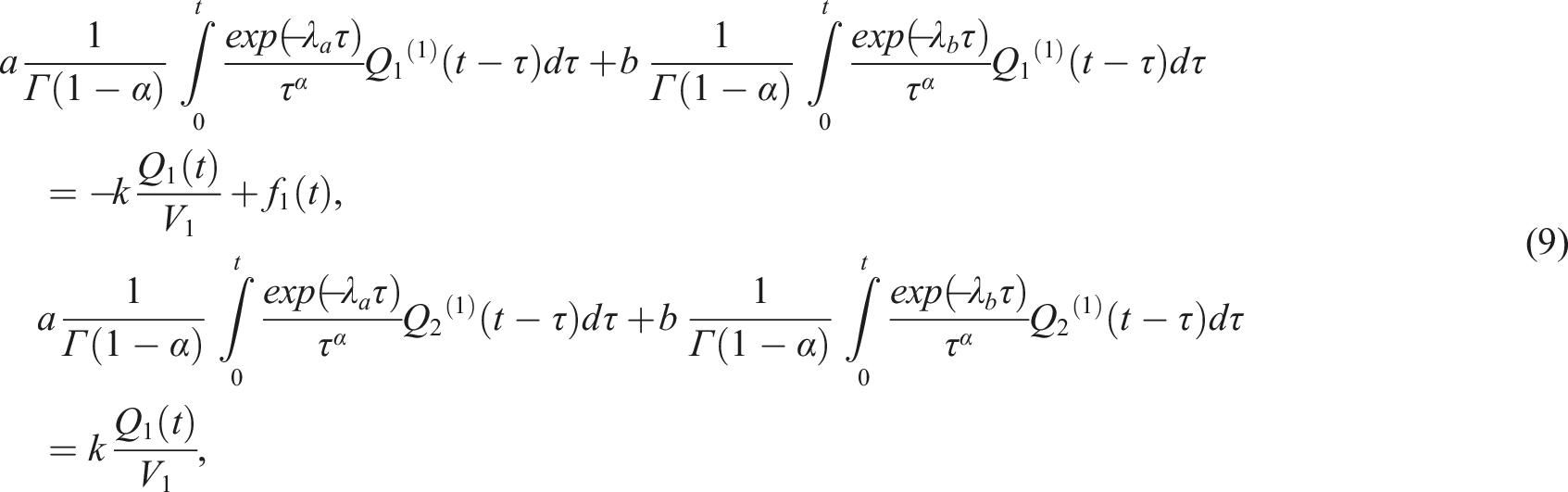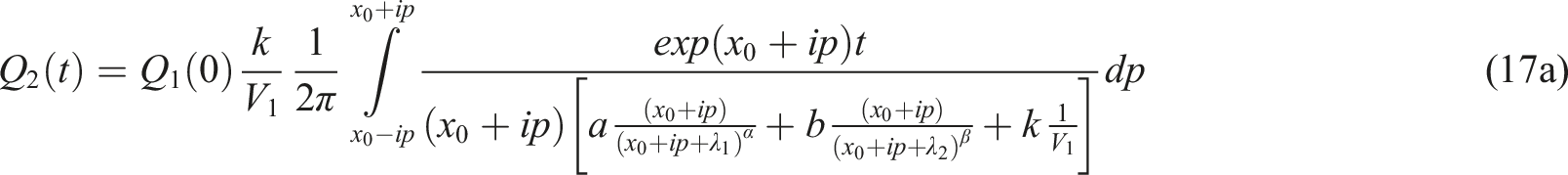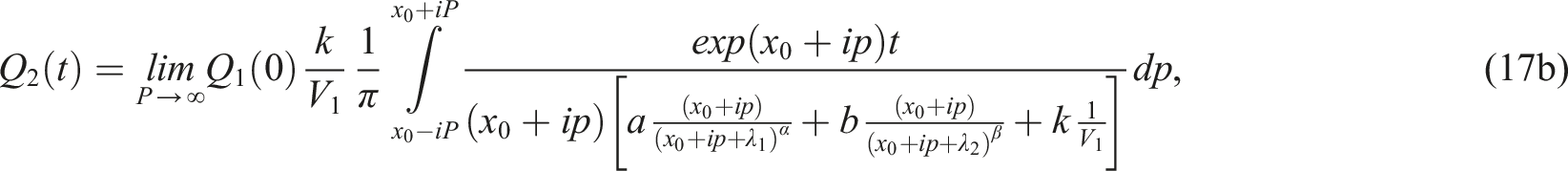Abstract
As a part of this study, a mathematical model based on two-compartment pharmacokinetic system with general fractional derivatives was developed. Its aim was to describe the release of BisGMA and TEGDMA monomers from composite core resin over three time periods. For this purpose, five Clearfil Photo Core resin samples were prepared and elution was measured using high performance liquid chromatography at 1, 7 and 28 days post-immersion in 75% ethanol. The findings confirmed good model fit to the data related to released monomer quantity.
