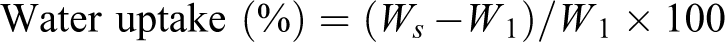Abstract
Wood polymer composite (WPC) was prepared using solution-blended high-density polyethylene (HDPE), low-density polyethylene (LDPE), polypropylene (PP), poly(vinyl chloride) (PVC), wood flour (WF) and polyethylene-co-glycidyl methacrylate (PE-co-GMA). The effect of nanoclay, SiO2 and ZnO addition on the properties of the composite was examined. The distribution of silicate layers, SiO2 and ZnO nanopowder was studied by X-ray diffraction (XRD) and transmission electron microscopy (TEM). The improvement in miscibility among polymers was studied by scanning electron microscopy (SEM). Fourier transform infrared spectroscopic (FTIR) studies reveal the interaction between polymer, wood, clay, SiO2 and ZnO. WPC treated with 3 phr each of clay, SiO2 and ZnO showed an improvement in mechanical properties, thermal stability and a decrease in water uptake capacity.
Introduction
Now a days, wood polymer composite (WPC) occupies a significant part in the field of composite. 1 It is used in all classes of materials ranging from household items to different exterior products viz. automobiles industries, different outdoor and indoor applications like decking, railing, fencing, docks, landscaping timbers, and many more. 2 They have relatively high strength and stiffness, low cost, low density, low CO2 emission, and are biodegradable and renewable. Moreover, they can also be used widely with other materials to improve various properties.
Nal (Phragmites karka), a conventional plant, is widely available as biowaste in the forest of Assam, India. They can be made into value-added material for the preparation of structural components by making composites with plastic materials. Sui et al. 3 has prepared WPC using nonconventional plant like sunflower hull sanding dust (SHSD) as the reinforcing agent and polypropylene (PP) as the matrix.
The disposal of plastic materials in the form of carry bags, boxes and packaging film causes a serious threat to the environment. Recycling and reusing is one of the processes to reduce this environmental pollution. But the poor mechanical properties of recycled plastic materials restrict their use for different applications. 4 These properties can be improved substantially if composites are made by combining these waste materials with cellulosic materials.
In order to improve the miscibility between organic polymer and inorganic wood fiber, certain compatibilizers are used. The compatibilizer enhances interaction between hydrophilic wood fibers and hydrophobic polymers and at the same time improves the interfacial adhesion among different thermoplastic materials. The uses of glycidyl methacrylate 5 and PE-co-GMA 6 as compatibilizer for making WPC have been reported.
Solution blending is one of the process used for blending of different kinds of plastics. The effectiveness of the blending process can be improved using a mixture of solvents. Although solution blending involves the use of some hazardous solvents, it is still useful in many cases. A high level of intercalation with partial exfoliation of nanofillers has been obtained only for solution blending process. 7 Chiu et al. 8 observed good dispersion of silicate layers when they studied PP/clay composite by solution blending method. Melt blending suffers from some disadvantages like stress development, temperature, and so on. 9 So, in order to obtain a good property development, it is assigned to develop the composite by solution blending method.
Nanocomposite technology with layered silicate as reinforcing phase has been the subject matter of many discussions in recent years. Due to the high aspect ratio of silicate nanolayers, it is ideal for reinforcement and enhancing the properties. 10 The performance of wood/polypropylene composite has been found to improve by the addition of nanoclay. 11
In the polymer composites, different types of metal oxide nanoparticles such as SiO2, TiO2, ZnO, and so on are widely used for improving the thermal, mechanical as well as other properties. These are nontoxic, stable and highly thermostable inorganic filler. Polymer-SiO2 and ZnO composites have been explored as technologically important due to their potential applications in electrochromic windows, fuel cells, chemical separation, electrochemical sensing, solar cells and in paint industries. 12 –14 Most of the cited reports have addressed the use of single nanoparticle in the preparation of composite. To the best of our knowledge, there are no reports regarding the use of multiple nanoparticles for making composites.
The present study is aimed to discuss the effect of SiO2 and ZnO nanopowder along with nanoclay on the thermal and mechanical properties of high-density polyethylene (HDPE)/low-density polyethylene (LDPE)/polypropylene (PP)/poly(vinyl chloride) (PVC) blend and wood flour (WF). The aim is also to study the effect of these nanoparticles on other properties like hardness and water uptake capacity of the composites.
Experimental
Materials
Both HDPE and LDPE (grade: PE/20/TK/CN) were obtained from Plast Alloys India Ltd. (Harayana, India). PP homopolymer (grade: H110MA, MFI 11 g/10 min) and PVC (grade: SPVC FS: 6701) were purchased from Reliance Industries Ltd. (Mumbai, India) and Finolex Industries Ltd. (Pune, India), respectively. The compatibilizer poly(ethylene-co-glycidyl methacrylate) (PE-co-GMA) and N-cetyl-N, N, N-trimethyl ammonium bromide (CTAB) were supplied by Otto chemicals, Mumbai, India, and Central Drug house (P) Ltd, Delhi, India, respectively. Nanomer (clay modified by 15–35 wt.% octadecylamine and 0.5–5 wt.% aminopropyltriethoxy silane; Sigma-Aldrich, USA), SiO2 nanopowder (5–15 nm, 99.5% trace metals basis; Aldrich, China) and ZnO nanopowder (<100 nm; Aldrich, Germany) were used as received. Nals (Phragmites karka) a type of nonconventional wood was collected from the local forest of Assam. Other reagents used were of analytical grade.
Preparation of wood samples
Nals (Phragmites karka) is available in the forest of Assam. It was collected and chopped into small strips. These were initially washed with 1% soap solution followed by washing with 1% NaOH solution and finally with cold water. The washed wood strips were oven-dried at 100 ± 5°C till the attainment of constant weight. These dried wood strips were grinded in a mixer, sieved and kept for subsequent uses.
Modification of SiO2 and ZnO
Ten grams each of SiO2 and ZnO was taken separately in two round-bottom flasks containing 1:1 ethanol–water mixture. It was stirred at 80°C for approximately 12 h; 12 g each of CTAB was taken in two separate beakers containing ethanol–water mixture and stirred at 80°C for 3 h. These mixtures were added slowly to the two flasks containing SiO2 and ZnO mixture under stirring condition. The stirring was continued for 6 h. The mixtures from both the flasks were then filtered separately and washed with deionized water for several times. It was collected, dried in vacuum oven at 45°C, grinded, stored in separate ampule and kept in desiccator to avoid moisture absorption.
