Abstract
Shape-memory polymer–magnetite (Fe3O4) composite samples were prepared by extrusion compounding and injection molding. Thermal diffusivity, conductivity and specific heat capacity were measured from 290 K to 340 K. Increasing filler fraction decreases specific heat capacity and increases thermal diffusivity. Experimental results show a good agreement with Agari–Uno and Hashin–Shtrikman models but not with Bruggeman model. The interconnectivity of the particles is very poor. Agari–Uno model leads to a high influence of polymer on thermal properties. Below 350 K mainly the polymer and above 350 K mainly the Fe3O4 influences specific heat capacity that shows drastic changes around 310 K (recovery temperature) and 357 K.
Introduction
Shape-memory polymers (SMP) are used in medical devices and biological microelectrical systems due to their wide range of temperatures for shape recovery, high recoverable strain and low costs in manufacturing. 1 –3 Therefore, intensive research is performed on the so called smart materials called SMPs, which are capable of responding to an external stimulus. 4 –7 One of the most extensively investigated groups of SMPs are polymers that show a shape transformation, when a certain switching temperature is exceeded.
The typical sequence for thermally stimulated SMPs is the application of an initial deformation to the polymer at an elevated temperature (predeformation), fixing the shape by cooling the predeformed SMP under strain to a lower temperature (storage) and then heating the material to recover the original shape (recovery). 2,8
Conventionally, the shape recovery in an SMP is actuated by external heating. If an external heating process is not feasible, another mechanism for triggering the shape recovery has to be considered, such as inductive heating; if magnetic particles are incorporated into an SMP, an inductive heating of SMPs by electromagnetic fields can be used to induce the shape memory effect. 9 –11 Because of their smaller size, heat generation in magnetic nanoparticles (as they are used in the study by Mohr et al. 9 ) under rotational magnetic field is very low due to Brownian and Néel relaxation losses. 10,12,13 Therefore, high magnetizing frequencies have to be used to generate an appropriate heat by power losses in applications with magnetic nanoparticles. However, in micro-sized magnetic particles, magnetization reversal losses are usually divided into hysteresis loss, eddy current losses and anomalous losses. Due to increased eddy current losses and increased number of domain walls micro-sized particles show higher heat generation by increased power losses under low magnetizing frequencies.
If the temperature in the SMP filled with magnetic particles is generated by inductive heating, the heat must be distributed through the polymer. However, if an external heater is used, the heat must be distributed through the composite, and the time required for this process is dependent on the thermal diffusivity of the material.
Thus, the shape recovery time must also be dependent on the thermal diffusivity of the composite. Although thermal properties of a number of particle-filled polymers are available, 13 temperature-dependent thermal properties of the SMPs are unknown. Therefore, in this work, the thermal properties of polyurethane SMP filled with magnetite (Fe3O4) particles are investigated.
Experimental
Materials
The SMP was a thermoplastic polyurethane MM4510 (Mitsubishi Heavy Industries, now SMP Technologies Inc., Tokyo, Japan) obtained in a pellet form. The glass transition temperature (
Three different grades of Fe3O4 (Minelco B.V., Moerdijk, The Netherlands) with mean particle sizes of 10 μm (trade name MagniF10), 50 µm (MagniF50) and 150 µm (MagniF150) were used. The particle size distribution, the composition, the thermal conductivity
No further processing additives for compounding or for injection molding or as stabilizers were used to enhance the thermal stability of the polymer during processing in these experiments.
Sample preparation
Different volume fractions (0%, 10%, 20%, 30% and 40%) of Fe3O4 filler were compounded with polymer matrix using a Thermo Haake Rheomix 600 (Thermo Electron, Karlsruhe, Germany) with mixing head operating at 50 r/min and at 473 K. Before compounding, the polymer granules were premixed with Fe3O4 and then fed into Thermo Haake Rheomix. The mixing time was 10 min. Later, the compound was dried in an oven at 353 K for 12 h directly before the preparation of samples by injection molding.
The test samples were prepared using a laboratory injection molding machine (Allrounder 320C 600-250, Arburg, Germany). The injection molding temperature was 473 K, and the mold temperature was 293 K. Samples for measuring the thermal conductivity were disks of 2 mm thickness and 50 mm diameter, while samples for thermal diffusivity experiments were small cylinders of 3 mm height and 10 mm diameter.
Figure 1 shows the morphology and the homogeneous dispersion of the Fe3O4 particles in the polymer.
