Abstract
In the present work, hydroxypropyl cellulose and hematite (α-Fe2O3) nanoparticles (HPC:Fe2O3NPs) composites in various ratios (20:0.5, 20:1, 20:1.5, and 20:2 wt:wt) were synthesised. X-ray diffraction (XRD), scanning electron microscopy (SEM), FTIR spectroscopy, and thermal analysis (DSC, and TGA) were used to examine the structural configuration and thermal properties of the produced composites. The IR crystallinity indices (TCI, LOI, and HBI) were also computed. XRD patterns reveal a mixture of amorphous HPC polymer components and Fe2O3NPs which identifies the relationship between the characteristics of the structure. Distributions and dispersions of Fe2O3NPs on the surface of HPC are apparent in SEM. FTIR absorbance spectra reveal that as the quantity of Fe2O3NPs increased alterations in the intensity, area, and band width were found indicating a change in the molecular configuration of HPC. Variations in the shape, size, and shifts in melting temperature to higher temperatures were found by DSC and TGA when the amount of Fe2O3NPs is increased which could be due to interactions between HPC and Fe2O3NPs. The antibacterial activity of the nanocomposites was tested against Gram-positive Staphylococcus aureus and Gram-negative Escherichia coli bacteria. The composites have good antibacterial activity, confirmed a high potential of antibacterial activity. The findings demonstrated that HPC:Fe2O3NPs composites lead to technological applications in a variety of medical applications.
Keywords
Introduction
Biocompatibility and biodegradable polymeric and polymeric nanoparticles materials have attracted the interest of many researchers due to their physical and chemical properties.1,2 Due to their critical technological significance, these composites have been used in a wide range of advanced medical and industrial applications.3,4
Hydroxypropyl cellulose (HPC) is a cellulose derivative and is a water-soluble semi-synthetic nonionic cellulose ether polymer. 1 HPC is a versatile, ecologically friendly polymer that has been used in food, cosmetics, and biomedical product packaging.5,6 HPC is employed as a thickening, tablet binding agent, modified release agent, and polymer coating agent. 7 HPC has been used to examine a variety of medications for use in mucous membrane delivery systems and to separate DNA by electrophoresis and microchip separation. 8
In a variety of sectors, metallic oxide nanostructures offer a wide range of biomedicine, and pharmaceutical industries applications.9,10 Moreover, the rising number of bacteria resistant to an increasing number of antibiotics has sparked interest in the development of antimicrobial materials including metal or metal-oxide nanoparticles. 11 Iron oxide is a mineral component that can be found in nature and is becoming increasingly used for a wide range of applications. 12 Hematite (α-Fe2O3), maghemite (γ-Fe2O3), and various oxides in the beta (β-Fe2O3) and epsilon (ε-Fe2O3) forms are all examples of iron oxide, which has a wide range of structural and magnetic properties. 13 Hematite can be found in rocks and soils and is one of the most frequent forms of iron oxides. 13 Due to their wide range of uses, iron oxide nanoparticles (Fe2O3NPs) have received a lot of attention in recent years. 14 have proven to be effective against a wide range of organic and inorganic pollutants. 14 Fe2O3NPs have a wide range of applications in biomedicine, including:15,16 visualization and diagnostics; magnetic resonance imaging and computed tomography; positron emission tomography; cancer therapy with magnetic hyperthermia; and cell or molecular separation and the development of biosensors for immunoassays; neuro-electronic studies; and biomedical imaging. Hematite (α-Fe2O3) nanoparticles have attracted a lot of attention because of low cost, low toxicity, excellent chemical stability, and tunable optical and magnetic properties. 17
Because HPC is a polymer formed on the bases of the polysaccharide’s main chain, researchers can learn more about the interaction of organic and inorganic elements by studying transitional temperatures under the influence of nanoparticles.18–20 Furthermore, the thermal efficiency of the final composite materials is essential to ensure that the nanocomposite product is compatible with a variety of applications. 21 Abu Krorra et al. 22 investigated the effect of consolidation materials on transparent papers, comparing hydroxypropyl cellulose to nanomaterials like zinc oxide nanoparticles and nanocellulose, as well as a hybrid mixture of HPC and ZnONPs and HPC and nanocellulose to improve the properties of both materials in the hybrid mixture.