Preparation of wood polymer nanocomposite
The minimum ratio of xylene and THF, at which a homogenous solution of HDPE, LDPE, PP and PVC was obtained, was optimized as 70:30. Six grams each of HDPE, LDPE and PP (1:1:1) were added slowly to 105 ml of xylene taken in a flask fitted with a spiral condenser at room temperature. This was followed by the addition of the PE-co-GMA (5 phr). The temperature of the flask was increased from room temperature to 130°C in order to make a homogenous solution. Now, another solution containing 3 g of PVC in 35 ml of tetrahydrofuran (THF) was prepared. The temperature of the polymer solution containing HDPE, LDPE and PP was brought down to 120°C. To this, PVC solution was added gradually and stirred continuously at 120°C (approximately) for 1 h. A known quantity of CTAB-modified SiO2 nanopowder (1–5 phr) and ZnO (1–5 phr) was dispersed in 15 ml of THF solution by sonication. This dispersed mixture was added gradually to the polymer solution under stirring condition. Oven-dried WF (40 phr) was added slowly to this solution. The whole mixture was stirred for another 1 h. The mixture was transferred in a tray, dried and grinded. The composite sheets were obtained by the compression molding press (Santec, New Delhi) at 150°C under a pressure of 80 MPa.
Polymer blend (HDPE + LDPE + PP + PVC), polymer blend/5 phr PE-co-GMA and polymer blend/5 phr PE-co-GMA/40 phr wood were designated as PB, PB/G5 and PB/G5/W40. WPC filled with 3 phr nanoclay and 1, 3 and 5 phr each of SiO2 and ZnO were designated as PB/G5/W40/N3/S1/Z1, PB/G5/W40/N3/S3/Z3 and PB/G5/W40/N3/S5/Z5.
Measurements
X-ray diffraction
The degree of nanoclay, SiO2 and ZnO distribution in the WPC was evaluated by X-ray diffraction (XRD) analysis. It was carried out in a Rigaku X-ray diffractometer (Miniflax, UK) using CuKα (λ = 0.154 nm) radiation at a scanning rate of 1° per min with an angle ranging from 2° to 70°.
Transmission electron microscopy
The dispersion of the silicate layers of nanoclay and SiO2/ZnO nanoparticles in WPCs was examined using transmission electron microscope ([TEM] JEM-100 CX II) at an accelerated voltage of 20–100 kV.
Scanning electron microscopy
The compatibility among different polymers as well as the morphological features of the WPC was studied using scanning electron microscope ([SEM] JEOL JSM–6390LV) at an accelerated voltage of 5–10 kV. Fractured surface of the samples, deposited on a brass holder and sputtered with platinum, were used for this study.
Fourier transform infrared spectroscopic studies
Fourier transform infrared spectroscopic (FTIR) spectra of WF, nanoclay, SiO2, ZnO nanopowder and WPC loaded with nanoclay/SiO2/ZnO nanopowder were recorded in FTIR spectrophotometer (Impact-410, Nicolet, USA) using KBr pellet.
Mechanical property
The tensile and flexural tests for polymer blend, PE-co-GMA treated polymer blend, and WPC loaded with different percentage of SiO2 and ZnO were carried out in a Universal Testing Machine (Zwick, model Z010) at a crosshead speed of 10 mm min−1 at room temperature, according to ASTM D-638 and D-790, respectively. Eight samples of each category were tested and their average values were reported.
Statistical analysis
All the data are expressed as means ± SD. All mechanical property results were statistically analyzed using one-way analysis of variance (ANOVA) followed by Tukey’s honestly significant difference (HSD) test. (All data have been given as supplementary file.)
Hardness
The hardness of the samples were measured according to ASTM D-2240 using a durometer (model RR12) and expressed as shore D hardness.
Thermal property
Thermal properties of polymer blend and the WPCs were measured in a thermogravimetric analyzer (TGA; TGA-50, Shimadzu, USA) at a heating rate of 10°C min−1 up to 600°C under nitrogen atmosphere at a flow rate of 30 ml m−1.
Water uptake test
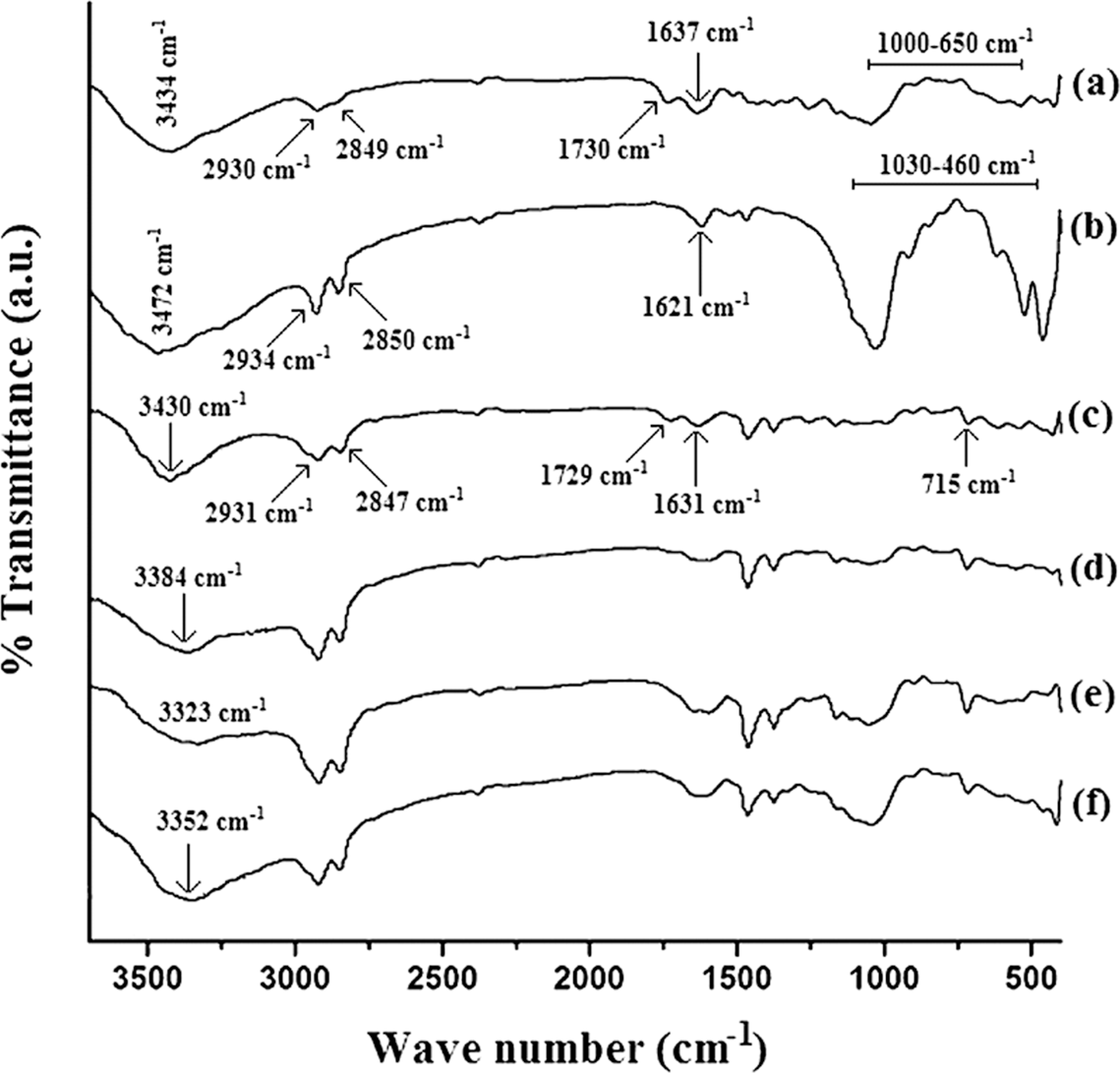
WPC samples were cut into 2.5 × 0.5 × 2.5 cm3 for the test. Percentage water uptake was measured by submerging the samples in distilled water at room temperature (30°C) for different time periods after conditioning at room temperature (30°C) and 65% relative humidity. It is expressed according to the formula,
where Ws is the weight of the water saturated specimen and W 1 is the weight of the oven-dried specimen.
