Abstract
Numerous water-treatment marital have been developed during the past few years, including adsorbents., the catalytic reduction behavior of carboxylated alginic acid derivatives has been investigated against harmful organic dyes including Methyl Orange (MO) and Congo Red (CR). Alginic acid was firstly oxidized through an easy addition of KMnO4 as an oxidizing agent. A carboxylated alginic acid (CAA) has been interacted with selected metal ions (Sn, Fe, Ni, and Zr) through coordination bonds at the value of pH = 4 to form the corresponding metal complexes namely: Sn-CAA, Fe-CAA, Ni-CAA, and Zr-CAA. The consistency of the coordination was confirmed by several spectroscopic techniques including FT-IR, XRD, SEM, and EDX. The catalytic reduction of these metal ion-based products was carried out against MO, and CR in the presence of NaBH4 as a reducing agent under UV irradiation. All catalysts-based metal complexes showed enhanced catalytic reduction against CR compared to MO. Among all those mentioned metal complexes Sn-CAA showed the best catalytic reduction of these dyes. The time is taken by the Sn-CAA for CR, and MO is 5 and 7 min respectively. Ni-CAA was classified as the second efficient product against both dyes, where the reduction process took 20 and 9 min respectively. The other two catalysts took a long time for CR and MO reduction. Zr-CAA showed more than 80% reduction of only CR dye within 20 min. Whereas, Fe-CAA did not show any significant sign of reduction against both the dyes after the same time. The order of higher catalytic reduction was illustrated as: Sn-CAA > Ni-CAA > Zr-CAA = Fe-CAA.
Introduction
It is imperative that one get access to water that is of high quality because water is one of the primary necessities necessary for human living The expansion of industry has resulted in the production and release of significant quantities of pollutants into the surrounding environment, which has caused a number of critical issues. When it comes to contaminants, heavy metals and dyes are the most prevalent and dangerous types. This is because the consumption of these substances has the potential to have a very long-lasting impact on the human body. The effluents of wastewater have been found to include a wide variety of synthetic dyestuffs and heavy metals. 1 Due to its potential impact on human health and safety, water treatment has prompted researchers to focus on the use of non-hazardous components in their search for an answer to the dye pollution problem in the water. It is therefore necessary to make use of environmentally friendly items as well. There are several naturally occurring sources of polysaccharide, some of which include cellulose, starch, chitosan, alginate, carrageenan, and pectin. Wood and seaweeds are two of the most common natural sources of alginic acid, which is one of the most common natural polymers. Because of its one-of-a-kind characteristics, such as biocompatibility, sustainability, low toxicity, and renewability, it has the potential to become an essential component in the development of innovative functional materials. In terms of its application as a foam for packing. 2
Nowadays, the use of biopolymer as a catalyst is increased due to minimizing the procedure expense and make several applications more practicable and environmentally friendly. The biopolymers such as chitosan (CS), 3 starch, 4 alginate and cellulose 5 etc. have been extensively utilized as heterogeneous green catalysts in the chemical transformations. Furthermore, supramolecular designs of usual carbohydrates in recent times attracted much attention as “smart materials” from bio and nano engineering perspectives. These polymers contain various functionalities that exhibit good catalytic applications. When looking at the structure of alginic acid it comprises of homopolymeric blocks of 1-4-linked β-D-mannuronic acid (M) and R-L-guluronic acid (G). Both the acidic units M and G were joined with areas of alternating structure in variable proportions of MG, GG and MM blocks. 6 Moreover, the eye-catching structures of Alginic acid AA are reactive carboxyl and hydroxyl functional groups. This feature made the composition as a template for synthesizing metal coordinate. The functionalization of the polymer material will rise its choosiness towards a specific ion. Permissible to efficiently exploit the hydroxyl groups in the alginic acid an oxidation reaction was performed by means of potassium permanganate (KMnO4), which alters the hydroxyl groups of AA into carboxyl groups namely carboxylated Alginic Acid (CAA).7–11 The choice of a biopolymer for instance AA has specific applications like photo-luminesce, biocompatibility and conductivity, which is really essential, and could be utilized in place of different technological purposes. As a result of the existence of carboxylic and hydroxyl group in its main sequence it shows exceptional films forming, membrane, fibers, and gels abilities. 12 The higher valence metal ions amalgamated materials remain fixated to prepare novel composite having respectable performances like removal and catalytic applications etc. 7 In this application, biopolymers, particularly polysaccharides, are highly sought after adsorbents due to their high adsorption capacity for metal ions and their biodegradability and biocompatibility, both of which provide significant advantages 13 There are many types of synthetic dyes used yearly in millions of tons by the industries. 14 During the dyeing process, many color pigments released into the water reservoir from paper, garments, and pharmaceuticals industries etc. to make water polluted and toxic for the surrounding environment. The strong color of azo dyes is because in their chemical formula they contain (–N = N–) groups.15,16 The azo compounds contain the biggest group of synthetic organic dyes and hugely utilized in food, paper, textile and cosmetic industries. Methyl Orang is a synthetic azo dye, which is one of the significant dyes that is typically used as a coloring agent in the textile and leather industries. MO is one of the important dyes that is generally used as a coloring agent in these sectors. It finds widespread application in the printing and paper production industries, as well as the pharmaceutical and food processing sectors, as well as in research facilities.17–20
The azo group present in MO lowers its biodegradability and makes it harmful to the ecosystem. It causes severe health problems like diarrhea, vomiting, and nausea. 21 For that reason, eliminating and reduction MO dye from water ecosystem is necessary. Previously, there were a lot of interest arises in hydrogenation of catalyst and heterogeneous catalysis.22,23 Amongst the disclosed industries, fabric industry arises as a first rank for coloration of fibers, clothes etc. in using dye molecules. Even though, it’s really tough to eliminate industrialized azo-dyes wastes from water and the utmost techniques utilized for treating the industrial effluents are catalysis, ion exchange, chemical coagulation, flocculation, precipitation, adsorption, biological degradation, membrane filtration, and adsorption using active carbon treatment, ozonation, denitrification process 24 electro-Fenton degradation 25 electrochemical sensor etc. Moreover, among them catalysis shows very good activity for reducing these dyes. As a continuation of our research for the fabrication of different types of hybrid nanocomposite materials for the eco-friendly applications,26–36 we giveaway easy and a simple process, it was done as follows potassium permanganate as an oxidizing agent for alginic acid For 30 min, the operation was carried out at a temperature of 45°C with intense mechanical stirring. Oxdayzing agent was only employed in concentrations up to 10 mM because of its explosive tendency. Centrifugation is used to extract carboxylated alginic acid CAA from the polymer solution. Water with a pH of between 6 and 7 was used to completely clean the material. To prepare the metal coordinates for CAA. For 24 h, the mixture was stirred at RT. Next, the dyes stated above are reduced, and the behavior of these complexes with various metals is explored in an unprecedented method to produce a reduction process completely in record time. The prime theme of our present work was to accomplish a catalytic effectiveness of natural products as a catalyst in the reduction of commercially available carcinogenic Congo Red (CR) and methyl orange (MO) as azo dyes from the aqueous solution functionalized and metal coordinated biomaterials as catalysts to reduce these harmful organic dyes.