The goal of this study was to produce composites of hematite (α-Fe2O3) nanoparticles and hydroxypropyl cellulose (HPC) in order to improve the structural, thermal, and antibacterial properties of HPC while maintaining high absorbency, non-toxicity, and environmental requirements, as well as friendliness and cost-effectiveness, all of which are important in many technological applications. HPC:Fe2O3NPs composites in various ratios (20:0.5, 20:1, 20:1.5, and 20:2 wt:wt; i.e., 2.5, 5, 7.5, and 10 wt%) were synthesized. XRD, SEM, FTIR spectroscopy, DSC, and TGA techniques were used to examine the structural configuration and thermal properties of the produced composites. The TCI, LOI, and HBI indices were also calculated. The antibacterial activity of the composites was tested using the agar diffusion method against S. aureus (Gram-positive) and E. coli (Gram-negative) bacteria.
Materials and experimental techniques
Hydroxypropyl cellulose powder with MW 100,000 and particle sizes of 20mesh was supplied from Alfa Aesar, GmbH & Co., UK. Iron oxide–hematite nanopowder (>100 nm particle size), MW 159.688, and MP 1538°C was provided from Strem Chemicals, UK.
To synthesize HPC:Fe2O3NPs composites, 20 mg HPC powder was mixed with different weights of α-Fe2O3 nanopowder (0.5, 1, 1.5, and 2 mg). The raw materials were treated in a small mini-mill (Phillips PW 4018/00) for 15 min at 3400 r/min to generate homogenous composites. 23
To produce HPC and HPC:Fe2O3NPs composite films, solution-casting method was used. 24 To prepare HPC thin film, 20 mg of HPC powder was dissolved in 100 mL of deionized water. A magnetic stirrer was used in a 50°C water bath for 4 h to achieve complete dissolution. To produce HPC:Fe2O3NPs composite films; 0.5, 1, 1.5, and 2 mg of α-Fe2O3 nanopowder were added to the HPC complete solution individually and vigorously stirred for 2 h at 50°C to avoid agglomeration. The solutions were cast into stainless steel Petri dishes and baked for 24 h at 60°C to produce thin films of ∼0.1 mm thickness. The dried films from the plates were removed and stored at 25 ± 2°C in a chamber with 50% relative humidity.
A PW-1480 (Netherlands) X-Ray Diffractometer with nickel-filtered CuKα radiation (λ = 1.54056 Å) was used to analyze the crystalline structures of the composites in the range of 2θ from 5° to 70° at a speed of 2°/min. With the Match 3+PDF4-2015 programme analysis model, the operating conditions were 35 kV and 20 mA.
A Quanta 250 Field Emission Gun-Scanning Electron Microscope (FEG-SEM) operating at 30 kV and ×5000 magnification was used to examine the surfaces of the composite films. Double-sided adhesive tape was used for sticking samples on a copper stub.
Fourier transform infrared (FTIR) absorbance spectra of HPC, Fe2O3NPs, and HPC:Fe2O3NPs were recorded in the range 4000–400 cm−1 using the KBr pellet technique23,25 on a Nicolet-380 FT-IR Spectrophotometer (UK) with a resolution of 4 cm−1 and accuracy ± 1%.
A DSC-50 Differential Scanning Calorimeter and a TGA-50H Thermogravimetric Analyzer (Shimadzu, Kyoto, Japan) were used to examine the DSC and TGA of the composites. The thermograms were conducted in a nitrogen atmosphere at temperatures ranging 20–750°C, with a heating rate of 10°C/minute and a flow rate of 20 mL/min. In tubs of pure aluminum, about 6 mg per sample was hermetically sealed.
The HPC and HPC:Fe2O3NPs composite films were tested for antibacterial activity against Gram-positive Staphylococcus aureus (ATCC-12600) and Gram-negative Escherichia coli (ATCC-11775) bacteria using the agar well diffusion method (NCCLS 1997). The agar plates (diameter of 90 mm) containing S. aureus and E. coli bacteria were incubated for 24 h at 35–37°C. The antibacterial action was determined by measuring the inhibition zone diameter (in mm) surrounding the well. All tests were repeated three times before calculating the mean inhibition zone diameter.
Results and discussion
X-ray diffraction analysis was used to confirm the crystal formation of the prepared samples. Figure 1 depicts the XRD patterns of HPC, Fe2O3NPs, and HPC:Fe2O3NPs composites. The XRD pattern confirmed the coexistence of amorphous and crystalline areas in the HPC polymer.
