Abstract
ZrO2/Na-β and ZrO2/H-β catalysts are prepared by the reflux impregnation method and are applied to the Meerwein–Ponndorf–Verley reduction of cyclohexanone and isopropanol. The catalysts are characterized by X-ray diffraction, scanning electron microscopy-energy dispersive spectrum, temperature-programmed desorption of ammonia, N2 isothermal adsorption and desorption, and inductively coupled plasma mass spectrometry. The products are identified by gas chromatography‒mass spectrometry and analyzed by gas chromatography. The experimental results showed that ZrO2/H-β exhibits higher catalytic activity for the above Meerwein–Ponndorf–Verley reduction because ZrO2/H-β has both Zr4+ and Al3+ ion acid centers, which provide the moderately strong acid needed for the reaction. Under optimized conditions, ZrO2/H-β(6) is used as the catalyst, and the conversion rate of cyclohexanone was 92.0% as determined by the area-normalized calculation based on cyclohexanone. The catalyst can be regenerated by programmed heating calcination. The conversion rate of cyclohexanone decreased to 79.4% after five recycling cycles, and its service life needs further study to be improved.
ZrO2/H-β possesses higher catalytic activity for the above Meerwein–Ponndorf–Verley reduction ascribing ZrO2/H-β with both Zr and Al Lewis acid sites. While H-β only has Lewis acid sites, Na-β and ZrO2/Na-β have acid–base bifunctional active sites without a good synergistic effect. The strength of the acid and its number of active centers have an important influence on the performance of Meerwein–Ponndorf–Verley. We found that the amount of moderately strong acid plays a vital role in the catalytic Meerwein–Ponndorf–Verley reaction of cyclohexanone and isopropanol.

Introduction
Reducing aldehydes or ketones via the Meerwein–Ponndorf–Verley (MPV) is an important method for producing the corresponding alcohols. 1 Later, under mild conditions, the MPV reaction with alcohol as the hydrogen source was applied to the selective hydrogenative reduction of the C=O bonds of aldehydes and ketones.2,3 Typical examples, such as the hydrogenation reduction reaction of the common α,β-unsaturated aldehydes and ketones, hydrogenate the C=O bond while retaining the unsaturated C=C bond. 4 Therefore, the MPV reaction is widely used in the industrial production of α,β-unsaturated alcohols.5–7 Due to the increased hindrance in ketones, the MPV reaction of ketones is often more challenging and often exhibits lower reaction rates than those of aldehydes under similar conditions. Therefore, cyclohexanone is typically employed as a template reactant. Metal alkoxides are commonly used as catalysts in traditional reactions, 8 and the reactions usually require stoichiometric or even excess of the alkoxide, which hinders the wide application of the MPV reaction. Looking to the future, changing the catalytic reaction based on stoichiometry into cyclable reactions is important for modern fine-organic-synthesis technology.
At present, homogeneous catalysts for the MPV reaction include single metal alkoxides, bimetallic alkoxides, chiral metal alkoxides, and lanthanide bimetallic alkoxides, for example, aluminum isopropoxide,9–11 praseodymium and neodymium, and complex ligands of vanadium. However, the preparation of the reaction catalyst generally requires alcohol and adoption of the metal reflux method in an anhydrous and oxygen-free environment. As can be imagined, the process is complex and strict requirements are needed for the equipment. The heterogeneous catalysts used in the MPV reaction mainly include metal oxides,12,13 zeolite molecular sieves, 14 supported mesoporous molecular sieves, and mesoporous molecular sieves grafted with metal alkoxides, such as magnesium oxide,15–18 zirconia,19,20 MX molecular sieves, H-β molecular sieves, 21 hydrotalcite, 22 Zr-β, Sn-β, 23 and Ti-β. In recent years, surrounding the investigation of catalysts for the MPV reaction, the modification of molecular sieves and the heterogeneity of homogeneous catalysts have gradually increased. In some studies, homogeneous catalysts loaded or grafted onto microporous or mesoporous molecular sieves have even been described. The development of new catalytic materials to realize effective catalysis and cycling is of far-reaching significance to expand the application of the MPV reaction and to improve its application value.
