Abstract
Development of novel antimicrobial phytochemical-bearing nanofibrous mats could be considered as a promising strategy to overcome against antibiotic resistance in wound healing. In this work, the electrospinning process was used to successfully create novel antimicrobial nanofiber mats made of a blend of electrospun chitosan/polycaprolactone (CS/PCL) loaded with M. communis leaf extract (MCLE) (15 and 30 wt.%). Field emission scanning electron microscopy (FESEM), X-ray diffraction (XRD) analysis, water contact angle (WCA) testing, attenuated total reflectance Fourier transform infrared (ATR-FTIR) spectroscopy, and mechanical properties testing were applied to evaluate physicochemical properties of the nanofiber mats. The FESEM images showed uniform, bead-free, and smooth nanofiber mats with good compatibility between MCLE and polymers. Image J software was used to calculate the average diameters of nanofibrous mats, and the average diameter increased significantly as the extract concentration increased. The existence of MCLE in the nanofibrous mats was verified by ATR-FTIR spectroscopy and XRD analysis. The tensile strength of the nanofiber mats was satisfactory (6.31–12.47 MPa). The incorporation of MCLE in CS/PCL nanofibers enhanced the scaffold’s hydrophilicity, as evidenced by a reduction in contact angle. Significant reduction up to 0.5 log of both Escherichia (E.) coli and Staphylococcus aureus count was observed upon exposure to CS/PCL nanofibers. The MCLE (15 and 30 wt.%)-incorporated CS/PCL nanofibers demonstrated a significant reduction of bacterial count up to 0.8 log for both bacteria. The results demonstrated that manufactured nanofibers could be considered as a promising dressing in wound dressing.
Introduction
Modern healthcare services are facing serious challenges since numerous antimicrobial agents have lost their therapeutic potential for infectious diseases, mainly because of the development of microbial resistance. 1 Due to the complexity of multidrug resistance, scientists have been obliged to look for novel antimicrobial agents from a variety of sources; for instance, medicinal plants. 2 People primarily resort to plant extracts for medical purposes due to their interesting effects in wound healing process including ant-inflammation, antibacterial, and antidiabetic activities. 3
The native Mediterranean shrub Myrtus communis L. (Myrtaceae) is an evergreen sclerophyll shrub. In traditional folk medicine, M. communis is applied as a hypoglycemic agent, disinfectant, and antiseptic agent. 4 It is also used as flavour in the food and cosmetic industry. 5 M. communis is rich in polyphenolic compounds, like tannins and flavonoids. Further, researches on phytochemical properties of M. communis have revealed that this plant contains essential oils. 6 Various biological activities like antibacterial, 7 anti-inflammatory, 8 and antioxidant 9 activities of have also been reported for M. communis making this valuable plant extract for application in wound healing.
Having used the electrospinning technique for encapsulation of plant material, scientists can potentially intensify their therapeutic effect. The process of encapsulation accelerates the remedial impact significantly by increasing the bioavailability of drugs and maintaining a constant concentration of medicine to the target area. 10 Melts, dispersions, and solution can all be used for electrospinning. 11 Fiber-forming technology based on electrospinning is commonly employed for phytochemical immobilization and preparation of wound dressings.12,13 Electrospinning is a versatile and efficient method for creating nanofibers. 12 It is a unique experimental approach for easily preparation of nanofibers for application in diverse field of medicine. 14 It is a method of producing nano/microfibers from viscoelastic polymers/copolymers solutions using high electrostatic force spinning. 15 Electrospun nanofibers have great characteristics such as high surface area, uniformity, porosity, unique chemical and physical characteristics. 16 Electrospun nanofibers have wide-ranging applications in wound dressing,17–21 catalyst,22–24 drug delivery,25,26 biosensor,27,28 tissue engineering,29–31 energy harvesting,32,33 air filtration,34,35 and etc. To date, scientists have produced electrospun fibers either from natural/synthetic polymers or their combinations. 36
