Abstract
The goal of this study is to improve the qualities of Saudi cotton fabrics to be used in a variety of industrial and medicinal applications. Cotton fabrics were treated by dipping in chitosan/ZnO-NPs solutions with various ZnO-NPs concentrations. XRD and SEM of crystal and morphological structure, thermal analysis (TG and DSC) of fabric stability for heat assessment, were used to characterize the fabrics. The produced textiles were tested for antimicrobial activity against Gram-positive Staphylococcus aureus, Gram-negative Escherichia coli (bacteria), and Aspergillus niger, Candida albicans (fungus) using agar diffusion method. According to the XRD data, the fabrics treated with chitosan and by increasing the concentration of ZnO-NPs had increased crystallinity. The successful deposition and strong binding of chitosan and ZnO-NPs to the fabric surface, and the obvious change in fabric morphology, were confirmed by SEM micrographs. The pyrolysis peak positions were changed to a higher temperature. The creation of the inhibitory zone indicated that the treated fabrics had bactericidal effects. The results could be useful in an open innovation approach to improving the qualities of treated textiles that point to their use in medical applications.
Keywords
Introduction
Rcently, one of the most active fields of academic study and the textile industry is the preparation and fixing of nanoparticles on fabric. 1 Products containing nanomaterials can be used in many industrial, medical and military applications. 2 Nanomaterials have been used in textiles because of their outstanding and innovative physical and chemical qualities.3,4 Nanomaterials are supposed to increase new functionality to medical fabrics, such as preventing the transmission of bacteria. 5 Many important works on natural fabric-reinforced composites using different methods have been reported.4,6 One approach for producing highly active surfaces with UV-blocking, antibacterial, and self-cleaning qualities is to coat the surface of textiles, apparel, and fabrics for footwear with nanoparticles. 7 Due to their large surface area, which allows for a lot of interaction with microbes, many types of nanoparticles have received much attention for their potential antimicrobial effects. 8 Zinc oxide nanoparticles are one of nanoparticle that can be employed in a variety of medical and fabric applications, including aesthetics, decontamination, and medical protecting garments. 9 ZnO-NPs were discovered to be particularly appealing due to their extraordinary potential for use as UV blockers and photocatalysts in solar cells, screens, photodetectors, and sun protection. ZnO-NPs offer a number of advantages over certain nanoparticles such as SiO2, MgO, TiO2, and CaO. 10 Several methodologies for integrating nanoparticles into textile materials have been proposed (one-step processes and multi-step processes). 11 On other hand, ZnO-NPs can be synthesized by several chemical methods such as chemical precipitation, hydrothermal method, pyrolysis, chemical vapor deposition, etc. These methods have some drawbacks that lead to the adsorption of some toxic chemicals to the surface which may have negative effects in medical applications. 12 Also; ZnO-NPs may have adverse effects on several organs including lungs, kidneys, liver, central nervous system, reproductive system, and fetal development in animal models. However, the toxicity induced by ZnO-NPs is multifactorial, and it is still unknown how toxic ZnO-NPs are to these organs. 13 Many researchers have studied the properties of nano-ZnO on textiles and determined that ZnO-NPs have a better antimicrobial impact than other metal oxides.5,10,14,15
Fabrics can provide a particularly favorable environment for the growth and proliferation of microorganisms such as bacteria and fungi, which, due to their inherent qualities, are often found in the environment.5,16,17 As a result, fabrics that prevent microbe growth are promising raw materials for sportswear, T-shirts, hosiery, shoe wall cladding, drapes, and upholstery for couches, chairs, and bed sheets. Tissue with antibacterial characteristics and biocides has been a major difficulty in the fight against harmful pathogens. 18 Cotton is a single-celled plant cellulose fabric that is utilized in the textile industry as a natural biomaterial. Cotton fabrics are widely used because of its great qualities, such as regenerating, degradability, softness, skin sensitivity, and hygroscopicity. 19 Also, cotton fabrics can provide an ideal environment for microorganism growth due to their enormous surface area and capacity to keep it moist.20,21
Chitosan is a linear polysaccharide composed of
In the current study chitosan and ZnO-NPs were introduced into pure Saudi cotton fabrics utilizing a novel, quick, and low-cost approach. The fabrics were immersed in chitosan and chitosan/ZnO-NPs solutions of four concentrations (10, 20, 30 and 40wt% of ZnO-NPs with respect to chitosan content). X-ray diffraction, scanning electron microscopy and thermal analysis techniques were used to study the effect of immersion on fabrics. Antimicrobial effectiveness against Gram-positive Staphylococcus aureus, Gram-negative Escherichia coli (bacteria), and Aspergillus niger, Candida albicans (fungus) was assessed using the agar diffusion method.
