Abstract
Hydrodegradable material as the replacement for plastic could be a better option than biodegradable material due to its solubility in water, which could extend the landfill’s lifespan. On the other hand, biodegradable material requires tightly controlled waste management and proper facilities for degradation purposes. The starch film is highly soluble in water, exhibiting poor mechanical strength. This study aims to improve the strength of starch film by adding cellulose, which was isolated from rice husk and polyvinyl alcohol (PVA). Results showed that Type 1 – cellulose was extracted from rice husk. With the addition of PVA to the starch/cellulose film, the homogeneity of the mixture improved, as illustrated in FESEM images. The mechanical strength of film also greatly improved from 2 MPa to 4.2 MPa when 3 g of PVA was used. The film showed the sign of degradation after being immersed in water for 24 h due to its high-water uptake property (∼89%). This starch/cellulose/starch film could be used as an edible wrap and single-use material for varied applications such as disposable plates.
Introduction
Plastic pollution is harmful to the environment as well as living organisms. It is due to the inappropriate way of plastic waste management and the overuse of single-use plastic, especially for packing applications. Attempts have been made to produce biodegradable materials as a replacement for plastic.1–3 Nevertheless, the degradation process would only occur under a well-controlled condition, where the microbial population, physicochemical of materials, and the aerobic/anaerobic environment are considered. 4 In real life, the degradation rate of biodegradable material could be significantly affected when it is disposed to the natural environment, such as landfill sites. The latest report released by Greenpeace East Asia 5 also indicated that tightly controlled waste management and proper facilities are required to perform in-situ biodegradation. Hydro-degradable material could be an alternative to biodegradable material. Cellulose, starch, and polyesters are commonly known as hydrodegradable plastic, which could be degraded by the simple hydrolysis process.6,7 According to Romero-Azogil et al., 8 the hydrolytic capability of the materials strongly depended on the hydrophilicity of the said materials. Besides, the interactive effects of the crystallinity, glass transition temperature close to 37°C, and hydrophilicity of the materials also determined the hydrodegrability. 9 Starch and polyvinyl alcohol (PVA) are highly hydrophilic, and thus they can be easily degraded in landfills, especially during the rainy season. This would increase the lifespan of landfills and avoid the formation of microplastic. Additionally, nontoxic PVA and starch are harmless to the environment. Nevertheless, the study on hydro-degradable material is minimal.
The starch-based film has been widely studied for food packing applications. Bhatia et al. 10 produced an antimicrobial starch-based film using a mixture of sodium benzoate, EDTA, and lysozyme. Domene-López 11 reported that the type of starch affected the mechanical properties of films, and the strength of starch-based films was within 4.48–8.14 MPa. Film produced from wheat starch exhibited the highest tensile strength, while rice starch showed the lowest strength. A similar find was also published by Żołek-Tryznowska and Kałuza 12 recently, where oat-starch-based film exhibited the lowest strength, 0.36 MPa, and the potato-starch-based film showed the highest strength value of 3.05 MPa. The tensile strength of cassava film was within 1.2–2.4 MPa, and it was strongly affected by the amount of glycerol used, which acted as the plasticizer. 13 Besides, an attempt also has been made to increase the strength of cassava starch film by using lemongrass oil. The result showed that the optimum strength of cassava starch film was recorded when 0.25% of lemongrass oil was used to fabricate the film, which was 0.52 MPa. This is at least two times stronger than the pristine film, which exhibited a strength value of 0.16 MPa. 14 However, studies revealed that poor mechanical strength is the weakness of starch-based films,15–17 and thus, work must be done to improve the strength of the starch-based film.
Cellulose is known as the filler, which enhances the strength of the material.18,19 Studies have been done to extract cellulose from agricultural waste such as rice husk, 20 palm waste, 21 and fibrous waste sugarcane bagasse. 22 However, the focus of these studies was on the quality of the extracted cellulose rather than the role of cellulose in enhancing the strength of the starch-based film. Additionally, the surface of composite cellulose/starch film was less homogenous due to the presence of cellulose, which resulted in a low mechanical strength. 23 On the other hand, PVA exhibited good film formation properties and was highly soluble in water, 24 making it an ideal polymer to fabricate hydro-degradable materials.
