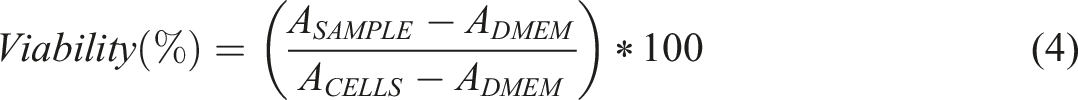Abstract
Nanofibrous scaffolds offer significant promise for wound healing due to their ability to absorb exudates, prevent microbial contamination, and enhance oxygen diffusion. However, challenges remain in fully realizing their clinical potential, as previous research has primarily focused on scaffolds made of two polymers or those encapsulating therapeutic agents within nanofibers. Additionally, scaling up fabrication while maintaining functionality presents a significant challenge. This study introduces a novel type of nanofibrous scaffold, combining poly (ethylene oxide) (PEO), poly (caprolactone) (PCL), and chitosan (CS) in various mass ratios, electrospun using Nanospider™ technology. The scaffolds featured fiber diameters ranging from 134 ± 37 to 148 ± 38 nm and exhibit high gram-per-square-meter values between 6.8 and 8.6 g/m2. An optimal balance of hydrophilicity was achieved, and the scaffolds demonstrated superior breathability with moisture vapor transmission rates ranging from 1904.3 ± 28.6 to 2005.7 ± 42.9 g/m2/day, outperforming commercial wound dressings. Additionally, a wide range of hydrolytic degradation rates (3.8 ± 1% to 73.2 ± 0.8%), elongation at fracture (21% to 0.8%), and Young’s modulus (106.7 ± 8.5 MPa to 170.7 ± 11.9 MPa) were observed. Surface-immobilized tetracycline (TET) significantly enhanced antibacterial efficacy, with inhibition zones exceeding 20 mm against Escherichia coli. Our findings confirm that scaffold properties can be effectively tailored by adjusting the PEO/PCL ratio, advancing customization for wound care. Post-fabrication soaking in TET solutions further boosts antibacterial performance and allows for tailored post-production adjustments. Compared to existing studies, this approach simplifies customization and improves the practicality of wound care solutions.
Keywords
Introduction
Wound healing is a complex and dynamic process crucial for restoring the skin’s barrier function and ensuring complete coverage of injuries. Factors such as infections, chronic conditions like diabetes, and the nature of the wound itself can significantly complicate this process, all of which require advanced therapeutic interventions. The management of extensive wounds, in particular, presents a medical challenge, requiring effective strategies to restore barrier function and achieve complete skin coverage. 1 Depending on the nature of the wound, a range of dressings may be considered, with conventional options such as gauze, bandages, and cotton wool being modified to provide protection against microbial contaminants. However, these traditional options often have limitations, such as adherence to the wound site, causing pain and further injury upon removal, and are generally suitable only for uninfected wounds with moderate exudate levels. 2 On the other hand, advanced wound dressings like hydrogels offer distinct advantages over traditional gauze. They are absorbent, non-stick, and capable of creating a moist microenvironment that promotes wound healing by keeping the wound bed hydrated, facilitating cell migration, and supporting tissue regeneration. Hydrogel dressings are designed to prevent wounds from drying out and to aid in the healing process.3,4 Despite these benefits, traditional hydrogel dressings in the form of films often lack sufficient breathability, leading to issues like wound infections and poor adaptability to wounds of varying shapes.5,6 Recent advancements in skin regeneration have led to the development of functional scaffolds that act as physical barriers against microbial infections, are non-stick, breathable, and maintain structural integrity while creating and sustaining a moist healing environment.7,8 Various fabrication techniques such as phase separation, freeze-drying, self-assembly, and 3D bioprinting are employed to create functional scaffolds, each of which requires specific parameters to achieve the desired characteristics and topographies.9–11 Among these fabrication techniques, electrospinning stands out for producing nanofibrous scaffolds with nanoscale fiber diameters. This method offers several advantages, including a high surface area-to-volume ratio, significant porosity, and the ability to incorporate various therapeutic agents, making it particularly well suited for wound healing applications.12–14
The electrospinning process involves applying an electric force to draw charged polymer fluids or melts into nanofibers. Different types of electrospinning processes are categorized based on the spinneret used, such as the traditional mono-tube needle, a tri-layer needle, an eccentric needle, or a concentric needle. 15 Needle electrospinning techniques offer precise control over fiber formation; however, they have significant drawbacks, such as the potential for needle clogging, cleaning challenges, and interference between jets, leading to operational complexities and frequent maintenance. Additionally, the layout of multiple needles in these setups can be intricate, requiring large operating spaces and making the process more cumbersome.16,17 In contrast, needle-less electrospinning techniques, such as free-liquid electrospinning, have been developed to address some of these limitations. Needle-less electrospinning systems can offer higher production rates and circumvent issues like needle clogging and the low manufacturing capability inherent in traditional needle-based processes.18,19 By eliminating the need for needles, these techniques can enhance the uniformity of fiber diameter and scaffold formation, which is often challenging in needle-based electrospinning due to the localized charge concentration at the needle tip. 20 The production of nanofibers via needle-less, high-voltage, free-liquid surface electrospinning processes, such as Nanospider™ technology, holds considerable promise for industrial applications owing to its distinctive attributes. This technology facilitates the fabrication of high-quality nanofibers characterized by minimal solvent consumption, continuous manufacturing capabilities, and compatibility with a diverse array of polymers. 21 Renowned for its adaptability, Nanospider™ technology offers ease of adjustment to optimize specific properties through the manipulation of various process parameters. 22
Nanofibrous scaffolds with interconnected porous networks not only support the overall wound healing process but also enhance tissue regeneration by providing structural support, promoting cell adhesion, and facilitating the exchange of vital molecules involved in the healing cascade.23–25 Studies have demonstrated that nanofibrous scaffolds can improve wound closure, reduce scar formation, and enhance overall wound healing outcomes. 26 By leveraging the unique properties of nanofibers, such as their biocompatibility and high drug absorption capabilities, these scaffolds hold immense potential for advancing wound care practices and addressing the challenges of chronic wounds.27–29 The selection of appropriate biomaterials is critical to the effectiveness of nanofibrous scaffolds in wound healing. 30
Chitosan (CS), a natural biopolymer, has garnered considerable interest due to its distinctive properties, such as its hemostatic, antimicrobial activity, and biocompatibility, making it a valuable material for creating advanced wound dressings. CS-based wound dressings have been shown to effectively manage exudates, prevent infections, and accelerate the healing process in various wound types, including diabetic ulcers and burns.31–33 Additionally, nanofibrous CS scaffolds have been observed to positively influence re-epithelialization and the regeneration of the granular layer of wounds, thereby enhancing the wound healing process.34,35 While CS-based nanofibrous scaffolds show promise in various applications, including wound dressings, they often encounter limitations due to their fragility. Furthermore, challenges persist in achieving spinability and reproducibility of CS to yield stable nanofibrous scaffolds using Nanospider™ technology. To overcome these challenges, CS is often blended with other synthetic polymers. Polyvinyl alcohol (PVA), polyethylene oxide (PEO), and polycaprolactone (PCL) have gained widespread adoption to facilitate CS electrospinning and reduce its fragility.36–39 These blends not only facilitate the electrospinning process but also enhance the biological performance of the resulting nanofibers, making them more effective in wound healing applications.40–42 Additionally, the incorporation of antibacterial, antimicrobial, or antioxidant agents into nanofibrous scaffolds has proven to be a key strategy in wound healing. These agents can include metallic nanoparticles, carbon-based composites, and herbal medicines, which offer unique pharmacological effects that combat inflammation and microbial infections in wound areas.43–50 Nanofibers loaded with antibacterial agents play a vital role in inhibiting bacterial growth and promoting wound healing. 51 The development of nanofibrous scaffolds loaded with antibiotics or other medical agents, such as tetracycline (TET), a broad-spectrum antibiotic, has garnered attention for their potential in rapid wound healing, encompassing infection control, inflammation reduction, and accelerating wound closure.52–58
Despite significant advancements in nanofibrous scaffolds, critical challenges persist in fully realizing their clinical potential. Previous research has often focused on binary polymer systems and encapsulation of therapeutic agents, leading to suboptimal drug release and reduced surface activity. Additionally, scaling up production while preserving scaffold functionality remains inadequately addressed. Our research aims to design, fabricate, and characterize nanofibrous scaffolds composed of three polymers, specifically examining how varying PEO and PCL compositions influence scaffold properties. We successfully electrospun a polymer blend of CS, PCL, and PEO into nanofibrous scaffolds using Nanospider™ technology, enabling large-scale production while maintaining desired functional properties. The scaffolds were characterized through SEM, FTIR, contact angle measurements, hydrolytic degradation, water vapor permeability tests, mechanical tests, and antibacterial activity assays, with cytotoxicity assessed using 3T3 fibroblasts. This approach advances beyond previous studies constrained by binary systems,38,39,41,59 complex multi-step processes, 48 or traditional needle electrospinning. 60 Additionally, our approach enhances antibacterial efficacy through surface immobilization of TET, preserving its activity and allowing for post-production customization. This innovative approach: (i) demonstrates the feasibility of industrial-scale production for wound healing, (ii) enables surface immobilization of TET for personalized care, and (iii) offers tunable hydrophilicity, hydrolytic degradation and mechanical properties by adjusting the PEO/PCL ratio.