Keywords
Introduction
Investigating composite resin properties is important for a better theoretical understanding of these materials as well as for their clinical applications. 1 Most composite resins comprise methacrylate monomer-based organic matrix, a photoinitiation system, and silane coupling agent as a treated filler. 2 The most widely used organic matrix is a mixture of two or more dimethacrylate monomers, such as bisphenol A-glycidyl methacrylate (BisGMA), triethylene glycol dimethacrylate (TEGDMA), urethane dimethacrylate (UDMA), and ethoxylated bisphenol A dimethacrylate (BisEMA). 3 In clinical practice, BisGMA remains the most popular due to its low volumetric shrinkage, good reactivity, and advantageous mechanical properties, as well as low diffusivity into tissues. 4
Under clinical conditions, 100% monomer-to-polymer conversion of any composite resin can never be achieved. As a result, the residual monomers as well as other substances can be eluted into the saliva, and liquids can leach these intact components from the restoration towards the pulp or oral cavity. Leaching is the process of extracting substances from a material by diffusion due to its constant contact with a solvent. 5 Incomplete composite resin polymerization and monomer leaching thus compromise clinical outcomes. Specifically, these adverse processes not only undermine the mechanical durability of the restoration,6–8 but can also negatively impact the material biocompatibility (e.g., by exerting cytotoxic, genotoxic, mutagenic or estrogenic effects). In some patients, allergic reactions can also occur, such as allergic contact dermatitis and asthma.9–13 Thus, to assess the biocompatibility of these materials, a better understanding of the eluted components is required.
Several factors contribute to the process of elution from composite resin materials, but the most significant are monomer double bond conversion (DC) rate, the extraction solvent chemistry, and the size and chemical characteristics of the released components. For example, as smaller molecules exhibit enhanced mobility, they tend to be eluted at a faster rate than larger molecules. 14 In prior studies, leaching of monomers and other products from composite resins was investigated using high performance liquid chromatography (HPLC) and gas chromatography/mass spectrometry (GC/MS).5–8 The obtained findings indicate that increasing the DC can reduce the quantity of monomers eluted from resin composite,6–8 even though elution of some monomers can be sustained over long periods.5,6,15,16 Considering the harmful effects of substances leached from composite resins for tooth restoration, these phenomena have been extensively studied.5–17 However, to our knowledge, a mathematical model describing the leaching of BisGMA monomer from composite resin has never been developed.
To address this gap in extant literature, in this work, we present a mathematical model corresponding to a two-compartment pharmacokinetic system and general fractional derivatives that can predict monomer release rate over time. This approach was guided by the recent advances in the pharmacokinetic theory.18–22
Materials and Methods
The main idea of the General fractional calculus is the definition of a general fractional integral
We state two conditions that the kernels in (1), (1a) must satisfy
Note that the Riemann-Liouville kernels18,19
There are many kernels that satisfy (2), (3) as shown in ref.19. We mention two of them. In ref.19 the following kernels are proposed
The proof the M and K used in (4) satisfy (2)2 is given in. 19
Our aim in this work is to formulate generalized two compartmental model for pharmacokinetics with General fractional derivative defined by (4).
