Abstract
In this work, γ-aminopropyltriethoxy silane (KH550) and γ-(2,3-epoxypropoxy)propyltrimethoxy silane (KH560) were used to modify BN and Al2O3, and described as KH550-BN and KH560-Al2O3 with amino and epoxy group on surface, respectively. KH550-BN and KH560-Al2O3 were filled in polypropylene (PP) matrix by twin-screw extruder. The thermal conductivity of composites increased with increasing catalyst dosage, material residence time in barrel and homogenization section temperature of twin-screw extruder. Hybrid fillers with semi core-shell structure were formed by KH550-BN
Introduction
Thermally conducting, electrically insulating polymer composites are widely used in LED lighting, automotive electronics, microelectronics, heat exchange equipment, etc.1–3 Usually, polymers have excellent insulating ability but low thermal conductivity. Al2O3, AlN, BN, SiO2, etc. Thermally conductive but insulating fillers can improve the thermal conductivity of polymer composites.4,5 A large amount of thermally conductive but insulating fillers are added to the polymer matrix to make the polymer composites possessing considerable high thermal conductivity (k), such as the k is higher than 0.6 W/(m.K). 6 Unfortunately, high loading of fillers often do harm to the mechanical properties of composites. Many researchers have made efforts to increase the k of polymer composites in the case of keeping low fillers volume percentage content in recent years. Orientating fillers in polymer matrix, 7 using special morphology fillers, 8 assembling of fillers 9 as well as enhancing the interface action between fillers and polymer 10 are effective methods to improve the k of polymer composites.
The hybrid of fillers with different geometries can often establish effective thermally conductive pathways in polymer composites. Karimzadeh et al. 11 studied the transfer of phonon energy in hybrid structure of pillared graphene and boron nitride film (PGBN). The temperature, length of PGBN structure, and the presence of vacancy defects at the interface of PGBN affected the interfacial thermal conductance. Gu et al. 12 fabricated hetero-structured boron nitride nanosheets@alumina (BNNS@Al2O3) fillers by the method of precipitation and high temperature calcination. The BNNS@Al2O3 heterostructured fillers had more significant thermal conductivity coefficient improvement than single Al2O3, BNNS and directly blending BNNS/Al2O3 fillers. Lee et al. 13 prepared hexagonal boron nitride/SiO2/glass fiber mixtures by calcining at 650 and 850 °C. They found that calcination temperature and the presence of glass fiber strongly affected the through-plane thermal conductivity of the PP composite. Shayganpour et al. 14 mechanically stabilized hematite/graphene nanoplatelet hybrids into freestanding films by hot-pressing them into a porous cellulosic membrane, the thermal conductivity of thermal interface material films arrived at 8.0 W/(mK). Lee et al. 15 constructed starfish surface-inspired graphene-copper metaparticles, in which multiple bundles of graphenes exist as protrusions on the surface of copper plate. They obtained carbon fiber composites with ultrahigh vertical thermal conductivity by filling low content of these metaparticles. Although hybrid thermally conductive fillers are effective in improving the thermal conductivity of composites, their preparation process is often very complex.
In this paper, KH550-BN and KH560-Al2O3 with amino and epoxy group on surface, respectively, were filled in PP matrix by twin-screw extruder. Hybrid fillers with semi core-shell structure were formed by KH550-BN
Experimental
Materials
BN (BNH03, 3–5 μm) was a product of Qinhuangdao ENO High-Tech Material Development Co, Ltd (China). Spherical Al2O3 (BAK-5, D50 5.5 μm) was purchased from Shanghai Bestry Performance Materials Co, Ltd (China). γ-Aminopropyltriethoxy silane (KH-550), γ-(2,3-epoxypropoxy) propyltrimethoxy silane (KH560) and 2,4,6-tris(dimethylaminomethyl)phenol (DMP-30) were purchased from Sinopharm Chemical Reagent (China). Polypropylene (K8303) was supplied by Sinopec Yanshan Petrochemical Co, (China). All reagents were used as received without any further purification.
Surface modification of fillers
400 g BN was added into a high-speed mixer (SHR-10C, Jiangsu Zhangjiagang Bell Machinery Co, Ltd, China). 4 g KH550, 0.98 g water and 5.1 g ethanol were added to a glass vessel, and the mixture was set at 30°C for 0.5 h to hydrolyze KH550. When the temperature of BN powders reached 110°C, the hydrolyzed KH550 solution was poured into the high-speed mixer uniformly and stirred for 15 min. The product was washed with absolute ethanol for three times, and then dried in a vacuum oven at 80°C for 8 h. KH550 surface treated BN powders (KH550-BN) were obtained.