Materials and methods
As mentioned in the introduction, CS is a well-known natural biopolymer with great potential for wound dressing due to its biocompatibility, biodegradability, low immunogenicity, and antimicrobial properties, which help prevent infections and promote cell growth. 61 Its reactive amino groups further enhance wound healing by facilitating drug bonding and delivery. However, CS has drawbacks, including fragility and difficulties with spinnability during electrospinning. 62 To address these challenges, literature suggests that blending CS with PCL or PEO improves the mechanical strength, spinnability, and biocompatibility of the scaffolds, creating an optimal environment for wound healing.38,39,41,59 PCL, with its flexibility, durability, and slow biodegradation, provides long-term structural support and enables controlled drug release. 63 However, PCL is highly hydrophobic and lacks cell-binding sites. 64 On the other hand, PEO enhances spinnability in needle-less electrospinning, aids in moisture retention, and promotes the formation of uniform nanofibers, though it has lower mechanical strength and is water soluble. 65 Our nanofibrous scaffold was designed to be easy to handle, non-toxic, and breathable, with the additional capability of drug bonding through its amino groups while effectively managing moisture. We hypothesize that adjusting the PEO/PCL ratio can tailor the scaffold’s properties, leveraging the distinct characteristics of PEO and PCL for various wound healing applications. The selected mass ratios (6/2/2, 4/4/2, 2/6/2) were chosen to systematically examine how variations in PEO and PCL quantity affect scaffold properties, while maintaining a constant CS content. This approach enabled us to isolate the impact of different polymer ratios on critical factors such as antibacterial efficacy, breathability, moisture management, and mechanical strength.
Fabrication of nanofibrous scaffolds
Nanofibrous scaffolds were electrospun from polymer mixture solutions using needle-less electrospinning technology (Nanospider™ NS-Lab 1WS500U, Elmarco, Czech Republic). This standalone electrospinning system integrates industrial production technology with the precision of a high-end laboratory instrument (see Figure 1). The Nanospider™ technology employs a spinning electrode in the form of a thin wire, along with a head that applies the polymer solution across the entire length of this wire. When subjected to a strong electric field, nanofibers are generated from a thin layer of polymer solution deposited on the underlying material at the opposite electrode. Numerous studies have reported that the morphology of electrospun fibers is significantly influenced by factors such as polymer concentration, collector-wire distance, spinning speed, applied voltage, temperature, and air humidity. By carefully controlling these parameters, the morphology of the fibers can be precisely engineered.
66
In this study, we optimized both the polymer solution preparation and the electrospinning process to successfully produce defect-free nanofibrous scaffolds with consistent nanofiber morphology and high productivity using Nanospider™ technology. We specifically compared polymer blend concentrations of 8 wt% and 6 wt% for PEO/PCL/CS. At 4 wt%, bead formation occurred instead of nanofibers, while concentrations above 8 wt% hindered the initiation of needle-less electrospinning. The viscosities of polymer solutions prior to electrospinning were measured using a HAAKE RotoVisco 1 (Thermo Scientific) at 24°C, with a RV1 spindle operating from 10 r/min to 4000 r/min for 90 s. Photographs of the Nanospider™ NS-Lab 1WS500U: (a) front view, (b) side view, and (c) the resulting nanofibrous scaffold (asterisk) deposited onto the underlying spun-bond (arrow).
For polymer solution preparation, two methods were compared. In the one-step method, solid polymers—PEO (Mn 100,000 g/mol, Sigma-Aldrich, USA), PCL (Mw 80,000 g/mol, Sigma-Aldrich, USA), and CS (Mw 30,000 g/mol, Glentham, UK)—were weighed in the ratios of 6/2/2, 4/4/2, and 2/6/2. The solvent mixture, consisting of acetic acid (AA, 99.98%, Sigma Aldrich) and formic acid (FA, 99%, Sigma Aldrich) in a 2/1 vol ratio, was added to dissolve the solid polymers. The polymer solutions were stirred at 200 r/min at room temperature for 24 h using a magnetic stirrer.
In the two-step method, each polymer was dissolved separately in the AA/FA solvent mixture (2/1, v/v) and stirred at 200 r/min for 24 h using a magnetic stirrer. The following day, the individual polymer solutions of PEO, PCL, and CS were mixed in the respective mass ratios (6/2/2, 4/4/2, 2/6/2). The combined solutions were stirred overnight using a magnetic stirrer for proper homogenization. The polymeric solutions PEO/PCL/CS were then transferred into a 500 mL reservoir connected to the head of the electrospinning apparatus. A voltage of either 40 or 65 kV was applied between the wire and the collector, with a distance of 150 or 180 mm, at a controlled temperature of 22°C and humidity of 10%. The resulting nanofibers were deposited onto a backing paper along a negative collector electrode at a velocity of 15 mm/min (see Figure 2). Schematic of needle-less electrospinning.
The resulting nanofibrous scaffolds were sheet-shaped, with dimensions of 50 × 600 cm. After electrospinning, the scaffolds were cut into A4-sized sheets and subjected to a stabilization process. This process involved heating the sheets overnight at 60°C, followed by removal of the scaffolds from the backing paper. The purpose of this step was to stabilize the scaffolds through cross-linking. After stabilization, the scaffolds were stored in a desiccator for further characterization.
Immobilization of tetracycline onto the nanofibrous scaffolds
The immobilization of TET (98.0%–102.0% HPLC, Sigma-Aldrich, USA) onto the PEO/PCL/CS nanofibrous scaffolds was carried out by immersing the scaffolds in a 0.5 wt% TET solution dissolved in ethanol. This process was conducted in a hermetically sealed chamber, where the scaffolds were placed in glassware containing the TET solution and shaken at 50 r/min using a laboratory shaker at room temperature. After 90 min, the scaffolds labeled as AB were removed from the TET solution and laid out on a silicone sheet to air dry at room temperature for 24 h. In contrast, scaffolds labeled as B underwent the same removal process but were thoroughly washed with ethanol before drying. These distinct treatments were designed to evaluate the antibacterial efficacy of the nanofibrous scaffolds, comparing those with the surface-bound TET component (sample_B) to those where TET was merely adsorbed onto the nanofibers (sample_AB).
