Abstract
This work presents the role of organoclay type (hydrophilic C30B vs hydrophobic C15A) and feeding mode (sequential vs. simultaneous) on a model ternary blend system poly (methyl methacrylate)/polystyrene/polypropylene (PMMA/PS/PP, 80/05/15). The rheological and thermal properties of these nanocomposites are linked to their morphology, which is mainly controlled by the preparation method and the nature of the organoclays. Using oscillatory shear rheology and dynamic mechanical analyses, both organoclays were shown to be mainly localized in the PMMA matrix. However, the more polar C30B showed a greater affinity toward the matrix. Studying the morphology using electron microscopy revealed that at 1 wt% of the organoclays, the original core-shell morphology of the blend was retained regardless of the feeding sequence. At 3 wt% of the organoclays, however, the core-shell morphology was only retained in the case of C30B-based nanocomposites prepared using sequential feeding mode. In the other cases, the increased solid-like behavior of the PS phase prevented the formation of a shell. Overall, it was shown that the feeding sequence and the affinity of organoclays towards different blend components determined their localization and therefore the eventual morphology of the nanocomposite.
Introduction
The complexity of material requirements for advanced applications rules out the suitability of many homopolymers thus creating a need for polymer blends and nanocomposites. However, most polymer blends feature multiphase morphologies due to a lack of favorable interactions to gain a sufficient enhancement in entropy. It is well recognized that both rheological behavior and the physico-mechanical properties of polymer blends are determined by their morphology.1–5 Controlling the morphology of multiphase systems with the aim of tuning their final properties for certain applications has been a major drive in this field of research. For instance, the last two decades have witnessed extensive research on ternary blends, especially those with core-shell type morphology.6–10 This is mostly due to the diverse applications they would offer once this morphology is achieved in a well-defined manner. Knowing that with three components, the morphology of the blend is mainly determined by the interfacial tensions,11–14 one can play with different chemistries, compatibilizers, and blending ratios to achieve a variety of morphologies.
More recently, polymer blending in combination with adding a nanofiller has been used as a method to design materials with novel structures for specific applications. The enhanced properties and extensive possibilities, these systems offer come at the cost of added complexity to the system. While with blends, one only needs to worry about tailoring the morphology for a certain application, the localization behavior of the nanoparticles in blend nanocomposites becomes a determining factor in final properties.15–17 The thermodynamic affinity of a nanofiller to certain blend components is certainly among the factors determining where they end up. But this factor only considers the one-to-one affinity of the nanofiller and each component and fails to provide a realistic prediction of how affinities might change, say, in a multi-phase system. Moreover, we need to consider cases where the nanofiller prefers to be in a more favorable phase (thermodynamics), but initially, it is localized elsewhere, for instance, due to processing conditions (kinetics). As shown by many researchers, as well as our previous work, kinetic parameters can serve as a gauge to define the localization of nanofillers in hybrid systems. Examples include the melting point and viscosity of the blend components, mixing time, as well as the feeding sequence. In practice, the sufficiently high viscosity of the components can lower the diffusion coefficient of the nanofillers to the point that Brownian forces no longer play an important role in determining the nanofillers' localization (at least not within normal processing time scales).18,19
Rostami et al. 20 previously reported on the morphological evolution and thermal and rheological properties of poly (methyl methacrylate)/polystyrene/polypropylene (PMMA/PS/PP) ternary blends upon the addition of Multi-walled carbon nanotubes (MWCNTs). After studying the ternary blend from the perspective of the interfacial tensions and the elasticity ratios of the components and how they interplay to determine the morphology we continued to study the effects of feeding sequence and MWCNTs composition. It was shown that at low MWCNTs loadings, the initial core-shell morphology remained mostly unchanged, while at higher loadings (1.5 wt%), the elasticity of the PS phase carrying a considerable amount of MWCNTs was too high to allow the encapsulation of the PP phase and the core-shell morphology was disrupted. These findings immediately triggered questions regarding the effect of the type and geometry of the nanoparticles, which served as inspiration for follow-up works, including the one here.