Results and discussion
XRD study
Figure 1 shows the XRD results of nanoclay, polymer blend, SiO2, ZnO and WPC loaded with clay (3 phr) and 1–5 phr each of SiO2 and ZnO. The diffraction peak appeared at 2θ = 4.11° with basal spacing of 2.15 nm for the organically modified nanoclay (curve a). Curve b represents the diffractrogram of polymer blend. The most prominent wide-angle XRD peaks that appeared at 2θ = 14.12 (200), 17.06 (040), 18.64 (211), 21.62 (110) and 24.02 (200) were for crystalline portion of different polymers present in the blend. 15 –17 Curve c shows a broad diffraction peak at 2θ = 23.5 for the amorphous SiO2 nanoparticles. The crystalline peaks appeared above 2θ = 30° (shown in curve d) were the characteristic peaks of ZnO nanopowder. 18 Curves e–g were for WPC loaded with nanoclay (3 phr) and 1–5 phr each of SiO2 and ZnO (1:1 mixture). The diffractograms of composites did not exhibit any characteristic peak of nanoclay, reflecting the delamination or exfoliation of silicate layers. With the increase in the level of incorporation of SiO2 and ZnO (1–5 phr), the crystalline peak intensity of the polymer blend that appeared in the range 2θ = 14–25° was found to decrease. Although the presence of amorphous portion of SiO2 reduced the peak intensities of ZnO nanopowder but still the intensities of peak corresponding to ZnO nanopowder increased with the increase in the concentration of ZnO in the composite. Similar effect of SiO2 on peak intensity was observed by Bao and Ma 19 while studying the XRD profile of exfoliated Na-MMT and SiO2 on polymethacrylic acid composite. From the study, it could be concluded that nanoclay layers were delaminated and SiO2 and ZnO particles were dispersed in the wood polymer matrix.
X-ray diffraction of (a) nanoclay, (b) polymer blend (PB), (c) nano-SiO2, (d) nano-ZnO, (e) PB/G5/W40/N3/S1/Z1, (f) PB/G5/W40/N3/S3/Z3, (g) PB/G5/W40/N3/S5/Z5.
TEM study
Figure 2 shows the TEM micrographs of WPC loaded with nanoclay (3 phr) and 1–5 phr each of SiO2 and ZnO. The dark lines correspond to the layers of nanoclay while the dark spots were for SiO2 and ZnO nanoparticles. The nanoparticles were found to be well distributed in the WPC till the addition of 3 phr each of SiO2 and ZnO beyond which a tendency to agglomeration (5 phr) was observed as shown in Figure 2(c). Park et al. 20 prepared nanosilica incorporated polypropylene/polypropylene elastomer blend and found that the silica nanoparticles were well dispersed within the blend. At higher concentrations of SiO2 and ZnO, the distances between the particles became less and this might have enhanced the tendency for agglomeration.

Transmission electron microscope (TEM) micrographs of (a) polymer blend (PB)/G5/W40/N3/S1/Z1, (b) PB/G5/W40/N3/S3/Z3, and (c) PB/G5/W40/N3/S5/Z5.
SEM study
SEM micrographs of polymer blend with and without compatibilizer, WPC and WPC loaded with nanoclay and mixture of different percentage of SiO2 and ZnO nanopowder are shown in Figure 3. Figure 3(a) and (b) shows the miscibility among different polymers before and after the addition of compatibilizer. The improved miscibility among polymers as shown by decreased segregation or roughness was due to the improvement in interfacial adhesion among polymers by the compatibilizer. 21 The addition of WF improved the smoothness of composites (Figure 3(c)). It increased further after the incorporation of WF, clay, SiO2 and ZnO to the polymer blend (Figure 3(d) to (e)). The surface smoothness of composites having 3 phr each of SiO2 and ZnO and nanoclay appeared to be more compared to those of the composites prepared with either 1 or 5 phr each of SiO2 and ZnO mixture and nanoclay. This might be due to the improvement in interaction among PE-co-GMA, nanoclay, SiO2, ZnO, wood and polymer. The decrease in smoothness at higher concentrations of SiO2 and ZnO mixture was due to the aggregation of SiO2 and ZnO. Wang et al. 22 observed the agglomeration of SiO2 nanoparticles at higher loading while developing poly (methyl methacrylate)-SiO2 nanocomposite film.

Scanning electron microscope (SEM) micrographs of (a) polymer blend (PB), (b) PB/G5, (c) PB/G5/W40, (d) PB/G5/W40/N3/S1/Z1, (e) PB/G5/W40/N3/S3/Z3 and (f) PB/G5/W40/N3/S5/Z5.
