Abstract
Montmorillonite intercalated with mixed alumina–iron (III) oxide pillars and promoted with hexamminecobalt (III) chloride was studied in selective catalytic reduction of NO with NH3. Pillaring process extended the interlayer spacing by 0.236 nm and increased specific surface area (SSABET) from 33 m2 up to 299 m2. Interlayer space width did not change after calcination process, but high temperature could cause decomposition of hexamminecobalt (III) – [Co(NH3)6]3+, possibly to Co2+. Promotion of hexamminecobalt (III) chloride to pillared clay did not significantly change SSABET. The intercalation process influenced positively selective catalytic reduction activity, but very high formation of N2O was observed. Pillared clay, promoted with [Co(NH3)6]3+, showed higher selective catalytic reduction activity and much lower N2O formation than unpromoted, pillared sample. Clay with interlayer cations exchanged for [Co(NH3)6]3+, without pillaring, showed no selective catalytic reduction activity, but still produced high amounts of N2O.
Introduction
The rapid growth in the concentration of toxic pollutants in air in last decades, mainly due to combustion processes, has become an ecologically serious problem. One of the major air pollutants are NOx. Several techniques might be used for the NOx abatement and one of them is a NH3-selective catalytic reduction (SCR) process (Grzybek et al., 2004; Liu and Ihl Woo, 2006; Samojeden and Grzybek, 2016; Samojeden et al., 2015). Montmorillonite, the main component of cheap and commonly occurring bentonite clay, is an example of possible SCR catalyst raw material. It belongs to the large specific surface area (SSA) clay minerals with 2:1 layer structure, whereas layers of tetrahedral silica sheets surrounds the octahedral sheet of alumina. Due to the excess of negative charge in octahedral and/or tetrahedral sheets, exchangeable cations, such as Na+ or Ca2+ are located between each individual crystal of smectite.
There are several possibilities of smectite modifications that could increase its SSA, cation exchange capacity (CEC), the number and strength of acid sites, etc. One of the well-known ways of smectite modification technique is pillaring. This modification prevents the interlayer space collapse due to the dehydration, increases SSA of the material (Chmielarz et al., 2003), and influences its acidity (Belkhadem et al., 2008). Thermal stability of pillared clay (PILC) can be enhanced by the introduction of mixed-oxide pillars instead of single-oxide pillars (Cañizares et al., 1999). PILCs can be also promoted with the oxides or hydroxides of various transition metals, like Cr3+ (Zhang et al., 2013), Cu2+ (Chmielarz et al., 2003; Motak, 2008), Fe3+ (Chmielarz et al., 2004), V5+ (Arfaoui et al., 2008), Co2+ (Chmielarz et al., 2003), Zr4+ (Boxiong et al., 2014), and others (Bosch and Janssen, 1988). Activity, selectivity, acidity, thermal stability, and temperature window of a catalyst are strongly dependent on the source of clay (Cañizares et al., 1999), pillars composition and height (Cañizares et al., 1999; Mishra et al., 2008), calcination conditions (Belkhadem et al., 2006), and promotor type, as well as the method of its introduction (Chmielarz et al., 2003; Mishra and Parida, 2006).
Single-oxide and mixed-oxide PILCs were the topic of the numerous studies that confirmed their suitability, not only in the nitrogen oxides removal processes from exhaust gases, using NH3 (Boxiong et al., 2014; Chmielarz et al., 2003, 2004) or hydrocarbons (Belver et al., 2004; Valverde et al., 2003) as reducing agents, but also in the other fields of catalysis: volatile organic compound decomposition (Mishra et al., 2008; Oliveira et al., 2008), methanol conversion (Mishra and Parida, 2006), hydrogenation (Marcos et al., 2016), isomerization (Khalfallah Boudali, 2014), or dehydrogenation (Xie et al., 2015).
The main reactions of SCR with NH3 are (Nedyalkova et al., 2013)
Parallel with the reduction of NOx, side reactions may occur that result in N2O formation (Busca et al., 1998), i.e.
Although several SCR studies have been performed on the alumina or iron oxide (III) PILC, the results of the SCR over mixed alumina–iron (III) oxide PILC are very limited, despite the fact that both types of PILCs have been verified as the efficient SCR catalyst (Chen et al., 1995; Chmielarz et al., 2003). The present paper deals with the synthesis of the montmorillonite pillared with mixed alumina–iron clusters, promoted with cobalt complex (hexamminecobalt (III) chloride), its characterization, and evaluation of catalytic properties in SCR with NH3 as reducing agent.