Organoclay-based nanocomposites have received increasing attention over the last two decades due to the unique and outstanding capability of these nanoparticles in enhancing barrier properties and flame retardancy, as well as physical and mechanical properties.21–27 In fact, these distinctive properties are directly linked with the microstructure of the nanocomposites, where the state of dispersion/exfoliation is predominantly determined by the interfacial interaction between organoclays and polymer matrices.28–33
To the best of our knowledge, the effect of organoclays nature (C15A and C30B) and their mixing order on the morphology of ternary blends with core-shell type morphology has not been investigated so far. Therefore, the main objectives of the present study are the following: (a) the investigation of the effect of the presence of plate-shaped nanoparticles on the core-shell morphology of the ternary polymer blends, (b) an effort to present simple approaches for the determination of nanoparticles localization in polymeric blends, and (c) tailoring blend nanocomposite morphologies through control of thermodynamic and kinetic parameters. X-ray diffraction and linear rheology were used to determine the localization and the intercalation/exfoliation state of the nanoclays in the nanocomposite samples. The corresponding changes in the morphology of the blend samples and the nanocomposites were probed using electron microscopy observations. Finally, the impact of these changes was studied on the thermal properties of the samples.
Experimental
Materials
The specifications of the used materials.

Sample preparation
Codes, compositions, and preparation modes for blends and their nanocomposites.

PMMA: poly (methyl methacrylate); PS: Polystyrene; PP: Polypropylene.
Characterization methods
In order to evaluate the dispersion state of the organoclays in the samples, X-ray diffraction (XRD) was performed at room temperature using an X-ray diffractometer (Philips model X’Pert, Netherland) in the low angle region of 2θ. The X-ray beam was a Co Kα radiation (λ = 1.78897 °A) using a 40 kV voltage generator and a 30 mA current. The diffractograms were scanned in the 2θ range of 2–10° at a rate of 1° min−1.
The viscoelastic behavior of the samples in the melt state was studied on MCR 301 rheometer (Physica Anton Paar, Austria) using a parallel plate geometry with a diameter of 25 mm and a 1 mm gap. Oscillatory shear measurements were performed under N2 at 220°C in the linear regime with an amplitude of 1%.
Field emission scanning electron microscopy (FE-SEM) S-4160 from Hitachi (Japan) at 15 kV was used to study the morphology of the samples. Transmission electron microscopy (TEM) images were obtained from ultrathin sections (∼200 nm) on Philips EM 208S (Netherlands) at 120 kV.
Results and discussion
X-ray diffraction results
Figure 1 shows the X-ray diffraction patterns of the C15A and C30B organoclays used in this study along with those of their corresponding nanocomposites. As it can be seen in Figure 1(a), C15A shows two characteristic diffraction peaks at 2θ = 2.8° and 2θ = 7.2°, corresponding to d-spacings of 3.15 and 1.22 nm, respectively. The peak at 2θ = 2.8° corresponds to the interlayer spacing of the surface-modified layers of montmorillonite while the one observed at 2θ = 7.2° is related to the unmodified layers. Meanwhile, the XRD pattern of C30B in Figure 1(b) shows a single broad peak at around 2θ = 4.7°, corresponding to a d-spacing of 1.86 nm.34,35 X-ray diffraction patterns of the organoclays and the nanocomposite samples: (a) C15A and the related nanocomposites; (b) C30B and the related nanocomposites.
As for the PMMA single polymer nanocomposites, the strong diffraction peak is absent suggesting that the C15A and C30B platelets were exfoliated in the PMMA. Given the polar nature of PMMA, this high level of interaction between the nanoparticles and the matrix, which leads to exfoliated PMMA/organoclay nanocomposites, is not unexpected.36,37 Furthermore, the XRD patterns for different binary and ternary blend nanocomposites containing 3 wt% of the organoclays are similar to those for the PMMA/organoclay nanocomposites with no distinct diffraction peaks observed. This implies that the PMMA matrix (as the main component) facilitated the exfoliation process when blended with the other blend components.
Dispersion of the organoclays in the nanocomposites via rheological measurements
The dispersion and/or exfoliation state of a nanocomposite can be assessed via linear rheology with the low-frequency dynamic response of the material as an indirect measure of chain mobility. Restrained mobility due to the formation of strong molecular interactions with nanofillers usually causes the storage modulus to deviate from the terminal behavior observed for polymer melts. When a sufficient number of chains are restricted, the storage modulus shows the so-called nonterminal behavior where it reaches a plateau at low frequencies (long time scales). The strength of the interactions as well as the number of elastically active chains are among the parameters that can be drawn from such rheological data. 38
Figure 2 presents the angular frequency dependence of storage modulus and complex viscosity for the PMMA and its nanocomposite samples containing 3 wt% of the organoclays. Both single polymer nanocomposites exhibit a strong solid-like behavior in the low-frequency range, which is a signature of the formation of three-dimensional physical networks between individual particles and/or particles and the matrix. The magnitude of this nonterminal behavior varies with the degree of dispersion and the concentration of the organoclays. In other words, a nanocomposite system containing well-dispersed nanofillers exhibits a stronger nonterminal effect compared to one with poorer dispersion at the same nanofiller content. However, the two nanocomposites studied do reveal rather similar nonterminal behaviors. (a) Storage modulus and (b) complex viscosity of poly (methyl methacrylate) in the presence of C30B and C15A.