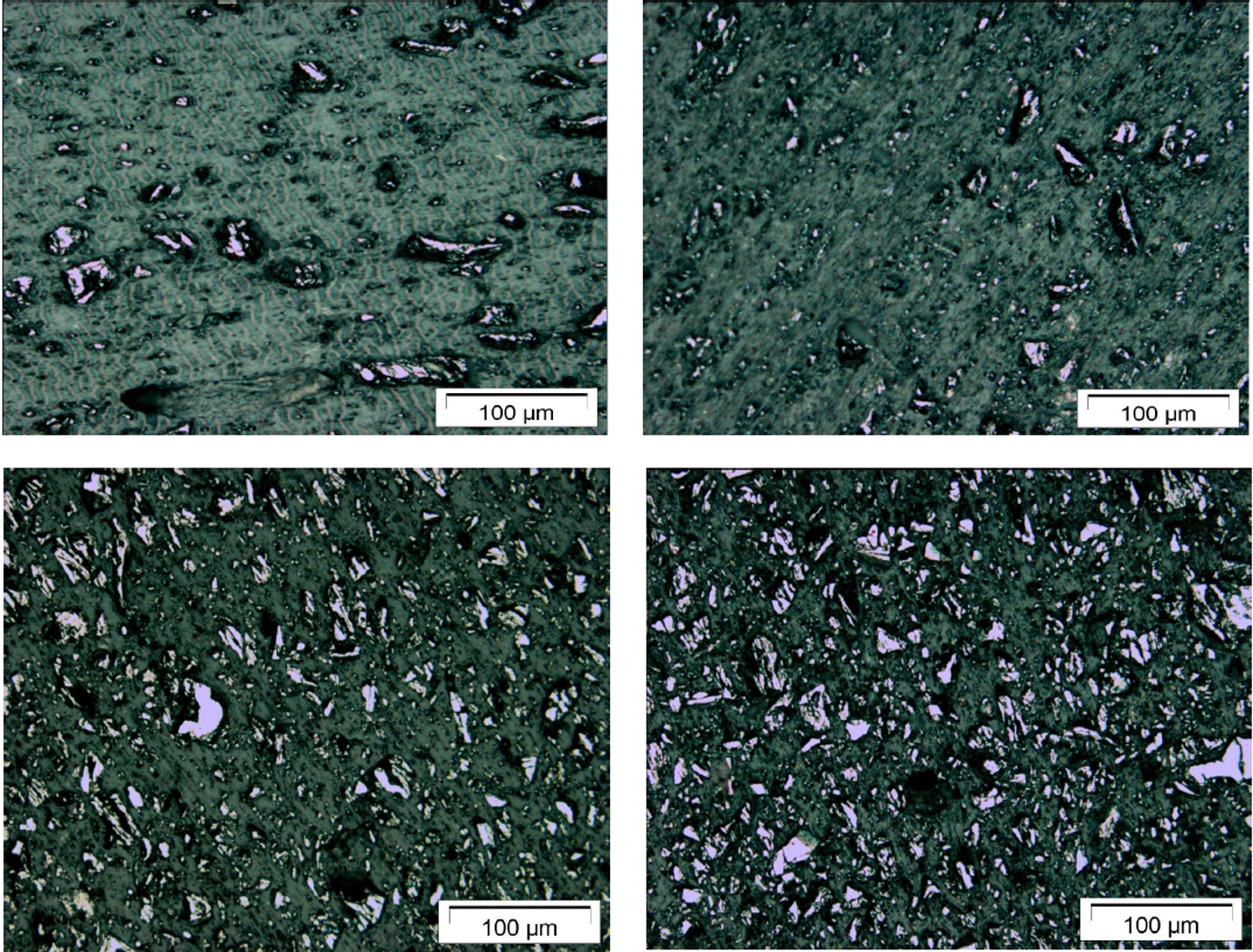
Micrographs of polyurethane–magnetite shape memory polymer (upper left: 10 vol%; upper right: 20 vol%; lower left: 30 vol%; lower right: 40 vol%.
Experimental methods
Using the guarded heat flow method, the thermal conductivity values were measured with a TCA Thermal Conductivity Analyzer (TCA-200LT-A, Netzsch, Selb, Germany). The injection molded samples with a diameter of
Using a differential scanning calorimeter DSC-2920 (TA Instruments, Alzenau, Germany), the specific heat capacity was measured. Measurements were carried out from 283 K to 383 K with a heating rate of 5 K/min. The differential scanning calorimeter was calibrated in the same temperature range using sapphire (Al2O3) sample as standard. The base lines of aluminum pan and sapphire reference were measured and compared with the heat flow measurements of the SMP samples. The accuracy of the measurements is 3%.
The thermal diffusivity values were measured by a transient method, closely related to laser-flash experiments. 17 The used transient technique is especially optimized for the measurements of polyphase aggregates. A detailed description of the apparatus is given by Schilling. 18 The accuracy of the measurement of the polyphase aggregates is 3%.
Theoretical considerations
Various theoretical models can be used to describe the thermal diffusivity of a homogeneous composite. 19 The equations are not necessarily developed for calculating the thermal diffusivity. But for reasons of mathematical analogy, these formulas can also be applied to heat conductivity and thermal diffusivity of composite materials.
Experimental results previously measured by the authors have shown that the models of Bruggeman (equation (1)), Agari–Uno (equation (2)) and Hashin-Shtrikman (equations (3) and (4) provide good congruence with experimental data. 19 –21
The model of Bruggeman
22
requires the thermal diffusivity of the polyurethane matrix (
in the model of Agari and Uno
23
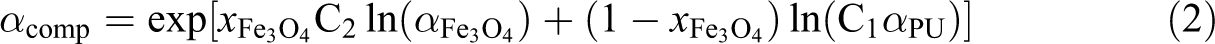
the microstructure of the composite is considered in the constant C1 describing the effect of crystallinity and crystal size of the polymer and C2 describing the ability of the composite to form conductive chains of particles.