Experimental
Chemicals and reagents
Potassium permanganate (KMnO4), hydrochloric acid (HCl), sulfuric acid (H2SO4). Sodium hydroxide (NaOH), potassium dichromate (K2Cr2O7), SnCl2·2H2O, NiCl2.4H2O, FeCl2.2H2O, ZrOCl2.8H2O. Sodium borohydride (NaBH4, 99%) Alginic acid (AA), were obtained from BDH chemicals Ltd Poole England. From Merck (Germany) MO and CR azo dyes were bought. Solvents and chemicals utilized were of analytical grades without further treatment. Deionized water obtained from Milli-Q Plus system (Millipore, Bedford, MA, USA).
Characterization
All spectrophotometric measurements recorded with UV-visible spectrophotometer (Shimadzu 1800) with a 10 mm (path width) quartz cell. Orion pH meter (model EA 940) was employed for the test solutions and pH measurements and a digital micropipette (Volac) for the preparation all solution. Deionized water obtained from Milli-Q Plus system (Millipore, Bedford, MA, USA) and used for the preparation of solutions. field emission scanning electron microscope (FESEM) (JSM-7600 F, USA) for morphological studies also for elemental studies were carried out with energy dispersive X-ray spectroscope (EDX) fitted to FESEM and X-ray photoelectron spectroscope. Fourier transform infrared (FTIR) spectroscope (Thermo scientific, USA) for collection of FTIR spectra data (Range: 500–4000 cm−1).
Preparation of coordinated M –CAA
For oxidizing alginic acid (5 g), it was treated with an oxidizing agent potassium permanganate having concentration of 10 mM. The process was done under strong mechanical stirring for 30 min at temperature range of 45°C. 11 Due to the explosive nature of potassium permanganate, it was used within the range of 10 mM as reported earlier. 15 The polymer solution goes under centrifugation to obtained CAA. The material was thoroughly washed with distilled water to reach the pH up to 6–7. The CAA dried in oven for 2 h at 60°C. To prepared the metal coordinates of CAA with Ni+2, Zr+4, Sn+2, and Fe+2 metals, two round bottom flasks were taken and each one contain 2 g CAA in 100 mL DI water. It was than treated with 0.2 mol of metal solution (NiCl2.4H2O, ZrOCl2·8H2O, SnCl2·2H2O and FeCl2.H2O). The mixture was stir continuously for 24 h at RT. For metal complexation, to keep the pH around four. The solution mixtures were treated with aqueous solutions of (1M) H2SO4 and NaOH. The final product obtained after filtration washed vigorously by DI water and keep in oven for 4 h for drying at 60°C.