Chitosan (CS) is a polysaccharide found in nature comprising a number of amino groups made by deacetylation of chitin polysaccharide. 37 CS is found in a broad variety of natural sources, including insects, fungi, and crustaceans. 38 It has gotten a lot of attention because of its remarkable biodegradability, antibacterial, nontoxicity, biocompatibility, and wound healing properties. 39 CS-based electrospun nanofibers has attracted great attention in antibacterial applications. The electrospinning of pure CS seems to be a serious issue since it provides a repulsive force among its ionic groups. 36 The researches have mostly addressed the electrospinning of CS with encapsulation of natural bioactive compounds including extract, 40 essential oils, 41 and phenolic agents. 42 Combining chitosan with the synthetic, biocompatible polymer, polycaprolactone (PCL), usually yields a copolymer combination with cell affinity, greater strength, stability and spinnability. 43 PCL is a synthetic aliphatic polyester that is biodegradable, nontoxic, biocompatible and hydrophobic. 44
In research done by Ge et al., Sabina chinensis leaf extract-loaded polycaprolactone/polyvinylpyrrolidone microfibers were fabricated via electrospinning technique. The antibacterial activity of the extract-loaded microfibers was outstanding against gram-positive and Gram-negative bacteria, with the inhibitory impact being stronger against gram-positive than Gram-negative bacteria. 45 In another study, Sadri et al. used electrospinning process to fabricate three different polymeric nanofiber composites comprising CS, CS/polyethylene oxide and CS/polyethylene oxide/green tea extract for preparation of wound dressing. According to their findings, acceptable antibacterial activities was observed for CS/polyethylene oxide/green tea nanofibers against gram-negative and gram-positive bacteria. Animal tests revealed that using CS/polyethylene oxide/green tea as a wound dressing rather than the other groups resulted in more efficient and quick wound recovery. 46
To our knowledge, no study has yet been published on the CS-based nanofibrous mats loaded with M. communis extract with the aim of investigating their antibacterial activity. The goal of the current work was to manufacture CS-based nanofibrous mats loaded with M. communis leaf extract (MCLE) and to evaluate their application as an antibacterial agent against pathogenic bacteria. We accordingly used a mixture of CS and PCL with or without M. communis leaf extract to prepare MCLE-free and MCLE–loaded nanofibrous mats following electrospinning method. The schematic representation of nanofibrous mats fabrication is showed in Figure 1. Nanofibers were characterized using field emission scanning electron microscopy (FE-SEM), attenuated total reflection fourier transformed infrared spectroscopy (ATR-FTIR), and X-ray diffraction (XRD). The water contact angle (WCA) and mechanical characteristics of the nanofibers were also evaluated. The antibacterial effects of the mats were investigated using two model microorganisms representing Gram-positive and Gram-negative bacteria. Schematic representation of the nanofibrous mats fabrication.
Experimental
Materials
Myrtus communis L. (Myrtaceae) leaves were bought at the market and identified by a botanist; then it was registered at Tehran University of Medical Sciences in the School of Pharmacy’s Herbarium (PMP-458). Polycaprolactone (PCL; with average Mn of 80000) and Chitosan (CS; degree of deacetylation 75–85%; medium molecular weight) were supplied from Sigma-Aldrich Co. (USA). Glacial acetic acid and formic acid were purchased from Duksan reagents and Merck, respectively. Staphylococcus aureus (ATCC 25923) and Escherichia coli (ATCC 8739) were preserved at −70°C at the Department of Drug and Food Control, Faculty of Pharmacy, Tehran University of Medical Sciences. Brain Heart Infusion (BHI) agar was purchased from (CONDA, Spain).
Preparation of Myrtus communis leaf extract
The maceration technique was used to make the extract from Myrtus communis L. The leaves of M. communis were ground by the mill and moved to the percolator. Subsequently, 90% ethanol was added as an extraction solvent. So, they were completely immersed in 90% ethanol. This was repeated in four periods of time with a total duration of about two weeks. At the end of each step, the resulting extracts were filtered, the filtrate was vaporized in a rotary evaporator, and the recycled solvents were poured back into the percolator. The extract was dried in room temperature and then it was freeze-dried to eliminate the solvent completely. The extract was stored in refrigerator.
Preparation of chitosan/polycaprolactone/Myrtus communis leaf extract solution
Solutions of CS and PCL were produced individually at concentrations of 1 and 10 wt.% in the mixture of solvents containing acetic acid/formic acid (at 1:1 volume ratio), respectively. CS and PCL solutions were subsequently blended in a ratio of 1:3 and mixed for 3 h. Finally, to prepare solutions containing M. communis leaf extract, MCLE (15, 30 wt.%) was added to the solutions of CS/PCL based on the weight of the polymers. These solutions were stirred overnight to obtain a homogeneous solution.