According to the Hashin-Shtrikman model,22 the thermal diffusivity of a composite which consists of a homogenous polyurethane matrix with low thermal diffusivity and a fraction x of spherical Fe3O4 particles with a significant higher thermal diffusivity, relates to the Hashin-Shtrikman lower boundary (HS−) and is given as follows: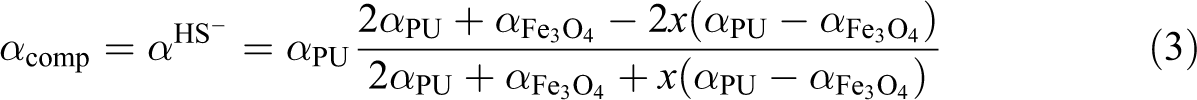
In contrast, if the matrix consists of a phase of high thermal diffusivity with embedded particles of low thermal conductivity, the Hashin–Shtrikman upper boundary (HS+) would describe the diffusivity of the composite as follows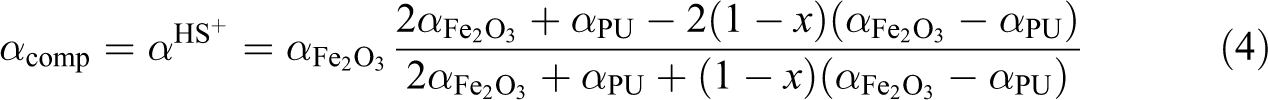
For HS− the particles are insulated from each other, while for HS+ the particles are interconnected into a three-dimensional network.
Using the model diffusivities HS−and HS+, the interconnectivity of the conducting phase 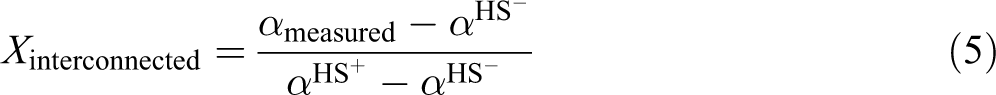
The as-determined interconnectivity is a relative measure to an ideally interconnected network of the phase with high thermal diffusivity. As the diffusivity is dominated by the smallest junctions of the path of high diffusivity, the resultant interconnectivity is dominated by these junctions. If, for example, the phase with high diffusivity is interconnected by point contacts, most part of this phase will not directly contribute to the overall diffusivity and the determined interconnectivity is dominated by point contacts and cannot be regarded as an ideally interconnected network. This especially holds true, if the difference between the diffusivities of different phases is huge.
Results and discussion