Morphological characterization
Scanning electron microscopy (SEM) using the VEGA TS 5130 instrument was employed to characterize the surface morphology of the nanofibrous scaffolds and to measure the diameters of the nanofibers. SEM images were acquired with an accelerating voltage of 30 kV. Prior to imaging, each nanofibrous scaffold was coated with gold via sputtering. SEM micrographs were then analyzed using the image analysis software VegaTC to measure the average diameter of approximately 100 fibers from each scaffold type. The gram per square meter (GSM) of the nanofibrous scaffolds was measured according to ISO 3801:1977.
Chemical analysis
Fourier transform infrared spectroscopy (FTIR) analysis was performed using attenuated total reflectance (ATR) with a Nicolet Magna-IR 560 spectrophotometer (Thermo Scientific). ATR-FTIR spectra were recorded over the wavenumber range of 3700–700 cm⁻1 using a split peak accessory. Each spectrum was the average of 64 scans, with a resolution of 4 cm⁻1. The FTIR spectra of PEO/PCL/CS nanofibrous scaffolds, with mass ratios of 2/6/2, 4/4/2, and 6/2/2, were analyzed before and after immersion in deionized (DI) water for 1 and 7 days.
The content of functional amino groups on the surface of the scaffolds was determined spectrophotometrically through a specific reaction with methyl orange (MO, Sigma-Aldrich, USA), which binds to primary amino groups.
67
Nanofibrous scaffolds were incubated in a 0.05% MO solution and 0.01 M phosphate buffer (Sigma-Aldrich, USA) at pH 4.7 for 1 h. After incubation, unbound dye was washed away, and MO bound to the amino groups was released by rinsing with a 0.1 M sodium carbonate solution (anhydrous, ≥99.5%, Sigma-Aldrich, USA). The concentration of bound MO, which indicates the available functional groups, was measured spectroscopically at 465 nm using Lambert-Beer’s law:
Hydrolytic degradation test
A hydrolytic degradation test was conducted to evaluate the stability and degradability of the nanofibrous scaffolds. The scaffolds were initially dried at 60°C for 24 h. Subsequently, they were immersed in DI water and incubated at 37°C for 7 days. During this period, the solid material was collected using a nylon filter (9 cm diameter) at 1, 2, and 7 days, and then dried to a constant weight at 60°C for an additional 24 h.
68
The percentage of remaining nanofiber mass g, representing the insoluble portion of the scaffolds, was calculated using the following equation:
Structural and morphological changes in the nanofibrous scaffolds were assessed through SEM imaging before and after hydrolytic degradation.
Contact angle measurement
Optical tensiometry, also known as contact angle goniometry, is a widely used technique for determining the contact angle of a liquid droplet on a solid surface, thereby characterizing its surface properties. 69 To assess the hydrophilicity of the nanofibrous scaffolds, contact angle measurements were performed at three different positions using a Kruss Drop Shape Analyzer DS4 at room temperature. A 2 μL droplet of DI water was placed onto the dry scaffold surface, and the average contact angle values were calculated from these measurements.
Characterization of mechanical properties
The mechanical properties of the nanofibrous scaffolds were evaluated using a uniaxial tensile test performed with a LabTest 2.010 system (LabControl, Czech Republic) at room temperature. Nanofiber scaffolds, sized 20 × 60 mm, were placed within a window frame and mounted onto the tensile test grips. Prior to initiating the test, the vertical ribs of the frame were cut. The scaffolds were stretched at a strain rate of 10 mm/min until failure. Stress-strain curves were recorded, and Young’s modulus was calculated from the slope of these curves within the strain range of 5%–10%.
Moisture vapor transmission rate measurement
Moisture vapor transmission rate (MVTR) of the nanofibrous scaffolds was measured according to the European Standard EN13726-2:2002, which outlines test methods for vapor-permeable film dressings.
70
Each scaffold was affixed to a Paddington cup with a 10 cm2 opening, containing 25 mL of water. The cup, along with its contents, was weighed and placed in an incubator at 37°C. To maintain low relative humidity within the chamber, a tray filled with 1 kg of freshly dried silica gel was placed at the bottom of the incubator. At predetermined intervals (ranging from 1 to 7 days), the entire cup system was reweighed, and the weight of moisture vapor loss through the nanofibrous scaffold was recorded. MVTR [g/m2/day] was calculated using the formula:
In vitro cytotoxicity evaluation
In vitro cytotoxicity of the nanofiber scaffolds was evaluated using two methods in accordance with ISO 10993-5. 71 Prior to testing, the nanofibrous scaffolds were rinsed three times in DI water for 10 min and dried overnight at room temperature to remove any residual solvents. The dried scaffolds were then sterilized on both sides using UV light for 20 min.
For the direct cytotoxicity assay, 3T3 fibroblasts (American Type Culture Collection, Manassas, USA) were seeded at a concentration of 6 × 10⁴ cells per well in sterile 24-well tissue culture plates. The cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM, StableCell™, Sigma-Aldrich, USA) supplemented with 5% fetal bovine serum (FBS, non-USA origin, Sigma-Aldrich, USA), 5% newborn calf serum (NBCS, USA origin, Sigma-Aldrich, USA), and 1% penicillin-streptomycin (P/S, suitable for cell culture, Sigma-Aldrich, USA) at 37°C in a 5% CO₂ environment for 24 h. After confirming cell layer consistency under a microscope, the supplemented DMEM was removed, and the scaffolds with controls (6 mm diameter circle-shaped) were carefully placed on the cell layer. Fresh supplemented DMEM was added, and the scaffolds were incubated in direct contact with the cells at 37°C in a 5% CO₂ environment for the next 24 h. Polyethylene high-density film (HDPE film, Sigma-Aldrich, USA) served as the negative control, while a 0.1% ZDEC-polyurethane (PU) film (ZDEC-PU film, Hatano Research Institute, Food and Drug Safety Center, Japan) was used as the positive control.
For the indirect cytotoxicity assay, 3T3 fibroblasts were seeded at a concentration of 1 × 10⁴ cells per well in sterile 96-well tissue culture plates. The cells were cultured in DMEM supplemented with 5% FBS, 5% NBCS, and 1% P/S at 37°C in a 5% CO₂ environment for 24 h. Eluates of the tested materials were prepared in supplemented DMEM at a concentration of 5 mg/mL and were incubated at 37°C in a 5% CO₂ environment for 24 h. After confirming cell layer consistency, the supplemented DMEM was removed, and the eluates from the tested scaffolds and controls were carefully added to the cell layer. The cells were then incubated with the eluates at 37°C in a 5% CO₂ environment for the next 24 h. Sodium dodecyl sulfate (SDS, ≥98.5% (GC), Sigma-Aldrich, USA) at a concentration of 0.2 mg/mL in supplemented DMEM served as the positive control.
Both direct and indirect cytotoxicity assays of the nanofibrous scaffolds were evaluated using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT, Duchefa Biochemie, Netherlands) assay. Sample aliquots (10 per each time point) of 100 µL were transferred to a 96-well plate, and the absorbance was measured at 570 nm against 650 nm using a microplate reader. Viability of nanofibrous scaffolds was calculated based on the absorbance values obtained from wells containing cells incubated with the scaffold/eluate (ASAMPLE), the mean absorbance of wells containing only DMEM (ADMEM), and the absorbance of wells containing non-affected cells (incubated with no material) (ACELLS):
Antimicrobial activity
The antibacterial activity of the nanofibrous scaffolds was evaluated using the Kirby-Bauer disk diffusion method against two bacterial strains: Escherichia coli (ATCC 25922) and Staphylococcus gallinarum (ATCC 35539), obtained from the Czech Collection of Microorganisms at Masaryk University. Both strains were prepared at a concentration of 1 × 10⁶ CFU/mL, cultured in a shaker incubator at 200 r/min and 37°C, and adjusted to an OD₆₀₀ of 0.5. The bacterial suspension was then evenly spread on sterile agar plates using a sterile swab. Nanofibrous scaffolds with TET, cut into 1 cm diameter circles, and controls (scaffolds without TET) were placed on the agar surfaces. After a 24-h incubation at 37°C, the presence of a clear zone of inhibition around the scaffolds was measured. The zone of inhibition, indicative of antibacterial activity, reflects the diffusion of TET from the scaffold and its effectiveness in preventing bacterial growth. The size of this zone correlates with the level of antimicrobial activity, with larger zones indicating greater effectiveness.