KH560 surface treated Al2O3 powders (KH560-Al2O3) were prepared by the same method. The mass of Al2O3, KH560, water, and ethanol were 2000 g, 20 g, 4.6 g, and 25.4 g, respectively.
Preparation of BN/Al2O3/PP composites with in-situ self-assembling
80 g KH550-BN, 320 g KH560-Al2O3, and a certain amount of PP and DMP-30 were uniformly mixed in the high-speed mixer. The volume fraction of BN and Al2O3 was set as 15.46% in all samples except special description. The mixture was added into a twin-screw extruder (SJSH-30, Nanjing GIANT Machinery Co., Ltd.), and the BN/Al2O3/PP composites were obtained by extrusion and granulation. The testing samples were prepared by injection (HTL90-F5B, Ningbo Haitai Machinery Manufacturing Co, Ltd). The The 
KH550-BN and KH560-Al2O3 pre-assembling
80 g KH550-BN, 320 g KH560-Al2O3, 4 g DMP-30 and 200 mL liquid paraffin were put into a three-necked round-bottom flask. The suspension was stirred at 110°C for different time under a N2 atmosphere. The product was filtered and washed with ethyl acetate, and dried in a vacuum oven overnight to get different pre-assembled degree KH550-BN and KH560-Al2O3 thermally conductive powders.
Characterization
Scanning electron microscopy
Scanning electron microscopy (GeminiSEM 500, ZEISS, Germany) was applied to examine the morphology of samples. The samples of composites were etched by laser (ZY-B04/B02, Changzhou Zhongyi Electronic Computer System Research Institute, China) before observation. All specimens were sputtered with a layer of gold before observation.
Thermal gravimetric analysis
Thermal gravimetric analysis was performed on TG-209-F3 (NETZSCH, Germany), 5–10 mg samples were heated to 900°C at a heating rate of 20°C.min−1 under N2.
X-ray photoelectron spectroscopy
X-ray photoelectron spectroscopy (ESCALAB 250, Thermo, USA) was employed to analyze the surface of pristine and modified fillers.
Thermal conductivity testing
The thermal conductivity of composites at room temperature were measured according to ASTM D5470 using Thermal analyzer (TPS 2500S, Hot Disk, Sweden). The double helix probe of 4 mm diameter was placed between two lamellar samples of 2 mm thickness, flat on both sides.
Mechanical properties testing
The tensile and Izod notched impact strength of composites were measured according to ISO 527–2: 1993 and ISO 180: 2000, respectively. The electronic universal testing machine (CMT4304, Shenzhen Sans, China) and impact strength tester (Shenzhen Sans, China) were used. The values reported were the medians from five specimens each.
Electrical resistivity measurements
The measurements were conducted on a digital high resistance test fixture PC68 (Shanghai Precision Instrument Manufacture, China) and a sample with a diameter of 60.0 mm and a thickness of 1.0 mm was used.
Results and discussion
Surface modification of BN and Al2O3
Figure 2 shows the XPS spectra of KH550-BN, KH560-Al2O3, and pristine BN and Al2O3. The corresponding data are provided in Table 1. The content of C and Si atom of KH550-BN and KH560-Al2O3 increased by comparing with pristine BN and Al2O3 powders, while the content of oxygen element of KH550-BN and KH560-Al2O3 was lower than that of pristine BN and Al2O3. The content of nitrogen element of KH550-BN was also lower than that of pristine BN. Because the residue of KH550 and KH 560 grafted on the surface of KH550-BN and KH560-Al2O3 was rich in silicon and carbon element. It can be concluded that KH550 and KH560 are grafted onto the surface of BN and Al2O3 powders, respectively. XPS spectra of powders (a)pristine Al2O3, (b) KH560-Al2O3, (c) pristine BN, and (d) KH550-BN. Element composition on the surface of powders.

Figure 3 shows the TG curves of KH550-BN, KH560-Al2O3, and pristine BN and Al2O3. The residual mass percentage (at 900 0C) of pristine BN and Al2O3 powders was 98.54% and 98.28%, respectively. The weight loss came from the free hydroxyl group on the surface of pristine BN and Al2O3 powders decomposing at high temperature. The residual mass percentage (at 900 0C) of KH550-BN and KH560-Al2O3 was 98.01% and 97.41%, respectively. The weight loss came from the decomposition of not only the free hydroxyl group but also the organic residua of KH550 or KH560 on the surface of KH550-BN and KH560-Al2O3 powders. The amount of residual silane agent grafted on surface of modified inorganic powders could be calculated by comparing the residual mass percentage difference between pristine and modified inorganic powders.