Results and discussions
FT-IR Fourier-transform infrared spectroscopy
After doing oxidation of the AA, we checked the formation of carboxyl group by FTIR. The peak at around 1745 cm−1 shown in Figure 1(b) is coming from carbonyl group present in AA after oxidation. At around 3000–3500 cm−1 we can clearly observed the fall in intensity of peak in Figure 1(b) from the parent material i.e. AA. Figure 1(a) which confirms the formation of the carbonyl group to AA. During the meantime we also treat the CAA with metals as well and to check the formation of the coordinate bond with metals. When the coordinate bond formed between metal and CAA the intensity at around 1745 cm−1 disappeared Figure 1(c)–(f), and again it shows broad peak at around 3000–3500 cm−1. This approves that protons that are in the carboxylic acid had changed and formed a harmonic bond with metals.37,38 FTIR: (a) AA (b) CAA (c) Sn-CAA (d) Zr-CCA (e) Ni-CAA (f) Fe-CAA.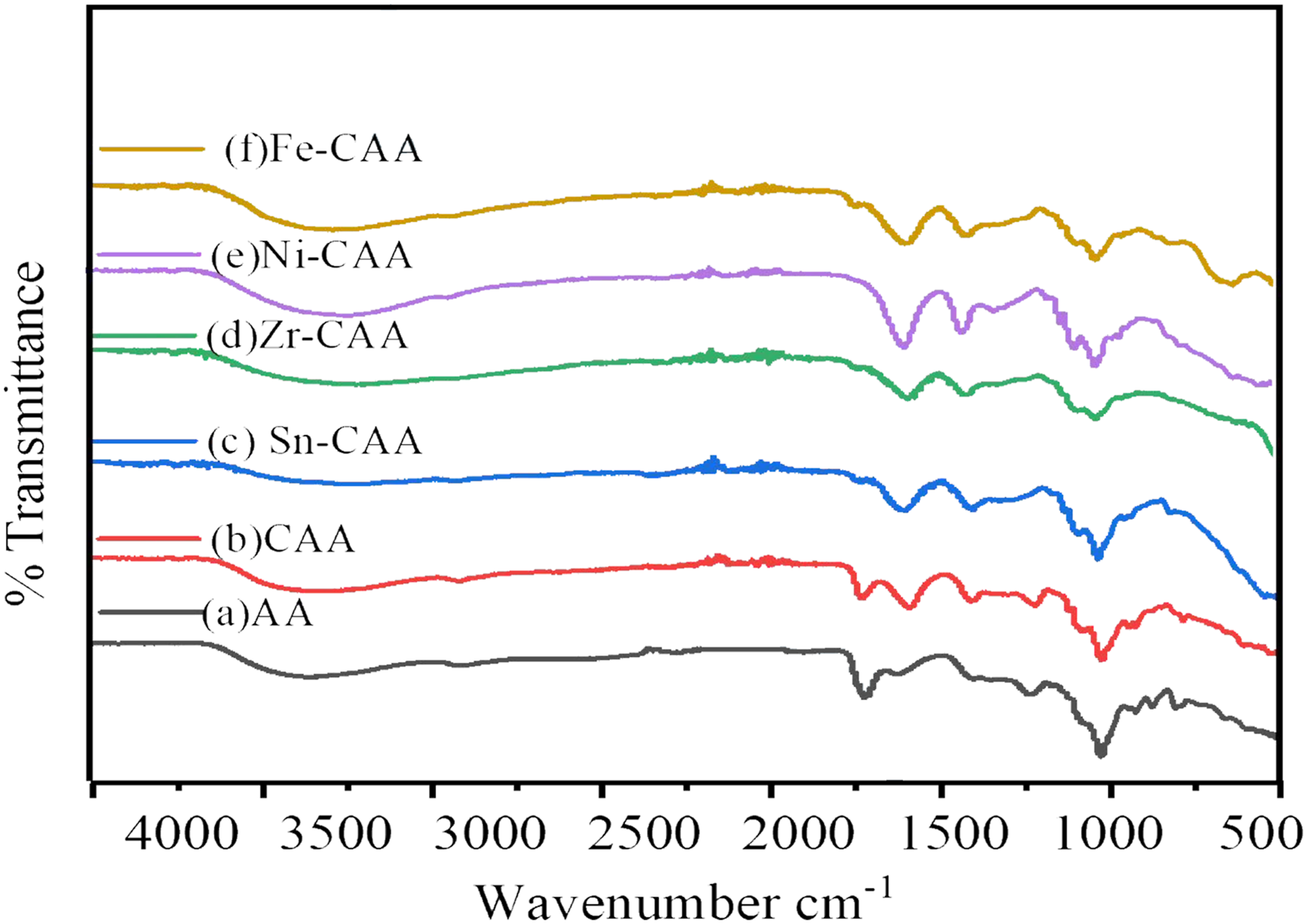
The X-ray diffraction (XRD) of CAA and M-CAA
To carry out X-ray diffraction pattern for studying the lattice structure of CAA and M-CAA adsorption materials. Figure 2 shows the XRD spectra of CAA (a), Zr-CAA (b), Sn-CAA(c) and (d) Ni-CAA respectively. The distinguished crystalline peaks of CAA(a) were located at 14.47° and 21.31°, respectively, representing the reflections of the (422) and (511) planes Figure 2(a).39,40 XRD patterns of (a) CAA and (b) Zr -CAA (c) Sn-CAA (d) Ni-CAA.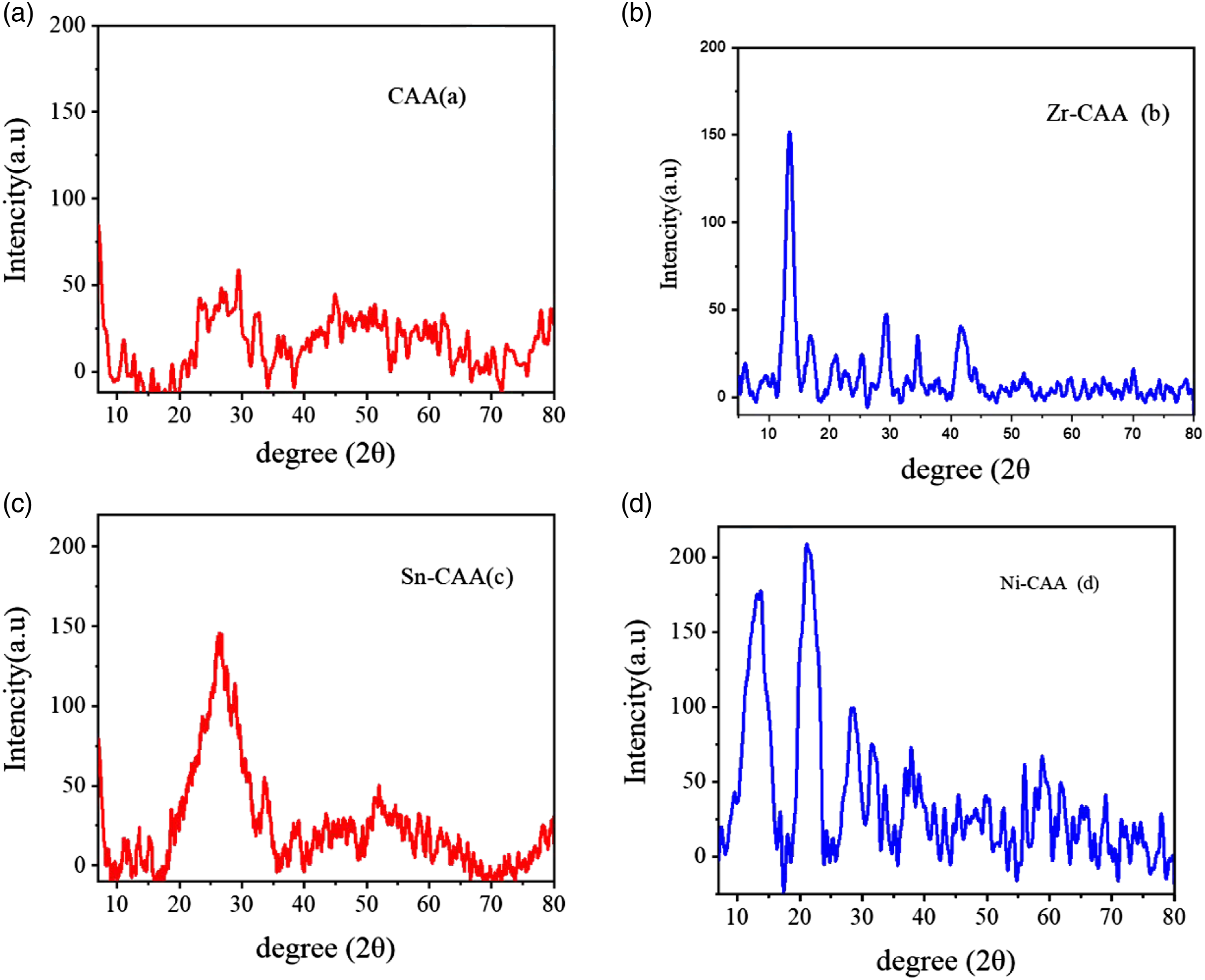
The XRD pattern analysis of Zr-CAA Figure 2(b) showed that there was a tetragonal phase. As well Sn-CAA and Ni-CAA Figure 2(a)–(d). The analysis also confirmed the semi-crystalline nature, which improved the structural stability of the manufactured M-CAA adsorbent material. The semi-crystalline nature of M-CAA may be deduced from the fact that its distinctive peaks occur at angles of 13°, 56°, 20.64°, 20.06°, 28.96°, and 36.40°.41,42
Scanning electron microscopy (SEM) image and EDAX for CAA, Sn+2 and Zr+4Ni+2, Fe+2
By checking the FE-SEM pictures shown in Figure 3, the morphology of net CAA and Zr-CAA as a selected example can be clarified. The SEM images a, b represents the CAA surface, which consists of, aggregated tiny particles in both lower and medium magnifications which is in accordance with the literature.