ZrO2 and H-β zeolite are effective catalysts for MPV reductions. Supporting ZrO2 on H-β zeolite increases the specific surface area of ZrO2, while the presence of dual Lewis acid sites enhances the activity of the catalyst. Here, we have prepared ZrO2/Na-β- and ZrO2/H-β-supported catalysts by impregnation. The MPV reduction of cyclohexanone with isopropanol was used as a probe reaction to investigate the catalytic activities of ZrO2/H-β with double Lewis acid active sites and ZrO2/Na-β with acid–base bifunctional active sites. We also explored whether there is a direct relationship between the acidity or alkalinity of the catalyst and the catalytic performance. At the same time, the active life was investigated to lay a foundation for an in-depth study of the MPV reaction.
Results and discussion
Characterization results of the catalysts
X-ray diffraction analysis of the catalysts
Figure 1(a) and (b) is the X-ray diffraction (XRD) patterns of the H-β and Na-β molecular sieves, H-Zr-β(n), and Na-Zr-β(n). Table 1 shows the relative crystallinity of the Na-β and H-β molecular sieves, H-Zr-β(n), and Na-Zr-β(n).

XRD spectra of unmodified and modified H-β and Na-β molecular sieves: (a) a: H-β; b: H-Zr-β(2); c: H-Zr-β(4); d: H-Zr-β(6); e: H-Zr-β(8). (b) f: Na-β; g: Na-Zr-β(2); h: Na-Zr-β(4); i: Na-Zr-β(6); j: Na-Zr-β(8). XRD: X-ray diffraction.
Relative crystallinity of Na-β and H-β molecular sieves before and after modification by loading with ZrO2.

Relative crystallinity was calculated using JADE 5.0.
As can be seen from Figure 1(a) and (b), the Na-β and H-β molecular sieves, H-Zr-β(n), and Na-Zr-β(n) exhibited characteristic diffraction peaks when 2θ was at 7.6° and 22.4°, which indicates that they retained the crystal form and skeletal structure of the β molecular sieves. With increasing loading, the characteristic peak became wider and the intensity decreased, and the above changes in the ZrO2/H-β series were more obvious than those in the ZrO2/Na-β series. According to the relative crystallinity data in Table 1 (from JADE 5.0), the relative crystallinity of Na-β molecular sieves decreased after ammonia exchange, and the relative crystallinity of molecular sieves loaded with ZrO2 also decreased. In the whole, when Na-β or H-β is loaded with ZrO2, the crystallinity of the molecular sieves is reduced.
Scanning electron microscopy-energy dispersive spectrum
Figure 2(a) and (b) presents the scanning electron microscopy (SEM) images of the Na-β and H-β molecular sieves before and after they were loading with ZrO2, while Table 2 shows the energy dispersive spectrum (EDS) data before and after the H-β and Na-β molecular sieves were loaded with ZrO2. As shown in Figure 2(a) and (b), the grain sizes of the Na-β and H-β molecular sieves were approximately 200 nm. After the ZrO2 had been loaded, there were some accumulated grains on the surfaces of the molecular sieves. With the increased loading, the accumulation became serious and even covered the grains of the molecular sieves, resulting in aggregation. It can be seen from the EDS data in Table 2 that the silicon/aluminum ratio (Si/Al) of the molecular sieves was approximately 12.5, which was consistent with the Si/Al ratio of the commercial molecular sieves that had been purchased. Moreover, on increasing the ZrO2 loading, the silicon zirconium ratio (Si/Zr) of the molecular sieves gradually decreased. The specific accurate data test is subject to inductively coupled plasma mass spectrometry (ICP‒MS).

SEM images of unmodified and modified H-β molecular sieves and Na-β molecular sieves: (a) a: H-β; b: H-Zr-β(2); c: H-Zr-β(4); d: H-Zr-β(6); e: H-Zr-β(8). (b) f: Na-β; g: Na-Zr-β(2); h: Na-Zr-β(4); i: Na-Zr-β(6); j: Na-Zr-β(8). SEM: scanning electron microscopy.
EDS data for the Na-β and H-β molecular sieves before and after modification by loading with ZrO2.

