Abstract
Poly(vinyl chloride) PVC materials are used in many industrial and household outdoor applications which exposed to sunlight ultraviolet. This exposure motivates photo-degradation of the PVC and hence, reduces its life time and leads to undesirable physical properties. Herein, three new Schiff bases containing a trimethoprim moiety were investigated as photo-stabilizers for poly(vinyl chloride) against photo-decomposition. Merely 0.5% by weight of these Schiff bases was mixed with the polymer to form blend films with thickness of (40 µm). The films were irradiated for a period of time from 0 to 300 h and the irradiation impact on the PVC stabilization was monitored every 50 h. The efficiency of these additives as photo-stabilizers was investigated as a function of different parameters during the course of irradiation, such as weight loss, growing carbonyl and alkene groups as well as the surface morphology of the prepared films. The changes during irradiation were obvious for the blank films in comparison to the films containing the new additives. Therefore, these Schiff bases were found to act as photo-degradation inhibitors through reducing the elimination of hydrogen chloride molecules and as free radical scavengers or peroxide decomposers.
Keywords
Introduction
Nowadays plastic polymers, in particular poly(vinyl chloride) (PVC), are extensively utilized in many industrial and outdoor applications. 1 The most important features of PVC are its excellent mechanical, physical, and chemical characteristics, in addition, to its low price production.2–5 Nevertheless, PVC has some of serious drawback which may arise from its ability to undergo thermal and photo-degradation.6,7 When exposed to UV light, PVC undergoes HCl molecules elimination reaction which leads to formation of carbonyl and polyene groups. This reaction change the chemical structure of the polymeric chain and affect the mechanical and physical properties of the PVC. 8 Photo-degradation could also lead to formation of free radicals, subsequently, increasing the cross linking between the polymer progression or breaking the chains to smaller units. All of that chemical changes generate unfavorable physical properties such as color changing, rigidity and discoloration.9,10 Owing to the employment of PVC in many important applications, the photo-stability theme of the PVC turns into a fundamental issue. Therefore, a wide range of UV absorbers, antioxidants, free radical scavengers have been synthesized or extracted from natural materials and tested as photo-degradation inhibitors for PVC.4,11,12,13 On the other side, desirable properties for photo-stabilizers are to be: non-volatile, UV light absorption, free-radical scavengers, harsh condition resistance, nontoxic and cheap production. 14 However, in the past two decades, many candidates have been prepared and examined as photo-stabilizers including Schiff’s bases, highly aromatic compounds, porous polyphosphates, organotin complexes and titanium dioxide.15–20 These compounds show high efficiency toward inhibiting photo-degradation of PVC after long time of UV light exposure. Nonetheless, the majority of these prepared stabilizers are inconsistent with the photo-stabilizers practical demands; for instance, some of them are easily degradable and others are carcinogenic. Currently, there are extensive works to develop new PVC photo-stabilizers regarding the application requirements. As part of our concern in the polymer stability field, the target of the present work is to highlight synthesis of Schiff bases containing a trimethoprim unit and test them as PVC photo-stabilizers. Such Schiff bases are highly aromatic, easy to prepare, nontoxic, non-volatile, UV-absorbent and cost-effective. These features may originate delicate and effective ability for PVC photostabilization. Recently, our research has been directed to investigate the use of newly synthesized aromatic compounds and those including Schiff bases as potential UV absorbers. We have made some progress in this field which is reported in the current work.
Materials and Methods
Materials and physical measurements
All the utilized reagents and chemicals have been purchased from Sigma-Aldrich, UK and used without any further purification. While the PVC with an average molecular weight of approximately 233,000 (a polymerization degree of 800) was purchased from Petkim Petrokimya, Turkey. Shimadzu 8400 Spectrophotometer, Japan was utilized to record fourier-transform infrared (FTIR) spectra. A Veeco instrument, NY, USA was used to record the atomic force microscopy (AFM) images. The Field-Emission Scanning Electron Microscopy (FESEM) (10 kV) images were recorded by TESCAN-MIRA3 (Kohoutovice, Czech Republic) technique at Sharif University of Technology, Iran. The energy dispersive X-ray (EDX) spectra were measured using Bruker XFlash 6 10, Tokyo, Japan, and before measuring EDX the film was coated by gold, 15 nm. QUV-accelerated weathering tester (Homestead, FL, USA at Al Mustansiriya University, Iraq) was employed to irradiate the polymeric films by UV light at 25°C. DIN 862 digital caliper micrometer (Vogel GmbH; Kevelaer, Germany) was used to measure the 40 µm thickness of the prepared films.