16
As a result, the amount of residual KH550 and KH560 grafted on surface of KH550-BN and KH560-Al2O3 was 0.53% and 0.87%, respectively. The TG curves of powders (a) pristine Al2O3, (b) KH560-Al2O3, (c) pristine BN, and (d) KH550-BN.
Effect of the amount of catalyst
The amount of PP and DMP-30.
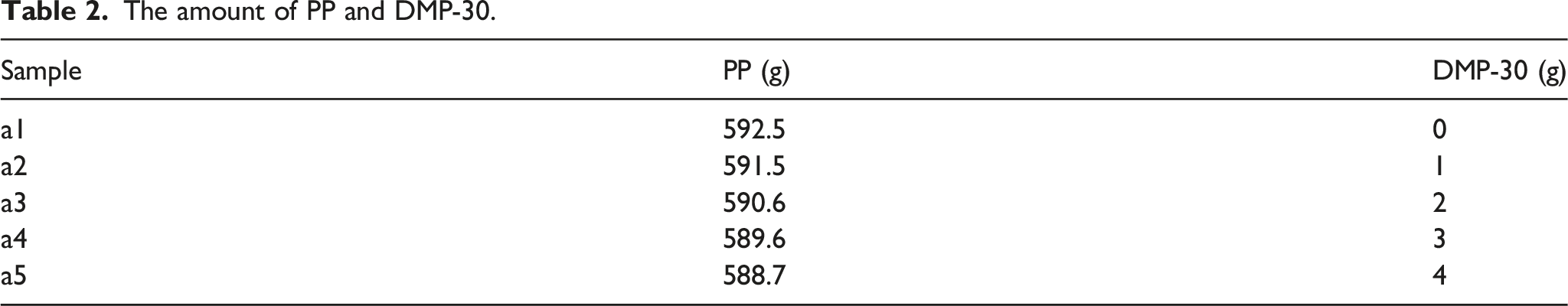
Figure 4 shows the effect of the amount of catalyst DMP-30 on the thermal conductivity of BN/Al2O3/PP composites. The thermal conductivity of BN/Al2O3/PP composites without DMP-30 is 0.51 W/(mK). The thermal conductivity of composites increased with increasing the amount of DMP-30. When the amount of DMP-30 exceeded 3 g, the increase of thermal conductivity of the composites gradually slowed down. The thermal conductivity of the composites reached 0.62 W/(mK) when the amount of DMP-30 was 4 g. The reason is that with the increase of the amount of catalyst, the activation energy of the reaction between amino and epoxy group decreases, and the reaction between amino group and epoxy group becomes easier and easier. It indicates that DMP-30 promotes the The effect of the amount of DMP-30 on the thermal conductivity of composites.
The effect of the amount of DMP-30 on mechanical properties and volume resistivity of composites.

Effect of the homogenization temperature
The effect of homogenization temperature on properties of composites.

Effect of the reaction time
The screw speed and the reaction time.

Figure 5 shows the influence of reaction time on the thermal conductivity of BN/Al2O3/PP composites. The thermal conductivity of the composite increased with the increase of the reaction time. The residence time of materials in the extruder became shorter with the increase of screw speed, and the reaction degree between the amino groups on the KH550-BN and the epoxy groups on the KH560-Al2O3 decreased. As a result, the amount of thermally conductive fillers with special morphology formed by The effect of the reaction time on the thermal conductivity of composites.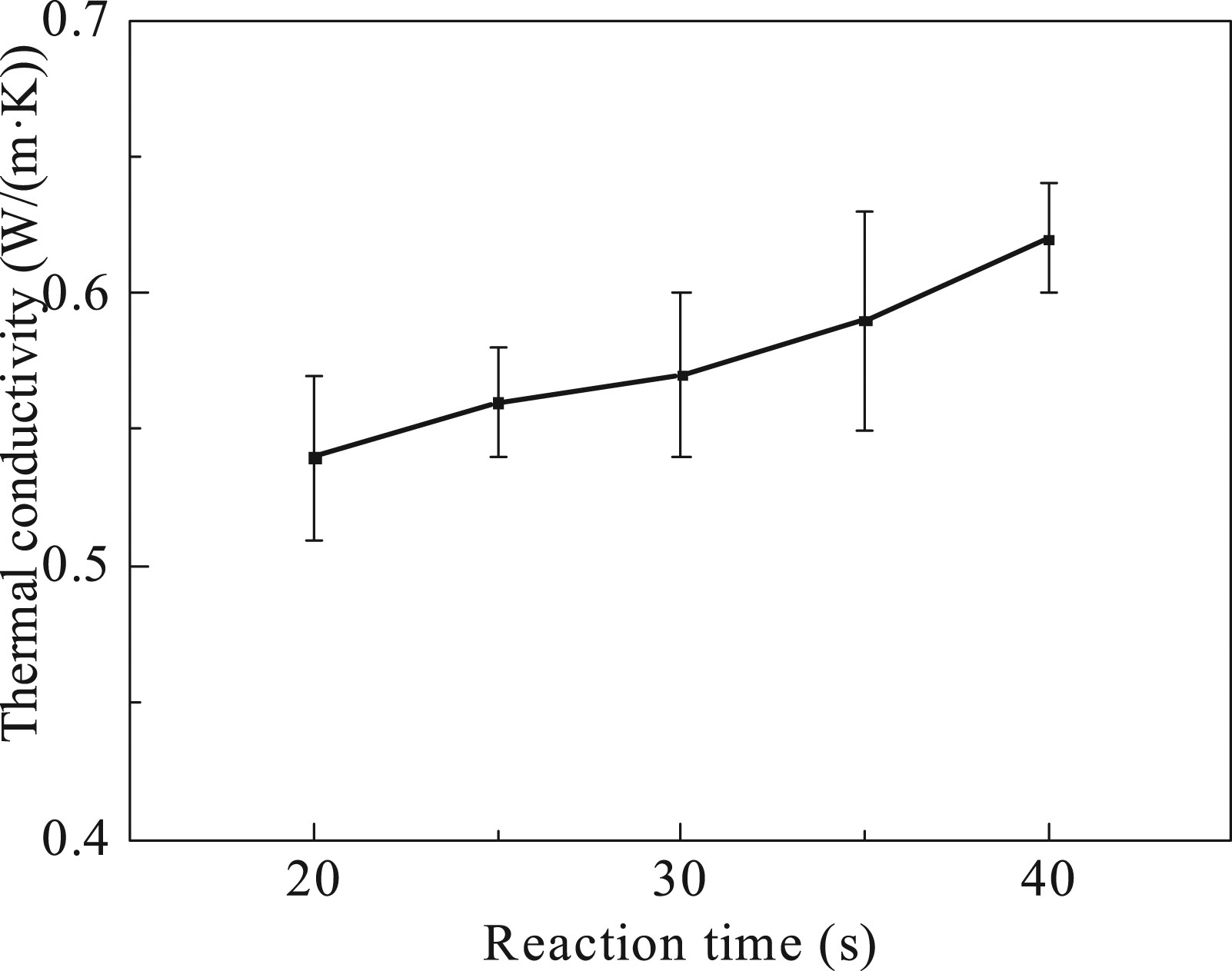