EDS: energy dispersive spectrum.
Acidity analysis of temperature-programmed desorption of ammonia
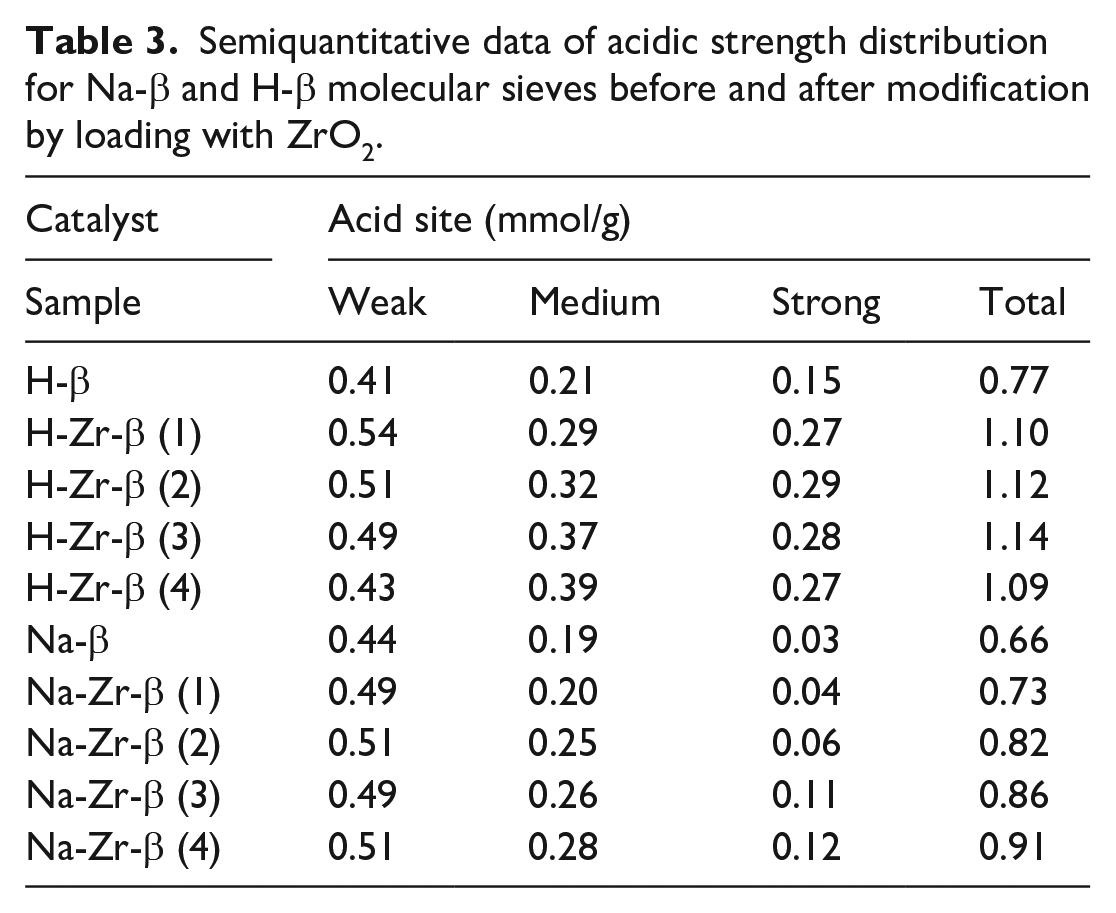
The strength of the acid or alkali and their numbers of active centers have an important influence on the performance of the MPV reaction. Here, the acidities of Na-β, H-β, H-Zr-β(n), and Na-Zr-β(n) were studied by temperature-programmed desorption of ammonia. Figure 3(a) and (b) shows the temperature-programmed desorption of ammonia (NH3-TPD) curves of Na-β, H-β, H-Zr-β(n), and Na-Zr-β(n). The NH3 desorption peaks at approximately 160 °C, 350 °C, and 500 °C can be attributed to weak, medium strong, and strong acid sites, respectively. As shown in Figure 3(a) and (b), there are a small number of strong acid centers and differences in the medium-strong acid strength of the ZrO2/Na-β and ZrO2/H-β molecular sieves and their distribution. Compared to ZrO2/H-β molecular sieves, the ZrO2/Na-β molecular sieves have fewer medium-strong and more weak acid sites. After analyzing the acid strengths and the distributions of the catalysts in Table 3 and combining the catalytic performance of the above catalysts for the MPV reaction of cyclohexanone and isopropanol, we found that the amount of moderately strong acid plays a vital role, providing enough acidity to catalyze the catalytic MPV reaction of cyclohexanone and isopropanol.