Statistical analysis
All experiments were performed in triplicate (at a minimum) to ensure accuracy and reproducibility. Results are expressed as the mean ± standard deviation (SD). Statistical analysis was conducted using the t test to compare data between scaffolds. Significance levels are indicated: p < 0,05 (*), p < 0,01 (**), and p < 0,001 (***).
Results and discussion
Morphology of PEO/PCL/CS nanofibrous scaffolds
Structural factors such as fiber diameter, porosity, and homogeneity are crucial in designing wound healing patches to optimize therapeutic outcomes. Finer nanofibers, with a higher surface area-to-volume ratio, enhance cell attachment and drug absorption, making them effective for wound care.72,73 A porous structure facilitates moisture retention, gas exchange, and nutrient transport, creating an ideal healing environment.74,75 Homogeneous nanofibers ensure consistent drug distribution and scaffold integrity, leading to reliable healing. 76 Conversely, larger fibers, non-porous structures, and non-homogeneous scaffolds can impair these processes and reduce wound healing efficacy. 77 Therefore, incorporating these structural features through electrospinning is essential for advancing wound care solutions. While needle-less electrospinning offers advantages such as scalability, it also presents several challenges. In this method, the polymer solution is exposed to a large surface area, causing multiple jets to form spontaneously due to surface tension. Controlling the number and uniformity of these jets is difficult, often resulting to non-uniform fiber diameters and inconsistent fiber structures. Additionally, not all polymers are compatible with needle-less electrospinning, as some require specific solvents or conditions that are not feasible in this system. The viscosity and conductivity of the polymer solution are crucial, as they must fall within a specific range to initiate and sustain multiple jets. If these parameters are not met, stable jet formation may fail, leading to interrupted fiber production or the formation of beads. 66
SEM images revealed that nanofibrous scaffolds electrospun under our proposed and repeatedly tested conditions (both process and materials) exhibited a homogeneous structure, providing a porous texture with no apparent beads or significant defects (see Figures 3 and 4). To produce defect-free nanofibrous scaffolds, the two-step method for polymer solution preparation proved to be more effective, resulting in higher-quality nanofibers compared to the one-step method (see Figure 3(a) versus (b)). In terms of polymer solution concentration, a consistent production of defect-free nanofibers was observed at a concentration of 6 wt%. At higher concentrations, macroscopic holes began to appear in the scaffolds (see Figure 3(c) versus (d)). For electrospinning with an electrode distance of 150 mm and a voltage of 40 kV, all scaffolds exhibited macroscopic holes (photographs not shown). When the voltage was adjusted to 65 kV at the same electrode distance, no macroscopic holes were observed in the nanofibrous scaffolds. Moreover, all nanofibrous scaffolds produced under these conditions achieved a higher GSM than those produced at 40 kV. When electrospinning was performed with an electrode distance of 180 mm and a voltage of 65 kV, all nanofibrous layers showed a lower GSM (see Table 1). Photographs and SEM images of electrospun nanofibrous scaffold PEO/PCL/CS from polymer solutions prepared by (a) one-step and (b) two-steps method. Polymer solution concentrations are shown in (c) 8 wt% and (d) 6 wt%. SEM images of nanofibrous scaffolds prepared from PEO/PCL/CS polymer mixtures with mass ratios of (a) 6/2/2, (b) 4/4/2, and (c) 2/6/2. The scaffolds were produced using the two-step preparation method at a polymer concentration of 6 wt% by needle-less electrospinning with a 150 mm electrode distance and a voltage of 65 kV. GSM and fiber diameters (fd) of nanofibrous scaffolds prepared by needle-less electrospinning under different electrode distance and voltage conditions. Results are expressed as mean ± SD.


Literature suggests that the ideal GSM for wound healing patches depends on their application and desired properties. Higher GSM patches provide robust protection, better absorption, and greater structural integrity, making them suitable for high-exudate wounds that require cushioning. 78 Conversely, lower GSM patches are lighter, thinner, more breathable, and flexible, making them ideal for wounds where mobility and comfort are prioritized. 79 The selection should balance the wound’s needs with patient comfort to achieve optimal healing outcomes.
Upon comparing fiber diameters, the highest value of 185 ± 53 nm was observed in scaffolds produced with an electrode distance of 150 mm and a voltage of 40 kV. In contrast, the lowest fiber diameter of 129 ± 40 nm was observed in scaffolds produced with an electrode distance of 180 mm and a voltage of 65 kV (see Table 1). Statistical analysis using the t test revealed no significant differences in fiber diameters between scaffolds with varying mass ratios of PEO/PCL/CS.
For further characterization and TET immobilization, nanofibrous scaffolds prepared using (i) the two-step method, (ii) a polymer concentration of 6 wt%, and (iii) an electrode distance of 150 mm with a voltage of 65 kV were selected. The viscosities of the polymer solutions for the 6/2/2, 4/4/2, and 2/6/2 compositions were 309 ± 6, 487 ± 9, and 546 ± 8 MPa·s, respectively. The structural morphologies of these scaffolds are shown in Figure 4 and Table 1.
In a previous study, 80 needle-less electrospinning was employed to produce wound healing patches from a single-component PCL system, resulting in a maximum GSM of 3.4 g/m2 and an average fiber diameter of 160 nm. In contrast, this study successfully fabricated nanofibrous scaffolds composed of three polymers (PEO/PCL/CS) using needle-less electrospinning, without the need for toxic solvents or cross-linking agents. Our scaffolds achieved finer fiber diameters, ranging from 134 to 148 nm, and demonstrated superior structural integrity and ease of manipulation, with GSM values between 6.8 and 8.6 g/m2. These results suggest that our multi-component system offers enhanced structural properties, making it more suitable for wound healing applications.
Chemical characterization of PEO/PCL/CS nanofibrous scaffolds
FTIR analysis was employed to characterize the structure and interactions within the PEO/PCL/CS nanofibrous scaffolds, focusing on identifying the presence of individual polymers. The FTIR spectrum was anticipated to show characteristic peaks corresponding to the functional groups of PEO, PCL, and CS.
PEO typically exhibits C–O–C stretching around 1100 cm⁻1, CH₂ wagging and twisting near 1342–1240 cm⁻1, and C–H stretching at 2891 cm⁻1.
81
PCL is known for its strong C = O stretching peak around 1720–1735 cm⁻1.
82
CS typically shows a broad O–H and N–H stretching between 3200 and 3600 cm⁻1, N–H bending (amide II) around 1550–1650 cm⁻1, and C–N stretching (amide III) near 1250–1310 cm⁻1.83,84 In polymer blends, these peaks may overlap, with potential shifts or changes in intensity due to interactions such as hydrogen bonding between the polymers.
85
The ATR–FTIR spectra of the PEO/PCL/CS nanofibrous scaffolds with different mass ratios are shown in Graph 1. ATR-FTIR spectra of nanofibrous scaffolds composed of PEO/PCL/CS with varying mass ratios (6/2/2, 4/4/2, 2/6/2). The spectra display peaks corresponding to different functional groups present in the scaffolds: 1724 cm⁻1 (confirming the presence of PCL), 1100 cm⁻1 and 2891 cm⁻1 (indicating the presence of PEO), and a broad peak between 3200 and 3600 cm⁻1 along with a sharp peak at 1590 cm⁻1 (confirming the presence of CS).