Materials and experimental techniques
Materials
Commercially available Saudi cotton fabric (150 g/m2) was used. The fabric was washed at 60°C for 15 min with a solution containing 2 g/L non-ionic detergent in a liquid ratio of 1:50, then washed with distilled water and dried in air.
Chitosan (Ch) crab shells 99.8%, very viscous poly(1,4-D-glucopyranosamine); 2-Amino-2-deoxy-(14)-D-glucopyranan, a deacetylated chitin with a particle size >100 mesh and MW 900,000D (CAS No.9012-76-4); powdered ZnO nanoparticles with a particle size of less than 100 nm (catalog No.544906); and acetic acid, were procured from Sigma-Aldrich (England). Chemicals were used without any additional purification or distillation.
Preparation of Ch/ZnO-NPs solutions
To make chitosan solution, 1g of chitosan powder was dissolved in 100 mL of a 1% w/v aqueous solution with 0.1 M acetic acid at room temperature. The mixture was stirred for 8 h at 600 rpm using a magnetic stirrer, and the insoluble masses were filtered out.
To prepare Ch/ZnO-NPs solutions, four weights of ZnO-nanopowders were added to the prepared chitosan solution: 0.1,0.2,0.3 and 0.4 g (concentrations of 10, 20, 30 and 40wt% with respect to the chitosan content). The mixed solutions were stirred continuously for 8 h at 600 rpm. Filtration was used to remove insoluble masses.
Functionalized of Ch/ZnO-NPs on cotton fabrics
Clean, dry cotton fabrics (CF) for each measurement were cut and soaked separately in the five prepared solutions at room temperature for 15 min. The fabrics were then taken from the solutions, squeezed out and dry at room temperature. Cotton fabrics treated with chitosan (CF/Ch) were labeled S1, and treated with chitosan-ZnO-NPs (CF/Ch/ZnO-NPs) were respectively labeled S2, S3, S4, and S5, at concentrations of 10, 20, 30 and 40wt% ZnO-NPs with respect to the chitosan content.
Physicochemical characterizations
The crystal structures of the untreated and treated cotton fabrics were investigated using an X-ray diffractometer from PANalytical Empyrean (Netherlands) with CuKα (λ = 1.5406 Å) radiation operating at 45 kV and 30 mA at a scanning speed of 5°/min over a range of 2θ from 5–75o. The percentage crystallinity index (%CrI) was calculated to describe the degree of crystallinity as:29-31
I002 is the primary diffraction peak intensity of the main lattice (002) at 2θ ≈ 23°, while Iam is the amorphous portion of cellulose diffraction peak intensity at 2θ ≈ 17°.
The surface morphology of cotton fabrics was studied using a scanning electron microscope Quanta FEG-250 (Netherlands) at 20 kV at magnifications of 400, 3000× and 6000×. The fabrics were mounted on an aluminium stand and sputtered with a thin gold layer.
Thermal stability of cotton fabrics was performed using a Thermogravimetric Analyzer TGA-50H and a Differential Scanning Calorimeter DSC-50 (Shimadzu, Japan) in an alumina cell at temperatures from 25–980°C in a nitrogen gas atmosphere at a flow rate of 20 mL/min and a heating rate of 10°C/min. 20 mg of each sample was examined for thermal analysis.