Consider the classical two compartmental model of pharmacokinetics, with different volumes of compartments
23
In
21
we proposed generalization of (5) where first derivatives on the left hand side of (5) are replaced by two General fractional derivatives defined by (4). In this way we obtained, instead of (5) the following system of the first order fractional differential equations
In Figure 1 we show the schematic view of clinical relevance of polymerized experimental specimen. Experimental model of the monomer elution.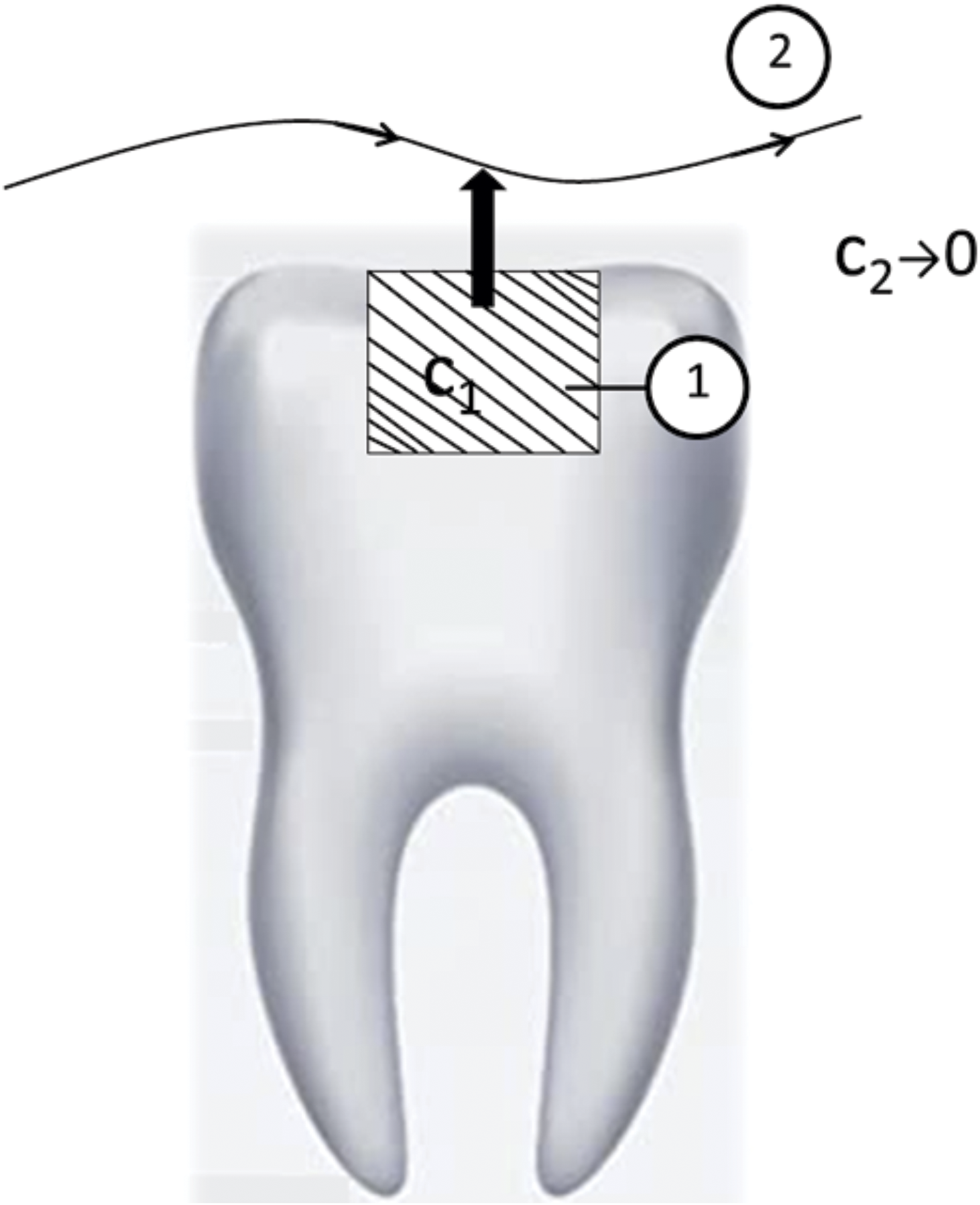
In the Figure 2 we present the two compartmental model that corresponds to the experimental model. In the model we assumed that the compartment 2 in which the monomer elusion is taking place has finite volume. Two compartmental model corresponding to monomer elution.