39
The images from (c, d) represent from medium to high magnifications of Zr-CAA (x = 7,500 and 60,000) respectively. The medium magnification shows the surface consists of sponge like shape; while the resultant high magnification image (x = 60,000) shows accumulative tiny spherical particles in the nanoscale. The complex mechanism between the selected metal cation and CAA can be used to explain the difference in morphological structure formed in these imaged compare to the reported images for pure CAA.
39
These tiny points indicate Zr are effective in coordination with CAA. The Zr-CAA images give a good evidence for the complexation process throughout metal ion chelation However, the reported CAA images undoubtedly demonstrate morphological features quite close to those found in the literature survey Furthermore, the synthesized Zr-CAA selected sample has been further investigated by Energy Dispersive X-Ray Analysis EDX to quantitatively defined the elemental composition. The Zr were undoubtedly spotted in Zr-CAA formulation. Zr alongside C and O detected in Zr-CAA, proposing the effective formation of Zr-CAA. C and O elements were detected due to the presence of carboxylic alginic acid. Zr was identified along with C, and O in Zr-CAA catalyst and the weight percentage for C, O and Zr in the form of Zr-CAA is 54, 44 and 2% respectively. SEM micrographs of (a, b) CAA at magnifications of (x = 750, 1500); (c, d) Zr-CAA at magnifications of (x = 7500, 60,000); (e) EDX analysis of Zr-CAA.
Catalytic study
Due to the diverse application of dyes and their unhealthy effect on the surrounding environment modification, we select two anionic dyes MO and CR for our study. Which common dyes used in many applications. We prepared a facile method for the reduction of these dyes. The CAA metal coordinate complex utilized as a catalyst in the presence of a reducing agent using UV-vis spectrometer technique in the wavelength range of 200–800 nm. To know the catalytic activity of the catalyst against model reaction of CR and MO we prepared fresh sodium borohydride having concentration of 0.1 M. We took 2.5 mL of dyes in cuvette, take the reading after that we add 0.5 mL of reducing agent sodium borohydride, and measured it. Then 10 mg of metal catalyst immersed in a cuvette and MO at 468 nm and CR at 495 nm. The time interval for the reaction was 1 min. The calculation of reduction of these dyes done according to time and percentage reduction equation as follows
43
Proposed mechanism of reduction of MO and CR.
Evaluation of M-CAA toward CR and MO
The selection of materials and their nature and chemical properties are important for many different applications, especially water purification. Therefore, it is necessary to choose materials with unique properties in terms of efficiency in their work as well as their cost and have an anti-bacterial feature. Many applications and researches applied to water purification. Nevertheless, all methods have a cost and may require high-cost devices, which makes their efficiency in the end less than Less than hoped for in the process of purifying the water from toxic material such as azo dyes. The method of catalytic reduction of dyes is a simple and easy method. In addition, many nanoscale materials used to remove dyes or even reduce them, but there are some disadvantages to them, which is the difficulty of removing them from the water, which requires difficult techniques or even filtering it to get rid of them. All these are defects of some of the materials mentioned. Polysaccharides such as chitosan or carboxymethylcellulose. They have been used to reduction dyes in many literature because of the properties that make it easy to get rid of them after use.in this study, in this work The carboxylated alginic acid CAA used as a safe organic biopolymer that contains many active groups that are important in interaction with metals as coordinate bonding for reduction harmful anionic azo dyes. Through the experiment in Figures 5(a) and 8(a) it was found that AA and (NaBH4) without modification it was found no effect on ionic dyes because they both carry the same electrical charge also, Only NaBH4 has been shown to be ineffective in the reduction of CR and MO dye in previous studies.
44
The reduction of these dyes is beneficial from a thermodynamic perspective, but it is exceedingly sluggish from a kinetic one. With time, the wide peak centering was seen to progressively decrease for CR and MO respectively. NaBH4 lowered the CR and MO dyes because of the presence of M-CAA, according to this study. UV-Vis spectra for reduction of C.R by NaBH4 with time in the presence of AA (a) C.R by NaBH4 with time in the presence of CAA (b).
On other hand CAA and (NaBH4). There were more effective groups such as the carbonyl group and hydroxyl, but in studying it with harmful dyes such as M.O and C.R, the result was unsatisfactory due to the presence of high electrical charges on many hydroxyl and carbonyl groups as Figures 5(b) and 8(b). Similar to the charge carried by the dyes thus there was no significant effect on the reduction reaction.