Antimicrobial activity tests
Untreated and treated cotton fabrics were tested for antimicrobial efficacy against Gram-positive Staphylococcus aureus (ATCC-12600) and Gram-negative Escherichia coli (ATCC-11775) as bacteria; Aspergillus flavus (ATCC-9643) and Candida albicans (ATCC-7102) as fungal pathogens. Antimicrobial activity was assessed using the agar well diffusion method.32,33 The medium was autoclaved at 120°C to sterilize it. Each sterile Petri plate received ≈30 mL of agar media with the required bacteria strains. The volume of the microbial inoculum is dispersed over the entire agar surface to inoculate the surface of the agar plate. Using a sterile cork drill, a well with a diameter of 6–8 mm is perforated aseptically and a volume (20–100 mL) of the antimicrobial agent of the necessary concentration is put into the well. The agar plates containing bacteria and filamentous fungi were incubated at 30–37°C for 24–48 h. It turns out that a zone is formed around the wells. The ability of microorganisms to resist the studied diseases was examined after 21 days. The antimicrobial effect was assessed by using a caliper to measure the inhibition zone diameter (IZD) in millimetres. All tests were performed in aseptic condition and repeated three times, with the mean IZD being computed. A standard 6 mm diameter tablet containing fibrofloxacin and ketoconazole (50 g/tablet) was used as an antibacterial control and the IZD was recorded.
Results and discussions
The schematic diagram of the untreated and treated cotton fabrics that were immersed in chitosan and ZnO-NPs solutions is illustrated in Figure 1. A schematic representation of the experimental techniques.
X-ray diffraction analysis
Figure 2 shows XRD patterns of untreated and treated cotton fabrics (CF/Ch/ZnO-NPs). XRD pattern of the untreated fabric displays strong diffraction peaks at 2θ = 15.14,16.90,22.96, and 34.86o, which correlate to the (101) XRD patterns of untreated and treated cotton fabrics (CF/Ch; S1 and CF/Ch/ZnO-NPs; S1-S5).
Figure 3 shows the percentage of crystallinity index (%CrI) determined from Equation (1). The cotton fabric treated with 40wt% ZnO-NPs was expected to have a greater %CrI (∼10%) than the untreated fabric, however this did not occur in other treated fabrics due to the harsh acid decomposition conditions.
30
A histogram showing the variance in %CrI for untreated and treated cotton fabrics (CF/Ch; S1 and CF/Ch/ZnO-NPs; S1-S5).