Trimethoprim Schiff bases synthesis
The Schiff bases were prepared as previously reported
21
by stirring of trimethoprim (2.9 g, 10 mmol) with a suitable aryl aldehyde derivatives (3-hydroxybenzaldehyde, 4-anisaldehyde, or 4-dimethylamino benzaldehyde) (10 mmol) in boiling mixture of methanol (25 mL) and glacial acetic acid (0.5 mL) for 6 h. After cooling, the precipitate was filtered, washed by methanol (15 mL) and recrystallized from methanol to obtain the pure targeted compounds 1–3 (Figure 1). Schiff bases compounds 1–3.
PVC Films Preparation
The blended PVC films were fabricated by mixing 0.5% by weight of the prepared Schiff bases, regarding the polymer weight, to obtain films with a thickness of 40 µm. Accordingly, the films were prepared by dissolving mixture of PVC (5.0 g) with the suitable synthesized aromatic Schiff bases (25 mg) in THF (100 mL). The mixture was left under stirring at room temperature until complete dissolution to acquire good homogenous solution. Afterwards, the solution was poured onto glass plates (4×4 cm2) and left overnight for dryness. The produced PVC films were dried at room temperature for 6 h, then in a vacuum oven for 24 h.
PVC films irradiation
Irradiation of the PVC samples was achieved by UV light (λmax = 365 nm; light intensity = 6.2 × 10−9 ein dm−3 s −1). Each sample was swiveled regularly to assure that it was exposed to the UV light equally from all sides. The films were irradiated for a period of time that extended from 0- 300 h. The irradiation impact on the PVC photostabilization was monitored every 50 h.
PVC functional groups indices evaluation
The photo-degradation process of the PVC leads to the formation of residues that contain carbonyl (C = O) and alkene (C = C) groups. The growth of the absorption band intensities of these two groups, arising from the photo-irradiation, can be monitored using FTIR spectroscopy and compared with that for the C–H bond as a reference.
22
Thus, indices (Is) of C = O and C = C groups were calculated using equation (1).
PVC weight loss evaluation
The weight loss, which emerged due to elimination of volatile molecules, was used to follow the photo-degradation of PVC films by weighing the polymer films pre and post irradiation and employing equation (2).
23
Results and discussion
Synthesis of the Schiff bases 1−3
Schiff bases 1–3 were prepared as previously stated 21 by reaction of trimethoprim moiety with the aryl aldehyde derivatives to obtain the required compounds Figure 1. Standard techniques were used to demonstrate the chemical structure of synthesized materials. 21