Electrospinning technique
In the electrospinning process, each prepared solution was poured into a syringe with an 18G blunt-ended needle. The filled syringe was put into the electrospinning apparatus (Side by Side Electroris®, lab-scale Dual Pump Electrospinning Machine) with high voltage power supply (Model: HV35P OV) made by Fanavaran Nano-meghyas Co. (Iran). The solution flow rate was set to 0.3 mL/h and a high voltage of 15 kV was used to the pumped solution. The distance between the collector and needle tip was 10 cm. The nanofibrous mats were collected on an aluminium foil-coated collector.
Characterizations
Morphological analysis
The morphologies of the nanofiber mats were carried out by Field Emission Scanning Electron microscopy (FESEM S4160, Hitachi, Japan). Image J software was applied to measure the average diameters of the nanofibrous mats.
Attenuated total reflectance Fourier transform infrared analysis
Attenuated total reflectance Fourier transform infrared (ATR-FTIR) spectroscopy (Bruker- Equinox 55, USA) was used to evaluate the chemical characteristics of the nanofibers. The analysis was performed in the wavenumber range of 600–4000 cm−1 with a spectral resolution of 1 cm−1 and 16 scans.
Water contact angle
The hydrophilic nature of the mats was determined by the WCA. A contact angle instrument (Veho VMS-004 Discovery Deluxe) was used to determine the WCA. The samples were attached on a glass slide. Then on the nanofibers, a drop of distilled water (15 μL) was placed. The WCA was taken 10 s after the droplet was placed and was evaluated using Image J software. Three measurements were carried out at various positions on the surface and the mean value was achieved.
X-ray diffraction analysis
X-ray Diffraction (XRD) analysis was done by a Bruker D8 Advance X-ray diffractometer (Germany) with Cu-Kα radiation (λ = 1.5418 Å) and operated at a current of 37 mA and a voltage of 28 kV. Data were collected with a scan speed of 1 sec/step and the range of 2θ = 5–80°.
Mechanical properties measurements
A tensile testing device (Model no. 5566, Instron Co., USA) with a 50 N load cell was applied to measure the mechanical characteristics of the nanofibrous mats at a speed of 5 mm/min. Before the mechanical tests, an appropriate paper square frame was used. The nanofibers were cut into a rectangular form with a width of 5 mm and a length of 30 mm. The square-shaped paper frame had an internal length of 2 cm. Both ends of the nanofibers specimen (5 mm by 30 mm) were attached to the top and bottom of the paper frame by an adhesive tape. The average of the three measurements was calculated and reported.
Antibacterial susceptibility test
Antibacterial activity of electrospun CS/PCL mats was evaluated in the presence or absence of M. communis leaf extract based on Clinical and Laboratory Standards Institute M100S (CLSI). 47 The pathogens used in this test were Staphylococcus (S.) aureus and Escherichia (E.) coli as Gram-positive and Gram-negative model bacteria, respectively. The nanofiber mats were cut into 2.5 × 2.5 cm2, sterilized under UV light for 2 h and were exposed to standardized bacterial suspensions (OD600 equals to 108 CFU/mL, diluted by PBS 1:100, pH 6). Bacterial suspension diluted in PBS (106 CFU/mL) without nanofiber mats at 1- and 2 h were considered as control samples. All tubes containing nanofibrous mats were put in an incubator shaker at 37°C for 1 and 2 h. At each of these times, serial dilutions were performed in 96-well flat-bottom Microplates. All experiments were repeated in triplicate. A sample of 10 μL of each dilution was cultured on BHI agar medium as spread culture. Subsequently, the plates were incubated for 24 h at 37°C and then the bacterial count was estimated using the Misra and Miles method.
The Data were reported as mean ± standard deviation (SD) of at least three independent experiments. Statistical analysis was performed by one-way analysis of variance (ANOVA) and Tukey’s post hoc test using GraphPad Prism v9.0.2.161. The p values below 0.05 (p < 0.05) was considered as statistically significant difference. First, all the test groups were compared one by one with the control groups and then the groups were compared in pairs. The asterisks on bar charts indicate the significant difference between each group and the control group (p < 0.05).