Catalytic reduction of CR
An anionic azo dye, which is widely utilized in various industries, is Congo red (CR). Because of its utilization in various fields like textile, cotton, silk, food, paper and pulp manufacturing factories, CR is very important in various azo dyes. It is utilization in medical field as a biological stain for the identification of amyloidosis, and as a pointer in acidic media.45,46 In addition to the extensive uses of CR, it also has toxic effects on wildlife. CR can cause vomiting, gastrointestinal irritation, skin and eye irritation, diarrhea, etc. In women it causes liver tumors for exposing long-term to CR.47,48 CR could report allergic reaction. The CR disposal rate is extraordinary in the use and manufacturing procedure. Traditional treatments cannot effectively remove CR. In addition, under anaerobic conditions, CR digested into human carcinogen called benzidine.48,49
The reduction of CR takes place in the existence of M-CAA and NaBH4. For reducing, 2.5 mL of 0.13 m
By adding 10 mg of M-CAA, the peak intensities at 493 and 344 nm begin to decline. As shown in Figure 6(a)–(d), the decrease in CR was recorded by measuring its UV-vis spectrum at regular time interval of 1 min between the two measurements. Adding of M-CAA reduces both peak intensities. However, as the intensity increases, a fresh peak arises at 252 nm, as displayed in Figure 6(a), (c) and (d). Therefore, the steady vanishing of peaks at 493 and 344 nm is because of CR reduction. Therefore, the arise of a new peak at 252 nm is due to the formation of free amino group compounds in the course of the reduction process of CR. The reduction CR was done against different metal catalyst like Figure 6 Sn-CAA (a), Fe-CAA(b), Ni-CAA(c)and Zr-CAA(d)respectively. Among them Sn-CAA shows the best result as it reduction the CR in 5 min while the others show long time for reduction. Fe-CAA took 20 min but did not show any effect of reduction, Ni-CAA took 9 min to reduce more than 95% and Zr-CAA took 27 min to reduce the CR up to 85%. UV-Vis spectra for reduction of CR by NaBH4 with time in the presence of M-CAA catalytic Fe -CAA (a), Sn-CAA (b), Ni-CAA (c), Zr-CAA (d).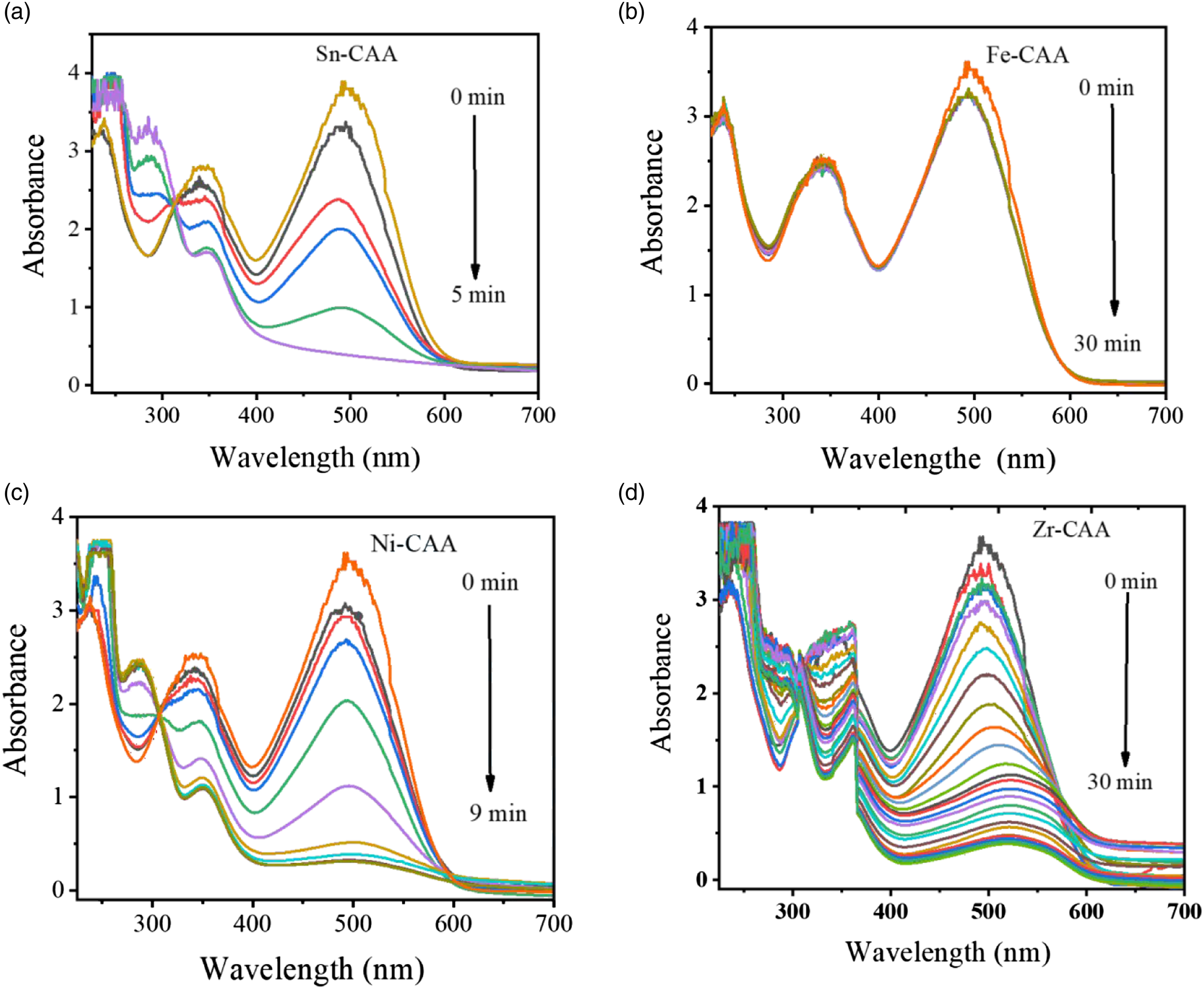
The percentage reduction of CR was measured as of the map of the UV-vis spectrum by means of equation (1). At this point, in the presence of NaBH4 and M-CAA, CR was reduced completely as shown in Figure 7(a). According to the earlier reports, NaBH4 alone does not show any effect on CR, even when utilized in excess.
50
Thus, the overall reduction process of the CR monitors the pseudo-first order kinetics. The calculation of the rate constant (Kapp) was based on the relationship between ln(At/A0) and the reduction time (as shown in Figure 7(b) by formula (1). The calculation done for Kapp from the slope of CR existed as Kapp = 0.6602 for Sn and Kapp = 0.427 of Ni and Kapp = 0.173. Zr and Kapp = 0.173 for Fe. Comparative percent reduction of CR, and (d) plot of ln (At/A0) versus time for CR. Experimental conditions (2.5 mL of aqueous dye solution + 0.5 mL of 0.1 M aqueous NaBH4 solution + 10 mg of M-CAA).