NH3-TPD curves for unmodified and modified H-β and Na-β molecular sieves: (a) a: H-β; b: H-Zr-β(2); c: H-Zr-β(4); d: H-Zr-β(6); e: H-Zr-β(8). (b) f: Na-β; g: Na-Zr-β(2); h: Na-Zr-β(4); i: Na-Zr-β(6); j: Na-Zr-β(8). NH3-TPD: temperature-programmed desorption of ammonia.
Semiquantitative data of acidic strength distribution for Na-β and H-β molecular sieves before and after modification by loading with ZrO2.
Specific surface area and pore structure analysis
The pore structure of the β molecular sieves changed before and after modification. Figure 4(a) and (b) shows the N2 adsorption-desorption isotherms before and after the Na-β and H-β molecular sieves had been loaded with ZrO2. Table 4 shows the Brunauer–Emmett–Teller (BET) specific surface areas, total pore volumes, and average pore diameters of the Na-β and H-β molecular sieves before and after they had been loaded with ZrO2. As shown in Figure 4(a) and (b), the adsorption isotherms of the Na-β and H-β molecular sieves before and after ZrO2 loading all belong to type IV isotherms, which indicated that there were both micropores and mesopores present in these catalysts at the same time. Their adsorption capacities were not much different, which showed that they had similar specific surface areas. On the basis of the data in Table 4, it can be seen more clearly that the specific surface area of Na-β treated with zirconium oxychloride decreased, while that of H-β that had received the same treatment did not show an obvious change, and the total pore volumes and average pore diameters of the Na-β and H-β molecular sieves also showed a similar change trend; there were no significant changes in the micropore volume before and after modification. We believe that the reason for this is that the high dispersion of zirconium oxychloride on the inner and outer surfaces played a major