Abstract
In this work, an alternative of bio-thermoset resin for composite applications was synthesized using unsaturated polyester resin (UPE) and epoxidized palm oil (EPO). The main purpose of this study is to reduce the dependency on petroleum and improve the toughness properties of UPE by partially introduced renewable source triglyceride, EPO in the UPE chain. The UPE resin blends with 10, 20, and 30 wt% EPO oil were synthesized by using benzoyl peroxide as an initiator. The samples were cured in an oven at 100°C and 160°C for 4 h. The interaction of the blend was studied by the FTIR test supported by the SEM test. The effects of EPO loadings in UPE on the mechanical, thermal, and thermo-mechanical properties were also being studied. The addition of EPO physically interacted in the system and acted as a lubricant. The higher the addition of EPO in UPE has increased the elongation at break and impact strength of the UPE resin, which indicated an increase in mobility and toughness of the UPE chain. The developed UPE/EPO blends resin also exhibited higher thermal stability than the neat UPE, thereby showing potential to be used in a wide range of composite applications.
Introduction
Most thermoplastics and thermosetting resins in current industrial use are derived from petroleum, imposing a limitation to the polymer industry due to the continuous depletion of crude oil, frequent oscillation in oil price, and various environmental concerns with sustainability, gas emissions, disposal, and recyclability. 1 This phenomenon has driven a lot of research on biopolymers and increase the use of renewable natural resources in different fields of applications as they are sustainable, eco-friendly, and cost-effective materials. Among the natural sources such as proteins, polysaccharides, and lignin, vegetable oils are the most widely used renewable source in the chemical and polymer industries including thermoset resin due to their inherent biodegradability, easy availability, low toxicity, and relatively low price. 2 The most reported starting materials for bio-based thermoset resin are soybean oil,3–5 linseed oil, 6 rapeseed oil, and canola oil. 7 However, it was found that the use of plant oil-based resin in composites has been limited due to their lower mechanical and thermo-physical properties compared to synthetic resin. 8 A promising compromise between sustainability and performance of bio-based resin was obtained by replacing part of a petroleum-based resin with natural plant oil.
Palm oil is an edible vegetable oil extracted from the mesocarp of the oil palm’s fruit. It is abundantly available in South East Asian countries, especially in Indonesia and Malaysia. Palm oil is mostly used in food as cooking oil, shortening, and margarine, while in the oleochemicals industry, functionalized palm oil is used in manufacturing soaps, cosmetics, detergents, and as raw material for biodiesel. In the last few decades, palm oil applications in the pharmaceutical, engineering, and industrial areas have been explored by many researchers. However, the use of epoxidized palm oil (EPO) as resin in composite matrix applications has not been explored and fully realized.
Unsaturated polyester resin (UPE) is one of the most important thermosetting polymers that are being used widely in fiber reinforced plastic (FRP) applications. UPE resins are more commonly used than other thermoset resin due to their attractive price-per-properties ratio. Unsaturated polyester resins possess some good properties such as good mechanical properties, excellent thermal stability, weather resistance, and resistance to a great number of chemicals. It is also easily used in the preparation of composite materials due to the simplicity and flexibility to modify their chemical structure that will allow changes in viscosities and curing time to be used in a large variety of applications, including housewares, transportation, sports equipment, electrical industries, and many more. However, due to the concern of petroleum depletion, synthesizing renewable-based polyester resin is gaining importance.
This study is focused on developing bio-based resin by replacing some UPEs with functionalized palm oil, which is an EPO processed by benzoyl peroxide as an initiator. The mechanical, thermal and thermo-physical properties of the developed bio-based UPE were discussed.
Methodology
Materials
Epoxidized palm oil (EPO) was received from Malaysian Palm Oil Berhad (MPOB) with 1049 g/mol molecular weight, 2.7–2.9% of oxirane oxygen content, 0.58 g I2/100 g sample of an iodine value, 1.0 mg KOH/g sample of acid value, and 0.3% moisture content. An unsaturated polyester resin (Reversol P 9539 NW) was purchased from Synthomer Malaysia Sdn Bhd with 45% styrene content, 300 cps viscosity, and 1.1 specific gravity at 25°C. Benzoyl peroxide (BP) initiator with 25% water content purchased from Acros Organics.