26
Diffraction peaks were discovered at 2θ = 14.93°,16.55°,22.56° and 34.50°, corresponding to the (110), (200), and (040) planes, respectively.26,27 The pattern of Fe2O3NPs yielded nine crystalline diffraction peaks indexed as (012), (104), (110), (113), (024), (116), (018), (214), and (300) planes at 2θ = 24.24°,33.26°,35.74°,40.94°,49.52°,54.05°,57.57°, 62.43° and 64.07°, respectively (JCPDS no.: 24-0072). These peaks are indicative of α-Fe2O3 rhombohedral crystalline formations.
28
The XRD patterns of HPC:Fe2O3NPs composites revealed eight peaks at 2θ = 24.05°,33.13°,35.62°,40.81°,49.44°,54.05°,62.35° and 63.92°, and their intensities began to expand as the amount of Fe2O3NPs grew up to 10wt%. These unique peaks revealed that hematite was prominent in the pattern of the produced nanocomposites, as well as the presence of Fe2O3NPs at various concentrations, which could result in structural heterogeneity in the HPC network and Fe2O3NPs crystals within the polymeric matrix.
29
XRD patterns of HPC, Fe2O3NPs and HPC:Fe2O3NPs composites.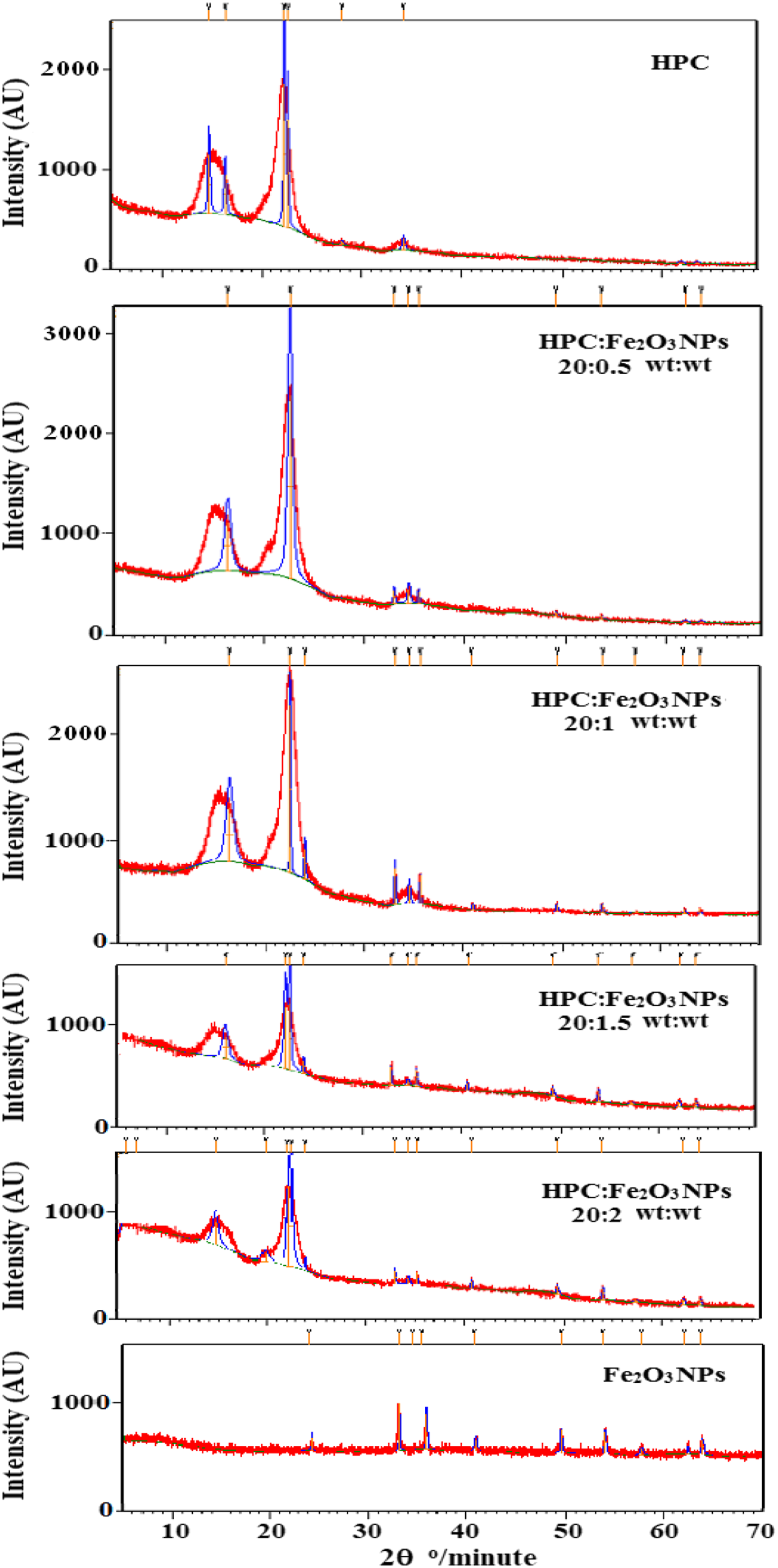
The crystallinity index (%CrI) is a statistic that has been calculated using the empirical relationship to assess relative crystallinity.24,27
I200 is the diffractogram height of the (200) plane as a fundamental peak and Iam is the height of the amorphous peak. The resulting patterns (Figure 1) show two peaks: a basic peak at 2θ = 22.55°–22.69° and a secondary peak at 2θ = 15.05°–16.55°. Figure 2 illustrates the fluctuation in %CrI of HPC and HPC:Fe2O3NPs composites. When the concentration of Fe2O3NPs in the composite was increased to 10wt%, the %CrI value decreased by ∼16% compared to the %CrI value of HPC. The results noticed structural changes in the polymer matrix where Fe2O3NPs were present.