This study aims to bridge the research gaps described above to reveal the role of PVA in improving the homogeneity of starch-based film. Cellulose was firstly extracted from rice husk (RH) via alkali-acid treatment. A varied concentration of PVA was added to the starch-cellulose mixture, and the performance of the films was evaluated in terms of mechanical strength and water uptake. FESEM images were also obtained to analyze the homogeneity of the PVA/starch/cellulose mixture.
Methodology
Materials
Cassava starch, NaOH (R&M Chemicals), NaClO (Bendosan), HNO3 (Bendosan), PVA (Acros Organic; 44.053 g/mol) with 87.0–89.0% degree of hydrolysis and 3.5–4.5cP (4% aq. Solution at 20°C), and glycerol (Bendosan) were used as received. RH was supplied by PLS Group, Sekinchan, Selangor.
Isolation of cellulose from rice husk
RH was washed and dried in an oven at 60°C for 24 h before being powdered to 250 μm, named as raw RH (RRH). 1M of NaOH was added to 30 g of RH and refluxed for 90 min at 80°C for delignification. The treated RH was rinsed using reverse osmosis (RO) water until pH7 and dried in an oven at 60°C for 24 h. 25 This was followed by bleaching, where 5% NaClO was added to the treated RH for reflux at 80°C for 100 min. Then, the RH was rinsed with RO water before acid hydrolysis for cellulose isolation purposes. This sample was labeled as alkali-treated RH (ALRH). Acid hydrolysis was conducted at room temperature by adding 2M HNO3 to the blenched RH for 30 min under stirring conditions. The isolated cellulose was collected, neutralized, dried in an oven before fabricating the hydrodegradable film. This sample was labeled as acid-treated RH (ARCH).
Fabrication of hydrodegrable film
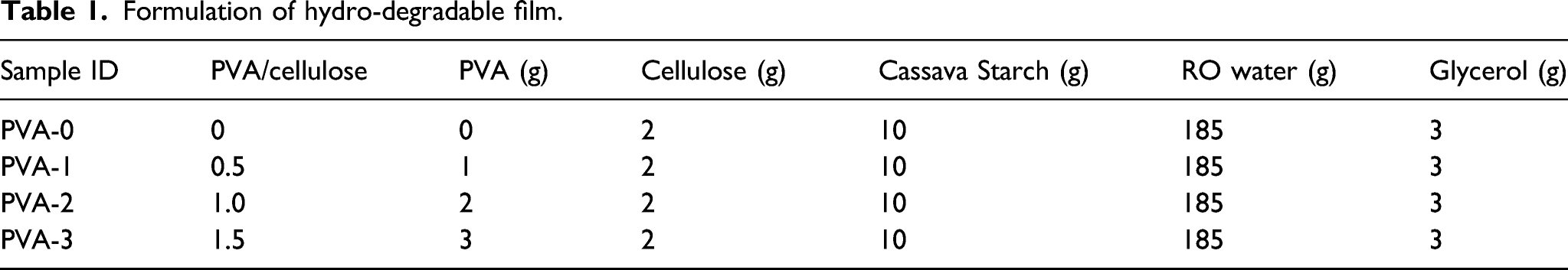
Formulation of hydro-degradable film.
Characterization of hydro-degradable film
The quality of isolated cellulose was analyzed using Fourier Transform Infrared Spectroscopy (FTIR) (Perkin Elmer Spectrum 100) and X-ray Powder Diffraction (XRD) (PANalytical Empyrean). The crystallinity of cellulose was calculated using equation (1) below. The morphologies of cellulose and hydro-degradable films were imaged using Field Emission Scanning Electron Microscopy (FESEM) (FEI model NOVA NANOSEM 230) while the strength of the film was evaluated using Instron 100 kN Universal Testing Machine at a cross speed of 0.5 mm/min. This complies with ASTM International.
29
Lastly, the water update test was conducted according to ASTM Interational.