Preparation of UPE/EPO biobased resin
Various amounts of EPO were blended in UPE resin, which at 10, 20, and 30 wt%. UPE resin was stirred on a hot plate at 55°C and EPO was gradually added and continuously stirred until they were thoroughly blended. Benzoyl peroxide was dried in a drier until a constant weight was obtained. The 1.5 phr dried benzoyl peroxide was then added to the UPE + EPO mixture and the mixtures were stirred and poured into 250 mm width x 250 mm length x 5 mm thickness aluminum closed mold and further cured at 100°C and 160°C for 4 h. The samples were then cut according to specific standard for further testing.9–12
Testing
The interaction was characterized using Perkin Elmer 1600 FTIR spectrophotometer. By using ASTM E1252, 5 mg of powder samples were mixed with about 95 mg of potassium bromide (KBr) and the mixture was pressed by using a hydraulic press to form a thin film at a thickness of 10–100 µm. The sample was scanned 16 times and the infrared spectra were determined to be in the 4000-300 cm−1 range.
Mechanical tests were further conducted using Lloyd Universal Test Machine model 4400. The test was performed according to ASTM D 3039 specified for tensile properties of polymer matrix composite materials. The test was applied at a crosshead speed of 5 mm/min and a gauge length of 150 mm (6 in). The impact test was evaluated according to ASTM D256-10, by using Toyoseiki Izod Impact Tester with capacity of 11 Joules. Notched is used to provide stress concentration area to promote brittle rather than ductile failure. This study was done at room temperature.
Thermal properties were conducted using thermogravimetric analysis (TGA) Perkin Elmer 7, USA. A sample of approximately 5 mg was heated at 10°C/min in a nitrogen purge stream from 30°C to 600°C. The flow rate of the nitrogen stream was 50 mL/min.
Thermo-physical analysis was conducted using Dynamic Mechanical Analyzer (DMA) (Perkin Elmer DMA7e), with three-point bending mode measurements (dual cantilever). The sample was prepared at the dimension of 60 mm length x 12 mm width x 5 mm thickness. The sample was heated from 30°C to 200°C with a heating rate of 2°C/min and a frequency of 1 Hz. Based on the DMA result, crosslinking density was calculated using equation E’= 3veRT, where E’ = storage modulus at (Tg + 40)°C, ve = cross-link density, R = gas constant (8.314 J.K−1.mol−1), and T = temperature (K).
Finally, the morphology of the system was evaluated using scanning electron microscopy (SEM) analysis on the tensile fractured specimens by using JEOL JSM-6390 LV, SEM machine. The tensile fracture sample from tensile test was cut at 5 mm height from the fracture surface for this test. The gold coating of the fractured surface was prepared, and the surface characteristic was studied and analyzed.