Experimental
Starting material
The main substrate was SAz-1, Ca montmorillonite “Cheto” Apache Country, Arizona, obtained from Source Clays Repository of the Clay Minerals Society. Chemical composition of the clay fraction <2 µm was provided by Mermut and Cano (2001): SiO2: 60.4%, Al2O3: 17.6%, TiO2: 0.24%, Fe2O3: 1.42%, FeO: 0.08%, MgO: 6.46%, CaO: 2.82%, Na2O: 0.063%, K2O: 0.19%, F: 0.287%, P2O5: 0.02%. Chipera and Bish (2001) presented the mineralogical composition of ‘as shipped’ material: 98% of smectite, 1% quartz, and 1% of other compounds. Borden and Giese (2001) measured CEC by the ammonia-electrode method, and it was equal to 123 meq/100 g of the clay. Differential thermal analysis, thermal gravimetric analysis, and derivative thermal gravimetric analysis of SAz-1 sample can be found in Guggenheim and Koster van Groos (2001).
Preparation of samples
Mixed solution of aluminum (III) chloride and ferric (III) chloride with molar ratio 9:1 Al3+ to Fe3+ of 0.33 M total concentration was prepared (0.0297 M AlCl3, 0.0033 M FeCl3). The solution was intensely stirred, and then 0.1 M NaOH was added dropwise to achieve the ratio of 2.5 mol OH− per 1 mol of combined Al3+ and Fe3+. The solution had undergone aging process (80℃ for 10 h) (Doff et al., 1988).
One gram of smectite was dispersed in 10 cm3 of deionized water, using ultrasonic dispersing equipment. The mixed alumina–ferric oligocations were added to suspension, to the ratio of 0.06 mol of the sum of Al3+ and Fe3+ per each gram of smectite. The suspension was mixed for 1 hour (10 minutes of fast and then 50 minutes of gentle swirling). The sample was rinsed with deionized water and decanted until the chloride test result was negative, dried and calcinated in air for 12 h at 370℃.
Untreated montmorillonite and intercalated calcined samples were dispersed (separately) and 25 cm3 of 0.01496 mol/dm3 hexamminecobalt (III) chloride was added. Suspension was shaken for 2 h and filtrated (Derkowski and Bristow, 2012), then air dried. This has resulted in exchange of interlayer cations for [Co(NH3)6]3+ in natural montmorillonite and adsorption of cobalt complex on the surface of intercalated, calcined sample. In this paper, the samples are designated as follows:
substrate montmorillonite – MONT substrate montmorillonite with [Co(NH3)6]3+ as interlayer cations – MONT_HEX intercalated montmorillonite – INT intercalated and calcined montmorillonite – INT_CAL intercalated and calcined montmorillonite promoted with [Co(NH3)6]3+ – INT_CAL_HEX.
Characterization
Oriented specimens of MONT, INT, and INT_CAL were analyzed in Thermo Electron X’TRA diffractometer using Cu-Kα radiation (glass slide method) to obtain (001) X-ray diffraction (XRD) reflection (λ = 0.15418 nm).
Specific surface area (SSABET) of the MONT_HEX, INT_CAL, and INT_CAL_HEX were calculated from N2 adsorption isotherms acquired at −196℃. Nitrogen physisorption on catalysts was performed using ASAP 2020 Micromeritics instrument after degassing at 105℃ for 24 h at ∼1 Pa vacuum. The adsorption–desorption isotherms of nitrogen were calculated by the standard Brunauer–Emmett–Teller (BET) procedure (Brunauer et al., 1938) from the p/p0 range = 0.05–0.25.
Filtrate after the process of [Co(NH3)6]3+ introduction was characterized by UV–vis, using 470 nm wavelength (Derkowski and Bristow, 2012), to determine remaining hexamminecobalt chloride concentration. It allowed the calculation of the amount of [Co(NH3)6]3+ that was introduced into the natural and pillared montmorillonite.