The FTIR analysis of nanofibrous scaffolds composed of PEO, PCL, and CS revealed characteristic peaks confirming the successful incorporation of these polymers. A prominent peak at 1724 cm⁻1 corresponds to the C = O stretching of PCL, confirming its presence. Notably, there was no significant increase in the intensity of this C = O stretching vibration band across the different scaffold compositions, indicating that the chemical environment of PCL remains relatively consistent regardless of the PEO or CS content. The C–O–C stretching vibrations at 1100 cm⁻1 and C-H stretching at 2891 cm⁻1 verify the presence of PEO, with the intensity of these bands increasing as the PEO mass ratio increased. Additionally, a broad peak between 3200 and 3600 cm⁻1, along with a sharp peak at 1590 cm⁻1, attributed to the amino, hydroxyl, and amide II groups in CS, confirmed its successful incorporation. Moreover, the FTIR data suggest potential chemical interactions, particularly hydrogen bonding, between PEO and CS. This is supported by the increasing intensity in the 3200–3600 cm⁻1 range as PEO content increases. Notably, this increase occurs despite a constant CS content across all scaffolds, indicating that the changes in peak intensity are primarily driven by amount of PEO. PCL, being hydrophobic, likely contributes to the scaffold’s mechanical strength through physical entanglement rather than direct chemical bonding. Additionally, possible ionic interactions between PEO and CS may further enhance the scaffold’s structural integrity.41,59,97
Further analysis of the FTIR results revealed notable changes during the solubility study, particularly in the spectrum of PEO/PCL/CS nanofibrous scaffolds after 7 days of hydrolytic degradation in DI water at 37°C. Specifically, a reduction in the characteristic peak associated with PEO (1100 cm⁻1) was observed, indicating the gradual dissolution of PEO from the scaffold during the soaking period, as shown in Graph 2. This information is crucial for designing scaffolds that retain their integrity and functionality throughout the intended application period, such as in wound healing. ATR-FTIR spectra of nanofibrous scaffolds made from PEO/PCL/CS with mass ratios of (a) 6/2/2 and (b) 2/6/2, both before and after hydrolytic degradation in DI water for 7 days at 37°C. The spectra highlight a peak attributed to C–O–C stretching, which signifies the presence of PEO.
The immobilization of molecules onto nanofibers is highly dependent on the presence of surface-accessible functional groups. Functional groups such as amine, imine, and carboxylic groups are crucial for making the surface active and suitable for molecule immobilization. Among these, primary amine groups, like those found in CS, are particularly favorable for antibiotic conjugation purposes. 86
The presence of surface-accessible amino groups was confirmed across all nanofibrous scaffolds. As shown in Graph 3., the variation in the mass ratios of PEO/PCL/CS did not significantly affect the presence of these amino groups. This is likely because the mass ratio of CS remained constant in all mixtures. Specifically, the quantification of available amino groups on PEO/PCL/CS scaffolds with mass ratios of 2/6/2, 4/4/2, and 6/2/2 were 86.9 ± 8, 82.6 ± 6, and 83.3 ± 4 nmol NH2/cm2, respectively. Statistical analysis using the t test revealed no significant differences in the number of amino groups between scaffolds with different mass ratios of PEO/PCL/CS. Quantification of available amino groups on PEO/PCL/CS nanofibrous scaffolds with different mass ratios (6/2/2, 4/4/2, 2/6/2). Error bars represent SD. (n = 3).
Hydrolytic degradation
Hydrolytic degradation is critical for the functionality of wound healing patches, influencing how the scaffold degrades in body fluids. The degradation rate must be carefully controlled: too rapid degradation may fail to provide adequate support for the wound, while too slow degradation can hinder healing or require surgical removal. 87
Materials like PCL, which degrade slowly, provide extended structural support, whereas PEO, a more hydrophilic and faster-degrading polymer, contributes to rapid hydrolytic degradation. 88 Balancing hydrolytic degradation is essential to ensure scaffolds provide effective support while degrading safely over time. 89
The hydrolytic degradation study of the nanofibrous scaffolds provided insights into their degradation behavior and weight loss profiles over 7 days in DI water. All scaffolds demonstrated noticeable degradation, as indicated by their weight loss. Specifically, scaffolds with PEO/PCL/CS mass ratios of 6/2/2, 4/4/2, and 2/6/2 experienced weight losses of 73.2 ± 0.8%, 54 ± 1.3%, and 35.8 ± 1%, respectively, within 7 days (see Graph 4). The study revealed a correlation between weight loss and PEO content: higher PEO content resulted in greater weight loss, indicating that PEO concentration directly affects the degradation kinetics of the scaffolds. The degradation did not plateau over the 7-day period, showing a continuous decrease with the most significant degradation observed on the first day of, indicating an initial burst degradation phase (see Graph 4.). Statistical analysis using the t test revealed significant differences in weight loss between scaffolds with varying mass ratios of PEO/PCL/CS. Hydrolytic degradation of nanofibrous scaffolds composed of PEO/PCL/CS with different mass ratios, immersed in DI water at 37°C for 1, 2, and 7 days. Significant statistical differences between the scaffolds with different mass ratios of PEO/PCL/CS were observed using the t test, with *** indicating p ≤ 0.001. Error bars represent SD. (n = 3).
SEM analysis revealed that the scaffolds maintained a partially porous structure throughout the dissolution period, as shown in Figure 5. Despite prolonged immersion in DI water, the scaffolds did not collapse into a homogeneous film, demonstrating their stability and durability in an aqueous environment. SEM images of thermal stabilized nanofibrous scaffolds with different mass ratios of PEO/PCL/CS immersed into DI water at 37°C for (a) 0 s, (b) 1 day and (c) 7 days.
In a study, 41 a two-component PCL/CS system was electrospun for wound healing applications, resulting in a hydrolytic degradation rate of approximately 90% after 7 days. In contrast, our PEO/PCL/CS scaffolds exhibited lower degradation rates, ranging from 73.2 ± 0.8% to 35.8 ± 1%, depending on the PEO/PCL mass ratio. The ability to tailor degradation rates by adjusting the PEO/PCL ratio enhances the adaptability of our scaffolds to specific wound healing requirements. Furthermore, the stability and durability of our scaffolds in an aqueous environment, achieved without toxic cross-linking agents, underscore their potential for biomedical applications, particularly in scenarios involving prolonged fluid exposure.