Results and discussion
Fourier transform infrared spectroscopy
Figure 1(a) shows the Fourier transform infrared spectroscopy (FTIR) peaks of neat UPE resin and EPO. As can be seen in Figure 1(a), several important peaks in neat UPE resin were observed, which are at 1727 cm−1 (C=O carbonyl group), 1629 cm−1 (C=C alkene group), and 1287 cm−1 (C-O ester group), while in EPO spectrum, a signal of 844 cm−1 can be observed, which is attributed to the oxirane ring (C-O-C). FTIR spectrum of UPE, EPO, and UPE/EPO resin.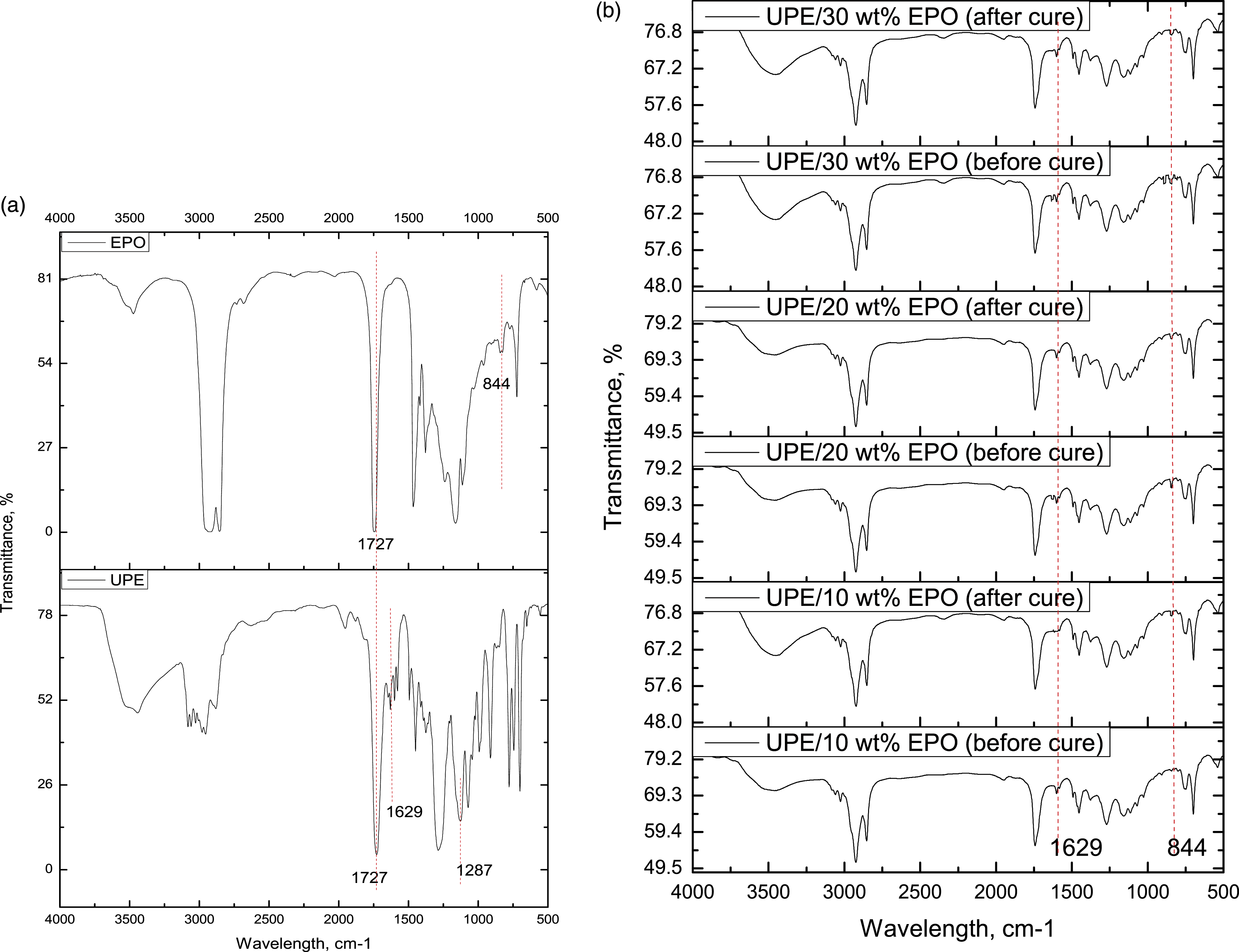
Figure 1(b) shows the comparison of uncured and cured resin systems at different EPO loadings. In the uncured UPE/EPO resin system shown in Figure 1(b), two important peaks can be observed, which are at 1629 cm−1 and 844 cm−1, indicating the existence of C=C and C-O-C oxirane functional group, respectively. In general comparison, the oxirane peaks at 844 cm−1 remained in the cured resin system indicating that there are no chemical reactions between the EPO and UPE resin. Thus, the palm oil added was acting as a lubricant in the system and interact physically with UPE resin. Besides, the C=C peaks at 1629 cm−1 also can be observed with slightly lower intensity in the cured resin systems. The slight decrement of C=C intensity peak in the cured resin system as compared to uncured resin indicated that the crosslinking process was taking place.13–15 The existence of the small peak C=C in the cured resin FTIR spectrum proved that the UPE/EPO blend resin system is not 100% cross-linked, mainly due to the existence of palm oil which does not chemically interact in the system. The crosslinking process was only taking place between UPEs resin by free radical chain-growth polymerization. The benzoyl peroxide formed radicals that initiated the crosslinking process of the UPEs. The free radicals attacked the C=C in UPE and styrene monomer and made UPE-styrene bridges.13,16 These chemical reactions occurred simultaneously during the curing process. The crosslinking mechanism of the unsaturated polyester resin is shown and illustrated in Figure 2. The free radical movements during polymerization were shown by the red arrow in the figure. Crosslinking mechanism of unsaturated polyester resin.
Scanning Electron Microscopy
Scanning electron microscopy (SEM) micrographs of tensile fracture surfaces of UPE/EPO resins at 10, 20, and 30 wt % EPO loadings are shown in Figure 3. As can be seen in Figure 3(a), the neat UPE had a flat and smooth featureless micrograph surface. This feature was suggested to be due to the brittleness properties of the UPE.