Catalytic reduction of MO
With the growth of industry, human beings are facing severe environmental problems because of releasing enormous number of pollutants into the water-based environment, particularly dyes and other organic molecules. The ejection of sewage into the ecological unit is a chief hazard to marine ecosystem and human health. The presence of azo (–N = N–) group in chemical structure of the dyes gives them deep color.15,16 Amongst organic synthetic dyes, azo group has established a major class of dyes and is extensively exploited by means of textile, food, cosmetic, paper and pulp manufacturing factories. MO is also anionic azo dye often utilized as an indicting material in laboratories and in the paper, textile, cosmetics, rubber and pharmaceutical industries etc.17,18
Emission of azo dye unwanted liquid from numerous industries, exclusively the textile industry, into the aquatic ecosystem pose a thoughtful warning. Therefore, the improvement of fresh methods intended for the decomposition of diazo has attracted boundless attention. The MO has intense peak around 468 nm. According to earlier reports, NaBH4 alone can reduce MO dye thermodynamically which is beneficial but the reaction is very gentle kinetically. 51 Lack of an appropriate catalyst, even within a few hours, the peak intensity at 468 nm peak of MO remained constant.52,53,67,68
MO was strong absorption peak in the UV-visible spectrum at 468 nm. In the absence of a catalyst, the peak intensity does not change for AA as shown in Figure 8(a). We than modify the AA to CAA and again check the UV for MO it also shows similar result like its predecessor Figure 8(b). UV-Vis spectra for reduction of MO by NaBH4 with time in the presence of AA (a) MO by NaBH4 with time in the presence of CAA (b).
In order to catalytically reduce MO, first put 2.5 mL of dye solution into the quartz cuvette and then add 0.5 mL of NaBH4 having 0.1 M concentration. The UV-vis spectrum of MO shows an intense peak at around 468 nm. Before adding the catalyst, the peak intensity of MO remains intact at 468 nm although, after introducing 10 mg of metal catalyst, the peak intensity at around 468 nm began to decrease.
As shown in Figure 9(a)–(d), the catalytic study of M-CAA was measured by assessing the UV-vis spectrum using a time span of 1 min. By adding M-CAA, the decrease in intensity of peak at 468 nm was observed. Therefore, the steady vanishing of the peak around 468 nm because of reducing the MO, usually in the existence of a vigorous catalytic agent, NaBH4 reduces MO molecules at the azo spot by producing low molecular free amino group compounds. Therefore, during MO reduction process, the formation of free amino compounds is the reason for the creation of a new peak at 250 nm. UV-Vis spectra for reduction of M.O by NaBH4 with time in the presence of M-CAA catalytic Sn -CAA (a), Fe -CAA (b), Ni-CAA (c), Zr-CAA (d).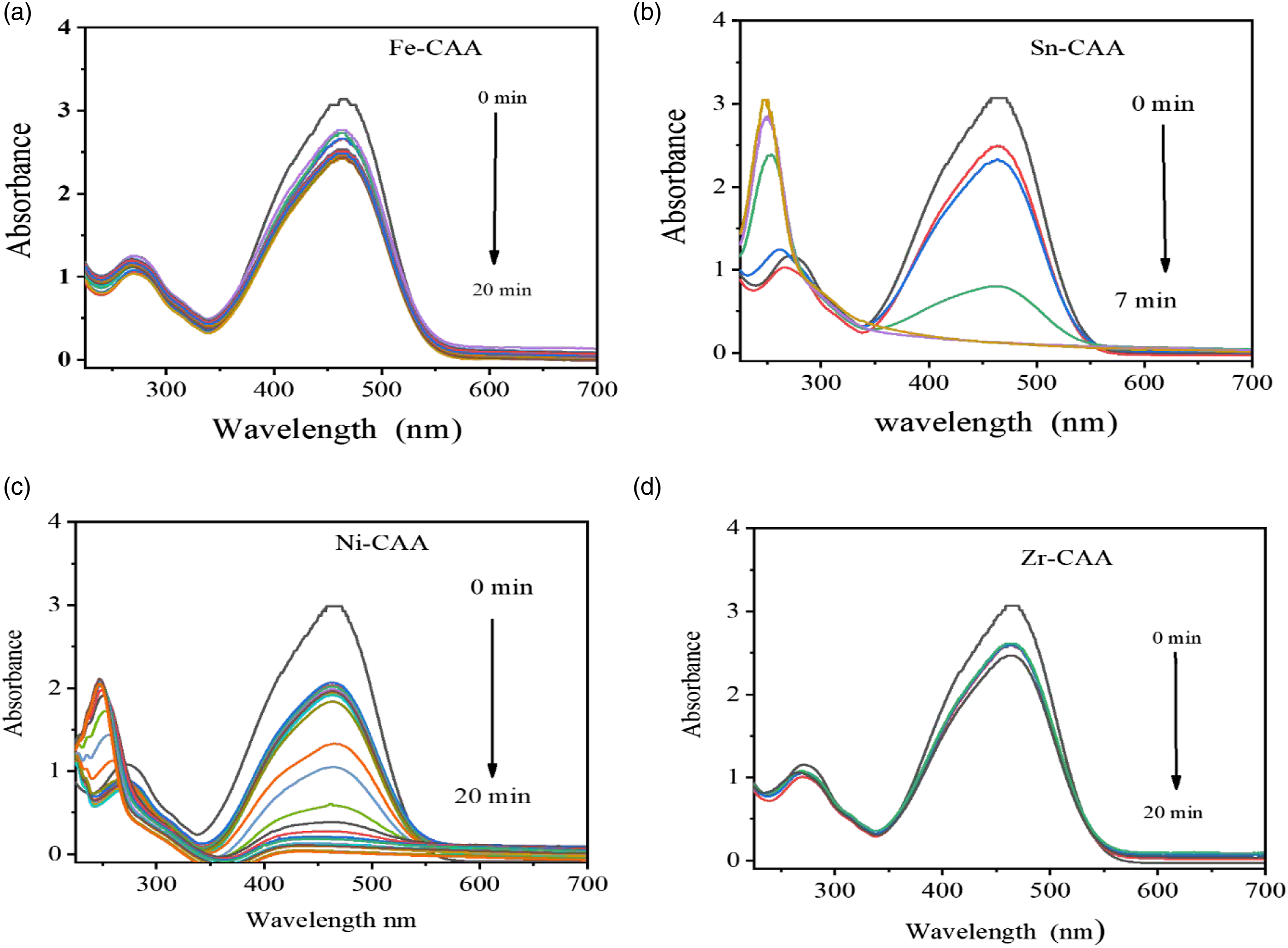
The effectiveness of (M-CAA) has been tested with the aforementioned metals. Above we see the figures for each metals and the extent of its effect on the dyes M.O, and we conclude from them that the Sn is the best metal, as we see that it did not take much time less than 7 min in the process of reducing MO dye compared to the rest of the metals (Ni, Fe and Zr) which take more time in Ni cause 20 min but for Fe and Zr not more effect as Figure 9(a) and (d).