SCR measurement
Each tested sample (0.5 g) was put into an SCR reactor and calcined in helium for 1 h in 500℃ (in situ) before the catalytic test. Experimental setup is presented in Figure 1. The reactor was heated to appropriate temperature (150–200–250–300–350℃, 10℃/min) and the temperature was measured by a thermocouple placed in catalyst bed. Gas concentration upstream of the reactor was: 800 ppm of NH3, 800 ppm of NO, and 3.5% of O2 in helium flow. NO and N2O concentrations after reaction were measured by gas analyzer (NDIR, provided by ABB). A catalyst was kept for 60 min at each temperature. Steady state was obtained after ca. 20 min.
Scheme of used SCR-NH3 equipment.
Initial and final NO concentrations were used to calculate NO conversion, according to equation (1)
Results and discussion
UV–vis analysis of the filtrate has shown that 2.7 mmol and 31.0 mmol of hexamminecobalt (III) cations were introduced into 100 g of INT_CAL_HEX and MONT_HEX, respectively. XRD patterns (Figure 2) proved that intercalation process caused a decrease of 2θ angle of 001 reflection. According to Bragg’s law, it corresponds to the expansion of interlayer space by 0.236 nm. The interlayer space width has not changed during calcination process.
Oriented crystal powders XRD patterns. (a) MONT (black) and INT (red). (b) Imposed XRD patterns of INT (red) and INT_CAL (azure).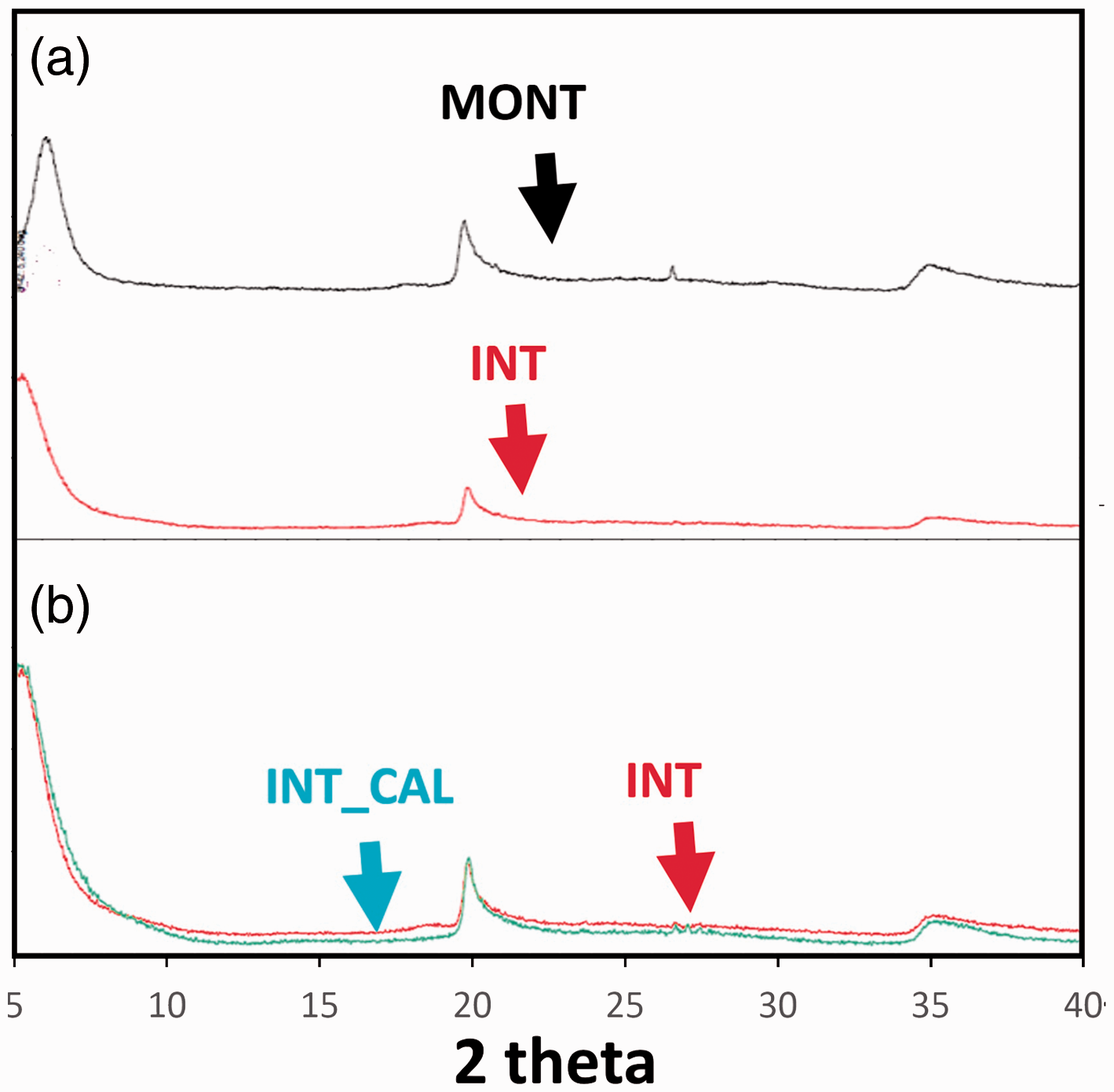
Calculated SSABET of the samples.