role. The skeleton exchange without zirconium ions shows that the impregnation treatment method did not seriously destroy the skeleton of the molecular sieves and did not obviously change the specific surface areas, total pore volumes, and average pore diameters of the β molecular sieves.
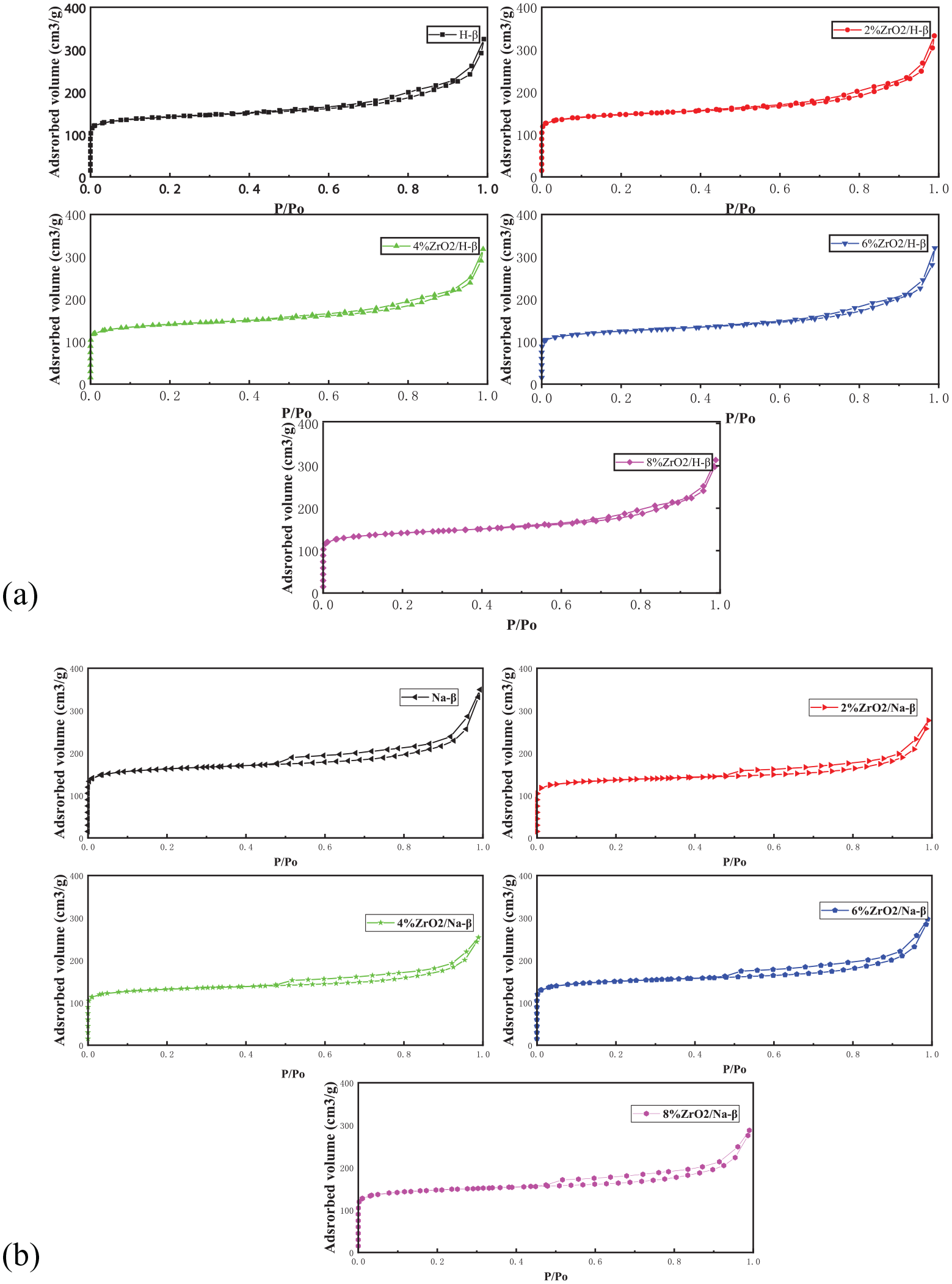
N2 adsorption–desorption isotherm curves of the Na-β and H-β molecular sieves before and after modification by loading with ZrO2.
Data on the BET specific surface areas, total pore volumes, and average diameters of the Na-β and H-β molecular sieves before and after modification.

BET: Brunauer–Emmett–Teller.
Inductively coupled plasma-mass spectrometry
The ratios of Si/Al and Si/Zr according to inductively coupled plasma-mass spectrometry (ICP-MS) analysis can be obtained. Furthermore, the number of acid sites can be estimated, and the acidity of the catalyst can be forecast. Table 5 shows the ICP‒MS data of Na-β, H-β, H-Zr-β(n), and Na-Zr-β(n). As seen from the data in Table 5, the Si/Al ratios of the Na-β molecular sieves and H-β molecular sieves are approximately 12–12.5, which is consistent with the Si/Al ratio of purchased examples. In the process of loading ZrO2, the Si/Zr ratio gradually decreased with increasing ZrO2 loading capacity, and the loading capacity was basically in line with expectations. At the same time, the number of acid sites present in ZrO2/H-β and ZrO2/Na-β also increased with increasing ZrO2 loading, and the activities of the catalysts were similar. Although the Si/Al and Si/Zr results obtained by EDS and ICP-MS are slightly different, the overall trends are the same; it is obvious that there are some errors in EDS analysis.
ICP-MS data for the Na-β and H-β molecular sieves before and after modification by loading with ZrO2.

ICP-MS: inductively coupled plasma-mass spectrometry.