17
The neat UPE was elastic and in fact, the crack propagated in a planar manner under the vertical tension applied. Low magnification SEM images showing the tensile failure surface of UPE and various blend resin: (a) neat UPE, (b) UPE containing 10 wt % EPO resin, (c) UPE containing 20 wt % EPO resin, and (d) UPE containing 30 wt % EPO resin.
Figures 3 (b)–(d) show SEM micrographs of tensile failure surfaces of UPE containing 10 wt %, 20 wt %, and 30 wt % EPO loadings, respectively. In contrast to neat UPE, the tensile fracture surface of UPE/EPO resin was rough. It was proven that adding EPO has resulted in a rougher UPE fracture sample surface. The roughness surface suggested that the mixture of resin and oil was non-homogenous.
11
Referring to the high magnification of SEM micrographs in Figure 4, it is clearly seen that the roughness surface consisted of discrete microstructures of dispersed phase scattered throughout the entire UPE matrix surface. This condition proved that all constituents of UPE and EPO were not homogenously mixed at the molecular level and formed phase separation (two phases). The phase separation occurred due to the different solubility of the UPE and the vegetable oil.12,18 Mehta et al. (2004)
19
who studied bio-based resin as a toughening agent for biocomposites explained that the small discrete particles (second phase) that were randomly distributed in the glassy brittle polyester resin matrix (first phase) were oil phase micro gel which was formed during curing. In this case, the second phase that existed in the UPE matrix was the EPO-rich phases which can be seen as a particle-like feature at the micrometer scale. The oil phase micro gel provided a rubbery structure to the UPE system. It acted as an impact modifier, hence absorbed impact energy and delayed catastrophic failure. It was found that the surface of UPE containing 30 wt % EPO (Figure 3(d)) was rougher and it had a larger number of the small resin pieces on the failure surfaces than 20 wt % (Figure 3(c)) and 10 wt % EPO (Figure 3(b)). The larger failure surface area was directly related to a higher failure energy dissipation.
11
High magnification SEM images showing the tensile failure surface of UPE and various blend resin: (a) UPE containing 20 wt % EPO resin and (b) UPE containing 30 wt % EPO resin.
Other than the homogeneity, SEM images may also explain the interfacial adhesion between the UPE and EPO. Referring to Figure 4, some of the particle-like oil phases remained bonded to each other and observed as a lamella structure on the tensile fracture surface. This condition explained the tensile specimen cracked at UPE-EPO interface. Thus, the interphase between the oil-like particle (second phase) and the UPE matrix (first phase) contributed to a higher stress concentration point of the UPE/EPO mixture, in which the crack propagated at the interfaces between the two faces.
Dynamic Mechanical Analysis (DMA)
Figure 5 shows the results on the storage modulus and loss factor of UPE/EPO resin at different percentages of EPO loadings. From the graph, it can be clearly seen that the addition of EPO has lowered the storage modulus of UPE over the entire temperature range. As the temperature increased, E’ was gradually decreased until the maximum loss at 95 to 100°C which was above the glass transition region, Tg (70–80°C). This condition indicated that the mobility of the UPE/EPO molecular chain was above Tg. At 35°C (below the glass transition temperature, Tg), it can be seen that the storage modulus was linearly decreased from 1590 MPa to 650 MPa at addition up to 30 wt % EPO in UPE/EPO resins. This result was expected since vegetable oils are intrinsically low modulus materials. In addition, the crosslinking density of the resin dropped at the increasing addition of EPO loadings. Thus, the addition of EPO has lowered the storage modulus and stiffness of the resulting UPE/EPO resin. The addition of up to 30 wt % of EPO in the UPE/EPO blend could be considered as an acceptable amount for load-bearing applications since the storage modulus was higher than 500 MPa.
11
Plot of storage modulus and loss factor versus temperature at different compositions of EPO in UPE resin.
Tg, maximum tan δ, peak width at half height, and crosslinking density results of UPE/EPO resin at different EPO loadings.

The decrease in crosslinking density with an increase in EPO loadings has also contributed to the decrement in the mechanical and thermal properties of the resulted blend resin. This is because decreasing in crosslinking decreases the number of binding chains and strain concentration which further increases the fracture strain and lowering the thermal behavior of the polymer. 12 Besides that, the peaks of the loss factor curve for all samples were broad. The peak factor (broadness of the tan δ peak) can be defined as the value of full-width at half-maximum (FWHM) as a qualitative assessment of the homogeneity of the polymer network and distribution of the molecular weight. 20 Referring to Table 1, the tan δ maximum value decreased from 0.9 to 0.6, while the FWHM value of neat UPE have increased from 27 to 31 with the addition of EPO up to 30 wt %, respectively. The decrement in tan δ maximum value indicated lower stiffness properties as decreasing value of storage modulus over loss modulus. The higher FWHM values as increased EPO loadings indicated broader peak value which suggested that UPE/EPO resin had a broader glass transition region which may be due to the heterogeneity of UPE/EPO blending system.