Surface morphological structure analysis
The SEM morphological change in the treated and untreated cotton fabrics was illustrated in Figure 4. It was observed from the images that there is a clear difference between the treated and untreated cotton fabrics in terms of the morphological shape of the fiber surfaces. In the SEM micrographs of untreated fabric, a rod-like structure with exceptionally smooth outer surfaces was detected, and each rod-like fabric was easily identified. In addition, SEM reveals the heterogeneity of the cotton fibers and a twisted ribbon-like structure that begins with the cellulose fibers.20,36 After processing the fabrics; it is evident from Figure 4 that the surface morphologies of the fabrics change dramatically. Moreover, the treated fabrics contain numerous ZnO-NPs structures on their fiber surface and the micrographs reveal sufficient amount of chitosan and ZnO-NPs deposition on the surface of the fabrics. The treated cotton fabrics were coarser with an elliptical-chitosan layer permeating the fabric and ZnO-NPs of various shapes and diameters dispersed across the fabric surfaces. When comparing the treated fabrics with the untreated one, the amount of chitosan polymer covering the fabrics and the amount of nanoparticles deposited on the surfaces of the fabrics increased with the increase in the concentration of ZnO-NPs in the solution. Also, the results show that chitosan and ZnO-NPs adhered to the surface of treated fabrics successfully and securely. Finally, immersing the cotton fabric in the solutions resulted in the formation of ZnO-nanostructures with a more homogeneous size and shape and better dispersion on the surface of the cotton fabrics. Moreover, cotton fabrics treated with chitosan and ZnO-NPs modified the morphological structure of the fabric and added a number of functions, including antioxidant and antibacterial capabilities. The obtained SEM micrographs were in agreement with those previously reported.19,23,36–39 SEM micrographs of untreated and treated cotton fabrics.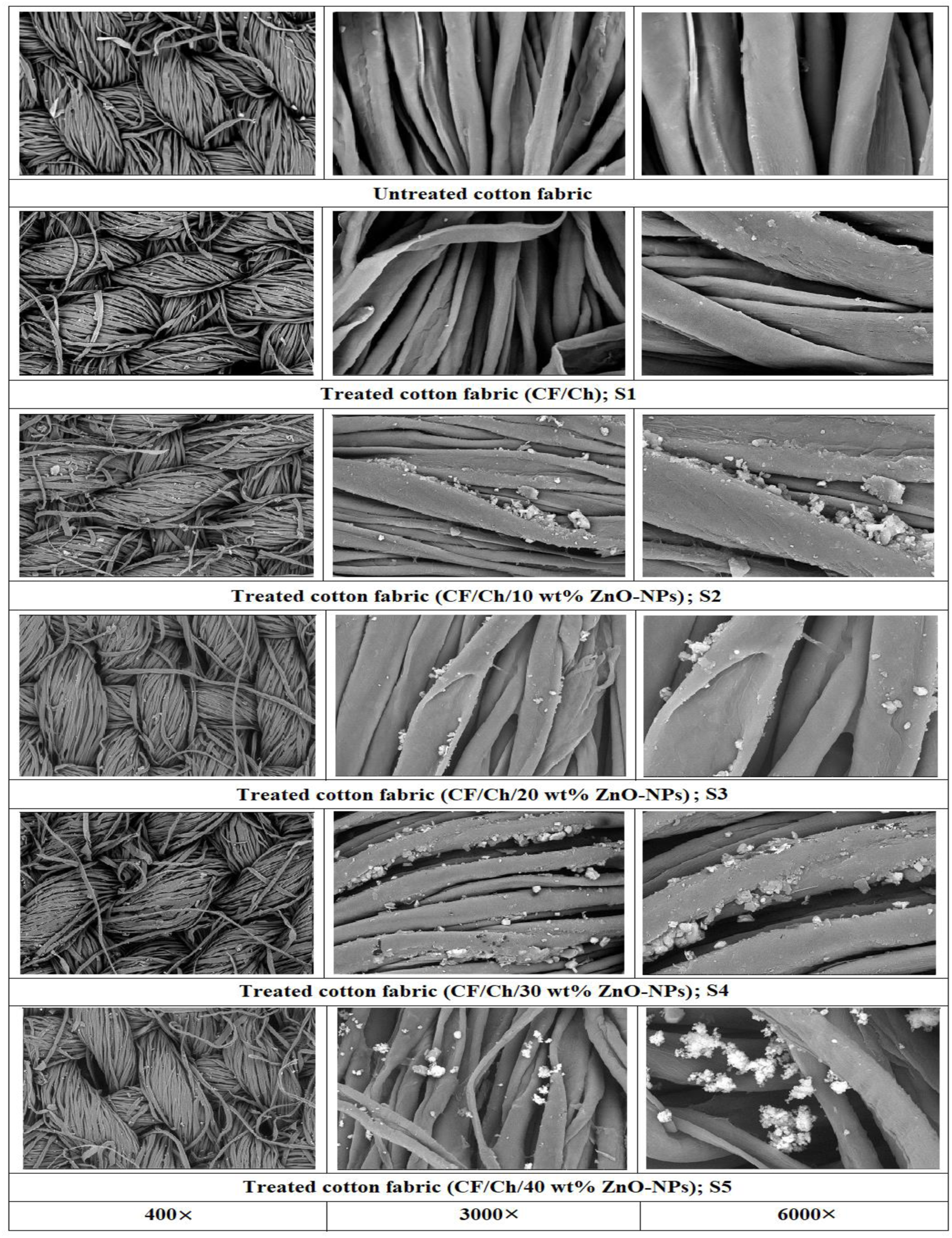
Thermal analysis
Thermogravimetric analysis (TG and DTG) for untreated and treated cotton fabrics (CF/Ch/ZnO-NPs; samples S1-S5) are shown in Figure 5. As seen by the TG thermograms, weight loss for cotton fabrics happens in two separate stages of disintegration. Water absorption from the polysaccharide structure caused the first stage of decomposition to occur below 100°C.