Transmission electron microscope-energy dispersive X-ray spectrum
The transmission electron microscope-energy dispersive X-ray spectrum (TEM-EDX) photographs and spectra of ZrO2/H-β (6) was obtained as shown in Figure 5. From TEM photographs, the crystal morphologies of ZrO2/H-β (6) was similar to those observed by SEM photographs from TEM photographs. The composition information was analyzed according to the energy spectrum microregion of different radial depths of particles (from center to edge). The Zr species in ZrO2/H-β(6) with enriched Zr species was mainly on its surface, not in the center of ZrO2/H-β(6), the Zr content of the surface is much higher than that of the middle. The results show that Zr is enriched on the surface of molecular sieves and skeleton exchange rarely occurs.

The TEM-EDX photographs and spectra of the catalysts ZrO2/H-β (6). TEM-EDX: transmission electron microscope-energy dispersive X-ray spectrum.
Results of catalytic performance
Comparison of catalytic activity
Table 6 shows the effects of the catalysts Na-β, H-β, H-Zr-β(n), Na-Zr-β(n), mixture of H-β and ZrO2, and mixture of Na-β and ZrO2 on the MPV reaction of cyclohexanone with isopropanol. According to the results, the H-β molecular sieves and H-Zr-β(n) catalysts have higher activity than the corresponding Na-β molecular sieves and Na-Zr-β(n) catalysts. As for the H-Zr-β(n) catalysts, the catalytic activity increased from 55.8% to 91.4% on increasing the ZrO2 loading, while for Na-Zr-β(n), their activity gradually increased from 3.5% to 88.5% with an increase of the ZrO2 loading. Under the same loading, the activity of Na-Zr-β(n) was relatively lower. This shows that the double Lewis acid center formed by aluminum and zirconium provided a more favorable active site for the MPV reaction, and the active alkali sites provided by Na-Zr-β(n), which had an acid–base bifunctional active center, did not show a sound acid–base synergy. However, the metal aluminate active sites were covered by alkali active sites, and the catalytic activity increased with an increase of metal zirconium active sites. In addition, we also found that the mechanical mixtures of H-beta or Na-beta molecular sieves and zirconia were used as the catalysts, which were not as active as the corresponding supported catalysts.
Effects of the catalysts Na-β, H-β, H-Zr-β(n), and Na-Zr-β(n) on the MPV reaction of cyclohexanone with isopropanol.

MPV: Meerwein–Ponndorf–Verley.
C, Conversion of cyclohexanone; reaction conditions: n(isopropyl alcohol)/n(cyclohexanone) = 20, T = 80 °C, t = 5 h, m(catalyst) = 6%(m(isopropyl alcohol) + m(cyclohexanone)).
Active life of the catalyst
With the H-Zr-β(6) sample as the catalyst, the MPV reaction of cyclohexanone with isopropanol was repeated after the catalyst had been dried and calcined, and the catalyst was reused five times. The conversion of cyclohexanone is shown in Table 7. We found that the activity of the reused catalyst was higher than that after being dried, which indicates that calcination removed the organic compounds from the surface and in the pores of the catalyst while being dried only volatilized the organic compounds on the surface and did not remove the organic compounds from the pores, which accounts for 15.1% of the mass of the catalyst by thermogravimetry(TG) and results in deactivation of the catalyst. Therefore, the activity of the catalyst is dependent on the active sites on the inner and outer surfaces. From Table 7, we can also see that the conversion of cyclohexanone decreased to 79.4% after the calcined catalyst had been used five times, while when the catalyst was dried and used for the first time, the conversion significantly decreased to 64.6% and then gradually decreased to 13.1% as the number of reuse cycles increased. These results reveal that the catalyst can be regenerated after calcination and that poisoning of the inner and outer catalyst surfaces is the main cause of deactivation. Thus, it can be concluded that the service life of the catalyst needs to be further studied and improved.
Effect of the—service life of the catalysts for the MPV reaction of cyclohexanone and isopropanol.

MPV: Meerwein–Ponndorf–Verley.
C, Conversion of cyclohexanone, reaction conditions: n(isopropyl alcohol)/n(cyclohexanone) = 20, T = 80 °C, t = 5 h, m(catalyst) = 6% (m(isopropyl alcohol) + m(cyclohexanone)).