The values of SSABET are similar to published results (Valverde, 2000) for montmorillonites, as well as for alumina-pillared smectites. SSABET increased 10 times after intercalation process, and did not change significantly after promotion with [Co(NH3)6]3+.
NO conversion is shown in Figure 3(a), while N2O concentration is presented in Figure 3(b).
(a) Conversion of NO for investigated samples in analyzed temperatures. (b) Upstream N2O concentration in experiment temperature range.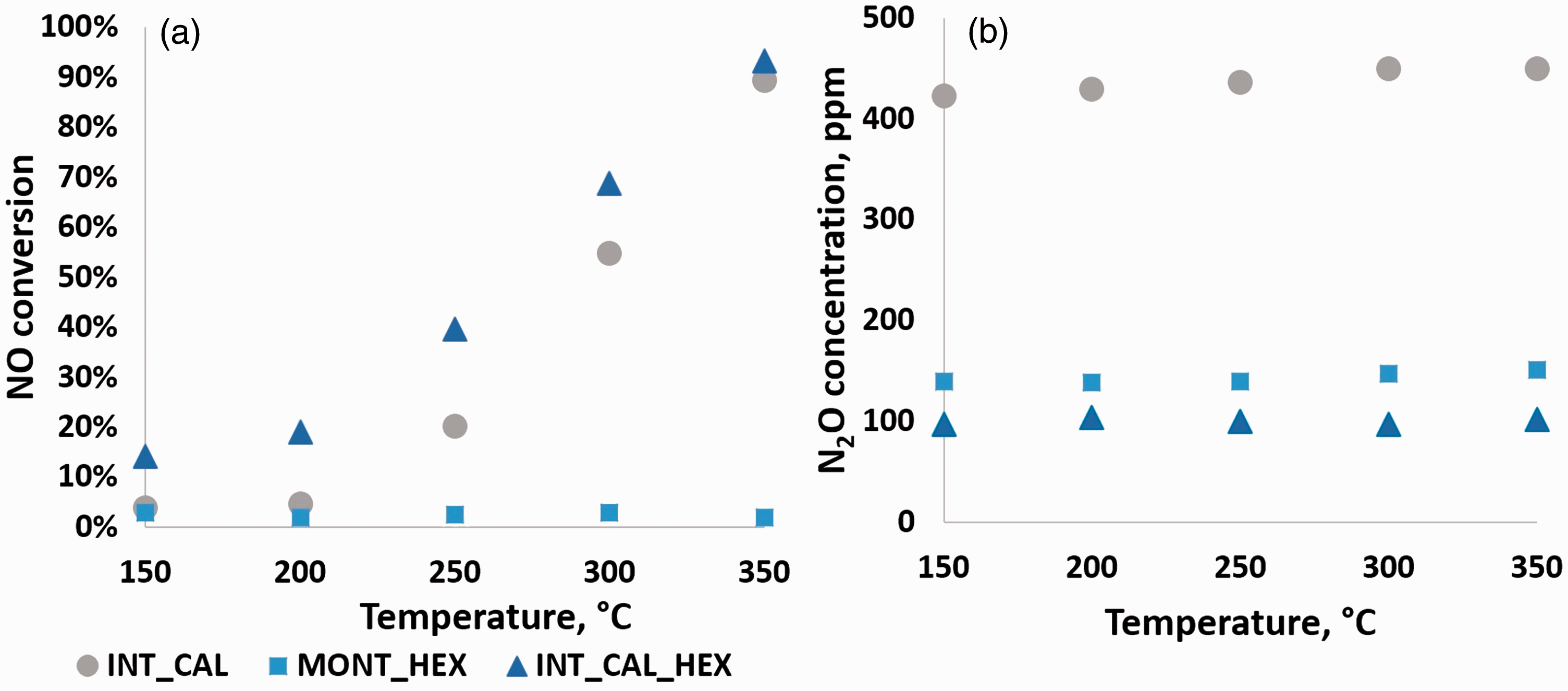
The highest conversion of NO (93%) was observed at 350℃ for INT_CAL_HEX sample. NO conversion for INT_CAL_HEX sample increased much more rapidly at lower temperatures than for INT_CAL. At 250℃ NO conversion was twice higher for INT_CAL_HEX than for INT_CAL, while at 350℃ the difference was only 4%. Chmielarz et al. (2003) reported the NH3-SCR results over unpromoted montmorillonite, up to 470℃, and showed its significant activity in NO reduction, even at low temperatures (i.e. 15% of NO conversion at 300℃, 25% at 470℃). For the whole temperature range, MONT_HEX was almost inactive, which is in contrast to Chmielarz et al.’s (2003) findings. One of the possible explanations could be that the introduction of high amounts of cobalt could decrease surface acidity, which decreased catalyst activity, as SCR reaction requires acidic sites for ammonia sorption (Busca et al., 1998; Grzybek, 2007). Other possible reason could be interlayer space collapse due to dehydration after calcination at 500℃ (30℃ higher than maximum temperature reported by Chmielarz et al. (2003)), which could results in decreased accessible surface area.
For all samples there were no noticeable changes in the formation of N2O at the studied temperature range. The lowest N2O concentration was observed for INT_CAL_HEX, at a constant level of about 100 ppm, which is rather high in comparison to typical SCR catalysts (Long et al., 2001; Long and Yang, 2000). The formation of N2O over MONT_HEX sample was similar to that of INT_CAL_HEX. Due to inactivity of MONT_HEX in NO reduction, it may be