FTIR study
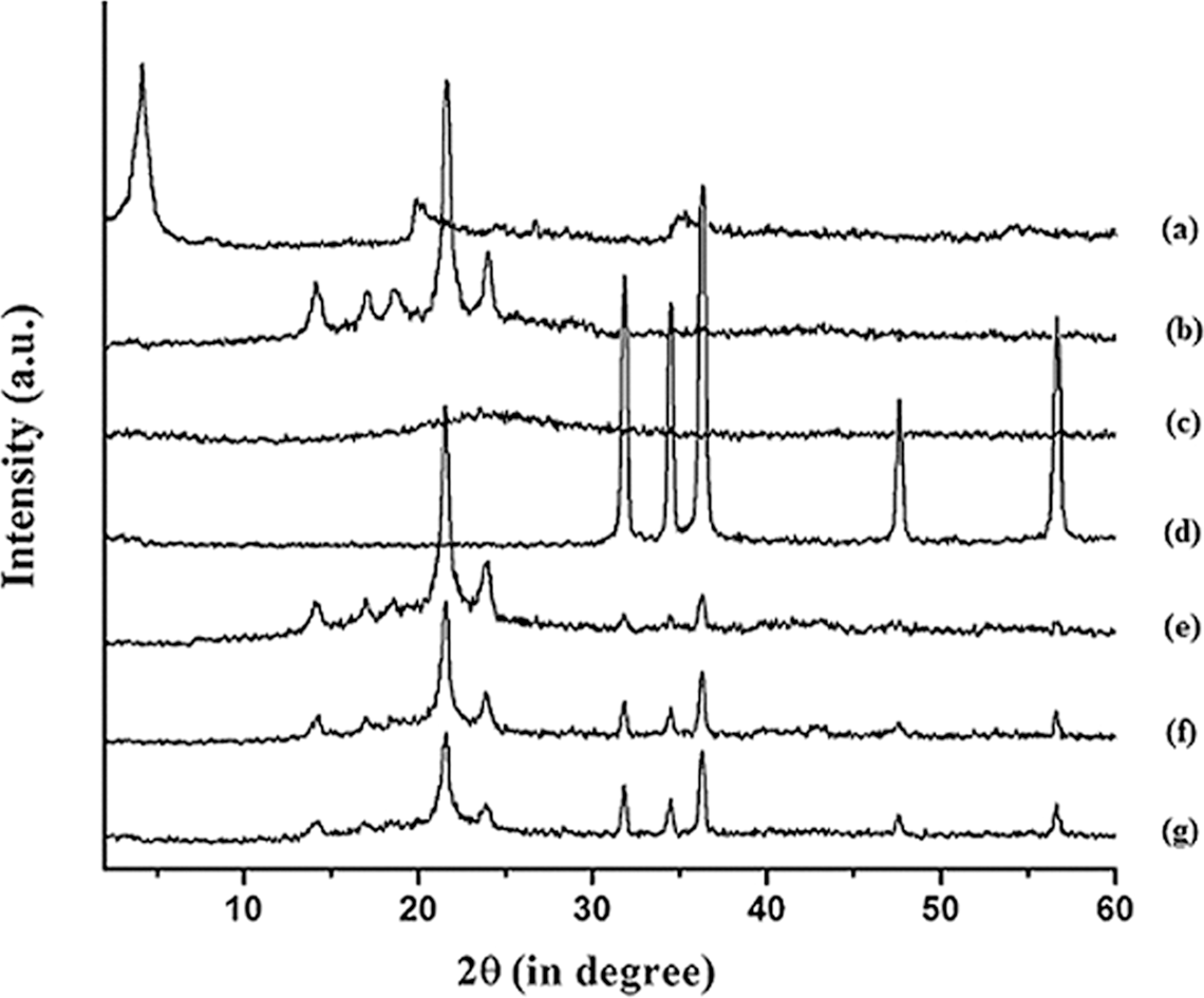
The FTIR spectra of CTAB, SiO2, CTAB-modified SiO2, ZnO and CTAB-modified ZnO are shown in Figure 4. In the spectrum of CTAB (Figure 4(a)), the absorption peaks at 2917 and 2847 cm−1 were assigned to asymmetric and symmetric stretching modes of –CH2 in the methylene chains. Presence of peaks at 1491, 1475 and 1396 cm−1 were for the asymmetric and symmetric CH3–N+ deformation modes of the CTAB head group, respectively. 23 The spectrum of unmodified SiO2 (Figure 4(b)) shows absorption peaks at 3472 and 1633 cm−1 for –OH stretching and –OH bending of hydroxyl group adsorbed on the particle surface. The other peaks that appeared in the range 1088–464 cm−1 were due to Si–O–Si group in the SiO2. 24 The peaks around 3424, 2931, 2852, 1633 and 1481 cm−1 and 1088–464 cm−1 were found on the spectrum of CTAB-modified SiO2 (Figure 4(c)). The intensity of –OH stretching in the modified SiO2 was found to decrease and shift toward lower wave number, indicating an interaction of the hydroxyl group absorbed on SiO2 surface with CTAB. The other characteristic peaks for CTAB were also present in the curve. FTIR spectrum of unmodified ZnO nanoparticles is presented in Figure 4(d). Absorption peaks at 3497 and 1635 cm−1 in the spectra was due to –OH (-stretching) and –OH (-bending) vibrations, while the peaks around 422 cm−1 was due to the vibration of metal–oxygen (M-O) bond as reported in the literature. 25 The FTIR spectrum of CTAB-modified ZnO is represented by the curve 4e. It was observed that the intensity of the peak for –OH group decreased to a desired extent. Besides this, two new peaks at 2918 and 2848 cm−1, which were due to the presence of –CH2 group of CTAB that appeared in the spectrum. The peaks at 1496 and 1476 cm−1 were due to the asymmetric CH3–N+ deformation mode of the CTAB head group. Another peak present at 1397 cm−1 was due to the symmetric CH3–N+ deformation mode of CTAB head group. These results indicated that long chain of CTAB group had been incorporated on the surface of the both SiO2 and ZnO nanoparticles. Further, both CTAB-modified SiO2 and ZnO were dispersed in a flask containing xylene and kept for checking of any particle settling. The dispersion of SiO2 and ZnO were found to be stable. This indicated that proper modification occurred.
Fourier transform infrared spectroscopic (FTIR) spectra of (a) N-Cetyl-N, N, N-trimethyl ammonium bromide (CTAB), (b) unmodified SiO2, (c) CTAB-modified SiO2, (d) unmodified ZnO and (e) CTAB-modified ZnO.