Figure 2 shows the temperature dependence of heat capacity of polyurethane SMP and of SMP composite in the temperature range from 280 K to 380 K. Additionally, the derivative of the specific heat capacity with respect to the temperature is shown for Fe3O4 of a mean particle size of 10 µm, because there were no significant differences measured for different particle sizes. In the whole measured temperature range, the specific heat capacity values of the unfilled polyurethane SMPs are clearly higher than the polyurethane SMPs filled with Fe3O4 particles. Below 300 K, the specific heat conductivity increases slowly, before a sudden rise of specific heat capacity appears in the temperature range from 300 K to 320 K. Regarding the derivative of specific heat capacity with respect to the temperature, the inflection point of all curves can be found at 310 K. This is below the
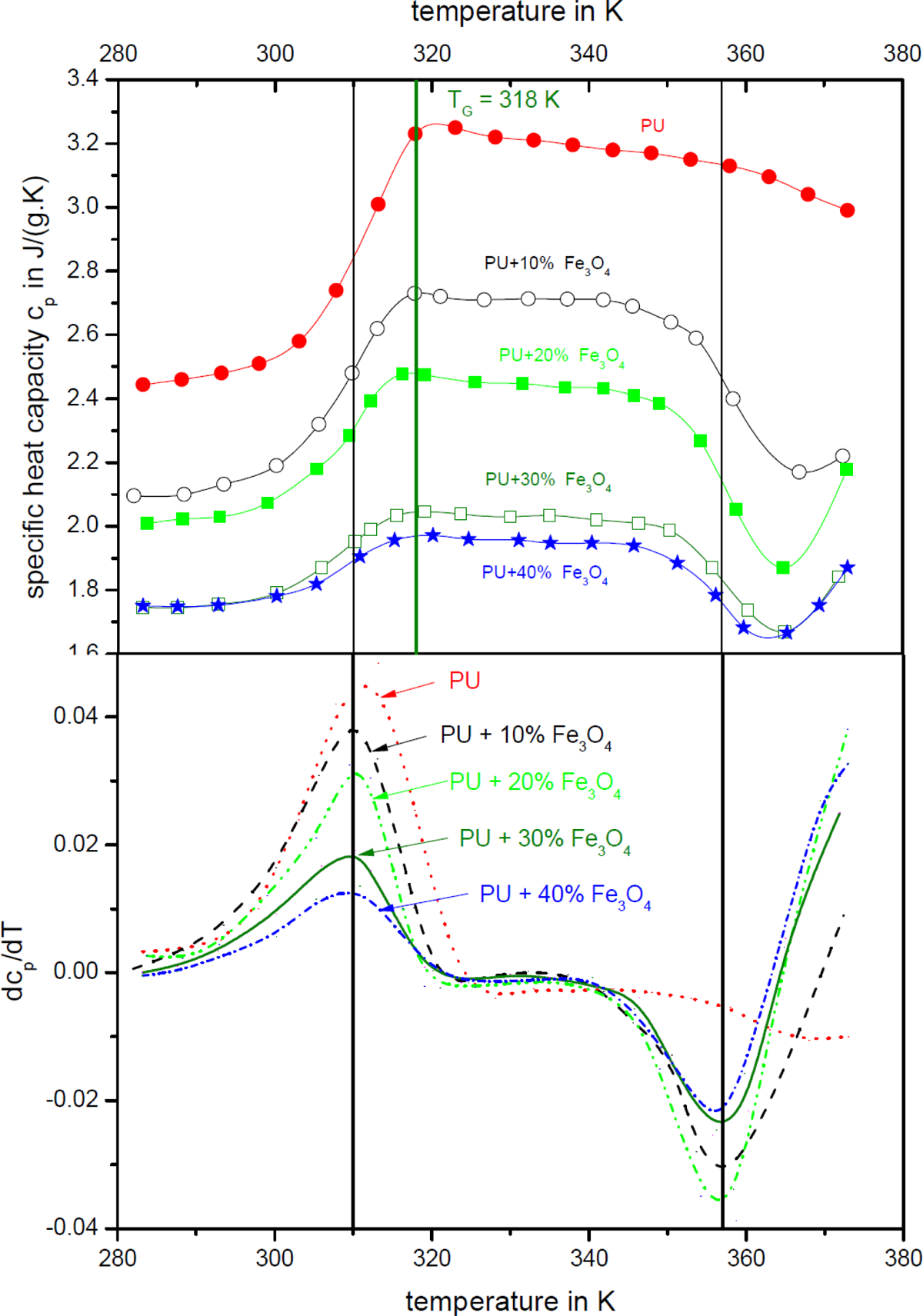
Upper graph: Temperature dependence of the specific heat capacity of magnetite (
With further increase in temperature up to 350 K, a weak decline of specific heat capacity can be observed before a drastic decrease is found between 350 K and 370 K followed by a minimum and a further increase in specific heat capacity. Again, in the derivative of specific heat capacity, it is found that the inflection point of all the curves, with the only exception of pure polyurethane, can be found at an identical temperature of 357 K. It is somehow astonishing that the temperature at which the minimum in the curve of specific heat capacity appears is varying with the Fe3O4 content. The higher the Fe3O4 content in the composite the lower is the temperature of the minimum. For the composite with 40 vol% Fe3O4, the minimum in the curve of specific heat capacity is found at 368 K. The position of the minimum of the unfilled polymer is assumed to be at higher temperatures and therefore not shown in Figure 2. These higher temperatures were not investigated because the shape memory effect of the polymer appears only in the shown temperature interval. This behavior leads to the assumption that the decrease in the specific heat capacity around the inflection point is mainly caused by the Fe3O4 filler. Moreover, the position and the depth of the minimum are caused by a superposition of the behavior of the polymer and the Fe3O4.