Wettability of PEO/PCL/CS nanofibrous scaffolds
The hydrophilicity of materials used for wound healing is a critical parameter that significantly influences their ability to manage wound exudate and maintain a moist environment conducive to proper healing. Assessing the hydrophilic properties of nanofibrous scaffolds is vital for evaluating their suitability in wound care, particularly regarding their capacity to facilitate fluid drainage while retaining optimal moisture levels. 90 Hydrophilic surfaces are generally preferred over hydrophobic ones in wound healing patches, as they promote a moist environment, which accelerates epithelialization and improves overall healing outcomes.91,92 Furthermore, hydrophilic surfaces adhere more effectively to wet surfaces, enhancing treatment efficacy. 93 Conversely, hydrophobic surfaces can disrupt moisture balance and inhibit cell migration, potentially hindering the healing process. 94
Contact angle measurements were performed to quantitatively assess the wetting behavior of water droplets on the nanofibrous scaffolds’ surfaces. As shown in Figure 6, these measurements provide valuable insights into how variations in scaffold composition, particularly changes in the proportions of PCL and PEO, influence their surface properties. Water contact angle measurement of nanofibrous scaffolds composed of PEO/PCL/CS with mass ratios of 6/2/2, 4/4/2, and 2/6/2, measured at 0 s, 1 min, and 5 min.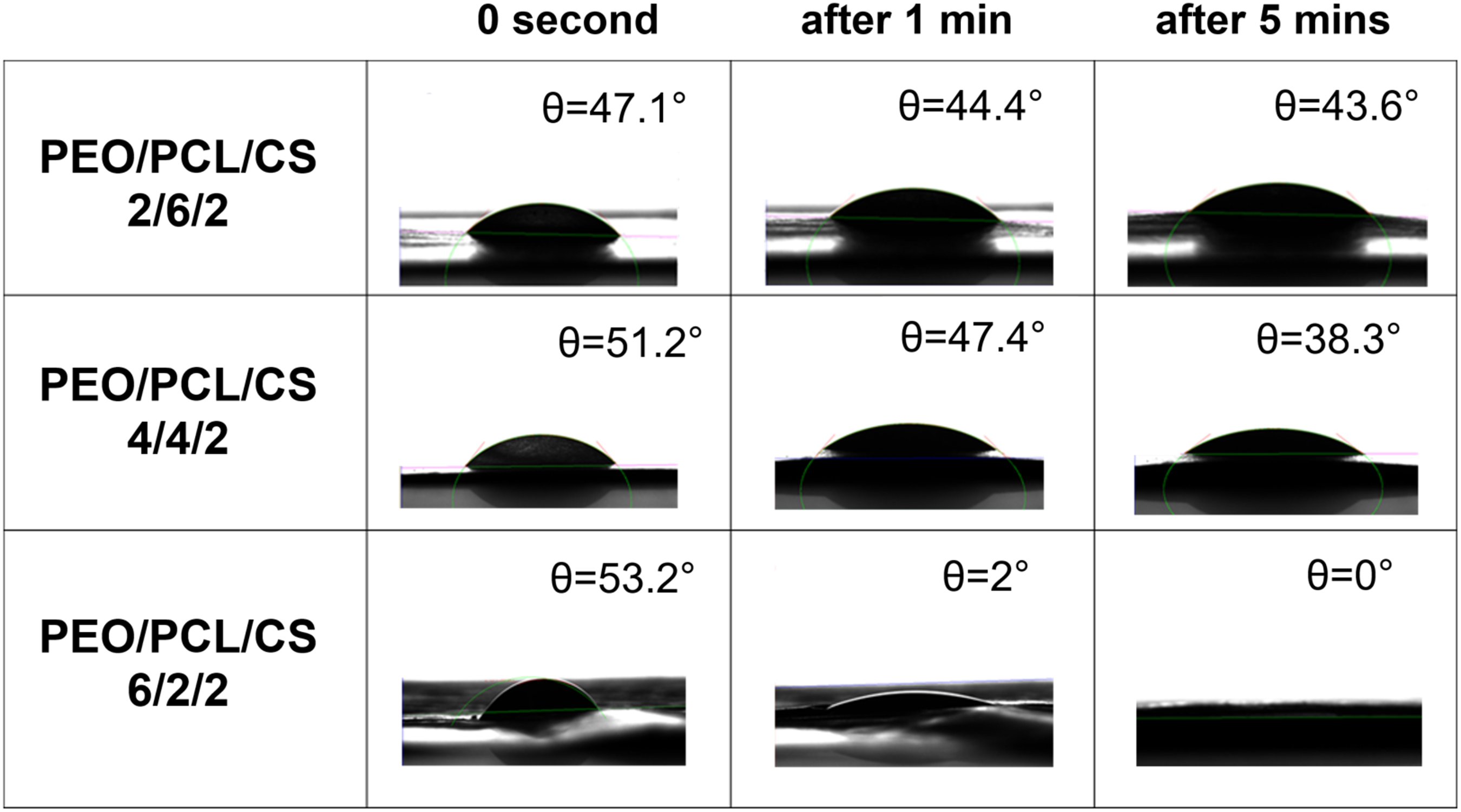
The static contact angle of water droplets on the PEO/PCL/CS scaffolds with mass ratios of 2/6/2 and 4/4/2 was measured at 42.9 ± 3.8° and 37.7 ± 3.3°, respectively, within 5 min of applying DI water, indicating moderate hydrophilicity. In contrast, the scaffold with a 6/2/2 mass ratio achieved a contact angle of 0.7 ± 1.3° within just 60 s, suggesting rapid surface wetting. The difference in contact angles after 5 min between the PEO/PCL/CS scaffolds with mass ratios of 6/2/2 and 2/6/2 was found to be statistically significant using the t test. This behavior can be attributed to the different mass ratios of PEO and PCL, as PEO is known for its hydrophilic properties, while PCL is more hydrophobic.
In a previous study, 48 the contact angles for a mesh of PCL nanofibers combined with CS/PEO nanofibers ranged from 55.5° to 107°, indicating a relatively hydrophobic surface. In contrast, our study achieved significantly lower contact angles, ranging from approximately 0 to 43°, depending on the PEO/PCL/CS mass ratio, indicating a more hydrophilic surface. The reduced contact angles and enhanced hydrophilicity of our scaffolds are expected to improve the functionality and therapeutic effectiveness of wound healing patches. 95
Mechanical properties of PEO/PCL/CS nanofibrous scaffolds
Mechanical properties of PEO/PCL/CS nanofibrous scaffolds with different mass ratios.
The Young’s modulus of the PEO/PCL/CS nanofibrous scaffolds with mass ratios of 2/6/2, 4/4/2, and 6/2/2 is 106.7 ± 8.5 MPa, 128.9 ± 7.2 MPa, and 170.7 ± 11.9 MPa, respectively. The decrease in Young’s modulus with increasing PCL content is due to PCL’s inherent flexibility, which reduces the overall stiffness of the scaffold. Despite this, the ultimate tensile strength increases with higher PCL content, as PCL enhances the scaffold’s ability to distribute stress and bear higher loads. 96 Additionally, the elongation at fracture for the PEO/PCL/CS 2/6/2 scaffold is 26 times greater compared to scaffold with higher PEO content. This significant increase in elongation at fracture is attributed to PCL’s high ductility and flexibility. As PCL content increases, the scaffold becomes more stretchable and can undergo larger deformations before breaking, resulting in much higher elongation at fracture. 97
Despite the different mechanical behaviors observed with varying PEO and PCL compositions, the mechanical properties of the scaffolds—such as Young’s modulus, ultimate tensile strength, and elongation at fracture—are comparable to those of native human skin. Native skin typically exhibits a Young’s modulus in the range of 6-140 MPa, an ultimate tensile strength between 2 and 16 MPa, and significant elongation at break.98,99 The scaffolds’ properties closely align with these values, making them suitable for applications in skin regeneration, where mimicking the natural mechanical behavior of skin is crucial.
Moisture vapor transmission rate of PEO/PCL/CS nanofibrous scaffolds
Maintaining optimal moisture content is crucial for effective wound healing, as a moist environment supports cell proliferation and function. 100 Scaffolds with an MVTR below 1000 g/m2/day may not effectively manage wound moisture, potentially leading to issues such as exudate buildup and patient discomfort.101,102 An ideal MVTR, ranging from 1000 to 2000 g/m2/day, strikes a balance between moisture retention and breathability, thereby enhancing the healing process by maintaining optimal moisture levels.103,104 Conversely, scaffolds with an MVTR above 2000 g/m2/day are suitable for wounds with high exudate levels, ensuring effective moisture management without excess buildup. 105
The MVTR of PEO/PCL/CS nanofibrous scaffolds was assessed using the standardized Paddington cup method. Graph 5. Depicts the results, where a lower proportion of the original medium corresponds to a higher MVTR. Initially, there was no significant difference between the nanofibrous scaffolds and a commonly used certified polypropylene/viscose-based patch cushion (commercial dressing patch). However, over a period of 7 days, the moisture content of the nanofibrous scaffolds decreased by approximately 55%–63%, compared to the commercial dressing patch, which retained only 13% of its original moisture. This suggests that the commercial dressing may not effectively release water vapor, potentially hindering the wound healing process by maintaining a consistently wet environment. Monitoring of moisture vapor leakage from the Paddington cup system through nanofibrous scaffolds composed of PEO/PCL/CS with different mass ratios, and a commercially available patch cushion, evaluated according to the EN13726-2 standard. Error bars represent SD. (n = 3).