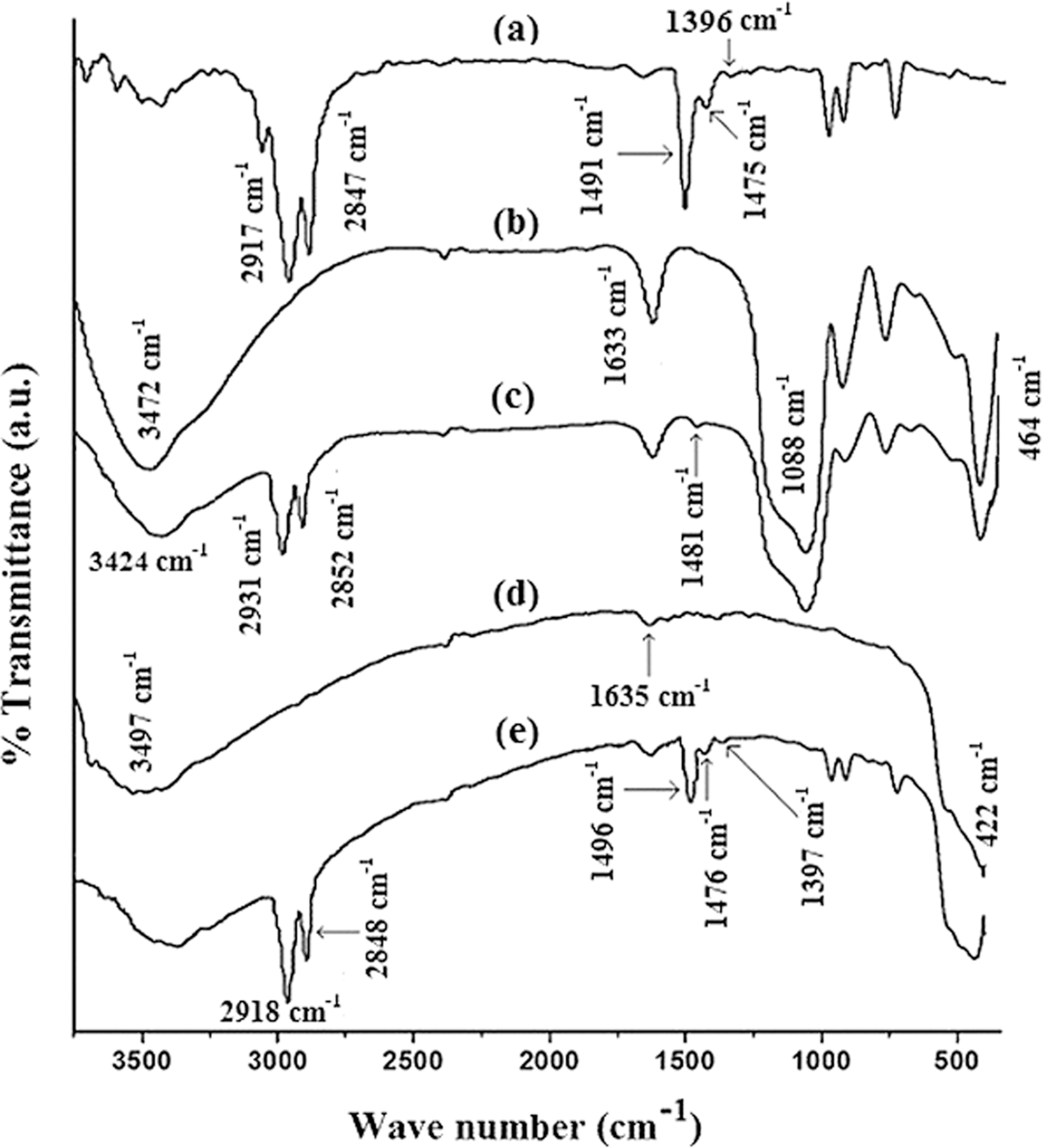
Figure 5 shows the FTIR spectra of wood, nanoclay, WPC and WPC loaded with nanoclay and different percentage of SiO2 and ZnO mixture. Curve a representing the wood sample shows the presence of bands at 3433 cm−1 for –OH stretching, 2930 and 2849 cm−1 for –CH stretching, 1730 cm−1 for C=O stretching, 1637 cm−1 for –OH bending, 1161 and 1045 cm−1 for C–O stretching and 1000–650 cm−1 for C–H bending vibration (out of plane). Organically modified nanoclay (curve b) exhibits the peaks at 3472 cm−1 (–OH stretching), 2934 and 2850 cm−1 (–CH stretching of modified hydrocarbon), 1621 cm−1 (–OH bending) and 1030–460 cm−1 (oxide bands of metals like Si, Al, Mg, etc.). PB/G5/W40 (curve c) shows peaks at 3430 cm−1 (–OH stretching), 2931 and 2847 cm−1 (–CH stretching), 1729 cm−1 (C=O stretching), 1631 cm−1 (–OH bending) and 715 cm−1 (–CH2 stretching).
Fourier transform infrared spectroscopic (FTIR) spectra of (a) Wood, (b) nanoclay, (c) PB/G5/W40, (d) PB/G5/W40/N3/S1/Z1, (e) PB/G5/W40/N3/S3/Z3 and (f) PB/G5/W40/N3/S5/Z5.
Figure 5(d) to (f) represent the FTIR spectra of WPC loaded with 3 phr nanoclay and 1, 3 and 5 phr each of CTAB-modified SiO2 and ZnO. From the figure, it was observed that the intensity of –OH stretching was decreased and shifted to 3384 cm−1 (curve d), 3323 cm−1 (curve e) and 3352 cm−1 (curve f) from 3434 cm−1 (wood). The decrease in intensity and shifting to lower wave number confirmed the bond formation between polymer and the hydroxyl groups of wood, nanoclay, SiO2 and ZnO. A similar decrease in the intensity of –OH absorption peak and shifting to lower wavelength was reported by Deka and Maji. 26 Moreover, the intensity of –CH stretching peaks at 2930 cm−1 and 2849 cm−1 (curves d–f) was found to increase. Similar increase in –CH peak intensities was observed by Awal et al. 27 The peak intensities (1088–464 cm−1) of metal oxide bonds of SiO2, ZnO and nanoclay (curves d–f) decreased to a considerable extent. All these suggested a strong interaction between wood, nanoclay, SiO2, ZnO and polymer.