While the specific heat capacity is dramatically reduced from the unfilled polyurethane SMPs to the composites with 10 vol%, 20 vol% and 30 vol% Fe3O4 contents, the specific heat capacity of composites with 30 vol% and 40 vol% Fe3O4 contents is quite similar. The reason for this behavior might be the formation of heat conductive pathways in the composites that appear at the percolation threshold of 30 vol%. 10,24
For the Fe3O4-filled polymers, an increase in the specific heat capacity was found in the whole temperature range.
24
In this investigation, a similar increase can only be observed for the composites at temperature higher than
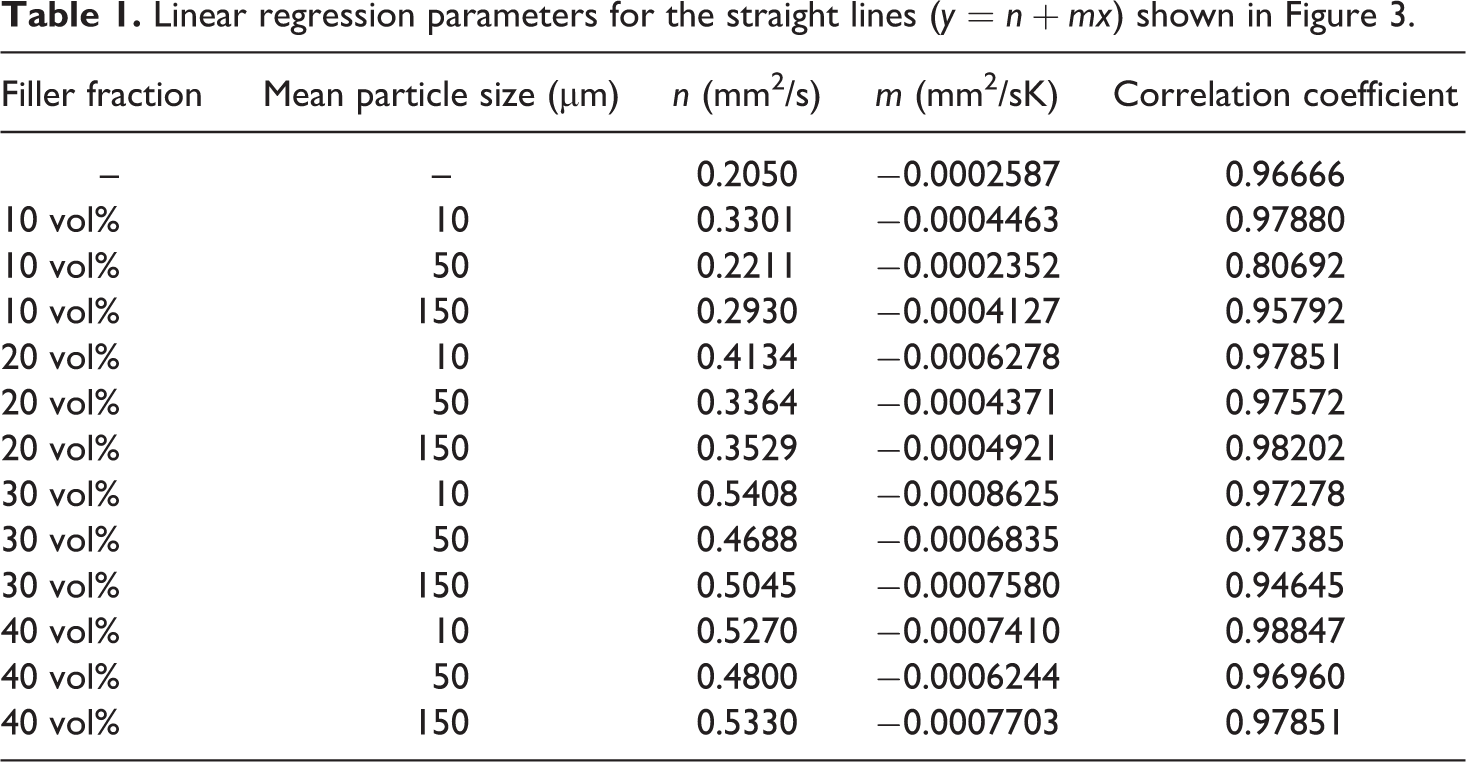
Such distinctive extremes, similar to the exhibited temperature dependency of specific heat capacity, are not visible in the temperature dependence of thermal diffusivity (Figure 3). It can be seen that the thermal diffusivity of all the samples decreases linearly with increasing temperature in the investigated temperature interval. The obtained regression data can be found in Table 1. The correlation coefficients of ∼1 indicate a very good linearity in the measurements values of the investigated temperature interval. This shows that the thermal diffusivity is not influenced by the
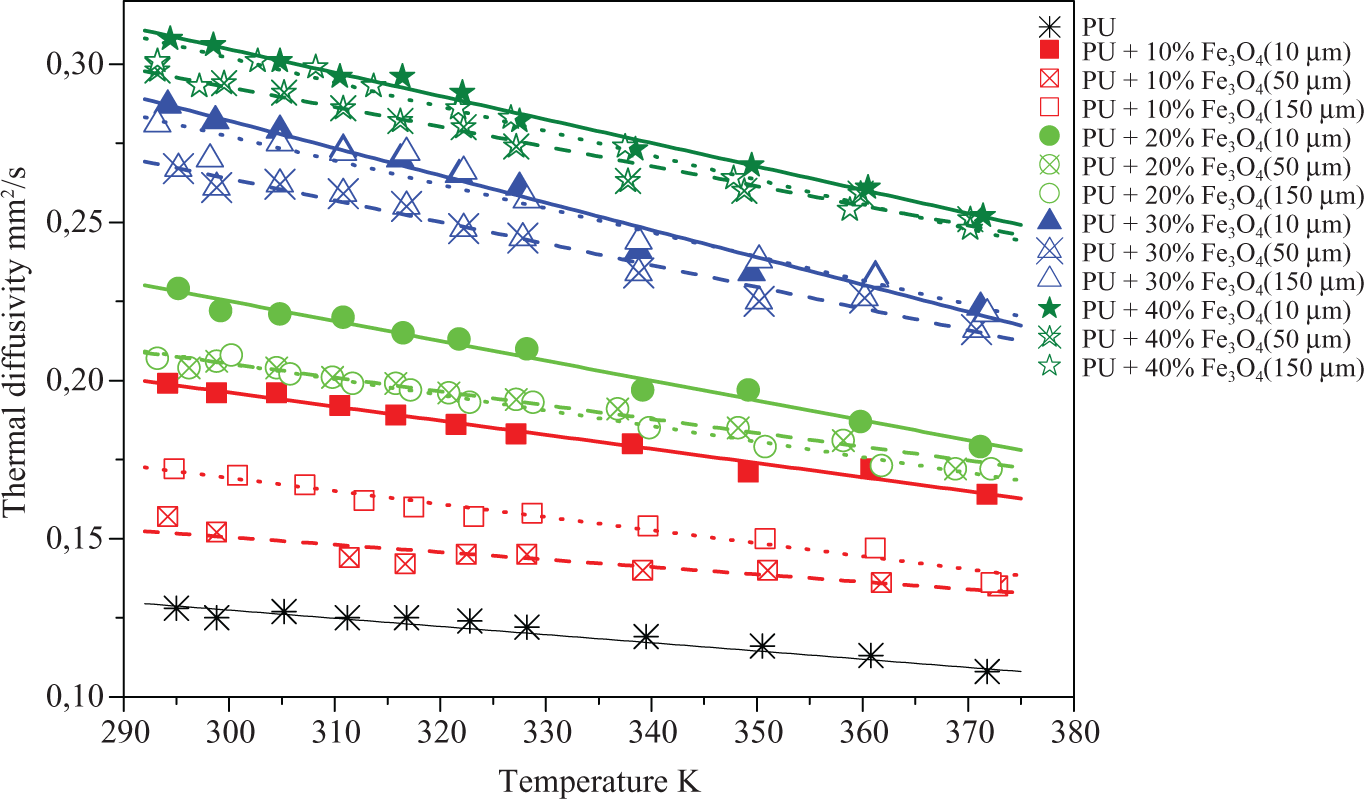
Thermal diffusivity of a polyurethane shape memory polymer filled with magnetite particles of different particle sizes (10 µm, 50 µm and 150 µm) and different volume fractions of magnetite (0%, 10%, 20%, 30% and 40%). Symbols represent measurement values and lines are regression lines ( Table 1).