Explicit form of the system of integro-differential equations that we will solve in this work, follows from (6) and reads
The initial conditions corresponding to (7) are
System (9) represents the generalized two compartmental model with GFD for radioactive decay.
Solution to the relevant system (7), (8)
We use the Laplace transform method in solving (7), (8). The Laplace transform of an exponentially bounded function
Using expression for the Laplace transform of the first derivative applied to
Note that (12) leads to
The limiting concentrations, in each compartment
The parameters in the model
Experimental
In the present study one composite resin material was tested: a light-cured core build-up material (Clearfil Photo Core, Kuraray Europe Gmbh, Düsseldorf, Germany) with filler loading 86.5% by weight and 68% by volume. For monomer elution measurements, cylindrical specimens 5 mm in diameter and 2 mm thick were prepared using standardized moulds.
Five samples were prepared. The composite resin light-cured material was used according to the manufacturer’s instructions. A mould was placed on a glass plate, filled with material, pressed with a Mylar strip to extrude excess and prevent oxygen inhibition layer. The material was polymerized according to manufacturer’s instructions. It was cured for 20 s using an LED light-curing unit (Bluephase, Ivoclar Vivadent, Schaan, Liechtenstein), operating at an intensity of 1,200 mW/cm2. The tip-to-surface distance was standardized to 1 mm and was maintained with a custom-made guide. Immediately after light curing, excess material was removed with a scalpel and each sample was polished using a series of Sof-Lex discs (medium, soft, super-soft, 3M ESPE, St Paul, MN, USA) for 30 s each, under water and standardized pressure. The samples were immersed 10 min post-curing.
Monomer elution
Each sample was immersed in 1 mL of 75% ethanol/water solution (HPLC Gradient Grade solvents) in a glass, dark vial and stored at 37°C. After 24 h, 7 and 28 days the solution was changed and HPLC measurements were done. The whole solution was taken for analysis. This procedure allowed us to treat each sample as immersed in infinite volume of the solution.
Qualitative and quantitative analysis was performed on an HPLC instrument (Thermo Fisher Scientific Inc., Waltham, MA, USA) equipped with an XDB-C18 column, 75 mm long, 4.6 mm inner diameter and 3.5 μm particle size (Zorbax Eclipse®, Agilent Technologies, Santa Clara, CA, USA). In front of the separation column, a pre-column was installed, 12.5 mm long, 4.6 mm inner diameter and 5 μm particle size (Zorbax Eclipse®, Agilent Technologies, Santa Clara, CA, USA). The mobile phase was a mixture of water and acetonitrile (HPLC Grade, Sigma–Aldrich, Dorset, UK) and a gradient was applied according to the following method (A: H2O, B: CH3CN): 40% B (0–4 min); 70% B (4–9 min); 100% B (9–11 min); 40% B (11.01–17 min). The flow rate was 1 mL/min and the injection volume was 10 μL. UV detection was performed at 205 nm (for monitoring the elution of BisGMA and TEGDMA) and 275 nm (for monitoring the elution of BisGMA). The compounds were identified by comparison of their retention times with those of the reference compounds under the same HPLC conditions. The retention times of the reference standards were 4.44 min (TEGDMA) and 6.67 min (BisGMA).