Mechanical property
Table 1 shows the flexural and tensile properties of polymer blends and WPC loaded with nanoclay and different percentages (1:1) of SiO2 and ZnO nanopowder. After the incorporation of compatibilizer, both flexural and tensile properties of the polymer blend increased. This was because of the increase in interfacial adhesion between the polymers due to which both flexural and tensile properties increased. The flexural and tensile properties of the composite were further improved after the addition of WF. The WF acted as a load carrier, reinforced the composites and increased the flexural and tensile properties. Nourbakhsh et al. 28 developed WF/polypropylene composite and found an improvement in mechanical properties due to reinforcement by WF. Moreover, the compatibilizer, PE-co-GMA, provided strong interfacial adhesion between wood and polymers through its glycidyl linkage and long olefinic chain. Sailaja 29 used PE-co-GMA as compatibilizer to improve the compatibility of wood pulp-LDPE composite. The properties of the WPCs were further improved after the incorporation of clay, SiO2 and ZnO nanopowder. At a fixed clay loading (3 phr), both flexural and tensile properties improved till the addition of 3 phr each of SiO2 and ZnO. At higher amount of SiO2 and ZnO (5 phr each) loading, the properties were found to decrease. The observed higher values might be due to the combined effect of nanoclay, SiO2 and ZnO. The silicate layers of nanoclay acted as a reinforcing agent and the long polymer chains inserted inside the gallery space restrict the mobility of the polymer chain. Lei et al. 30 found an increase in mechanical properties of wood/HDPE composite after the incorporation of nanoclay. CTAB-modified SiO2 and ZnO improved the interaction between clay, wood and polymer through its surface hydroxyl group and cetyl group, respectively. Higher the dispersion of nanoparticles, higher would be the interaction. At higher level of SiO2 and ZnO loading (5 phr each), the agglomeration occurred and hence the interaction of SiO2 and ZnO with the clay, polymer and wood decreased. Liu and Kontopoulou 31 observed an increase in the mechanical properties of thermoplastic olefin/nanosilica composite at lower loading of nanosilica. The increase in tensile modulus of polyacrylonitrile due to the interaction of ZnO nanoparticles was reported in the literature. 32
Flexural, tensile and hardness properties of polymer blend and WPC loaded with nanoclay and different percentages of SiO2 and ZnO.a
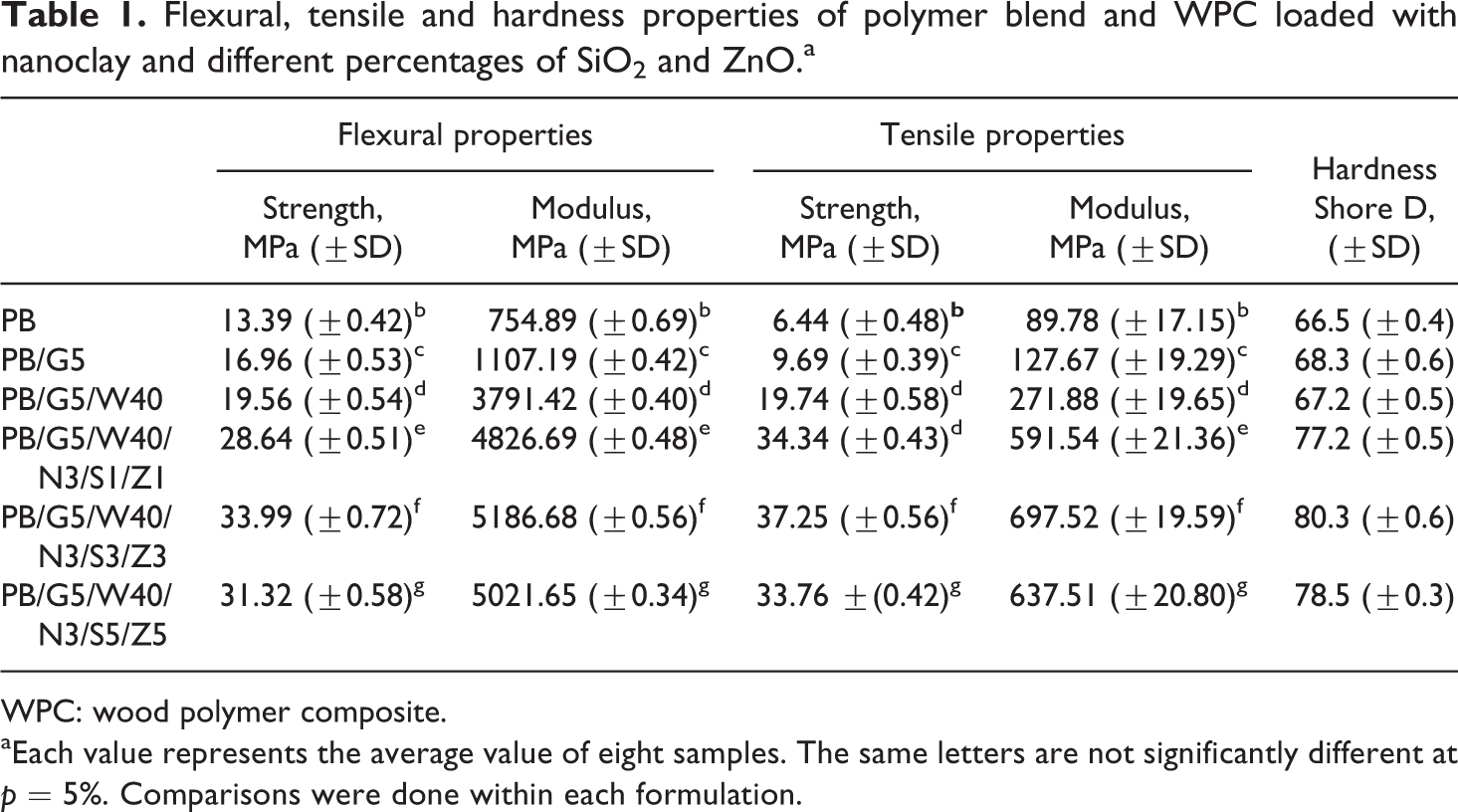
WPC: wood polymer composite.
aEach value represents the average value of eight samples. The same letters are not significantly different at p = 5%. Comparisons were done within each formulation.
Hardness results
Table 1 shows the hardness results of polymer blend and WPC loaded with clay and different percentage of SiO2 and ZnO. Addition of compatibilizer increased the hardness value of the polymer blend. The hardness value was found to decrease on the addition of WF to the polymer blend. But after the incorporation of nanoclay, SiO2 and ZnO, the hardness value of the WPC was found to increase again. The improvement was due to the combined effect of decrease in mobility of intercalated polymer chain and increase in reinforcing action of SiO2 and ZnO. At a fixed clay concentration (3 phr), the hardness value was found to be maximum at 3 phr each of SiO2 and ZnO loading but decreased after that. The reason for this was stated as earlier.