30
The first step was followed by a relatively constant weight from 100 to 240°C. The thermograms revealed that all fabrics behaved similarly, with the second decomposition stage starting at ≈240°C and the final decomposition temperature hovering around 500°C. Cotton fabrics' thermal stability improves marginally following processing at temperatures between 240 and 500°C, which could be related to cellulose disintegration and depolymerization.
40
The most significant weight loss was found at ≈242°C for the untreated fabric and at around 273, 262, 259, 257 and 253°C for the treated fabrics (samples: S1, S2, S3, S4, and S5, respectively). This means that treated cotton fabrics are more thermally stable than untreated fabric and have a smaller organized cellulose area. TG and DTG thermograms of untreated and treated cotton fabrics (CF/Ch: S1 and CF/Ch/ZnO-NPs: S1-S5).
The major decomposition stage is produced in the range of 242–393°C, resulting in a weight loss of ∼58% for untreated fabric; and 273–392°C, resulting in a weight loss of ∼46% (S1); 262–376°C, resulting in a weight loss of ∼49% (S2); 269–401°C, resulting in a weight loss of ∼47% (S3); 257–403°C, resulting in weight loss of ∼44% (S4); and 253–420°C, resulting in a weight loss of ∼48% (S5). In this decomposition stage, due to the breaking of the glycosidic linkages of cellulose, the degree of polymerization was lowered and the organic fraction disintegrated, suggesting the removal of CO2 and H2O as well as a variety of hydrocarbon derivatives. Fabrics begin pyrolytic at a slower rate of weight loss beyond 500 to 980°C, as all volatile content is extracted, causing residual charcoal to develop in the fabric. The existence of ZnO-NPs and the inorganic component in the fabric was revealed by unobserved decomposition over 600°C. The outcomes were consistent with those previously reported.41–43 The peak temperature values corresponding to the second decomposition stage were detected using DTG curves (Figure 5). After treatment with chitosan and 40wt% ZnO-NPs (S5), the peak position shifted by ∼5% to a high temperature, from ∼343 to ∼357°C, indicating an increase in crystal size. This trend shows that the treated fabrics have a higher thermal stability, which could be owing to an increase in the number of hydrogen bonds between the cellulose chains, which could result in a higher cellulose pyrolysis temperature. Furthermore, each fabric has one transition peak, indicating that the fabrics contain only one type of crystal (cellulose crystal Ib), as determined by the XRD data.
30
The overall percentage weight loss of treated samples S4 and S5 reduced near the end of the thermal decompositions (Figure 6). The data also show that following the thermogravimetric test, the residual percentage of these treated fabrics increased, indicating that they are more stable than the untreated fabric. Histograms showing the variation in total % weight loss and residue % data for untreated and treated cotton fabrics (CF/Ch; S1 and CF/Ch/ZnO-NPs; S1-S5).
DSC thermograms for untreated and treated cotton fabrics are shown in Figure 7. The curves did not identify any endothermic peak beyond 100°C and up to 300°C, indicating that no crystallization or phase change occurred during the heating and that the fabric was thermally stable in the appropriate temperature range.
44
The DSC thermogram of the untreated fabric shows a single prominent endothermic transition at ≈339°C, which is attributed to the thermal degradation of the cotton fabric’s cellulose content. The primary endothermic peak for the treated fabrics migrated towards higher temperatures, approximately 336, 338, 347, 350, and 357°C (samples: S1, S2, S3, S4, and S5, respectively), suggesting higher thermal stability. For cotton fabric treated with chitosan and 40wt% ZnO-NPs, a maximum displacement of ∼5% was recorded. Changes in DSC thermograms show the existence of ZnO-NPs with chitosan over the treated fabrics. The temperature change is due to non-cellulosic components and lignin cross-linking. This suggests that cellulose particles have a higher thermal molecular mobility and the cotton fabric has a higher thermal stability following processing.