Results and discussion
Morphology and diameter distribution of the nanofibrous mats
The CS/PCL nanofibers without and with variable amounts of MCLE were characterized by FESEM in order to obtain precise examination of the morphology and size of the nanofibers. The FESEM images and diameter distribution of the mats are shown in Figure 2, indicating that the morphology of the nanofibrous mats was uniform, smooth, and bead-free and no MCLE particles were present on the surfaces of the nanofibers. These results demonstrated that the MCLE was well loaded into the nanofibers. ImageJ software determined average diameters of nanofibers from FESEM images as 64.9 ± 18.1, 77.7 ± 20.2, and 92.1 ± 34.3 nm for CS/PCL, CS/PCL/MCLE (15 wt.%), and CS/PCL/MCLE (30 wt.%), respectively. One-way ANOVA was used to compare and to evaluate the statistically significant difference between the groups. p < 0.05 was considered to be significant. The impact of MCLE content on the mean diameter of the nanofibers analysed by One-way ANOVA showed that the difference in mean diameter of nanofibers was statistically significant (p < 0.05). This increase could be due to hydrogen bonding between the polymeric matrix and MCLE active ingredients, increasing shear viscosities and thus impeding effective drawing operations during nanofibers production.
48
In this respect, Samprasit et al.,
49
prepared thiolated CS/polyvinyl alcohol nanofibers comprising various amounts of α-mangostin and found that by increasing the α-mangostin content, the nanofiber diameter increased. Nanofibrous mats morphology and diameter distribution of the M. communis leaf extract (wt.% MCLE)-loaded CS/PCL nanofiber mats with different amount of MCLE; (A1, A2) 0 wt.% MCLE, (B1, B2) 15 wt.% MCLE and (C1, C2) 30 wt.% MCLE. Scale bars represent 1 μm.
Attenuated total reflectance Fourier transform infrared analysis
ATR–FTIR spectra of CS/PCL, CS/PCL/MCLE (15 wt.%), CS/PCL/MCLE (30 wt.%) and MCLE are shown in Figure 3. The figure represents the comparison between the ATR–FTIR spectra of the CS/PCL/MCLE nanofibers and MCLE. MCLE related ATR–FTIR spectra showed a peak at 1038 cm−1 (O–H deformation), 1244 cm−1 (C–O asymmetric stretching in cyclic polyphenolic compounds), 1360 cm−1 (C–O stretching of the ester group), 1446 cm−1 (O–H bending vibrations), 1610 cm−1 (C = C stretching vibrations), 1724 cm−1 (C = O stretching vibration), 2931 cm−1 (C–H and CH2 vibration of aliphatic hydrocarbons) and a broad peak at around 3298 cm−1 (O–H stretching vibrations).
50
The presence of PCL was confirmed by the peaks appeared at 2866 cm−1 associated with symmetric CH2 stretching, the peaks at 2943 cm−1, 2943 cm−1, and 2941 cm−1 related to asymmetric CH2 stretching, and the peaks at 1724 cm−1, 1724 cm−1, and 1722 cm−1 associated with C = O stretching in the spectra of the CS/PCL, CS/PCL/MCLE (15 wt.%), and CS/PCL/MCLE (30 wt.%) nanofibers, respectively (Figure 3).
51
The shifts in the wavenumbers suggest a possibility of hydrogen bonds formed between the CS/PCL nanofibers and MCLE. Moreover, the peak at 1466 cm−1 related to CH2 bending of polysaccharide and the band in the range of 3200–3600 cm−1 associated with O-H and N-H stretching of the polymer could verify the presence of CS in the corresponding spectra.