TGA
Initial decomposition temperature (Ti ), maximum pyrolysis temperature (Tm ), decomposition temperature at different weight loss (%; TD ) and residual weight (RW, %) for polymer blend and WPCs are shown in Table 2. Ti value of the polymer blend increased after the incorporation of compatibilizer and WF. The addition of nanoclay, SiO2 and ZnO nanoparticles further increased the Ti value. It was found to be maximum at 3 phr loading of nanoclay and at 3 phr each of SiO2 and ZnO loading. At higher concentrations of SiO2 and ZnO, the Ti value decreased again.
Thermal analysis of polymer blend and WPC loaded with nanoclay, SiO2 and ZnO.
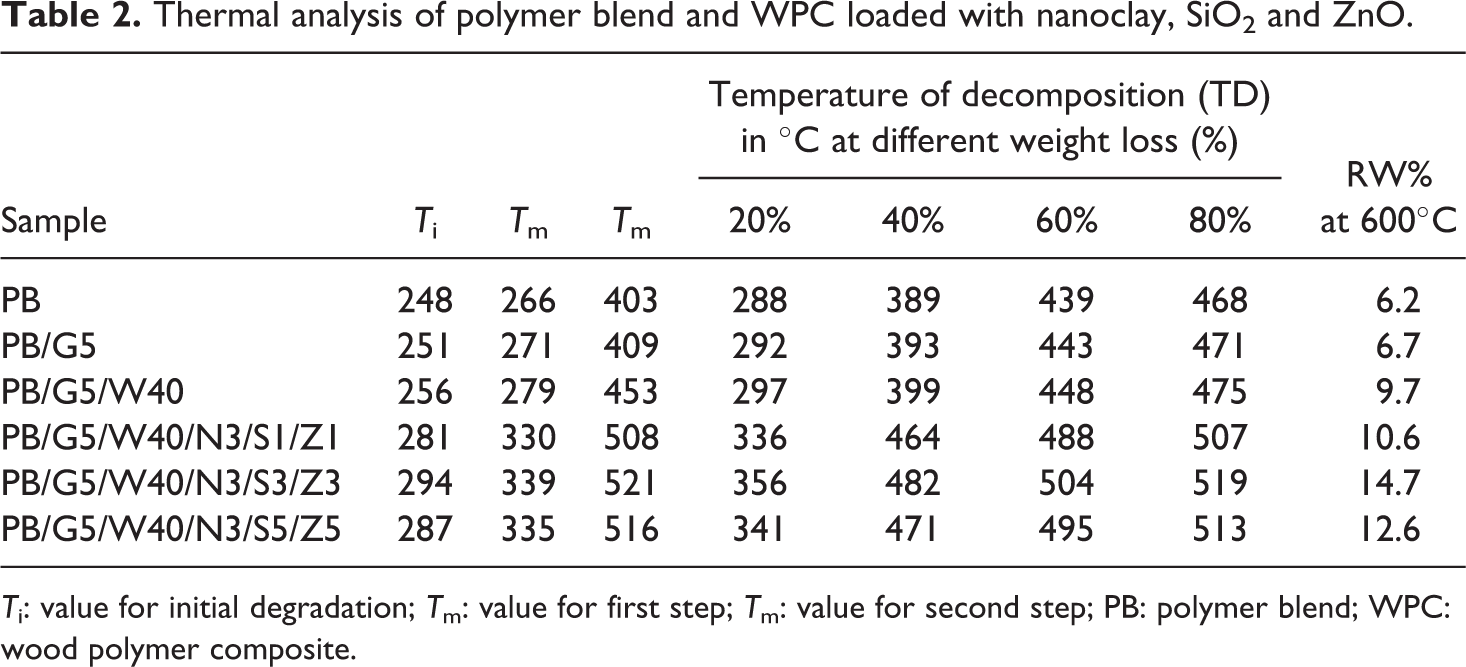
T i: value for initial degradation; T m: value for first step; T m: value for second step; PB: polymer blend; WPC: wood polymer composite.
Both the polymer blend and the composites showed two decomposition peaks. The Tm value for the first step in both WPC and WPC loaded with clay, SiO2 and ZnO was due to the depolymerization of hemicellulose, glycosidic linkage of cellulose, thermal decomposition of cellulose 33 and dehydrochlorination of PVC, while the peak for second step was due to decomposition of HDPE and PP. 34 –36 The Tm values of the composites for both the steps were found to follow the similar trend as those of Ti values.
The percentage weight loss in samples at different temperatures is listed in Table 2. TD values of composite containing nanoclay, SiO2, ZnO were more compared to either polymer blend or polymer/wood composites. The higher the percentage of ZnO and SiO2, the higher was the decomposition temperature.
The compatibilizer enhanced the interaction between the polymer blend and WF and as a result the thermal stability improved. Araujo et al. 37 reported the improvement in thermal stability after inclusion of compatibilizer to HDPE and natural fiber composites. The silicate layers present in nanoclay of the composites provided a long tortuous path which delayed the diffusion of the decomposed volatile products through the composite. The improvement in thermal stability of WF and polypropylene composite after addition of nanoclay was reported by Ashori et al. 38 Moreover, the incorporation of CTAB-modified SiO2 and ZnO nanopowder might play a role in enhancing the thermal stability by interacting with clay, wood and polymer through its surface hydroxyl group and cetyl groups, respectively. At higher loading of SiO2 and ZnO, the interaction of SiO2, ZnO and clay with wood and polymer might decrease due to agglomeration and hence a reduction in thermal stability was observed. Bailly and Kontopoulou 39 developed thermoplastic olefin/nanosilica composite and found an increase in thermal properties due to the incorporation of nanosilica. The increase in thermal stability was also reported by He et al. 40 while studying the thermal behavior of PET-ZnO nanocomposite.