30
In addition, the decrease in the %CrI of the composites could be explained by the change in the internal processes caused by the induced action of Fe2O3NPs on the HPC structure. As a result, there were inconsistencies in the HPC network setup. A histogram showing the variance in of the crystallinity index (%CrI) for HPC and HPC:Fe2O3NPs composites.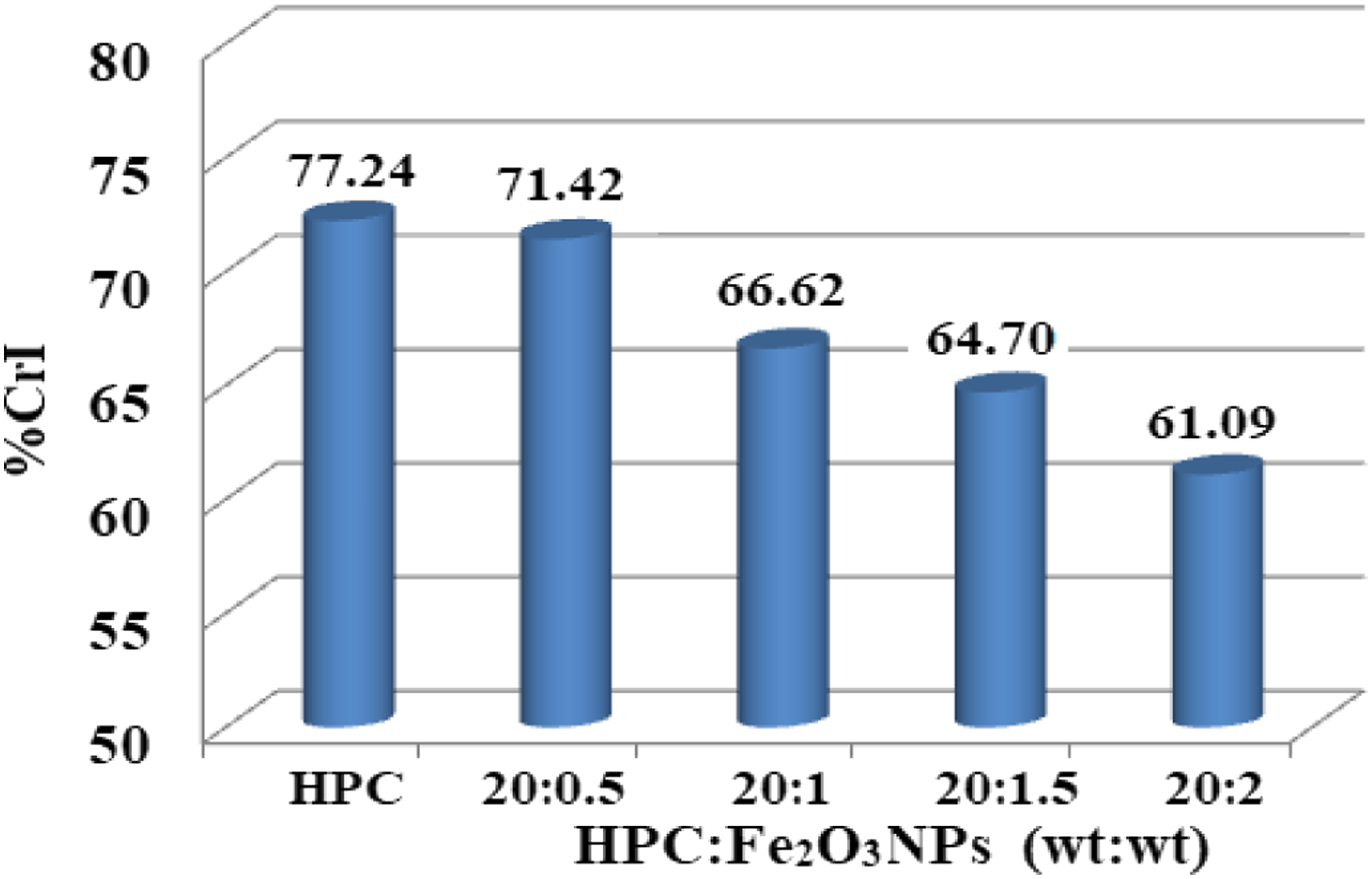
The surface morphologies of HPC and two selected HPC:Fe2O3NPs composite films (20:1, and 20:2 wt:wt) were examined using a SEM and the micrographs are shown in Figure 3. The HPC has a perfectly smooth, flat, compact, and uniform surface with no cracks or pores (Image A). Micrographs B and C of the composites 20:1 wt:wt (Image B) and 20:2 wt:wt (Image C) reveal that, nanoparticle fusion has roughened and heterogeneized the surface of HPC and show a lot of white aggregates. With increased Fe2O3NPs concentration, the HPC base increased the uneven distribution and aggregated particles (sizes and quantity). This observation might indicate that Fe2O3NPs were found incorporated into the HPC matrix, and that the size of the complex was limited to the nanoscale.
31
Furthermore, the homogenous structure indicates a good mixing potential between HPC and Fe2O3NPs, which were strongly coupled to the HPC matrix and affected the physical and chemical properties of HPC.
32
SEM micrographs of the surface of HPC and HPC:Fe2O3NPs composite films. Image A: HPC; Images B and C: 20:1 and 20:2 wt:wt composites (Magnification ×5000).
In the wavenumber range 4000–400 cm−1, Figure 4 represents the FTIR absorbance spectra of HPC, Fe2O3NPs, and their composites (HPC:Fe2O3NPs). From the HPC absorbance spectrum, it is seen that:2,33 broadness O–H peak detected at ∼3346 cm−1 corresponds to the polymeric connection of the hydrogen bound O–H stretching vibration, which could be