Specifically, scaffolds with a higher mass ratio of PEO retained 44.9 ± 1.2% of their original moisture content after 7 days, whereas those with a lower PEO mass ratio retained 37.3 ± 0.8%. Furthermore, no statistically significant differences were observed between the nanofibrous scaffolds with varying mass ratios of PEO/PCL/CS, as shown in Graph 5.
Recalculating the proportion of the original medium to the MVTR using formula 3 yielded values of 1904.3 ± 28.6, 1941.4 ± 35.7, and 2005.7 ± 42.9 g/m2/day for nanofibrous scaffolds with PEO/PCL/CS mass ratios of 6/2/2, 4/4/2, and 2/6/2, respectively. In contrast, the commercial dressing patch demonstrated less effective moisture management, potentially impeding the healing process. This highlights the superior performance of our scaffolds in maintaining optimal moisture levels for enhanced wound care. Notably, the MVTR values of our scaffolds exceed those reported in the literature, 59 where a two-layer scaffold (PCL nanofibers as the first layer and CS/PEO nanofibers as the second layer) achieved MVTR values of approximately 1252–1452 g/m2/day. These results indicate that our nanofibrous scaffolds are highly effective in moisture management, making them suitable for exudative wounds, where precise moisture control is crucial.
Cytotoxicity of PEO/PCL/CS nanofibrous scaffolds
Cytotoxicity assessment is a critical step in developing safe and effective wound healing patches. Materials must be non-toxic and biocompatible, ensuring that they support healing without harming cells.106,107 Common tests, such as MTT assays and direct contact evaluations, measure cell viability by observing how materials interact with cells. Compliance with ISO 10993 standards, particularly ISO 10993-5 for in vitro cytotoxicity testing, is crucial to ensure that materials meet safety and performance requirements, instilling confidence in their clinical use. These assessments are essential for confirming the safety of wound healing patches for patient care.108,109
To assess the suitability of the nanofibrous scaffolds for wound healing, a cytotoxicity study was conducted using the MTT assay. This method evaluates cell viability and metabolic activity, offering valuable insights into the biocompatibility of the scaffolds. Mouse fibroblast 3T3 cells were used as a model system due to their relevance in mimicking skin cell behavior in response to biomaterials. Graph 6. Presents the results of the cytotoxicity tests, showing the response of 3T3 fibroblasts when cultured in both direct and indirect contact with the scaffolds. The results are highly promising, indicating minimal cytotoxic effects of the nanofibrous scaffolds on the cultured cells. Viability consistently exceeded the 70% threshold recommended by ISO 10993-5, confirming the biocompatibility of the scaffolds. The MTT assay in direct contact demonstrated high cell viability, affirming that the PEO/PCL/CS nanofibrous scaffolds are suitable for wound healing applications. These findings align with existing literature that emphasizes the importance of direct methods for evaluating the safety of wound healing patches.
110
Statistical analysis using the t test revealed no significant differences in cytotoxicity between scaffolds with varying PEO/PCL/CS ratios, further demonstrating the consistent biocompatibility and safety of these scaffolds. In vitro cytotoxicity assessed by the MTT assay of 3T3 cells seeded in (a) direct and (b) indirect contact with PEO/PCL/CS nanofibrous scaffolds after 1 day of culture. Error bars indicate SD. (n = 3).
Antibacterial efficacy of PEO/PCL/CS nanofibrous scaffolds
Incorporating antibacterial properties into nanofibrous wound healing patches is crucial for preventing infections that could impede the healing process. 111 These patches are designed to combat bacterial contamination on contact, thus reducing inflammation and the risk of systemic infection while supporting tissue regeneration.112,113 Evaluating their efficacy against common bacterial strains is essential to ensure effective infection prevention and wound healing.114,115
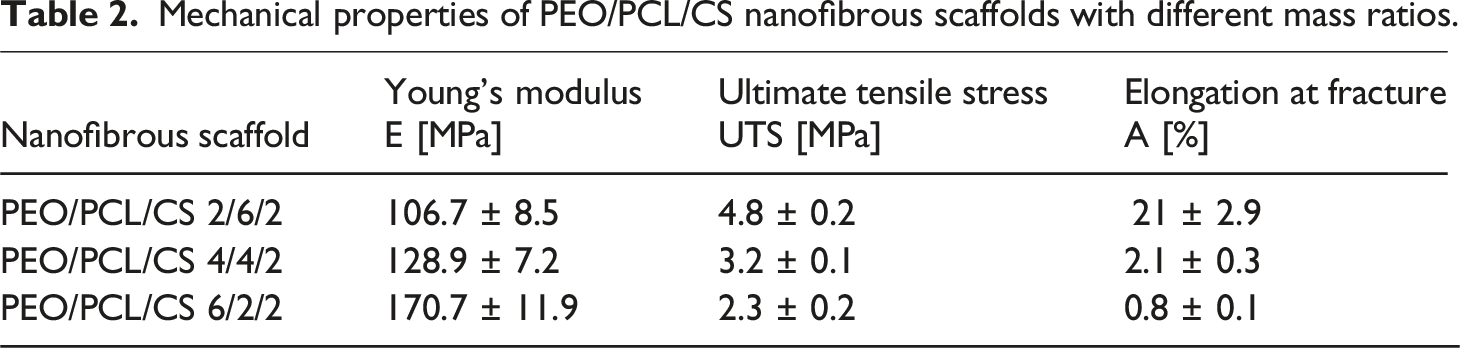
To assess the antibacterial efficacy of our nanofibrous scaffolds, we performed microbiological testing using the Kirby-Bauer method on E. coli and S. gallinarum. The size of the inhibition zone is indicative of antimicrobial activity, with a larger zone reflecting greater potency. The results revealed minimal antibacterial activity for the control scaffold without TET. In contrast, the nanofibrous scaffolds with immobilized TET demonstrated significant antibacterial activity, particularly against E. coli, with inhibition zones exceeding 20 mm. The inhibition zone was notably smaller for S. gallinarum, indicating antibacterial activity directly under or near the scaffolds, as shown in Figure 7. Photographs of inhibition zones (asterisks) around nanofibrous scaffolds with different compositions of PEO/PCL/CS, as determined by the Kirby-Bauer test: (a) Staphylococcus gallinarum and (b) Escherichia coli.