43
DSC thermograms of untreated and treated cotton fabrics (CF/Ch; S1 and CF/Ch/ZnO-NPs; S1-S5).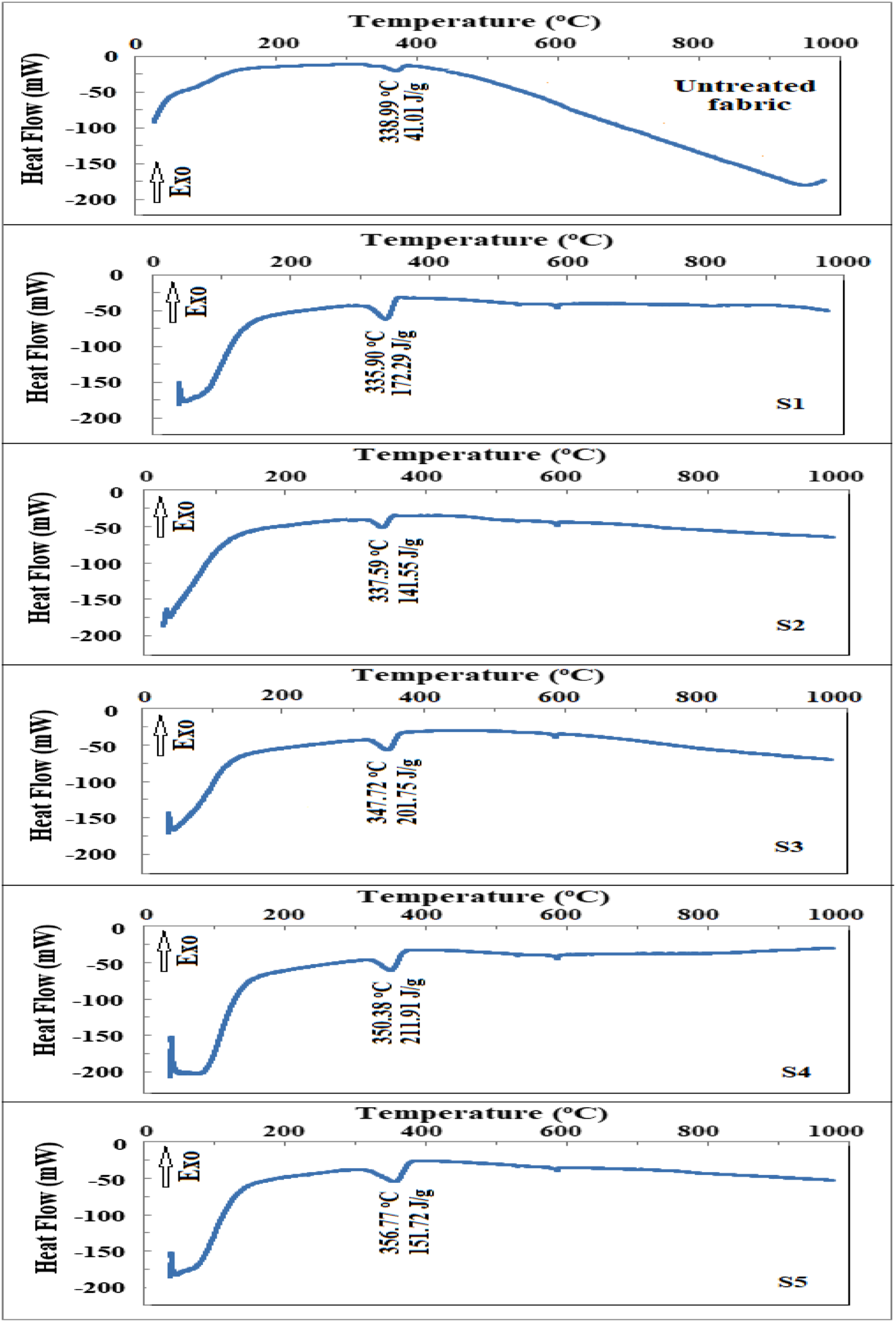
Antimicrobial activity
Using the well-agar diffusion method, the antimicrobial activities of untreated and treated cotton fabrics against Gram-positive S. aureus, Gram-negative E. coli (bacteria), A. Flavus, and C. albicans (fungus) pathogens were visualized in Figure 8. The result of untreated cotton fabric reveals no antibacterial properties.1,9,45–47 Also, treated cotton fabrics did not show antifungal action against A. flavus and C. albicans. Photographs of plates showing the antimicrobial activity of untreated and treated cotton fabrics (CF/Ch; S1 and CF/Ch/ZnO-NPs; S1-S5). against various pathogenic microorganisms.