Reference standards of BisGMA and TEGDMA (>98% purity; IvoclarVivadent, Schaan, Liechtenstein) were used to produce stock solutions of 1,000 μg/mL each. The stock solutions were diluted in acetonitrile to produce the final calibration solutions 2.5, 5, 10, 15 and 20 μg/mL. The peak area for each monomer was plotted versus concentration using linear regression analysis to quantify monomer concentration in the sample solutions. The limit of quantification (LoQ) and detection (LoD) were calculated for a signal to noise ratio of 10 (S/N = 10) and 3 (S/N = 3), respectively. The LoQwas 0.325 μg/mL for TEGDMA and 0.442 μg/mL for BisGMA. The LoDwas 0.097 μg/mL for TEGDMA and 0.132 μg/mL for BisGMA.
Experimental results
The experimental cumulative BisGMA and TEGDMA release profiles from Clearfil Photo Core are represented in Figure 3 and Figure 4 as an average of five samples where Q2B is mass of released BisGMA and Q2T is mass of released TEGDMA. BisGMA and TEGDMA released profiles verified the initial burst release followed by slow release in a later time period. This behaviour is a consequence of different diffusion gradient of monomer molecules on the surface of experimental samples and monomer molecules entrapped in the polymer network of experimental samples. This finding is in agreement with previous studies on composite resin.7,8 And it is most likely due to higher BisGMA content. The quantity of the released BisGMA in μg. The quantity of the released TEGDMA in μg.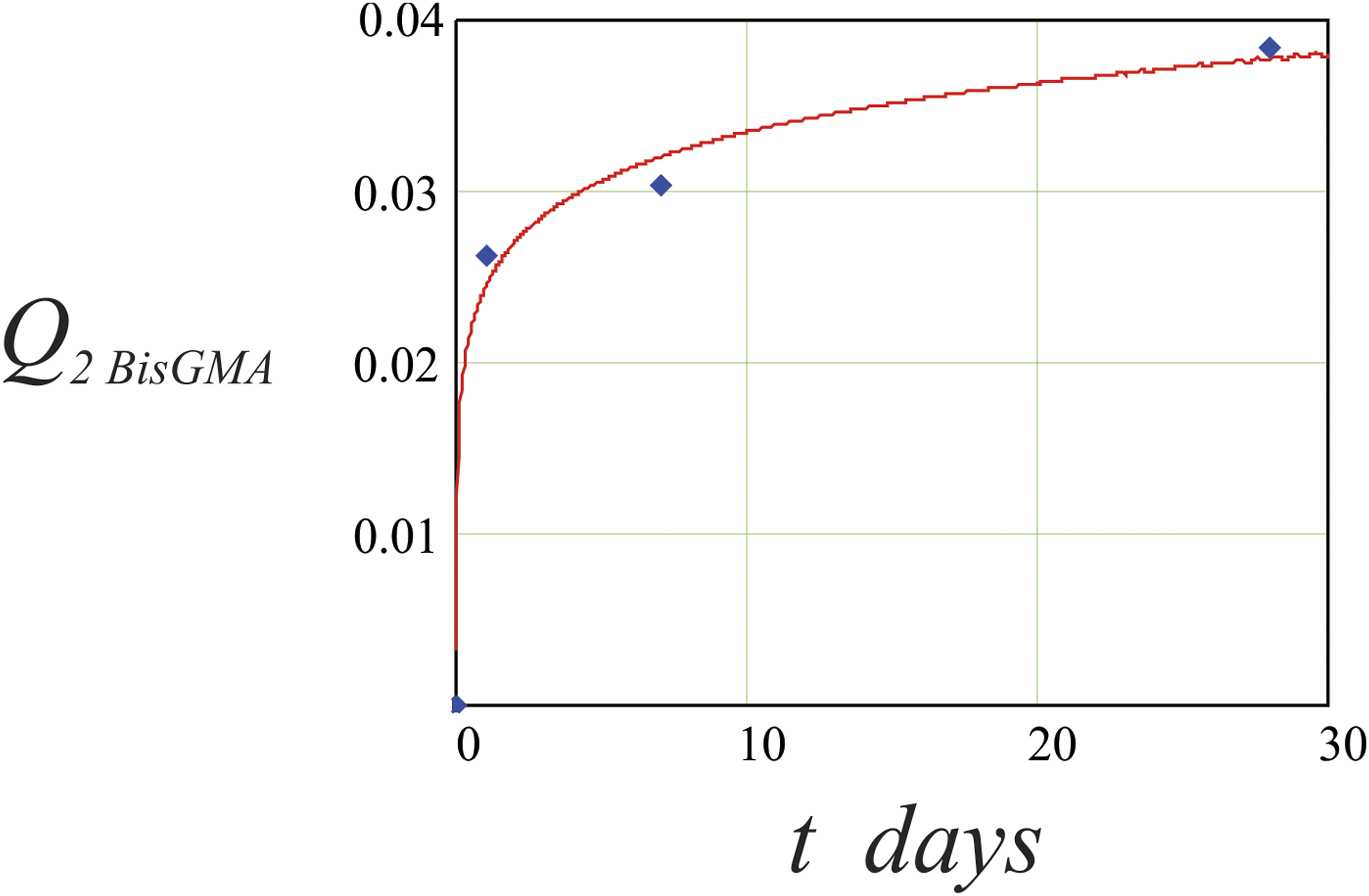

In this Section we present the results of numerical inversion of (12). From (12) for we obtain
By using the argument principle, it can be easily shown that the function
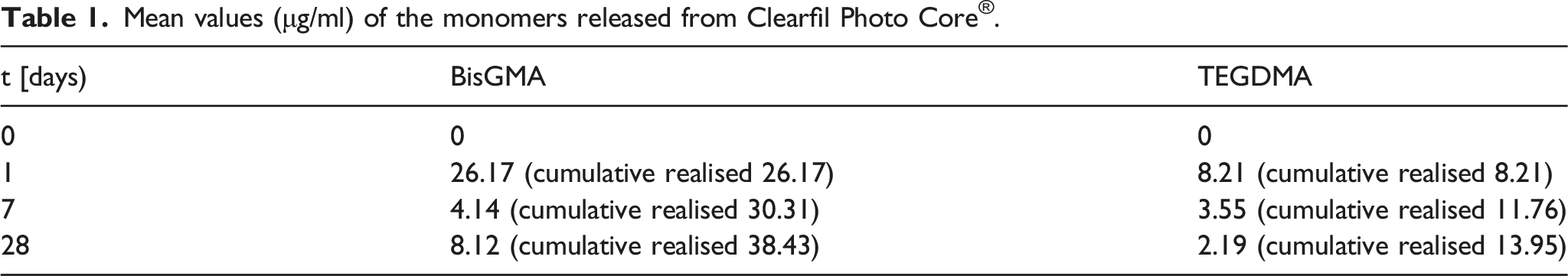
Mean values (μg/ml) of the monomers released from Clearfil Photo Core®.
Discussion
In the present study, the elution of BisGMA and TEGDMA monomers from commercially available composite resin material was evaluated at three time intervals (i.e., 24 h, 7 days and 28 days post-immersion). Its novelty stems from the development of a mathematical model that precisely describes the dynamics of monomer release from composite resin. While other authors have proposed different mechanisms underlying this process, as leaching was measured after 28 days only 6 (i.e., rather than at three different time intervals, as was done in this research), the obtained results represent the total amount of leached monomers, rather than delineating the evolution of this process over time.