Analysis of the mechanism
Based on our research, a possible mechanism for the ZrO2/H-β- catalyzed MPV reaction of cyclohexanone and isopropanol to give cyclohexanol has been proposed (Scheme 1). It has been reported that Lewis acidic sites in the catalyst were essential for the MPV reaction. 23 For the ZrO2/H-β catalysts, the Lewis acidic sites mainly originated from Zr4+ (ZrO2) and Al3+(AlO4−). First, isopropanol was adsorbed onto the catalyst and interacted with the acidic sites of ZrO2/H-β, resulting in its dissociation into the corresponding alkoxide. Meanwhile, the substrate cyclohexanone was adsorbed onto adjacent sites, and the carbonyl groups in cyclohexanone were activated by the Zr4+ and Al3+ sites on ZrO2/H-β. Next, hydrogen transfer occurred between the dissociated alcohol and the activated cyclohexanone via a concerted process involving a six-membered ring intermediate. Finally, the above intermediate was converted into cyclohexanol via intramolecular transesterification. Isopropanol was then converted into acetone during this process after losing two hydrogen atoms.

Reaction mechanism for the MPV reduction of cyclohexanone and isopropanol on ZrO2/H-β. MPV: Meerwein–Ponndorf–Verley.
Conclusion
Employing the reflux-impregnation method, the catalysts Na-Zr-β(n) and H-Zr-β(n) were prepared. Because H-Zr-β(n) had both Zr4+ and Al3+ acid centers, it had more advantages because of the distribution of medium-strength acid sites. In the MPV reduction of cyclohexanone with isopropanol to cyclohexanol, Lewis acid centers affect the catalytic efficiency of the reduction, and the synergistic effect of acid and base centers will also affect the catalytic reaction. Here, H-Zr-β(n) showed higher catalytic activity, while Na-Zr-β(n), with the synergistic effect of acid and base did not show the expected activity.
With H-Zr-β(6) as the catalyst, the mole ratio of cyclohexanone to isopropanol was 1:20, and the amount of catalyst was 6% of the total mass of the reactants. The reaction took 5 h at 80 °C. Calculated by area normalization on the bases of cyclohexanone, the conversion of cyclohexanone was 92.0%. Postreactive catalysts can be regenerated by calcination. After the catalyst had been reused 5 times following calcination, the conversion of cyclohexanone decreased to 79.4%. However, the catalyst rapidly lost part of its activity after only being dried and recycled once. This shows that poisoning of the catalyst mainly resulted from the deactivation of the active sites on the inner and outer surfaces caused by the blockage of the pores by the physical adsorption of water, carbon dioxide, and organic compounds. Furthermore, prolonging the cycle life of the catalyst and expanding the scope of the MPV reaction increase the benefits of this process. In the follow-up, we will also report on the microwave-assisted research in this area.
Experiment
Experimental reagents
Cyclohexanone, analytical purity, Tianjin Beichen Founder Reagent Factory; isopropanol, analytical purity, Tianjin Yongda Chemical Reagent Co., Ltd; zirconium oxychloride octahydrate, analytical purity, Sinopharm Group Chemical Reagent Co; H-β molecular sieves (Si/Al = 12.5) and Na-β molecular sieves (Si/Al = 12.5), Catalyst Plant of Nankai University, Tianjin. All reagents were not purified further before use.
Preparation of the catalyst
Preparation of ZrO2/H-β
The loading was carried out using the mass ratios of zirconia to H-β molecular sieves of 2%, 4%, 6% and 8%. The H-β molecular sieves were immersed in zirconium oxychloride aqueous solution at 80 °C for 12 h. The water was removed by rotary evaporation and the obtained white solid powder was dried at 105 °C in an oven for 4 h. Finally, the white powder was subjected to temperature-programmed calcination (heating to 260 °C at a rate of 10 °C/min from room temperature, maintaining the temperature at 260 °C for 2 h, then heating to 550 °C at a rate of 15 °C/min and calcinating it at 550 °C for 6 h). After being ground, the obtained white powders were denoted as H-Zr-β(n), n = 2, 4, 6, 8, which represents the percentage content of ZrO2.
Preparation of ZrO2/Na-β
H-β was replaced with Na-β, and according to the above steps, the resulting samples were recorded as Na-Zr-β(n), n = 2, 4, 6, 8, where n represents the percentage content of ZrO2.