Mechanical properties
Figure 6 shows the change in notched Izod impact strength and elongation at break of neat UPE at different percentage of EPO loadings. As can be seen, the addition of EPO has slightly increased the impact strength and elongation at break of the UPE/EPO resin. From the analyzed data of 10, 20, and 30 wt % of EPO loadings in UPE, the Izod impact strength has increased 4.06%, 15.10%, and 22.90%, respectively. These results showed that higher impact energy absorption was achieved in UPE/EPO resin at higher EPO loading. The improvement in impact behaviors may be due to a long flexible and resilient network structure exhibited by the palm oil epoxy system (EPO).
21
The flexible character of the fatty acid chains increased the mobility of the UPE/EPO resin and thus contributed to higher resistance to crack initiation and propagation.
22
The high mobility of UPE/EPO resin is related to the lower stiffness properties of the UPE when EPO was added. The improvement in Izod impact properties due to the addition of EPO is in agreement with the increasing ductility in elongation at break results. The improvements in Izod impact properties due to the addition of EPO were similar with the results studied previously by Haq et al. (2011)
6
on the effects of UPE/EML blends and nanoclay. As can be seen in the same figure, the elongation at break value also increased from 1.46% (neat UPE) to 1.90% (30.18% increment) at 10 wt % of EPO loadings, about 2.12% (45.51% increment) at 20 wt % of EPO loadings, and about 2.36% (62.12% increment) at 30 wt % of EPO loadings. According to the studies conducted by Gitta Silverajah et al. (2012)
23
on the mechanical properties of poly(lactic acid)/epoxidized palm olein blend, the presence of EPO reduces intermolecular forces and increases the mobility of PLA chain, thereby enhancing the flexibility and extensibility of the PLA/EPO blend. The same phenomenon occurred in this study. The flexibility of EPO has acted like a plasticizer in UPE and contributed to higher ductility. Thus, higher EPO loadings in UPE produced lower tensile modulus and tensile strength, but higher in the percentage of elongation of UPE/EPO resin. Elongation at break and Izod impact properties.
Figure 7 showed the tensile modulus and tensile strength of UPE at 10, 20, and 30 wt % of EPO loadings. From the figure, it can be seen that the addition of triglyceride EPO has decreased the tensile modulus and tensile strength of the neat UPE. The blend of 90/10, 80/20, and 70/30 of UPE/EPO resins has decreased the tensile modulus by 40.71%, 42.39%, and 69.12%, respectively. While in tensile strength, the blend of 90/10, 80/20, and 70/30 of UPE/EPO resins has decreased the tensile strength by 27.96%, 51.30%, and 55.58%, respectively. The reductions in tensile modulus were attributed to the low stiffness of the triglyceride (EPO).
6
The flexibility of EPO in the UPE chain has disturbed the stiffness properties of the original UPE. The low stiffness property of EPO has generated deformation in the UPE/EPO blends sample when the load is applied. The reduction in tensile modulus due to the addition of EPO also indicated less brittle and higher flexibility of the UPE/EPO resin. The decrease in tensile strength indicated that the UPE/EPO blend resin can be easily fractured and failed at lower stress when higher EPO was introduced into the system. The low tensile strength properties were due to the poor crosslinking between UPE and EPO. The interphase between polymer and oil created a stress concentration point that decreased the strength properties of the polymer. The stress concentrators initiated micro crack, and the crack propagated by breaking the bonding chains at the crack tip until a fatal fracture occurs.
20
This result revealed that UPE becomes more elastic with the addition of EPO, which has less strength, but it breaks at higher deformation (higher elongations). The same findings have also been reported elsewhere.
22
Tensile modulus and tensile strength properties.