36
After incorporation of MCLE, a characteristic peak at 1610 cm−1 was observed related to C = C stretching vibrations indicating successful incorporation of the MCLE into the nanofibers. The relative intensity of the peak slightly increased with increasing in the amount of MCLE. ATR–FTIR spectra of (a) free MCLE (b) pristine CS/PCL and MCLE-incorporated blend nanofibers bearing (c) 15, and (d) 30 wt.% of MCLE.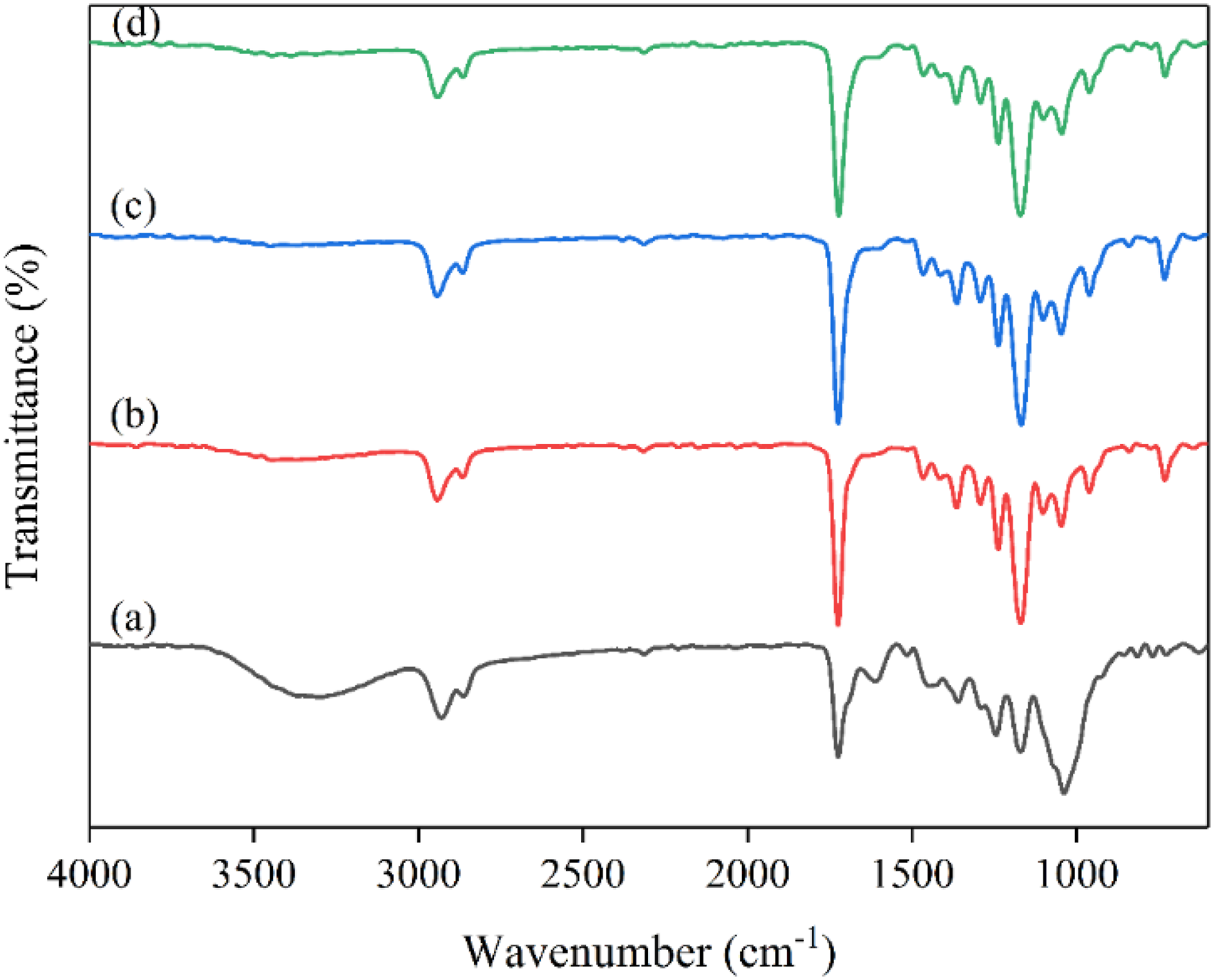
Water contact angle
An appropriate dressing should be able to absorb the exudates of the wound and retain moisture at the wound site. This feature could be characterized by the surface wettability and hydrophilicity of the dressing. WCA was used to show the nanofibers’ hydrophilicity. The results are presented in Figure 4. The average WCA for the CS/PCL, CS/PCL/MCLE (15 wt.%), and CS/PCL/MCLE (30 wt.%) nanofibers were 79.33 ± 5.77°, 54.67 ± 5.03°, and 49.67 ± 1.15°, respectively. Addition of MCLE to the CS/PCL nanofibers reduced the WCA. The effect of MCLE content on the WCAs analysed by One-way ANOVA showed that the difference in the WCAs was statistically significant (p < 0.05). The incorporation of MCLE resulted in a decrease in WCA, indicating an increase in hydrophilicity. This could be related to the existence of the compounds in MCLE like polyphenolic compounds, which can interact via hydrogen bonding with water molecules. Arbade et al., fabricated poly(ε-caprolactone) nanofibers incorporated with different amounts of Emblica officinalis (EO) extract. According to their results, the WCA of the nanofibers reduced as the amount of EO increased.