Figures 3 and 4 depict the melt linear viscoelastic results obtained for the ternary blend and the corresponding nanocomposites (prepared by simultaneous and sequential feeding modes) with C15A and C30B, respectively. At low frequencies, all the ternary blend nanocomposites exhibit a pronounced solid-like behavior in storage modulus as well as a complex viscosity upturn. Since the continuous phase (matrix) in these samples is PMMA, the enhancement of the storage modulus indirectly suggests that the nanoplatelets are localized in the matrix where they have formed a percolating network. Effect of C30B on the (a) storage modulus and (b) complex viscosity of the ternary blend prepared using different feeding modes. Effect of C15A on the (a) storage modulus and (b) complex viscosity of the ternary blend prepared using different feeding modes.

For nanocomposites containing C30B, the nonterminal response is slightly stronger compared to the one containing C15A. This suggests that in C30B-based nanocomposites, more nanoparticles remain in the PMMA matrix because of the stronger interactions between these hydrophilic nanoparticles and the polar side groups of PMMA. A similar argument can be used to understand the effect of sequential versus simultaneous feeding mode. With both types of nanoclays, the solid-like behavior is stronger when the nanocomposite was prepared using the sequential feeding procedure, meaning that more nanoclays ended up in the PMMA matrix. DMA curves from the binary and ternary blends and their nanocomposites prepared using simultaneous and sequential feeding are also presented in Supplementary Figures S1 and S2. The incorporation of organoclays into the binary blend led to additional energy dissipation, which was manifested in a decrease in the height of the tan δ peak. It was also found that out of the three components, organoclays influenced the chains mobility of PMMA to a larger extent, showing their stronger presence in this phase, 19 in agreement with our previous findings. The obtained results are also in agreement with the viscoelastic data.
Direct morphological characterizations
Morphology observations of neat ternary blend and its nanocomposite samples
It has previously been explained how phenomenological models can be helpful in predicting the morphology of PMMA/PS/PP ternary blends based on the interfacial tension. 20 The predictions of these models were confirmed by SEM observations. The SEM images of the neat blends are reproduced here as a reference, so that we can follow how the morphology evolves upon the addition of the two types of organoclays.
Figure 5(a) shows a FE-SEM micrograph of the cryofractured surface of the ternary blend sample prepared by simultaneous mixing, where a core-shell type morphology is observed. This is more clearly visible in the FE-SEM micrograph of the same sample with the PS phase etched out (by cyclohexane, 24 h), as shown in Figure 5(b). The particles surrounded by a dark hollow ring (etched region) provide evidence for the formation of core-shell type morphology, where PP cores are encapsulated by PS shells and dispersed across the PMMA matrix. We can also detect a few scattered PS droplets. Similar morphologies have been reported for PMMA/PS/PP ternary blend samples by Valera et al.
6
Field emission scanning electron microscopy micrograph of the cryofractured surface of (a) TB-0-0-Sim, (b) TB-0-0-Sim with the polystyrene shell etched out.
Figure 6 displays FE-SEM micrographs of the C30B-based nanocomposite samples prepared by simultaneous (Figure 6(a) and (b)) and sequential (Figure 6(c) and (d)) feeding modes. At 1 wt% of hydrophilic C30B (Figure 6(a) and (c)), the original core-shell type morphology of the ternary blend samples almost did not change for either feeding mode. At 3 wt%, however, only the sequential feeding sample retained a core-shell morphology, whereas that prepared by simultaneous feeding displayed a coarser morphology consisting of individual particles of PP and PS scattered across the PMMA matrix (Figure 6(b)). This suggests that at higher loadings of C30B, some of the nanoparticles end up in the core-shell droplets leading to a new network. Consequently, the enhanced solid-like behavior of the C30B-containing PS phase hinders its ability to form shells around the PP cores. Field emission scanning electron microscopy micrographs of (a) TB-0-1-Sim, (b) TB-0-3-Sim, (c) TB-0-1-Seq and (d) TB-0-3-Seq.