Water uptake results
The water uptake of polymer blend, PE-co-GMA-treated polymer blend and WPC loaded with clay and different percentages of SiO2 and ZnO are shown in Figure 6. On addition of PE-co-GMA compatibilizer, the water uptake capacity of the polymer blend decreased. The decrease in the water uptake capacity of PE-co-GMA-treated polymer blend was due to the increase in interfacial adhesion between the polymers by the compatibilizer. After the addition of WF to the blend, the water uptake capacity increased suddenly. The hydrophilic nature of WF caused an increase in the water uptake capacity. The water uptake was again decreased after the addition of clay, SiO2 and ZnO nanoparticle. WPC loaded with 3 phr each of clay, SiO2 and ZnO showed lowest water uptake followed by WPC with 3 phr clay and 5 phr each of SiO2 and ZnO, and WPC with 3 phr clay and 1 phr each of SiO2 and ZnO. The silicate layers of the clay provided tortuous path and increased the barrier property for water transport. Deka and Maji 5 obtained similar increase in barrier properties of WPC after the incorporation of clay. The presence of SiO2 and ZnO nanopowder also provided a barrier to the passage of water. The strong affinity of water molecules toward nano-SiO2 and ZnO particles restricted its free motion and reduced the diffusion coefficient of water. The distribution nature of modified nanoparticles further improved the resistance and retarded the motion of water molecules through the composites. Moreover, the water absorption of inorganic particles reduced after the modification of particles by organic surfactant. Better the distribution of nanoparticles, the higher was the barrier property. The incorporation of SiO2 nanoparticles increased the water barrier properties of polyvinyl alcohol–based hybrid coatings. 41 The agglomeration of nanoparticles took place at higher concentration SiO2 and ZnO nanoparticles and as a result increased water uptake capacity was observed.
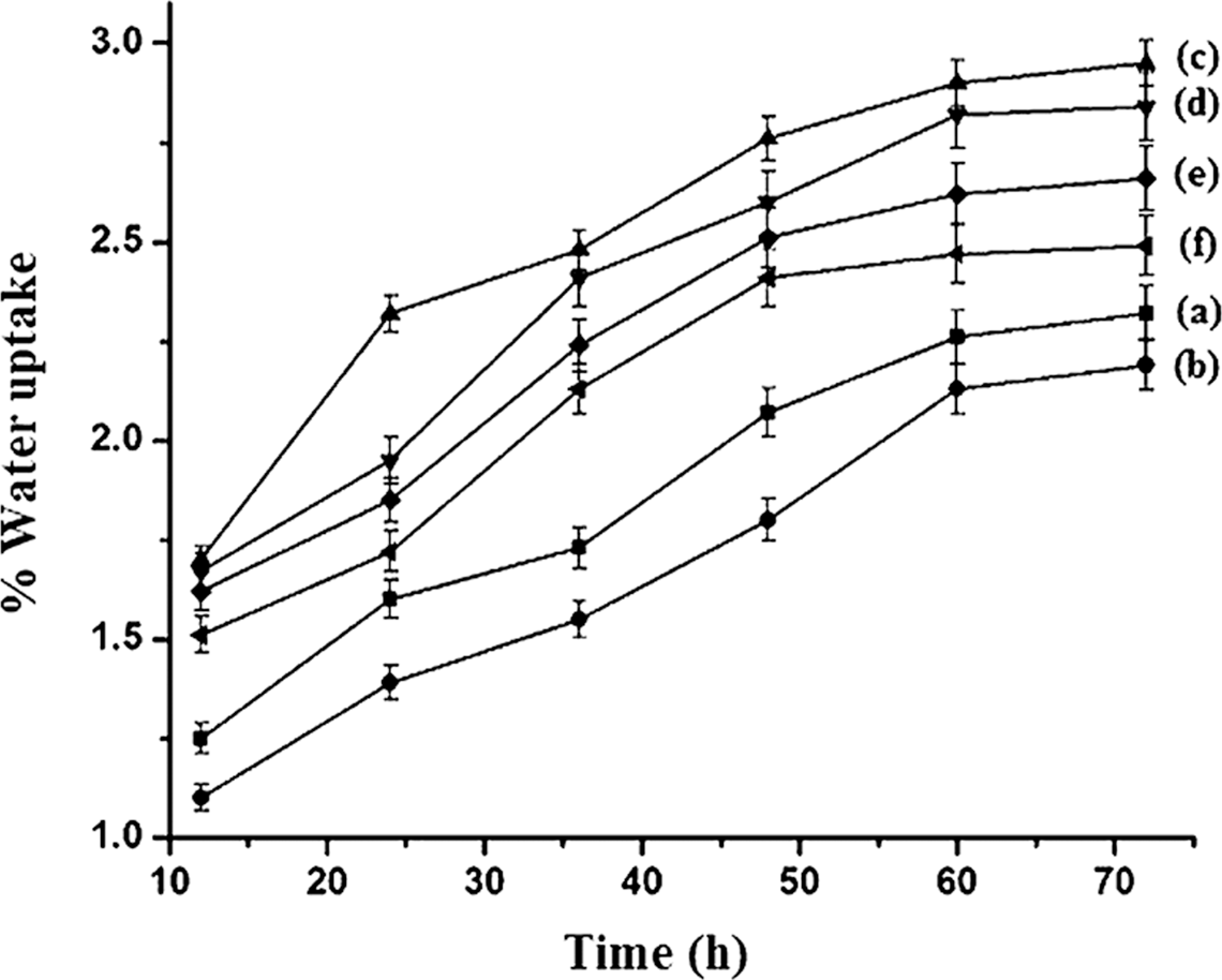
Water absorption of (a) polymer blend (PB), (b) PB/G5, (c) PB/G5/W40, (d) PB/G5/W40/N3/S1/Z1, (e) PB/G5/W40/N3/S5/Z5 and (f) PB/G5/W40/N3/S3/Z3.
Conclusions
The optimized ratio of xylene and THF for solution blending of HDPE, LDPE, PP and PVC (1:1:1:0.5) was 70:30. The compatibility among the polymers and WF was improved using PE-co-GMA compatibilizer as revealed by SEM study. The distribution of silicate layers of nanoclay and SiO2/ZnO in WPCs was examined by XRD and TEM study. The shifting of peak corresponding to –OH stretching to lower wave number and decreased intensity confirmed the reaction between polymer, wood, SiO2, ZnO and nanoclay. Surface modification of SiO2 and ZnO nanoparticles by cationic surfactant CTAB was examined by FTIR studies. The interactions among wood, PE-co-GMA, SiO2, ZnO and nanoclay were also studied by FTIR. At higher concentration of SiO2 and ZnO loading, the particles became agglomerated. WPC loaded with nanoclay, SiO2 and ZnO enhanced the mechanical, hardness, thermal properties and reduced the water absorption capacity. WPC loaded with 3 phr each of clay, SiO2 and ZnO exhibited maximum improvement in properties.
Footnotes
Funding
This work was supported by Council of Scientific and Industrial Research (CSIR), New Delhi (grant number: 01(2287)/08/EMR-II).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.