Characterizations of molecular sieve catalysts
XRD patterns of the catalysts were obtained using a D8 ADVANCE X-ray diffractometer (Bruker, Germany) with Cu-Kα radiation at 40 kV and 40 mA, 2θ scanning range 4°–52°, scanning speed 2°/min (2θ).
The acidities of the samples were recorded by temperature-programmed desorption of ammonia (NH3-TPD) on an AutoChem II 2920 automatic chemical adsorption meter (Micromeritics instruments co., LTD, America). Samples of 50–100 mg were weighed, placed into a U-shaped quartz tube, and heated from room temperature to 300 °C for drying pretreatment at 10 °C/min according to the temperature programming. The mixture was purged with a He gas flow (30 mL/min) for 1 h, cooled to 50 °C, and injected with a 10% NH3/He mixture (30–50 mL/min) for 1 h until it was saturated. Next, the system was switched to a He gas flow (30 mL/min) for purging for 1 h to remove the weak physical adsorption of NH3 on the surface. Finally, the temperature was raised to 800 °C at 10 °C/min for desorption, and degassing was detected by thermal conductivity detector (TCD).
BET specific surface areas, pore volumes, and pore size measurements were measured using a Micromeritics Instrument Co., Ltd. Gemini VII 2390 fully automatic specific surface area analyzer. The carrier gas was high-purity He (99.99%), and high-purity N2 (99.99%) acted as the adsorbent. For single-point and multipoint analysis of the BET specific surface area, the average time of adsorption and desorption at each P/P0 point of each sample was 5 min. The attachment rate was 200 mmHg/min, and the equilibration time of the equilibrium method was 5 s. The BET specific surface areas, total pore volumes, and average pore diameters were calculated by the Gemini VII software for Windows.
The morphology of the catalyst was measured using a JSM-7610F Plus scanning electron microscope (JEOL, Japan). The working distance was 8 mm, the objective diaphragm was 100 μm, the sample current was 5 nA, the acceleration voltage was 15 kV, the magnification was 20,000 times, and the counting rate was 2 cps. The spectral element analysis range was Be4~U92. Furthermore, micro-observation and analysis of TEM-EDX was made on the JEM- F200 (JEOL, Japan). The probe current was 7.47500 nA, the acceleration voltage was 200 kV, the magnification was 600,000 times, and the counting rate was 208 cps. The spectral element analysis range was B5~U92.
The determination of Al and Si in the catalysts was performed using an Agilent 7800 full spectrum direct reading spectrometer (ICP‒MS) from Agilent, USA. Sample preparation: The prepared zirconium-modified molecular sieve catalysts were dissolved in HF and diluted to the detectable concentration range. High frequency power 1300 W, auxiliary air flow 0.7 L/min, pump speed: 29 r/min, integration time: 15 s, and carrier gas pressure: 221. 05×103 Pa, injection Volume: 1.85 mL/min. Maximum absorption wavelength: Al: 396.152 nm; Si: 251.612 nm.
Catalytic MPV reduction of cyclohexanone with isopropanol
Typical MPV reaction steps are as follows: the catalyst (0.79 g) and isopropyl alcohol (12.12 g, 0.201 mol) were added into a 100-mL three-neck round-bottom flask filled with a thermometer, a constant pressure funnel and a spherical reflux condenser, the reactants were heated and stirred during heating to 80 °C. Cyclohexanone (0.98 g, 0.01 mol) was slowly added via the constant pressure funnel into the three round-bottom flasks, and the reaction mixture was stirred for 4 h at 80 °C. Subsequently, the product was cooled to room temperature and filtered under vacuum. Then, the filtrate was retained in the sampling bottle for gas chromatography analysis, and the filter cake was dried at 80 °C for 12 h for recycling.
Product analysis
The product was identified by GC/MS-QP2010 (Shimadzu, Japan), and qualitative analysis of the product was performed on a GC2014 instrument (Shimadzu, Japan). The peak area normalization method was used to calculate the content of each component in the mixture. The formula was as follows.

Footnotes
Acknowledgements
The authors thank Science Compass Labs for testing services and the Elsevier Agency for help with language touch-ups.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