In the case of the sequentially mixed samples (Figure 6(c) and (d)), it is plausible that the migration of hydrophilic C30B nanoplatelets from PMMA to the core-sell particles is impeded by kinetic factors, and thus, C30B is mostly localized in the matrix. Overall, it can be suggested that localization can be directed towards a core-shell morphology using sequential feeding mode. In fact, this observation is not too far from intuition, since sequential feeding allows the initial dispersion of the nanoparticles within the matrix, to which they show a greater affinity compared to the other two components added afterwards. Therefore, in this case both kinetics and thermodynamics favor core-shell morphology (PP-PS) across a PMMA-clay nanocomposite.
As it can be seen in Figure 7(a) and (c), both nanocomposite samples containing 1 wt% of hydrophobic C15A retained their core-shell morphology (regardless of the sequence of feeding). However, in the case of the nanocomposites containing 3 wt% of this nanofiller, the morphology is no longer core-shell with coarsely dispersed, individual particles of PP and PS in the PMMA matrix (Figure 7(b) and (d)). This indicates that at higher loadings of hydrophobic C15A, a relatively larger portion of the nanoplatelets is present in the core-shell droplets, intensifying the solid-like behavior of the PS phase and completely hindering shell formation. Finally, a slighter reduction in the number of the core-shell droplets is observed with sequential mixing, which is due to the weaker presence of the nanoclays in the shell. Field emission scanning electron microscopy micrographs of (a) TB-1-0-Sim, (b) TB-3-0-Sim, (c) TB-1-0-Seq and (d) TB-3-0-Seq.
In summary, above a given nanocaly content (between 1 and 3 wt % in this case), the PS phase fails to encapsulate the PP cores due to increased elasticity. Subsequently, the number of core-shell droplets decreases. In a schematic representation, Figure 8 summarizes the effect of organoclays on the morphology of the ternary blend in the two mixing modes employed. Schematic representations of (a) TB-0-0-Sim, and the effect of organoclays on its morphology by feeding mode (b) simultaneous and (c) sequential.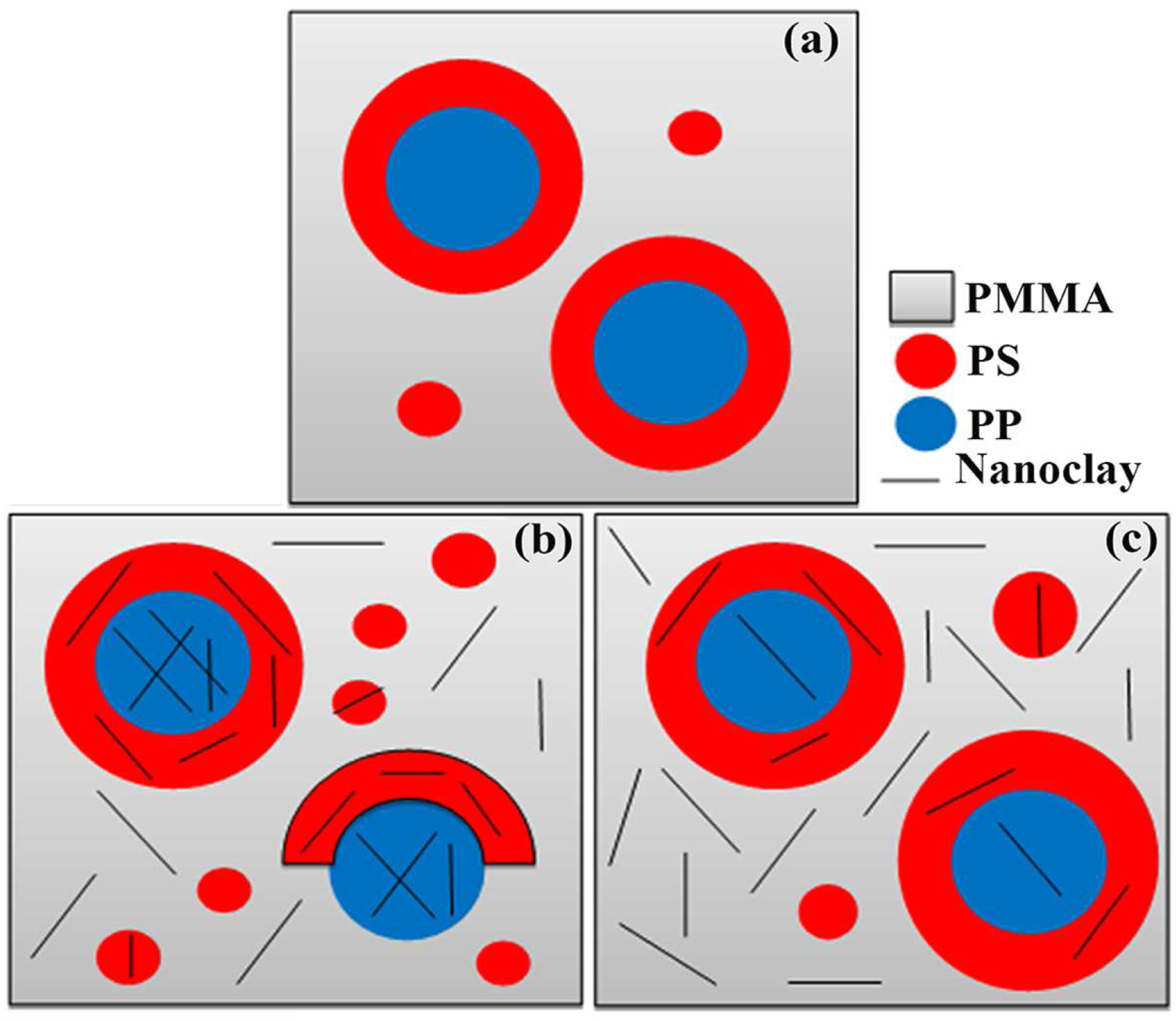
A qualitatively similar trend has been found with MWCNT-based nanocomposites based on the same ternary blend. Given the percolation threshold is typically much lower with MWCNTs, the morphology in the case of the simultaneous feeding mode was already disrupted at 1 wt%. 20 Our findings thus suggest that, regardless of the nanoparticle type, the final morphology can be predicted if the percolation threshold of the specific nanoparticle used is known.
Transmission electron microscopy images of ternary blend nanocomposites