30
2 cm x 2 cm film was immersed into 200 mL of RO water for 24 h.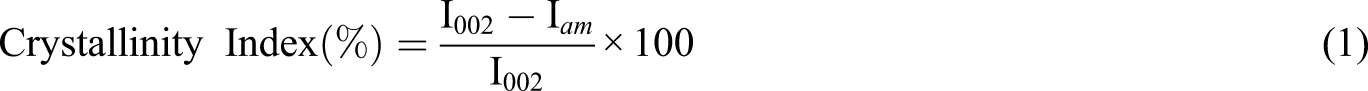
The initial 
Result and discussion
Characterization of cellulose extracted from rice husk
Figure 1 presented the FTIR spectra of RRH, ALRH, and ACRH. The abundance of R-O-R’ (ether bond) and R-O (acetyl bond) at 1700 cm−1 to 1740 cm−1 in Figure 1(a) are the signature bonds for lignin and hemicellulose.31,32 This peak was disappeared in ALRH, and ACRH samples as alkali treatment removed both lignin and hemicellulose. A similar finding was reported by Ikramullah et al.
33
where hemicellulose and lignin on typha fiber were removed by using 5% of NaOH. The presence of cellulose in ALRH and ACRH was proved by the absorbance bands at ∼898 cm−1 and ∼1056 cm−1 to ∼1161 cm−1.
34
This indicates the C-H rocking vibration and C-O stretching vibration, which are the characteristic peaks of cellulose. The peaks at ∼1640 cm−1 found at ALRH and ACRH represented the bending of O-H bonds of water absorbed in the structure.
35
The peaks at ∼1340 cm−1 indicated the presence of OH deformation vibration.
36
Notably, the O-H stretching of ALRH and ACRH, within the range of 3000 cm−1 to 3750 cm−1, is more prominent than RRH. This is due to the presence of cellulose, which consists of abundant OH groups in the structure. Fourier transform infrared spectroscopy spectra for (a) RRH (b) ALRH and (c) ACRH.
The XRD pattern also confirmed the presence of cellulose and FESEM image shown in Figure 2(a) and (b). The presence of a fibers-like structure in Figure 2(b) showed that both lignin and hemicellulose were successfully removed. Characteristic peaks of cellulose were observed at 2θ = 22.59°, 2θ = 15.63° and 2θ = 34.90° in the XRD spectrum, Figure 2(a). This agrees with the findings reported by Trilokesh et al.,
37
where the isolated cellulose from jackfruit peel showed similar peaks, as shown in Figure 2. According to Madureira et al.
38
and Owolabi et al.,
39
these peaks indicated the crystalline structure of Type-1 cellulose, which is the native form of cellulose. The crystallinity index of cellulose is 78.98%, which is higher than Ahmad et al.,
31
where 63.9% was reported. This may be due to the different types of rice husk used for extraction. On the other hand, Hanani et al.
25
reported maximum crystallinity of 52.4%, and it may be due to a lower concentration of HNO3 used, which was 0.5 M. Generally, high crystallinity index is desired as it exhibits excellent mechanical properties such as high tensile strength and high rigidity. (a) X-ray powder diffraction diagram and (b) FESEM image of ACRH at 500x (scale length 10 μm).
Performance of starch/cellulose/polyvinyl alcohol films
Figure 3 shows the FESEM micrographs of the top surface and cross-sectional structure of PVA-0 and PVA-3. The surface is shown in Figure 3(a) was inconsistent where the cellulose particles were clearly seen. This indicates that the isolated cellulose from the rice husk was successfully embedded into the starch/glycerol mixture. The cross-sectional images of PVA-0 shown in Figure 3(b) were not homogenous. A compact mass of particles was observed on the top of the cross-sectional area while the substructure was slightly dense with numerous nodules. This structure is not desired as the non-uniform structure resulted in poor mechanical strength. On the other hand, the FESEM image of PVA-3 was homogenous and consistent in both surface and cross-sectional areas, as illustrated in Figure 3(c) and (d). This indicated the formation of strong bonding between PVA and cellulose, which contributed to high strength property. This agrees with the strength data presented in Figure 4, where the tensile strength was enhanced from 2 MPa for PVA-0–4.2 MPa when the mass of PVA increased to 3 g in PVA-3. Meanwhile, the tensile strain was increased from 7.5 MPa for PVA-0–17 MPa for PVA-3. It is hypothesized that the mechanical strength of the film could be further increased to its optimum value if the amount of PVA was increased beyond 3 g.40,41 However, the water uptake of the film could be further reduced, which was undesirable for a hydrodegrable film. Thus, 3 g was the maximum of amount PVA used in this study. Field emission scanning electron microscopy image of PVA-0 (a) surface, 200x (b) cross section, 500x, and PVA-3 (c) surface, 200x (d) cross section, 500x; with scale length 10 μm. Mechanical strength of polyvinyl alcohol films.