By performing measurements at several time points, it is possible to more precisely determine the release dynamics, as well as obtain the amount of monomer released at every stage, as indicated by other authors. This method was also applied in prior investigations,6–8,24 and their findings indicate that the release of some monomers does not cease even after 12 months. 24
As expected, in the present study, eluted monomer concentration was the highest in the first 24 h post-immersion (acute release phase) and its quantity gradually declined over the study period, concurring with the findings reported in extant literature.5–8,14–16 Moreover, focus was given to BisGMA and TEGDMA monomers due to their potential toxicity and widespread use in both research and practice.6–13,24
The elution of components from composite resin is influenced by several factors, including the monomer conversion degree, the composition and solubility of the extraction solvent, and the size and chemical characteristics of the leachable species. 14 The composite resin in focus of the present study comprised primarily BisGMA and TEGDMA cross-linking monomers, along with small amounts of other monomers. On the other hand, filler content varied considerably in the 86.5 Wt% to 68 Vol% range. Moreover, at 26 μg/mL, the BisGMA elution rate was considerably greater than 3.37 μg/mL measured for TEGDMA. These findings are in line with those obtained in previous studies involving composite resin6–8 and can be attributed to the higher BisGMA content in the matrix, as confirmed by subjecting uncured materials to HPLC analysis. For this reason, small amounts of TEGDMA are often added to the matrix to dilute highly viscous BisGMA, thus achieving an optimal balance between curing reactivity and mechanical properties of the polymer. 25
The detected amounts of leached monomers support the fact that BisGMA leaching is more pronounced even though both materials exhibit the same leaching dynamics. Specifically, the highest rate is observed within the first 24 h, after which the eluted quantity gradually decreases, but the process does not cease after 28 days. 24 These findings are in accordance with those reported by other authors.6–8,24 In one such investigation, a two-compartment mathematical model was developed to describe the dynamics of gentamicin leaching, 21 while according to our knowledge this is the first two-compartment model that describes monomer elution where V2 tends to zero.
In extant research on monomer elution, artificial saliva, distilled water, ethanol, methanol and acetonitrile were typically adopted as solvents. As monomer leaching rate was considerably higher when ethanol was used as extraction solution, 24 and the United States Federal Drug Administration (FDA) recommends 75% ethanol–water solution as a clinically relevant food–oral simulating liquid, it was the preferred choice in several studies.6–8,24 Moreover, as ethanol is capable of penetrating into the resin matrix and swelling the polymer network, it promotes the release of unreacted monomers. 7 For the aforementioned reasons, it was adopted as the extraction solution in this study.
As sample surface may also influence the outcome, it is worth noting that in the present study the composite specimen surface area was approximately 95 mm2, which is comparable with the average surface area of large restorations in clinical practice.6–8,24
However, when interpreting the findings reported here, some limitations of this study should be noted. In particular, due to its in vitro nature, the conditions within the oral environment could not be fully replicated, such as temperature changes, pH environment variations, as well as constant saliva changes, all of which would impact the restorations comprising composite resins.
Thus, additional research is needed to address these shortcomings. Moreover, as the experiment terminated after 28 days, studies of longer duration are needed to establish for how long the leaching would persist. It would also be beneficial to evaluate and compare monomer elution from composite resin materials under conditions that more closely emulate clinical application.
Clinical significance
Residual monomers (Bis-GMA, TEGDMA) were eluted from composite resin materials at all time periods and it has also been related to possible toxicological effects.
Our results show that the highest amount of eluted monomers concentrations was detected in 24 h. The amount of eluted monomers decreased with time. The mathematical model based on two compartmental pharmacokinetic system with General fractional derivatives excellently describes the quantity of released monomers.
Additional research is needed to see how long the residual monomers leaching lasts and after how long it stops.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work of TMA is sponsored by Faculty of Technical Sciences, University of Novi Sad.