Monitoring of the PVC photodegradation by FTIR spectrophotometry
The main defect of the PVC plastic outdoor applications is photo-oxidation which occurs during the UV light exposure in presence of oxygen. This results in formation of small fragments containing of carbonyl (C = O) as well as developing alkene (C = C) groups.
22
These two functional groups produce sharp infrared peaks at about 1720 cm
FTIR spectra of the PVC films ( FTIR spectra of the blended PVC+1 (
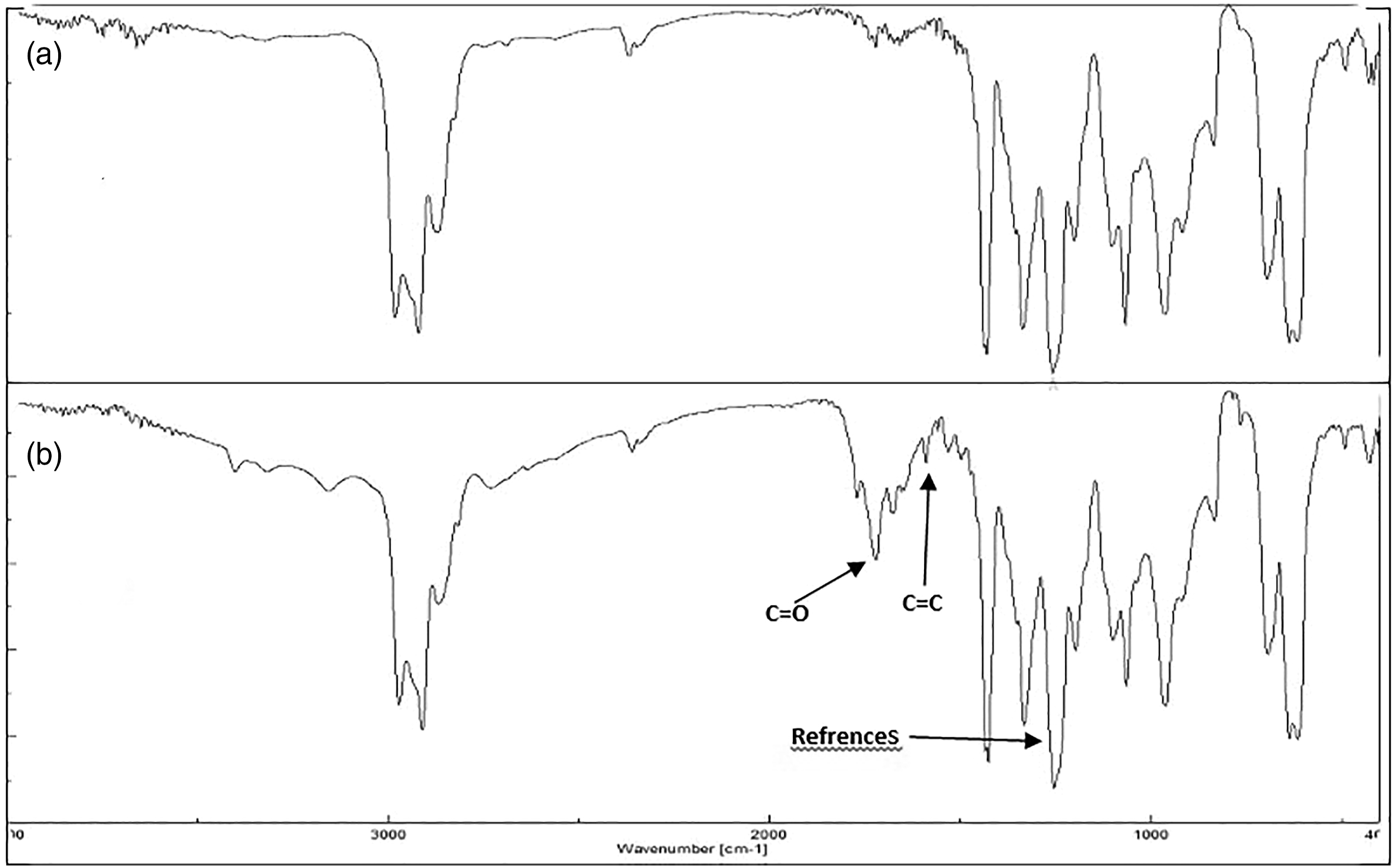
The figures indicate that the intensity of the C = O and C = C groups have grown significantly post-irradiation which displays the PVC photodegradation. Therefore, the absorbance intensity of the C = O (AC=O) and C = C (AC=C) were observed over the irradiation time and compared with that for the C–H (AC–H) at 1328 cm−1. The functional groups indices (IC=O and IC=C)) were calculated by applying equation (1) and plotted as a function of irradiation time (Figures 4 and 5). The profile displays higher indices (IC=O and IC=C) of the irradiated PVC blank films after every 50 h relative to that for the blend (PVC + Schiff bases 1–3). For instance, at the end of irradiation (300 h) the (IC=O and IC=C), for the blank PVC, were found to be 1.19 and 1.18, respectively. While the indices for both functional groups, post to the same irradiation period, were 0.6–0.7 and 0.59–0.7, respectively. In general, these findings indicate clear evidence that the IC=O and IC=C indices were significantly reduced when Schiff bases were added. The conjugated systems and the heteroatoms (oxygen, and nitrogen) of the prepared additives presumably act as a UV-absorbers or blockers. Moreover, the figures indicate that Schiff base 1 has much more stabilization effect, in comparison to the other compounds. This result could be attributed to the structure of compound 1, since it has a phenolic hydroxyl group which may be more efficient as free radical scavenger than the others. Change in the IC=O index for PVC films. Change in the IC=C index for PVC films.

Evaluation of the weight loss caused by UV irradiation of PVC
Long-term exposure of the PVC to UV irradiation eliminates several small fragments with low molecular weight by-products. Depending on the duration of exposure, irradiation of PVC causes loss in the weight of the material.