52
(a) Images of water droplets on electrospun nanofibers containg different contents of MCLE (a) pristine CS/PCL (0 wt.% MCLE), (b) 15 wt.% MCLE and (c) 30 wt.% MCLE content, respectively; (b) its corresponding contact angle.
X-ray diffraction analysis
The XRD patterns of free MCLE, naked CS/PCL, CS/PCL/MCLE (15 wt.%), and CS/PCL/MCLE (30 wt.%) nanofibers are shown in Figure 5. Diffraction pattern of MCLE represents a broad peak at 2θ = 20.41° indicating the amorphous nature of the extract. The CS/PCL nanofibers showed two characteristic peaks at 2θ = 21.31° and 24.11°, corresponding to the characteristic peaks of PCL.
45
The CS peaks are found to overlap with the PCL peaks observed in this result. These two peaks shifted slightly to high angles when MCLE was loaded onto the CS/PCL nanofibers (the characteristic peaks were at 2θ = 21.82° and 24.20° for CS/PCL/MCLE (15 wt.%) nanofibers and were at 2θ = 21.44° and 24.85° for CS/PCL/MCLE (30 wt.%) nanofibers). In addition, by increasing the content of MCLE to 30 wt.%, the intensity of the characteristic peaks increased. The full width at half maximum (FWHM) of the angle increased with increasing the content of MCLE. The values of FWHM were 0.49°, 0.78°, 1.18° for the CS/PCL, CS/PCL/MCLE (15 wt.%) and CS/PCL/MCLE (30 wt.%) nanofibers, respectively. These results imply that the MCLE was successfully loaded in CS/PCL nanofiber mats. XRD patterns of (a) MCLE (b) pristine CS/PCL and MCLE-incorporated blend nanofibers having (c) 15, and (d) 30 wt.% MCLE content.
Mechanical properties
In the interaction with the wound, the mechanical properties of the wound dressing are critical. Having an appropriate mechanical strength of nanofibers is basic criteria for durability and handling in wound dressing. Figure 6 depicts the stress-strain curves of the nanofibers. Tensile stress, Young’s modulus and tensile strain for the nanofibers are shown in Table 1. The tensile strength of the nanofibrous mats reduced by increasing the MCLE content. All the samples had ultimate tensile strengths (UTS), ranging from 6.31 to 12.47 MPa, with CS/PCL nanofibers having the highest tensile strength. All of the samples had a Young’s modulus of 28.06–53.71 MPa. As the MCLE content increased, the Young’s modulus of the nanofibers decreased in a dose-dependent manner. The influence of MCLE content on tensile stress, Young’s modulus, and tensile strain was investigated, and one-way ANOVA revealed a statistically significant difference between tensile stress values (p < 0.05). Also, the difference between Young’s modulus values was significant as p < 0.05. However, no statistically significant difference was seen between the tensile strains. Nanofibrous dressings with tensile strength of 0.8–18.0 are adequate and sufficiently durable for wound dressing and dermal cell culture.
53
Our results revealed that the nanofibers loaded with MCLE have the potential candidate as a wound dressing material. Shokrollahi et al.,
54
fabricated multilayer nanofibrous patch containing chamomile loaded carboxyethyl CS/poly(vinyl alcohol) and PCL and found that the strength of the nanofibers reduced by increasing the amount of chamomile. Tensile stress–strain curves of samples: CS/PCL, CS/PCL with 15 wt.% MCLE, and CS/PCL with 30 wt.% MCLE nanofiber mats. Mechanical properties of the MCLE (0, 15, and 30 wt.% MCLE)-loaded CS/PCL nanofibers.

Antibacterial susceptibility test
Antibacterial activity is considered as one of the critical elements for a wound dressing formulation. Antibacterial wound dressings are potential candidates to lower infection and therefore expediting the wound healing. The prepared nanofibers were tested for antibacterial activities against S. aureus and E. coli using CLSI standards.