Of particular interest was the difference in antibacterial activity between samples AB and B, which differ in their post-TET immobilization treatment (see section Immobilization of tetracycline onto the nanofibrous scaffolds). The distinct immobilization methods were used to assess the antibacterial efficacy of nanofibrous scaffolds with surface-bound TET (sample_B) compared to those where TET was merely adsorbed onto the nanofibers (sample_AB). As shown in Graph 7, the results clearly indicate that despite the removal of TET by washing, some antibacterial activity persisted, albeit at a reduced level. This suggests that TET binding involves more than just simple surface adsorption. Specifically, for the nanofibrous scaffold with a PEO/PCL/CS mass ratio of 6/2/2, the difference in inhibition zones between samples AB and B was 7 mm, while for the PEO/PCL/CS 2/6/2 scaffold, the difference was 13 mm against E. coli. This underscores the effectiveness of the immobilization method in sustaining antibacterial effects, even after washing. Antibacterial activity of nanofibrous scaffolds with PEO/PCL/CS mass ratios of 6/2/2, 4/4/2, and 2/6/2 against Escherichia coli after 24 h, as assessed by the Kirby-Bauer test. Significant statistical differences were observed using the t test between nanofiber scaffolds with TET immobilized in different ways (samples_A and AB). Significant differences were also noted between scaffolds with varying mass ratios of PEO/PCL/CS. ** indicates p ≤ 0.01, and *** indicates p ≤ 0.001. Error bars represent SD. (n = 3).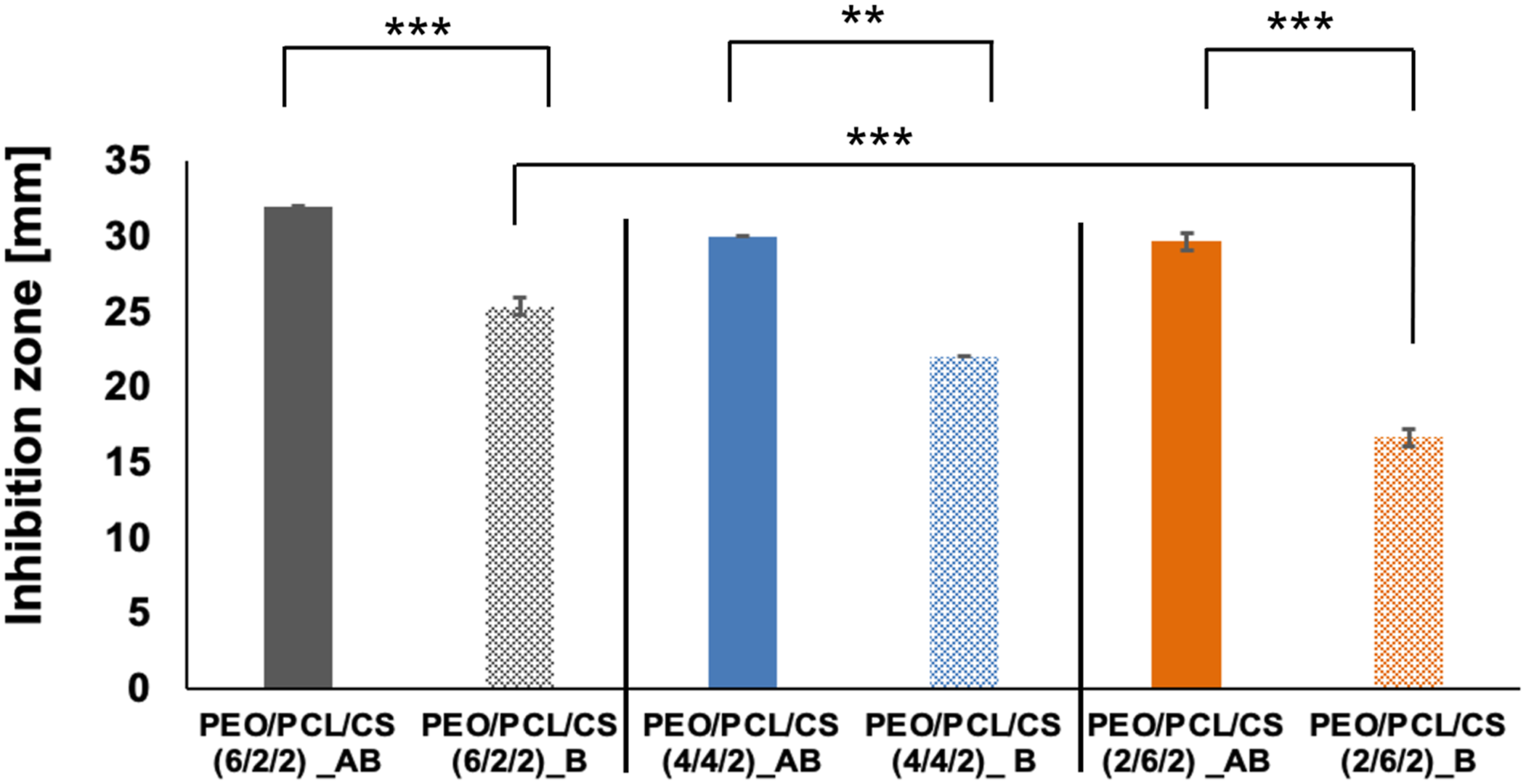
Moreover, statistical analysis using the t test revealed significant differences in antibacterial activity between scaffolds with different mass ratios of PEO/PCL/CS. Notably, scaffolds with higher PEO content exhibited better antibacterial efficacy compared to those with lower PEO content, albeit at the expense of PCL. TET, a multifunctional compound, contains chemical groups capable of interacting with counterparts in PEO, CS, and PCL. The observed differences in antibacterial activity can be attributed to the varying stability and strength of the bonds formed between TET’s chemical groups and the hydroxyl groups in PEO or the carbonyl groups inherent in PCL. Additionally, the density of attachment sites plays a crucial role in modulating antibacterial activity.
Discussion
In nanofibrous scaffolds composed of PEO/PCL/CS, the interactions between these polymers are crucial in determining the scaffold’s properties. Hydrogen bonding between PEO and CS enhances structural integrity by increasing intermolecular interactions. Additionally, the presence of amino groups in CS may facilitate ionic interactions with PEO, particularly in moist environments, further reinforcing the scaffold’s structural framework. While PCL is hydrophobic and does not participate in strong chemical interactions with the other components, its physical entanglement with the flexible chains of PEO significantly enhances the scaffold’s mechanical strength and flexibility. The combination of these materials through hydrogen bonding, ionic interactions, and physical entanglements results in a scaffold that effectively balances enhanced mechanical strength, structural stability, and bioactivity, making it highly suitable for wound healing applications.
Detailed comparison of PEO/PCL/CS nanofibrous scaffolds in this study versus previous studies.

Conclusion
In summary, this study introduces a versatile approach to fabricating PEO/PCL/CS nanofibrous scaffolds using needle-less electrospinning (Nanospider™ technology). Our findings demonstrate that combining PEO, PCL, and CS in nanofibrous scaffolds achieves an optimal balance of hydrophilicity, superior moisture vapor transmission rate, and adjustable hydrolytic degradation and mechanical properties. The ability to tailor scaffold properties—such as hydrolytic degradation, mechanical strength, and hydrophilicity—by adjusting the PEO/PCL ratio allows for specific adaptations based on wound type.
Our hypothesis that scaffold properties could be effectively adjusted by manipulating the PEO/PCL ratio was validated, demonstrating significant improvements over existing methods. Compared to other studies, our scaffolds offer enhanced customization capabilities, achieved using needleless electrospinning technique. Post-fabrication soaking in TET solutions further enhances antibacterial performance and allows for tailored adjustments. This approach is more efficient than producing multiple scaffold variants with different TET concentrations. Instead, scaffolds are immersed in a TET solution just before application, enabling direct adaptation to the patient’s wound needs. This method streamlines the customization process, providing on-demand adjustments and significantly improving the practicality and efficiency of wound care solutions.
Future work will focus on in vivo studies and detailed TET release profiles to fully assess the clinical applicability of our scaffolds. The successful transition from laboratory to industrial-scale production represents a significant milestone, with the potential to address challenges associated with chronic wounds and complex skin injuries, paving the way for advanced wound care products in clinical settings.
Footnotes
Acknowledgements
This work was supported by the Ministry of Education, Youth and Sports of the Czech Republic and the European Union - European Structural and Investment Funds in the frames of Operational Programme Research, Development and Education - project Hybrid Materials for Hierarchical> Structures (HyHi, Reg. No. CZ.02.1.01/0.0/0.0/16_019/0000843)
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministerstvo Skolstvi, Mladeze a Telovychovy; CZ.02.1.01/0.0/0.0/16_019/0000843.