Linear regression parameters for the straight lines (
With increasing filler fraction, the negative slope of the regression line is tripled. This can be explained by the temperature dependence of thermal diffusivity of Fe3O4 as well as by the softening of the polymer with increasing temperature.
A decrease in thermal diffusivity was already found by Hofmeister
16
for two different grades of natural Fe3O4 in the temperature range from 300 K to 750 K. Thus, the influence of the Fe3O4 in the composite should also lead to a reduction in the thermal diffusivity of the composite. This influence must be larger if the Fe3O4 fraction in the composite is higher. Furthermore, a stronger decrease in the Young’s modulus exists for the high filler fractions when compared with the low filler fractions,
26
which is mainly due to the softening of the polymer. Young’s modulus
An increase in filler fraction leads to an increase in the thermal diffusivity. In contrast, it is somehow unexpected that different mean particle sizes cause differences in thermal diffusivity. Especially, for the low filler fraction of 10 vol% Fe3O4, it is recognizable that the composite with
Apparently, the larger number of contacts in the polymer filled with 10 µm particles compared with the composites with 150 µm or 50 µm particles led to an increase in thermal diffusivity. The larger diffusivity of composites with 150 µm when compared with composites with 50 µm particles, however, suggests that the diffusivity depends on the number of contacts and gaps between the particles. Other thermal diffusivity measurements of Al2O3–Ni composites 27 are showing an increasing diffusivity with increasing filler particle sizes, and also thermal conductivity measurements of polymer particle composites are showing rising diffusivity with increasing particle sizes. 28 Contrarily, composites containing polypropylene filled with 50 vol% iron silicon particles are showing only a small dependence of thermal diffusivity on particle sizes. 21 With increasing particle size in that work, a weak increase in thermal diffusivity was found and a dependence of thermal diffusivity on the number of contacts and gaps between the particles is neglected.
Figure 4 shows the interconnectivity of a selection of investigated materials according to equation 5. The highest interconnectivity of around
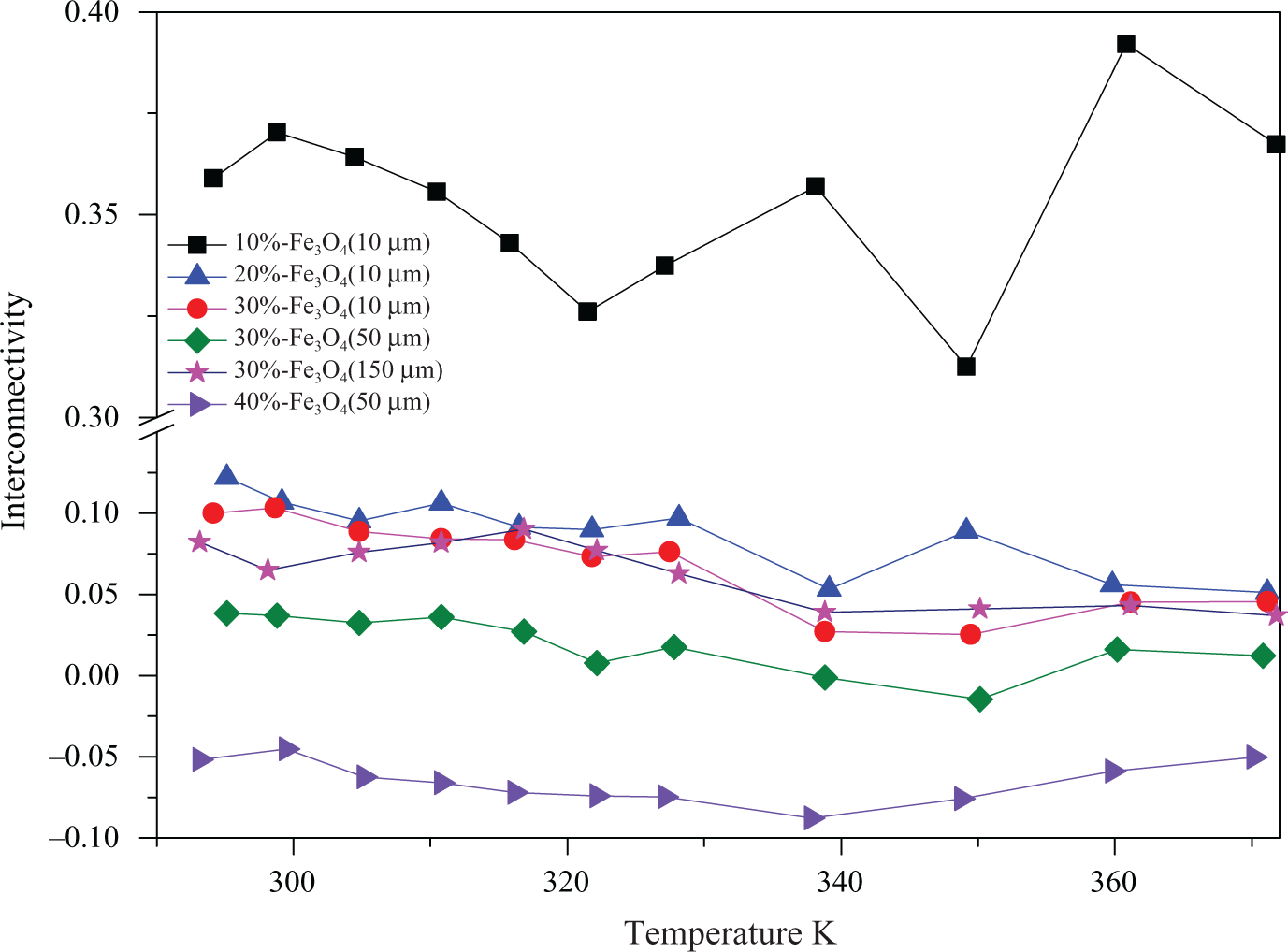
Temperature dependence of interconnectivity
The consistency of the measurement values using theoretical models (equations (2) and (3)) at room temperature
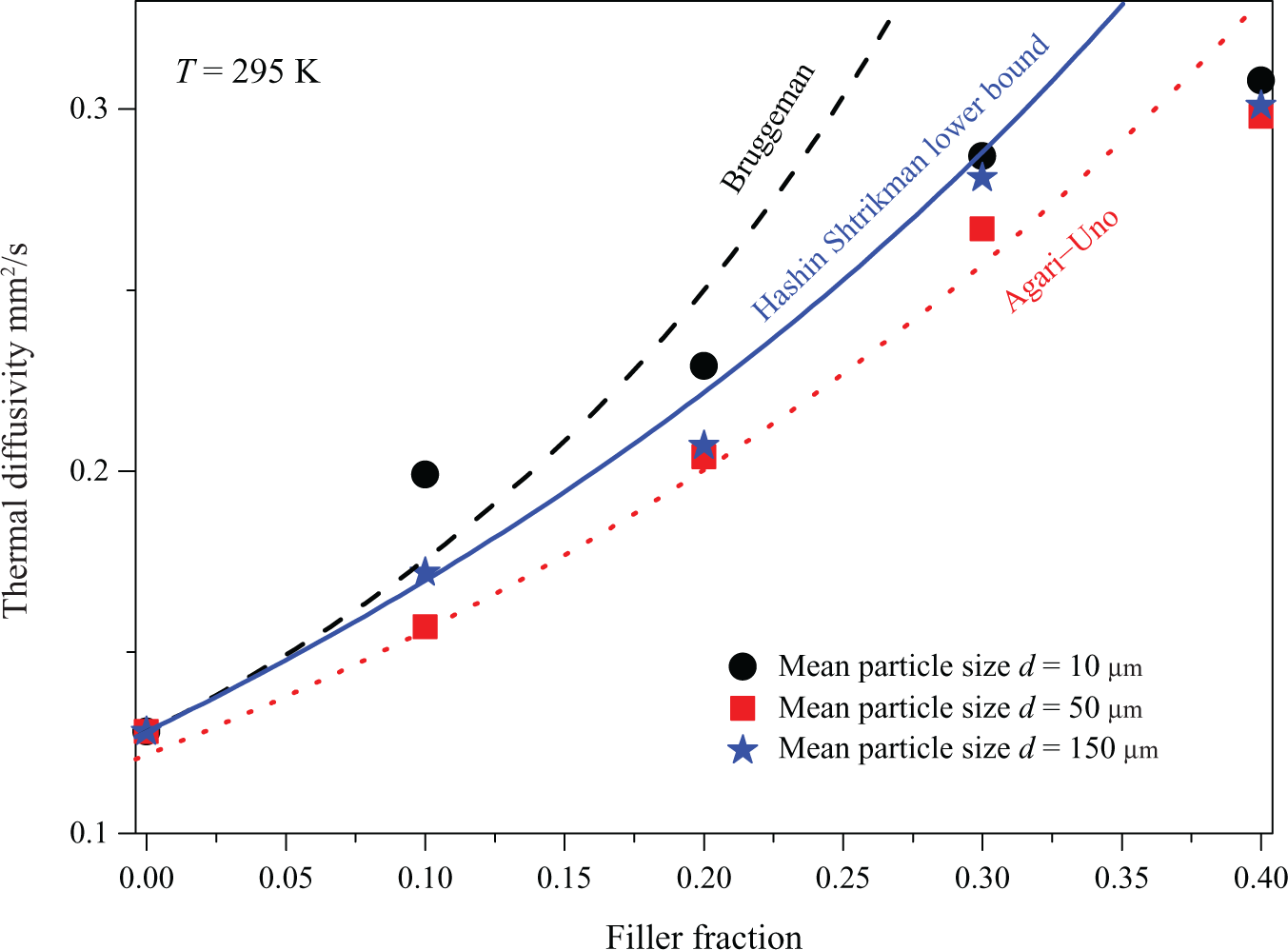
Dependence of thermal diffusivity of SMP–Fe3O4-composite on Fe3O4 fraction and mean particle sizes at a temperature of
Finally, the thermal conductivity of pure SMP and different composites in the temperature interval between 305 K and 340 K can be seen in Figure 6. In this temperature range, the