attributed to the humidity absorbed by the KBr during the pellet formation; C–H stretching vibration identifies at ∼2901 cm−1 and a shoulder at ∼2760 cm−1; at about 1655 and 1648 cm−1, a splitting two peaks due to C–O stretching mode arise, and they may also appear from deformative water molecules; the peak at ∼1431 cm−1 is attributed to O–H, C–H, and –CH2 deformation; the peaks at about 1372 and 1327 cm−1 are caused by –CH2 wagging as well as C–H and O–H bending vibration modes; O–H bending and C–H wagging were given to the peak at ∼1272 cm−1; C–O asymmetric stretching vibration was responsible for the peak at ∼1164 cm−1; C–O stretching vibrations were ascribed to the peaks at around 1134, 1059, and 1035 cm−1; C–O deformation and –CH2 rocking caused the peak to occur at ∼897 cm−1; and O–H twisting was responsible for the peaks of 670, 617, and 560 cm−1. As shown in Figure 4, the spectrum of Fe2O3NPs displays substantial absorption peaks at 3448, 1111, 546, and 475 cm−1. The broad peak at ∼3448 cm−1 is attributed to hydroxyl group that bind to the iron surface and corresponded to the hydrogen-bonded OH stretching vibration, usually water; and Fe–O stretching and bending vibration modes are ascribed to the strong peaks at about 546 and 475 cm−1. The FTIR spectrum of Fe2O3NPs detected is similar to that found by Morsy et al.
34
In addition, the FTIR absorbance spectra of HPC:Fe2O3NPs composites revealed many similar peaks at the same locations as those recorded for pure HPC. The heights of the assigned groups' peaks at respective wavenumbers were used to indicate the variance in group band intensities for varying Fe2O3NPs concentrations. This is due to the presence of Fe2O3NPs on the HPC network, as well as the creation of hydrogen bonds between the Fe2O3NPs and HPC, which reduces the degree of intermolecular hydrogen bonding in HPC.