47
As shown in Figure 7, the CS/PCL nanofiber reduced the growth of S. aureus (about 0.3 log/53%) and (about 0.5 log/66%) significantly compared to the control at both 1- and 2-hour incubation times, respectively (p < 0.05). Significant growth reductions were observed for E. coli with (about 0.2 log/37%) and (about 0.3 log/45%) at the same time points, respectively (p < 0.05). Colony count of (a) S. aureus and (b) E. coli (CFU/mL) in the presence of (A) pristine CS/PCL, (B) CS/PCL/MCLE 15 wt.% and (C) CS/PCL/MCLE 30 wt.% after 1 and 2 h treatment. The asterisks indicate statistically significant difference between the test groups and the control group (fiber-free bacterial suspension) (*p < 0.05, **p < 0.01, ***p < 0.001).
The antibacterial activity of CS can be attributed to two primary mechanisms. Firstly, positively charged CS can interact with negatively charged functional groups on the cell surface, changing its permeability. This inhibits vital substances from entering the cells and/or removing the essential solutes from the cell. The second mechanism is related to the attachment of CS to cellular DNA which would inhibit microbial RNA synthesis. Antimicrobial activity of CS could also be the consequence of a mixture of both mechanisms. 42
The 15 wt.% MCLE-loaded CS/PCL nanofiber reduced the growth of S. aureus (about 0.6 log/73%) and (about 0.7 log/81%) significantly compared to the control at both 1- and 2 h incubation times, respectively (p < 0.05). Growth reductions at the same intervals were also significant with (about 0.3 log/47%) and (about 0.3 log/50%) for the E. coli, respectively (p < 0.05). The growth of S. aureus was reduced (about 0.7 log/78%) and (about 0.8 log/84%) upon 1- and 2 h exposure to 30 wt.% MCLE loaded CS/PCL mats significantly in comparison with the control, respectively (p < 0.05). Furthermore, significant growth reductions were also observed for E. coli at the same time points mentioned above with (about 0.3 log/46%) and (about 0.3 log/50%), respectively (p < 0.05). However, there was no statistically significant different between 1- and 2-h incubation with the nanofibers in terms of bacterial growth (p > 0.05). In the nanofiber-treated S. aureus groups, an increase in antimicrobial effect was observed by increasing the concentration of the MCLE which could be attributed to the synergistic antimicrobial effect of CS and MCLE extract (p < 0.05). Unlike S. aureus, there was no significant difference between the samples incubated with E.coli. This could be related to the unavailability of MCLE at the appropriate antimicrobial concentration due to differences in the structures of these two bacteria (p > 0.05). Essential oils and extracts of Myrtus communis L. have been reported to exert antibacterial properties by disrupting the permeability of bacterial cell wall and membrane, releasing cellular contents outside the cell. This phenomenon interferes with essential membrane functions such as electron transfer, nutrient absorption, and enzyme activity. 55 Gram-negative bacteria are frequently reported to be more resistant to plant-based extracts and essential oils, since Gram-negative bacteria have a hydrophilic cell wall structure that is mostly made up of a lipo-polysaccharide (LPS) that prevents hydrophobic oil from penetrating and essential oils from accumulating in the target cell membrane. 56 Because of this, Gram-positive bacteria have been found to be more sensitive to M. communis extracts than gram-negative bacteria. Many studies have reported antibacterial activity of MCLE and essential oils against pathogenic bacteria. In a study performed by Taheri et al., 57 the hydroalcoholic leaf extract of M. communis demonstrated potent inhibitory effects on S. aureus (MIC: 0.2 mg/mL) and Vibrio cholera (MIC: 2 mg/mL), with weak antibacterial effect against E. coli (MIC: 8 mg/mL) and no effect on Pseudomonas aeruginosa. Thus, these findings confirmed that the fabricated dressings could be utilized as a potential antibacterial agent for wound dressing.
Conclusions
In this study, novel antimicrobial nanofibers of CS/PCL loaded with different concentrations of MCLE (15 and 30 wt.%) were successfully fabricated via electrospinning. Uniform, bead-free, smooth nanofibers were obtained, which showed excellent compatibility between polymers and MCLE. By increasing the amount of extract, average diameter of the nanofibrous mats increased considerably. ATR–FTIR spectroscopy and XRD analysis confirmed the presence of MCLE in the nanofibrous mats. The hydrophilicity of the nanofibers increased by adding MCLE. The tensile strength of the nanofibers was in the range required for wound dressing applications. Incorporation of MCLE at certain concentrations into the nanofibrous mats enhanced antibacterial properties against S. aureus, significantly. The current research showed a promising potential of the MCLE-incorporated CS/PCL nanofibers in wound dressing applications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from Iran National Science Foundation (INSF, Grant number: 4003236).