Figure 9 shows the mean IZD values of treated cotton fabrics. The treated fabrics show distinct and considerable zones of inhibition against S. aureus and E. coli. When the concentration of ZnO-NPs was raised, the IZD increased, indicating a tendency to improve antibacterial efficacy.
47
The results reveal that the treated fabrics had a larger inhibition area for E. coli (Gram-negative) growth than for S. aureus (Gram-positive) culture, probably due to changes in cell wall structure because the size of a cell wall of bacteria determines its resistance.
23
As a surface layer, S. aureus has a 20–80 nm thick wall, but E. coli has a thin layer less than 10 nm thick.
48
The fabric with the largest IZD around it after being dipped in a solution containing chitosan and 40wt% ZnO-NPs (S5) had a better antibacterial action. The results show that sample S5 had antibacterial IZD of 25 ± 0.35 and 26 ± 0.42 mm against S. aureus and E. coli, respectively. The researchers observed a number of mechanisms, which include:48,49 (1) due to the presence of nanoparticles, active oxygen species are produced, (2) due to electrostatic forces, particle adhesion on the surface of bacteria damages the membranous cell wall, (3) cell membrane penetration, and (4) the interaction of active oxygen species with the cellular membrane. Furthermore, Gouda et al.
50
reported that, nanoparticle permeability is mostly caused by oxidative strain and causes of cell membrane dispersing. The formation of the inhibitory zone then clearly suggests that the combined fabric with chitosan and ZnO-NPs has antiseptic activity, which could be related to the impact of ZnO-NPs and increasing their concentration on the membrane of the organism.
1
The results are consistent with those previously reported.1,43,47 Histograms showing the variance in IZD for treated cotton fabrics (CF/Ch; S1 and CF/Ch/ZnO-NPs; S1-S5).of both S. aureus and E. coli bacteria.
Conclusions
To improve the multifunctional capabilities of the cotton fabrics, chitosan and ZnO-NPs were applied to the surface of cotton fabrics using a solution immersion approach. XRD analysis confirmed the formations of ZnO-NPs crystal phases. In comparison to the untreated fabric, the crystallization of the fabric improved after treatment with chitosan and ZnO-NPs (samples: S4 and S5), indicating a reorganization of the cellulose structure. The deposition of nanoparticles on the fabrics’ surface was shown by SEM micrographs. SEM for treated cotton fabrics modified the morphological structure of fabrics and added a number of activities, including antibacterial and antioxidant capabilities. Changes in the TG and DSC thermograms indicate the existence of ZnO-NPs with chitosan over the treated fabrics. According to the DSC results, the position of the main endothermic peak migrated towards higher temperatures, ≈350 and ≈357°C for samples S4 and S5, respectively, indicating that these fabrics are more thermally stable than the untreated fabric. Cotton fabrics treated with chitosan and ZnO-NPs have significantly improved thermal stability. All XRD, SEM, TG, DTG, and DSC data showed that chitosan and ZnO-NPs were well dispersed and fixed on the surface of cotton fabrics.
The inhibitory zones were used to demonstrate the antibacterial activity of cotton fabrics against Gram-positive S. aureus and Gram-negative E. coli bacteria. Antibacterial activity against E. coli was higher than that against S. aureus in the treated textiles. More effective antibacterial activities were observed in fabric coated with chitosan and 40wt% ZnO-NPs. The antibacterial activity of ZnO-nanostructures produced on cotton fabrics showed that they have the potential to be used as an antimicrobial agent.
Finally, the results reported in could be relevant in the textile industry, where both chitosan and biosynthetic ZnO-NPs are being used to improve the qualities of processed textiles in an open creative method. To supply Saudi cotton fabrics for industrial and medicinal research, much work remains to be done. These fabrics coated with chitosan and ZnO-NPs is a new modified fabric that will be used in a number of applications in the future, including wound treatment, bed liners, virus masks, medical bandages, and storage containers to be widely adopted.
Footnotes
Author’s contributions
All authors contributed equally: Validation, writing, reviewing and editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the University of Jeddah, Jeddah, Saudi Arabia, under grant No.(UJ-20-129-DR).
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.