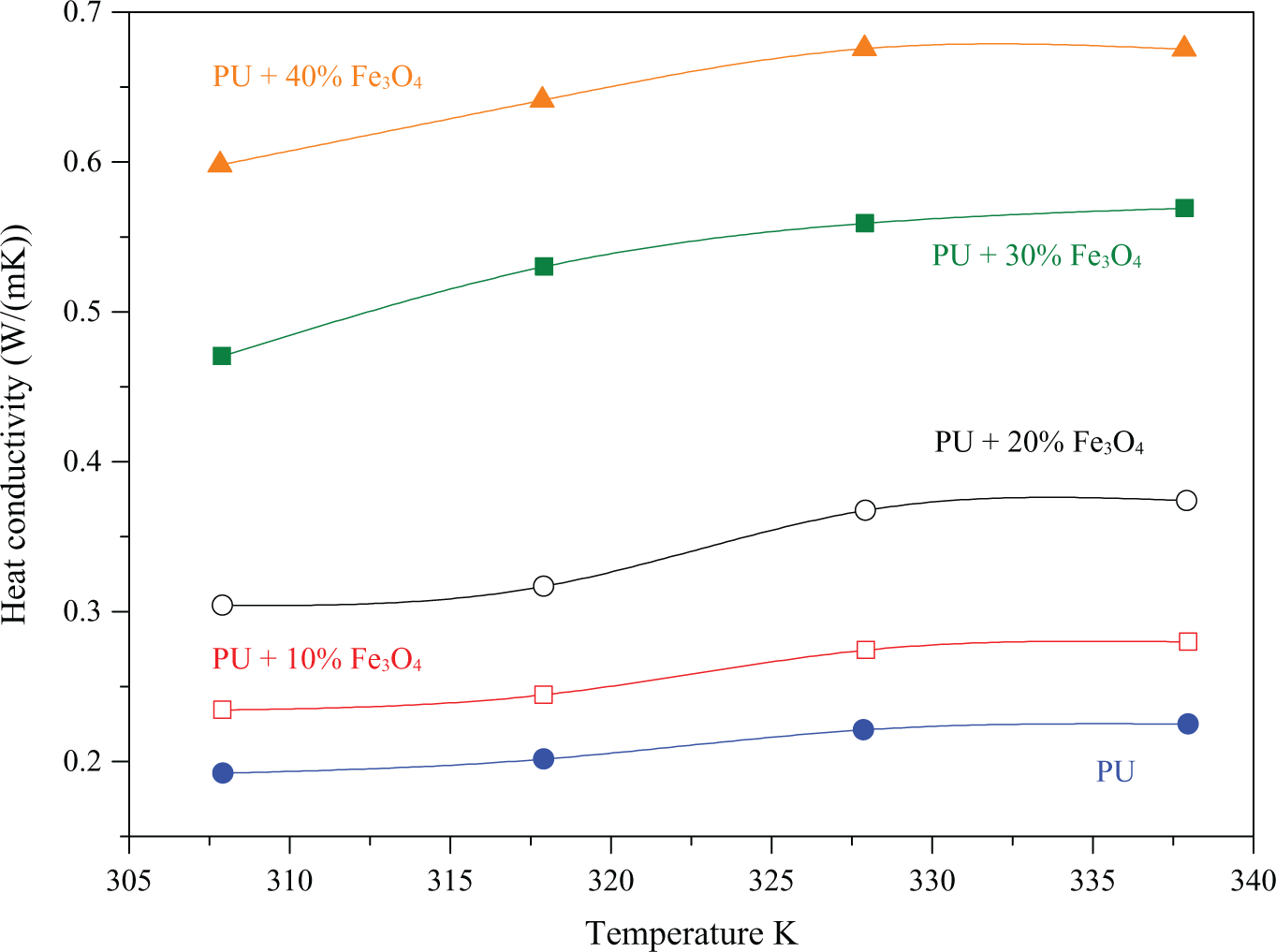
Temperature dependent thermal conductivity of SMP–Fe3O4 composite. The magnetite particles are having a mean particle size of
Conclusions
Thermal diffusivity and conductivity of an SMP can be increased by Fe3O4 as a filler material. With rising filler fraction, these properties also ascend. While thermal conductivity is increased with rising temperature, the thermal diffusivity of an SMP and SMP–Fe3O4-composites decreases. For filler fractions around or higher than the percolation threshold, changes in the thermal properties are more significant than for filler fractions below the threshold. This can be attributed to the formation of conductive pathways of filler particles in the polymer. The formation of conductive pathways is very poor which can be seen in very low interconnectivity values. The interconnectivity can be estimated using the model conductivities calculated with the Hashin–Shtrikman model. This model describes the measurement values as rather good. Although the physical meaning of the fit parameter in the Agari–Uno model is unclear, this model can also describe the measurement values and leads to an identical result, namely poor interconnected particles and a major influence of the polymer onto thermal properties. Data of specific heat capacity are influenced significantly by
Footnotes
Funding
This research was supported by the German Science Foundation (DFG).