The apparent rate constant (Kapp) calculation was based on the relationship between ln(At/A0) and the reduction time as shown in Figure 10 by formula (1). The calculation done for Kapp from the slope of MO was Kapp = 1.44 for Sn and Kapp = 0.0413 of Ni and Kapp = 0.14. Zr and Kapp = 0.139 for Fe. (a) comparative percent reduction of MO, and (b) plot of ln (At/A0) versus time for MO. Experimental conditions (2.5 mL of aqueous dye solution + 0.5 mL of 0.1 M aqueous NaBH4 solution + 10 mg of M-CAA).
Catalytic reduction of mixed solution of MO and CR with Sn-CAA
Catalyst-based M-CAA
Clearly, the quantity of metal bound was more in Sn than in Fe, Ni, and Zr, which is why the reduction process happened more rapidly. However, Sn-CAA required less time to completely reduce both dyes than other metals, 7 and 5 min for CR and MO, respectively. This may be owing to the differing actions of Fe, Ni, and Zr. The time required for the reduction of CR and MO by NaBH4 in the presence of Sn-CAA was smaller than that reported in previous reports of supporting materials.52,54–61,65,66 To ensure its efficacy as a reducing agentWe mixed the two dyes with 10 mL of 0.1 mM of each CR and MO solution, and then added 2.5 mL of the mixed in the UV cuvette, (0.1 M) 0.5 mL NaBH4 and 10 mg of Sn-CAA for analysis. Under the same conditions of testing the two dyes separately, a decrease in the absorption of the UV spectrum is observed every minute. Therefore, when the two dyes are mixed, we have one peak, and it is broad and large, which indicates that the two peaks overlap because they are both close to aggregate absorption (465 nm and 495 nm) MO and CR, respectively. When the two dyes are added with Sn-CAA, they will affect the reduction process in a short time of less than about 6 min. We can see that the peak intensity decreases at 476 and 344 nm, and with increasing, at 252 nm A new peak appears with very high intensity, because free amino compounds are formed during the reduction of CR and MO, which shows the effectiveness of Sn-CAA in the reduction process, and even the two dyes together show excellent performance, as shown in Figure 11. The apparent rate constant (Kapp) calculation was based on the relationship between ln (At/A0) and the reduction time (as shown in Figure 11(c) by formula (1). The Kapp measured from the slope of CR was Kapp = 0.598. In the presence of Sn-CAA catalyst, UV absorption spectra of mix solution M.O and C.R as a function time. (a) percent removal of M.O and C.R in the mixed solution with time (t). (b) ln(At/A0) and K app value (c).
Recyclability
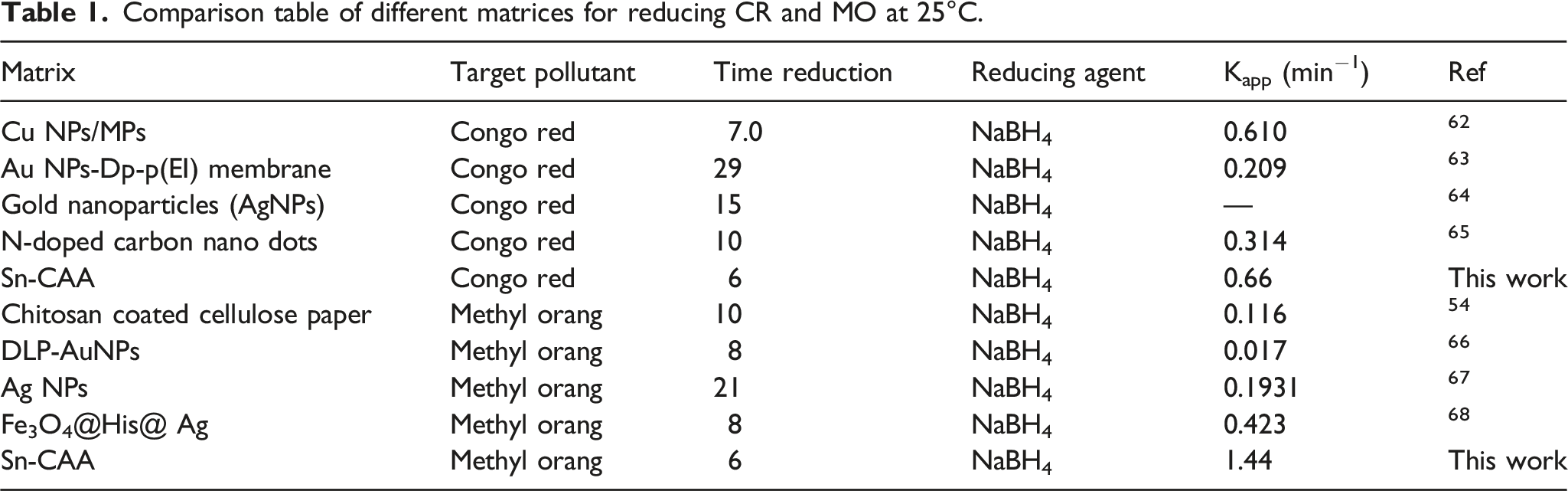