The dispersion degree of 3 wt% of C30B and C15A in the PMMA matrix observed by TEM is shown in Figure 9(a) and (b), respectively. It is clear that both organoclays have a good degree of dispersion in the PMMA matrix,
39
in agreement with our XRD and rheology results. Transmission electron microscopy micrographs of (a) PMMA/3%C30B, (b) PMMA/3%C15A, (c) TB-0-3-Sim and (d) TB-3-0-Sim. PMMA: poly (methyl methacrylate).
Typical TEM micrographs from the ternary blend samples containing 3 wt% of hydrophilic C30B and hydrophobic C15A prepared using simultaneous feeding mode are shown in Figure 9(c) and (d), respectively. It can be seen that C15A nanoplatelets have disrupted the core-shell morphology of the ternary nanocomposites and are now present in all the blend components, while C30B is preferentially distributed in the matrix.
Conclusions
We studied the morphological evolution of PMMA/PS/PP (80/15/05) ternary blend upon the inclusion of two types of organoclays (hydrophilic C30B and hydrophobic C15A), prepared by either simultaneous or sequential feeding modes. Depending on their localization, mainly controlled by the sequence of feeding, organoclays were shown to play different roles in the morphological development of these blend nanocomposites. Microscopy observations revealed that organoclays contents above 1 wt% tend to disrupt the initial core-shell morphology, especially in the case of simultaneous feeding. Using linear rheology, we found that the effect of organoclays’ localization on the elasticity ratio and the interfacial tension between the blend components was responsible for these observations. This offers a simple means of predicting the localization behavior of nanoclay particles in ternary polymer blends. Intercalated/exfoliated organoclays intensified the solid-like behavior of the PS phase, which hindered the formation of a PS shell in samples prepared using simultaneous feeding. The results of this work highlight the importance of considering both thermodynamic and kinetic parameters in tuning the morphology and thermomechanical properties of ternary polymer blends.
Supplemental Material
Supplemental Material - Morphology and physico-mechanical properties of poly (methyl methacrylate)/polystyrene/polypropylene ternary polymer blend and its nanocomposites with organoclay: The effect of nature of organoclay and method of preparation
Supplementary Material for Morphology and physico-mechanical properties of poly (methyl methacrylate)/polystyrene/polypropylene ternary polymer blend and its nanocomposites with organoclay: The effect of nature of organoclay and method of preparation by Amir Rostami, Mehdi Vahdati, Mohsen Nowrouzi, Mohammadreza Karimpour, and Amir Babaei in Polymers and Polymer Composites.
Footnotes
Author’s note
Mehdi Vahdati is now affiliated with the University of Strasbourg, CNRS, Institut Charles Sadron, UPR 22, Strasbourg, France.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