2
FTIR absorbance spectra of HPC, Fe2O3NPs, and HPC:Fe2O3NPs composites.
The IR crystallinity ratio was calculated using the total crystalline index (TCI).
35
TCI is proportional to the crystallinity degree of cellulose. At 1427 and 896 cm−1 the lateral order index (LOI) was determined, which correlate to the quantity of crystalline component and amorphous region in cellulose.
35
The hydrogen bond intensity (HBI) of cellulose was shown to be connected to the crystalline structure and degree of homogeneity of the molecules as evaluated by chain movement and bonding distance, i.e., crystallinity.
36
The following relationships were used to determine the TCI, LOI, and HBI of HPC and the produced composites.
37
IR crystallinity indices (TCI, LOI, and HBI) of HPC and HPC:Fe2O3NPs composites. The percentage changes in the IR crystallinity indices as well as the hydrogen bond energy (EH) for HPC:Fe2O3NPs composites.

The most often used techniques for thermal characterization of composites are DSC and TGA.26,38,39 DSC was employed in the temperature range of 20–600°C to determine the thermal changes of HPC, Fe2O3NPs, and the produced HPC:Fe2O3NPs composites and the thermograms are shown in Figure 6. Table 2 shows the differences in transition temperatures and heat related with fusion (ΔH). The thermograms for HPC and HPC:Fe2O3NPs composites exhibit a broad band in the region of 54–58°C, which represents the first order thermodynamic transition (Tth), with ΔH in the range of −14 to −21 J/g, indicating the miscibility of the systems.38,39 The second endothermic stage represents the phase transition temperature (Tm) resulting from the relaxation of the chain segments in the amorphous part of HPC. HPC decomposition temperatures were reported to be around 360 and 389°C, with ΔH of −120.41 and −114.62 J/g.
29
The decomposition peaks for HPC:Fe2O3NPs composites migrated toward higher temperature values when Fe2O3NPs were present and their concentration was increased (Table 2). The shape and area of the decomposition peaks were different, which could be attributed to a change in crystallinity and/or the arrangement of the molecules in the composites examined. A reduction in ΔH and a rise in Tm values were also observed. The interactions between HPC and Fe2O3NPs in the amorphous phase of HPC can cause a reduction in the crystalline structure by increasing the degree of cross-linking. Furthermore, disorder developed in the crystals, resulting in a decrease in phase change enthalpy.
40
The change obtained can be attributed to the variation in internal mechanisms that occurred as a result of the induced influence of different Fe2O3NPs compositions on the configuration of the HPC structure. DSC thermograms of HPC, Fe2O3NPs, and HPC:Fe2O3NPs composites. Transition temperatures and associated heat of fusion for HPC, and HPC:Fe2O3NPs composites.