The recyclability of Sn-CAA as a coordination bond was determined. Following the same experimental procedure as before to reduce C.R and M.O. We used the same carboxylated Sn-CAA, washed with water, and reused in seven reductions of C.R and M.O solutions. In the course of seven uses of coordinated carboxylated alginic acid, a reduction of more than 90% was observed. In Figure 12 you can see the time taken to reduce the CR and MO solutions to more than 90% in the first, second, third, fourth, fifth, sixth, and seventh cycles. For the MO solution, in the 90% reduction process, the reduction was completed in less than 7 min. The first cycle takes 7 while 9, 11, 13, 13, and 15 min for the other cycles. In the case of C.R, 90% of the reduction reaction can be completed in 5 min, while most of the conversion in the repeated cycle is completed in 5, 7, 10, 11, and 11 min. We have observed that the reduction time increases when using the same Sn-CAA, which indicates a decrease in catalytic performance. Furthermore, a comparison table regarding the using of different matrices against the reduction of CR and MO at 25°C has been listed in Table 1. Re-use of Sn-CAA catalyst for 7-times reduction of M.O and C.R as a function of time. Comparison table of different matrices for reducing CR and MO at 25°C.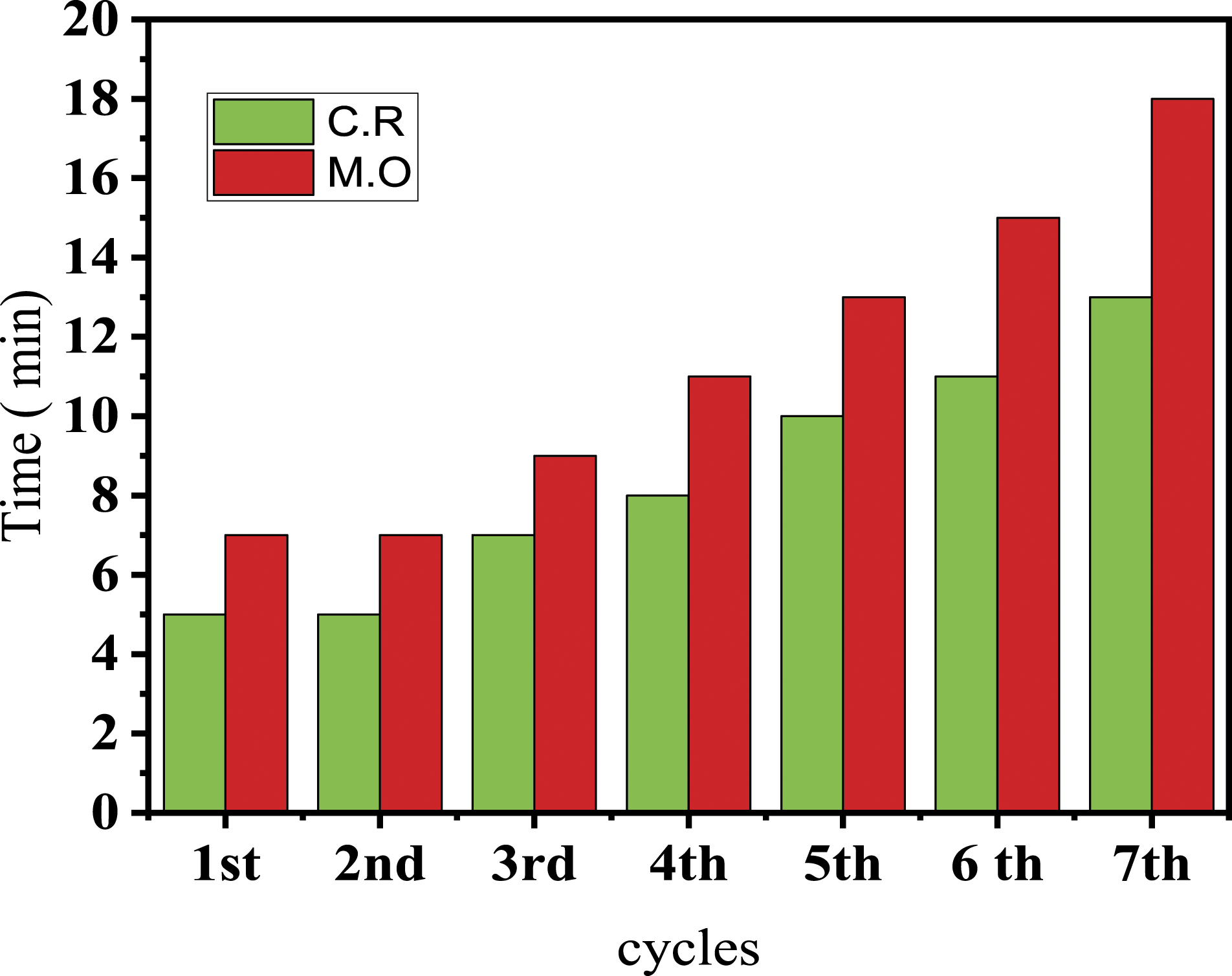
Conclusion
Safe, low costly and ecofriendly modified alginic acid derivatives in the form of M-CAA were successfully prepared as coordinate materials including different metal ions (Sn, Fe, Ni and Zr). The desired materials were prepared by easy method using two steps. The final products were confirmed by common characterization techniques including FT-IR, XRD, SEM and EDX analyses. Based on the given results, the biopolymer bonded to metals is considered as a promising material as well The behavior of some metals after their bonding to the biopolymer was studied Moreover, these materials were applied as moderate new materials for the catalytic reduction of (M.O)dyes. Sn-CAA showed the best catalytic reduction for MO (5 min) Whereas, Fe-CAA Zr-CAA and Ni-CAA showed the longest catalytic reduction time against the same dyes The calculation done for Kapp from the slope of MO was Kapp = 1.44 for Sn and Kapp = 0.0413 of Ni and Kapp = 0.14. Zr and Kapp = 0.139 for Fe. Respectively. While in CR case Sn-CAA showed the best catalytic reduction only (7 min). The calculation done for Kapp from the slope of CR existed as Kapp = 0.6602 for Sn and Kapp = 0.427 of Ni and Kapp = 0.173. Zr and Kapp = 0.173 for Fe. The optimum conditions for all the used catalysts were measured. Sn-CAA catalytic reduction showed an efficient recyclable product after four cycles then gradually become less efficient. Furthermore, Sn-CAA showed an excellent result for purifying natural water from toxic organic wastes as an example for real test sample.
Footnotes
Acknowledgements
The authors also thank Dr SH Abdel-Hafez for his kind support and continuous help to the research team to finalize this work as required.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Taif University Researchers Supporting Project number (TURSP-2020/244), Taif University, Taif, Saudi Arabia.