Recently, Oyeoka et al. 42 reported a maximum strength of 13.83 MPa for a film fabricated from a PVA/Gelatin/cellulose nanocrystal (CNC) mixture. This mixture consisted of 10 g PVA/100 g water, 7.5 g gelatin/100 g water, and 5 wt% CNC. The PVA/Chitosan film produced by Jahan et al. 43 exhibited 8 MPa of tensile strength without crosslinker. The strength data reported in this study is slightly lower than the reported works. This may be due to the use of starch, which is inherently low in strength, and the low mass of PVA used, which was ∼3 g in 200 g of mixture for PVA-3. Nevertheless, the strength reported in this study is better compared to some of the pure starch films produced by Żołek-Tryznowska and Kałuza, 12 which was within 0.36–3.05 MPa.
Water update data of hydro-degradable film.

Relation between water absorption property and strength of the film was revealed by Jain et al.,
47
where high water update of PVA-based composite resulted from poor mechanical strength. This also explains the high-water update and low strength of PVA-3 compared to the finding reported in the literature. Nevertheless, the high water uptake is the desired property for a hydro-degradable material. It could be a good indicator to reveal the degradation rate of the film after being exposed to water. Figure 5 presented the images of PVA films, which were immersed in water for 24 h. PVA-0, which showed 89.536 ± 0.527% of water update as tabulated in Table 2, was deformed and broke into a few pieces after being immersed for 24 h in water. With the addition of PVA in samples PVA-1, PVA-2, and PVA-3, the level of deformation was reduced. This is due to the slightly reduced water update in the sample, as shown in Table 2. A complete piece of swollen samples can be seen in PVA-2 and PVA-3. The photo of (a) dry sample and swollen samples (b) PVA-0 (c) PVA-1 (d) PVA-2 and (e) PVA-3.
The water uptake of PVA film has closely related to the ratio of PVA/starch used in the study. Studies done by Salleh et al. 48 reported that an increase of PVA in PVA/starch fiber (SF) film reduced the % of water absorbed was uptake from ∼27% to ∼16% when the ratio was increased from 10%PVA/90% SF to 50%PVA/50% SF. A similar finding was also reported by Juhari et al. 49 The sample’s weight gain, which was prepared from 50%PVA/50% sago starch, was ∼0.2 g after it was exposed to water, while the 10% PVA/90% sago starch sample showed a weight gain of ∼0.4 g when it was exposed to the water. These explained the slight reduction in water uptake when the ratio of PVA/starch was increased from PVA-0 to PVA-3 in this study.
The PVA/cellulose/starch film produced in this study exhibited moderate strength with high water uptake properties. It may not be suitable for food packaging material due to high water updates, but it could be used as an edible wrap. 50 PVA is also documented as an edible polymer by Moreira et al. 51 and U.S. Food and Drug Administration. 52 Besides, the non-toxic and high-water uptake properties also made PVA/cellulose/starch film the potential substrate for controlled-release fertilizers and the single-use material for varied applications such as disposable plates, food packaging, and coating for fresh food and vegetables.
Conclusion
The performance of starch/cellulose/PVA film was revealed in this study. PVA improved the homogeneity of starch/cellulose/PVA mixture, as observed in FESEM images. The strength of the film was increased from 2 MPa to 4.2 MPa when 3 g of PVA was used. The water uptake of PVA/starch was slightly reduced due to the presence of PVA thus, the degree of degradation of film in water was slightly reduced. Nevertheless, the water uptake of ∼89% was considered high compared to the literature data. Thus, this PVA/starch film could be used as an edible wrap and single-use material for various disposable plates.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the SEGi University via grant number (SEGiIRF/2020-4/FoEBE-36/99) is highly appreciated.