Thermal gravimetric analysis was used to investigate the pyrolysis and stability of polymers.26,38,39 TGA and DTGA thermograms for HPC, Fe2O3NPs, and HPC:Fe2O3NPs composites throughout a temperature range of 20–750°C are shown in Figure 7. The first weight loss was detected in the range 20–150°C; the second weight loss was observed in the range 250–400°C; and the third weight loss was discovered in the range 400–750°C. The three stages of weight loss imply that there are numerous degrading processes occurring. The % weight losses, as well as the midpoint temperatures at first order thermodynamic transition and the second phase transition temperature are included in Table 3. According to the TGA thermograms, the first loss step was characterized by endothermic peaks in the range 47–52°C and was completed between 70 and 93°C with small % weight losses due to the evaporation of water physically adsorbed on the substances. The second loss stage, which begins above 250°C, represents the weight loss caused by the decomposition of the composite’s structure. Melting and degradation of the morphological components that make up the HPC structure formations are responsible for the second phase, which is characterized by an endothermic peak at ∼315°C. The values of the second step midpoint melting temperatures for HPC containing Fe2O3NPs were greater than those for pure HPC (Table 3). The increased melting temperatures of the composites could be attributed to the increased amount of Fe2O3NPs in the HPC network, and the enhancement of additional thermal stability. According to this data, Fe2O3NPs are better separated in the HPC matrix. After thermal deterioration, Figure 8 depicts the changes in total% weight loss of the investigated composites. It has been demonstrated that raising the proportion of Fe2O3NPs in the composite to 10 wt% reduces total% weight loss by ∼9%. This indicates that the composite 20:2 wt:wt has the highest residual% value (Residue% = 100 - Total% weight loss) at the end of the thermal disintegration, suggesting that the thermal stability of this nanocomposite has been strengthened and it is more resistant to fire hazards. When the amount of Fe2O3NPs added to the composites raised, the composite became more thermally stable, resulting in more Fe2O3NPs diffusion in the HPC network. TGA and DTGA thermograms of HPC, Fe2O3NPs, and HPC:Fe2O3NPs composites. TGA data for HPC, and HPC:Fe2O3NPs composites. Total % weight loss and residue % of HPC, Fe2O3 NPs, and HPC:Fe2O3 NPs composites.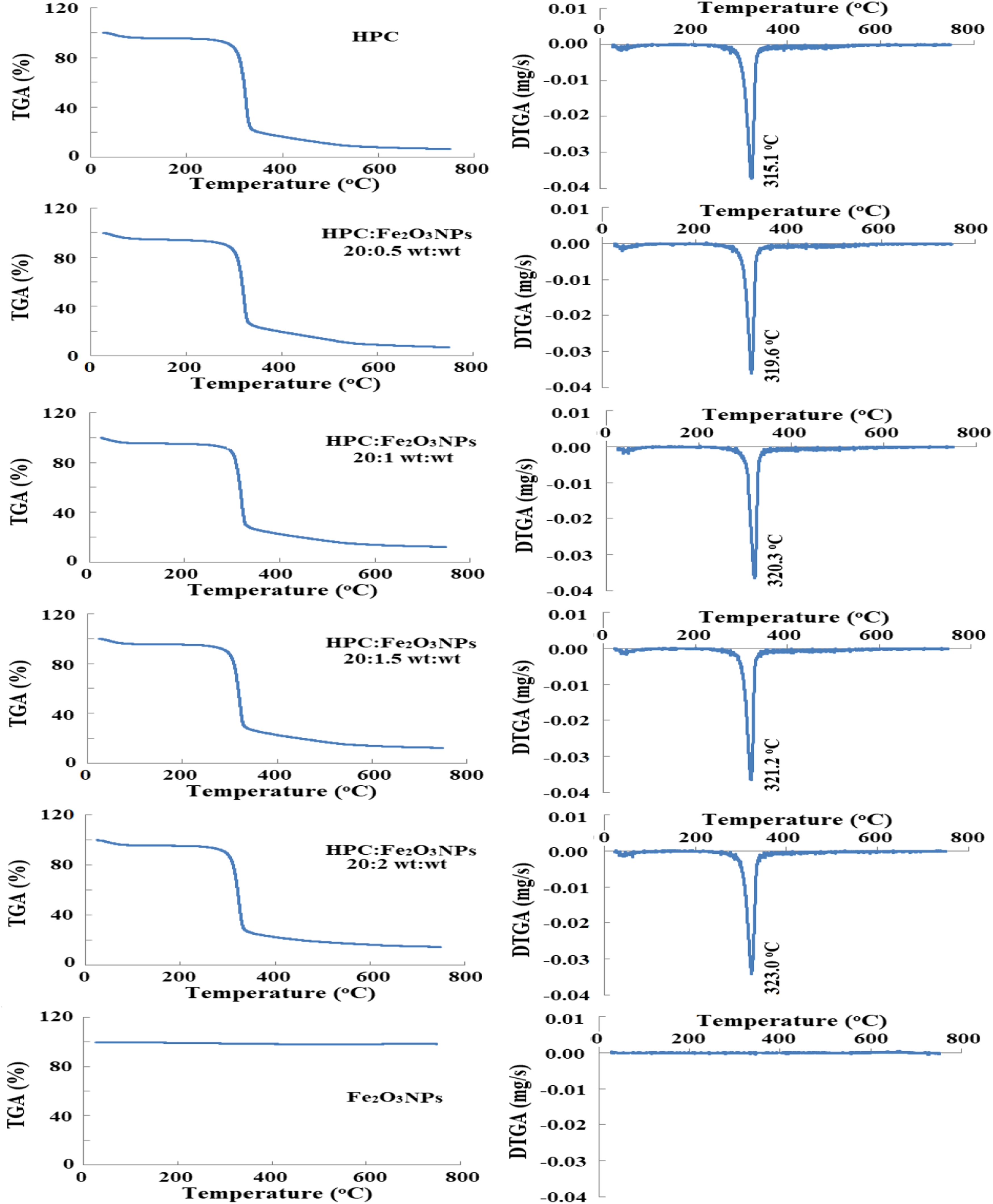


The differential thermogravimetry (DTGA) curves of the produced composites are also shown in Figure 7. Peaks in the 200–400°C range are shown curves, which were most likely caused by HPC pyrolysis. The degradation temperatures of HPC and HPC:Fe2O3NPs composites are estimated using the maximum peaks of DTGA, as observed in the figure. The addition of Fe2O3NPs in the composite enhanced the decomposition values observed, implying that the HPC process has deteriorated. This implies that during degradation, heat is emitted near the end of the temperature curves. The infrared active groups of Fe2O3NPs composites are linked to the change in observed melting temperature values. This could be due to the presence of Fe2O3NPs on the HPC matrix and the formation of a hydrogen bond between the Fe2O3NPs and HPC, which reduces the number of hydrogen bonding molecules in HPC. 38
The measurement of the zones free of bacterial growth was used to assess the materials' potential antibacterial action.