concluded that N2O formation was resulting from NH3 oxidation. INT_CAL showed higher N2O formation. N2O concentration for this sample increased from 420 ppm to 450 ppm with temperature increase (150℃ to 350℃). At low temperatures, only low amount of NO was converted, thus the only source of N2O could have been ammonia oxidation. This suggests that the studied catalyst could be considered as efficient, low-temperature-window catalyst for NH3 oxidation rather than for SCR.
According to Figure 3, N2O concentrations for INT_CAL were much higher than for MONT_HEX and INT_CAL. It suggests that cobalt promotion [Co(NH3)6]3+ or the Co2+ generated by its decomposition during the calcination at 500℃ (Fripiat and Helsen, 1966) inhibits N2O formation. Decrease in N2O formation after cobalt promotion is in agreement with the results reported by Chmielarz et al. (2003), where proper amount of Co (molar ratio of Co/Si is equal to 0.002 for Al-PILC and Ti-PILC, obtained from X-ray fluorescence analysis) enhanced selectivity towards N2 over Al- or Ti-PILC.
Conclusions
The studied Al–Fe-PILC promoted with cobalt showed high conversion of NO (∼90%) with significant formation of N2O, and belongs to high-temperature window catalysts group. Relatively small addition of [Co(NH3)6]3+ (or possibly Co2+ by complex decomposition) to the Al–Fe-PILC (2.7 mmol per 100 g of sample) has considerably decreased N2O formation (ca. 4 times) and slightly enhanced NO conversion. Montmorillonite promoted with cobalt complex was almost inactive in SCR, possibly due to the decrease of surface acidity, but still showed activity in NH3 oxidation. Alumina–iron oxide (III) intercalated montmorillonite may be thus a promising low-temperature ammonia oxidation catalyst with high selectivity to N2O.
Footnotes
Acknowledgements
This paper was first presented at the 15th Ukrainian–Polish Symposium on Theoretical and Experimental Studies of Interfacial Phenomena and their Technological Applications, Lviv, Ukraine, 12–15 September 2016.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by Grant AGH No. 11.11.210.373.