The effect of screw speed on mechanical properties and volume resistivity of composites.

In-situ self-assembling of thermally conductive powders
The pre-assembling time of samples.

Figure 6 shows the SEM images of pristine BN/Al2O3 powders (Figure 6(a)), and KH550-BN/KH560-Al2O3 pre-assembling 65 min powders (Figure 6(b)). In Figure 6(a), Al2O3 and BN are independent of each other. While in Figure 6(b), BN is distributed on the surface of Al2O3, and a half coated hybrid thermally conductive fillers are obtained. SEM images of (a) pristine BN/Al2O3, and (b) KH550-BN and KH560-Al2O3 pre-assembling 65 min.
The effect of KH550-BN and KH560-Al2O3 pre-assembling time on the thermal conductivity of the composites is shown in Figure 7. The thermal conductivity of the composites increased with the increase of the pre-assembling time. When the pre-assembling time exceeded 65 min, the increase of thermal conductivity of the composites slowed down. The reason might be that with the increase of pre-assembling time, the reaction degree of amino groups on KH550-BN surface and epoxy group on KH560-Al2O3 surface gradually increased, and more hybrid thermally conductive fillers with special morphology as shown in Figure 6(b) were formed. The effect of KH550-BNand KH560-Al2O3 pre-assembling time on thermal conductivity of composites.
The SEM images of impact sections of samples etched by laser are shown in Figure 8. Figure 8(a) and (b) were KH550-BN/KH560-Al2O3 SEM images of composites filled with: (a) KH550-BN/KH560-Al2O3 
Conclusions
X-Ray photoelectron spectroscopy and thermal gravimetric analysis revealed that KH550 and KH560 were successfully grafted onto the surface of BN and Al2O3. SEM images showed that hybrid fillers with semi core-shell structure were formed by KH550-BN
Footnotes
Acknowledgements
The authors are grateful for the support from Anhui Science and Technology Department, China (17030901076).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:funding of Anhui Science and Technology Department.