Thermal properties
Figure 8 represents the thermal stability and decomposition behavior of UPE, EPO, and UPE/EPO resin at different percentage of EPO loadings. From the first derivatives of the TGA thermograph as shown in Figure 8(b), it was clearly noted that UPE/EPO resin experiences two-stage thermal decomposition when heated up to 600°C. The first stage decomposition occurred at 150–200°C was believed to be due to the decomposition of the low molecular weight benzoyl peroxide initiator. This is because some of the two free radicals may eventually hit each other and remain as a molecule. It was generally believed that curing agents and catalysts showed lower thermal stability as the thermal stability is proportional to molecular weight.4,11 The second stage decomposition occurred at 330 to 360°C is the major decomposition which was attributed to the thermal degradation of thermally stable UPE, EPO, and UPE/EPO components. Referring to the weight loss data in Table 2, EPO had higher thermal stability as compared to the neat UPE as the onset decomposition temperature of EPO was 358.87120°C and the maximum decomposition temperature was 400.56311°C. These temperatures were higher than the onset temperature of cured UPE, which was 337.59472°C, and the maximum decomposition at 387.64060°C. Thus, UPE/EPO blends formed intermediate thermal stability between the cured UPE and EPO as tabulated in Table 2. This phenomenon proved that the presence of EPO in the UPE polymer matrix had improved the thermal behavior of the neat UPE. The reassembling EPO has created a protective physical barrier on the surface of the material, which hindered the permeability of volatile degradation products out from the blend and eventually helped to delay the degradation of the blend.22,23 Referring to the results, the decomposition of the blends up to 30 wt % EPO loadings commenced close to 340°C and rapidly continued until maximum degradation at 402°C. It can be confirmed that UPE/EPO blends had excellent thermal stability.
24
TGA micrograph: (a) derivatives thermogram and (b) of UPE, EPO, and UPE/EPO resins of different compositions. Weight loss analysis by TGA of UPE/EPO resin.

According to the results, increasing EPO loadings in UPE from 10 wt % to 30 wt % led to a slight drop in the thermal stability of the resulted UPE/EPO resin as the onset degradation temperature was dropped from 342.76237°C to 340.16333°C. However, the maximum degradation temperature of the UPE/EPO resin was increased from 377.67283°C to 402.48388°C. This phenomenon was highly related to the physical interactions that occurred between UPE and EPO in the UPE/EPO system. The decreasing onset degradation temperature at higher EPO loadings (20 and 30 wt% EPO) indicates a decrease in the crosslinking of the UPE/EPO system which was increasing the bond cleavage. In addition, the high EPO loadings had allowed excess unreacted EPO to be trapped in the system in which has increased the maximum thermal degradation; as the excess EPO has acted as a protective physical barrier on the surface of the material which hindered the permeability of volatile degradation products out from the blend and eventually helped to delay the maximum degradation of the resin blend.
Conclusions
This manuscript has discussed the properties of new biobased UPE resin by blending 10, 20, and 30 wt% EPO. This study has revealed that functionalized epoxidized palm oil did not chemically react with UPE resin and remained in the system. As shown in FTIR, SEM, DMA, and tensile results, higher loadings of EPO allow higher excess palm oil to be trapped in the system which led to a lower crosslinking density of the resulted UPE/EPO resin. EPO has acted as a plasticizer in UPE and improved the toughness and elongation at break of the resulted UPE/EPO resin by 22.90% and 62.12%, respectively. However, the improvements in toughness compromised in the stiffness properties. The addition of EPO decreased the stiffness and strength of the UPE/EPO blend resin by 69.12% and 55.58%, respectively. Thermal analysis through TGA results indicated an increase in thermal stability from 337.59°C to 342.76°C as 10 wt% EPO was added in UPE. However, increasing the loadings of EPO up to 30 wt% has decreased the thermal stability and glass transition temperature of the UPE/EPO resin due to lower crosslinking density properties. Overall, the EPO/UPE blends show a promising step for a new alternative of bio-based polymer. According to the results, it is suitable enough for indoor applications with low strength applications. However, further modification is highly recommended to improve the properties for other applications.
Footnotes
Acknowledgments
The authors would like to thank the Ministry of Higher Education for providing financial support under Fundamental Research Grant Scheme (FRGS) No. FRGS/1/2019/STG07/UMP/02/2 (University reference RDU1901105), additional financial support under Internal Research grant Universiti Malaysia Pahang, RDU1903136, and Internal Research Grant Universiti Teknologi Malaysia, Q.J.130000.7125.01H24.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Universiti Teknologi Malaysia (Q.J.130000.7125.01H24), Universiti Malaysia Pahang (RDU 1903136) and Ministry of Higher Education, Malaysia (RDU1901105).