41
The antibacterial activity of HPC:Fe2O3NPs composites against Gram-positive S. aureus and Gram-negative E. coli bacteria were visualized using the well-agar diffusion method in Figures 9 and 10. The results show that composites containing 2.5–10wt% Fe2O3NPs have well-formed inhibitory zones surrounding them. In the instance of HPC (Figure 9), no inhibitory zones are seen.
32
The diameter of the inhibition zones for S. aureus and E. coli increased when the quantity of Fe2O3NPs was increased showing a tendency to improve antibacterial efficacy (Figure 10). The results demonstrate that Gram-positive bacteria like S. aureus are more sensitive than those of Gram-negative bacteria E. coli.
42
The current findings shows that the investigated nanocomposites inhibited S. aureus growth more than E. coli growth, most likely due to changes in cell wall structure, as the size of a bacteria’s cell wall impacts its resistance.
43
According to many studies,42–44 oxide nanoparticles show antibacterial efficacy against Gram-positive (including S. aureus) and Gram-negative (including E. coli) bacteria. The HPC:Fe2O3NPs composites produced are potentially active against bacteria, suggesting that such Fe2O3NPs could be employed as therapeutic tools. Photographs of plates showing the antimicrobial activity by agar diffusion method of HPC, and HPC:Fe2O3NPs composites against S. aureus (Gram-positive) and E. coli (Gram-negative) bacteria. A histogram showing the variance in the inhibition zone diameters for HPC:Fe2O3NPs composites of both S. aureus (Gram-positive) and E. coli (Gram-negative) bacteria.

Conclusions
Structural characteristics of HPC, Fe2O3NPs, and their composites (HPC:Fe2O3NPs) were studied using XRD, SEM, FTIR, IR crystallinity indices, and thermal investigations. According to XRD patterns, the crystallinity index falls as the concentration of Fe2O3NPs increases, indicating structural changes in the HPC matrix as a result of Fe2O3NPs proliferation. According to the SEM micrographs, the Fe2O3NPs were arranged in an uneven distribution and particle aggregation, which could be owing to the lack of attractive forces between HPC and Fe2O3NPs. The chemical bonding and molecular configuration of HPC were detected by the variation in the intensity of absorbance as the Fe2O3NPs concentration was increased, as evidenced by the FTIR absorbance spectra. The shape and extent of the degradation endothermic peaks varied according on the DSC values. According to TGA and DTGA investigations, the composites decomposed at about 315–323°C. As the amount of Fe2O3NPs increased, the total% weight loss decreased significantly. These findings reveal that HPC:Fe2O3NPs composites produced were more thermally stable, fire-resistant, and had a higher dispersion of Fe2O3NPs in the HPC network. The composites produced show substantial antibacterial activity against S. aureus (Gram-positive) and E. coli (Gram-negative) bacteria. According to the findings, increases in Fe2O3NPs concentrations up to 10wt% result in greater inhibition zones around both materials. The results suggest that the HPC:Fe2O3NPs composites have antibacterial activity against S. aureus and E. coli bacteria, demonstrating their ability to restrict bacterial development at the implant site. Finally, Fe2O3NPs-biopolymer composites have hybrid properties of both HPC and Fe2O3NPs which could find potential applications in catalysis, nanobiotechnology, and therapeutics.
Footnotes
Author’s contributions
All authors contributed equally: Validation, writing, reviewing and editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.