23
Therefore, the degree of damage can be determined through the weight loss percent (%) by implementing equation (2), and it is plotted as a function of irradiation time (Figure 6). Changes in the weight loss of PVC films.
Figure 6 indicates that the blank PVC film undergoes weight loss notably faster than the films with the trimethoprim Schiff bases 1, 2 and 3 additives. For example, the weight loss percent (%) at the end of irradiation exposure was estimated to be 0.14% for the PVC + 1, 0.51% for the PVC + 2, 0.64%, for the PVC + 3 and eventually 1.42% for the blank PVC film. From these findings, we can understand that Schiff base 1 is being more effective as photostabilizer in comparison to the other compounds. This observation is in concordance with the aforementioned calculations of the functional groups indices (Is) (Figures 4 and 5).
Surface morphology of the PVC films
The irradiation and photodegradation of PVC material creates damage and irregularities due to the appearance of defects on the surface of the polymer. This damage results originally from HCl elimination (dehydrochlorination) and chain scission which can be visualized by optical microscopy technique.19,20 The microscopic images of the pure PVC films, with a magnification power of ×400, show that the surface of the non-irradiated films was regular with few defects, (Figure 7). By contrast, the optical image of the pure PVC films, post 300h of irradiation, demonstrates an appearance of grooves, dark spots, cracks, and color change.
29
For the PVC containing trimethoprim Schiff bases 1–3 the damage was less obvious than that noticed on the pure PVC film surface post to the same period of irradiation. These outcomes revealed that the Schiff bases 1–3 additives reduce the HCl elimination rate and consequently, improve the photostability of the PVC. Microscopic images of the pure and blended PVC pre and post exposure to UV light with a magnification power of (400 ×).
Field-Emission Scanning Electron Microscopy of the PVC
Field-Emission Scanning Electron Microscopy (FESEM) is an effective technique to capture high-resolution images and provides detailed information about the homogeneity, shape, cross-sections and particle sizes of materials. Therefore, it is utilized widely to inspect the PVC surface morphology. Accordingly, the FESEM images for pure PVC were examined pre- and post-irradiation in parallel with those irradiated films containing trimethoprim Schiff bases 1–3 (Figures 8 and 9). The image of the non-irradiated pure PVC film revealed a smooth, homogeneous and regular surface. While the surfaces of the irradiated films post 300 h irradiation time, indicate clear damage like cavities, spots and irregularities (Figure 8). The damage appears mainly due to chain cross-linking and elimination of HCl as well as removal of tiny fragments.18,29 Nonetheless, the defects are more prominent in the pure film in comparison with the films involving trimethoprim Schiff bases 1–3 (Figure 9). FESEM images of pure PVC (a) pre-irradiation, (b) post-irradiation. FESEM images of (a) PVC+1, (b) PVC+2, (c) PVC+3 after 300 h irradiation.

Atomic force microscopy topographic images of the PVC
Atomic force microscopy is used to investigate the smoothness, roughness and the homogeneity of the materials surfaces.
30
Hence, the PVC films surfaces were examined by that techniques as shown in Figure 10. The AFM images display that the surfaces of the post-irradiated pure PVC films were rougher than pre-irradiated ones (Figure 10). On the other hand, the trimethoprim Schiff bases 1–3 PVC blends show uniform and normal surfaces in comparison to the irradiated standard PVC film. Furthermore, the smoothness of the PVC surface films was analyzed through applying the roughness factor (Rq). A high Rq value indicates significant irregularity and low degree of smoothness due to dehydrochlorination and bond-breaking of the polymeric chains. In this context, the roughness factor value of the PVC post-exposure to UV light was Rq = 295.2, while that of the PVC films containing Schiff bases 1, 2 and 3 had high degrees of smoothness Rq = 4.191, 5.02 and 16.28, respectively. These results support the proposition that the trimethoprim Schiff bases act as a photostabilizer for the PVC materials. In addition, Schiff base 1 has decreased the Rq of the irradiated PVC by 70.1 fold which is very significant in comparison with published results of using Schiff bases as photostabilizers.
29
Table 1 shows the improvement in the roughness factor (Rq) of PVC films containing various additives. The atomic force microscope images of the pure and blended PVC films after 300 h irradiation. The improvement in the roughness factor (Rq) of PVC films containing various additives.

Energy-dispersive X-ray mapping
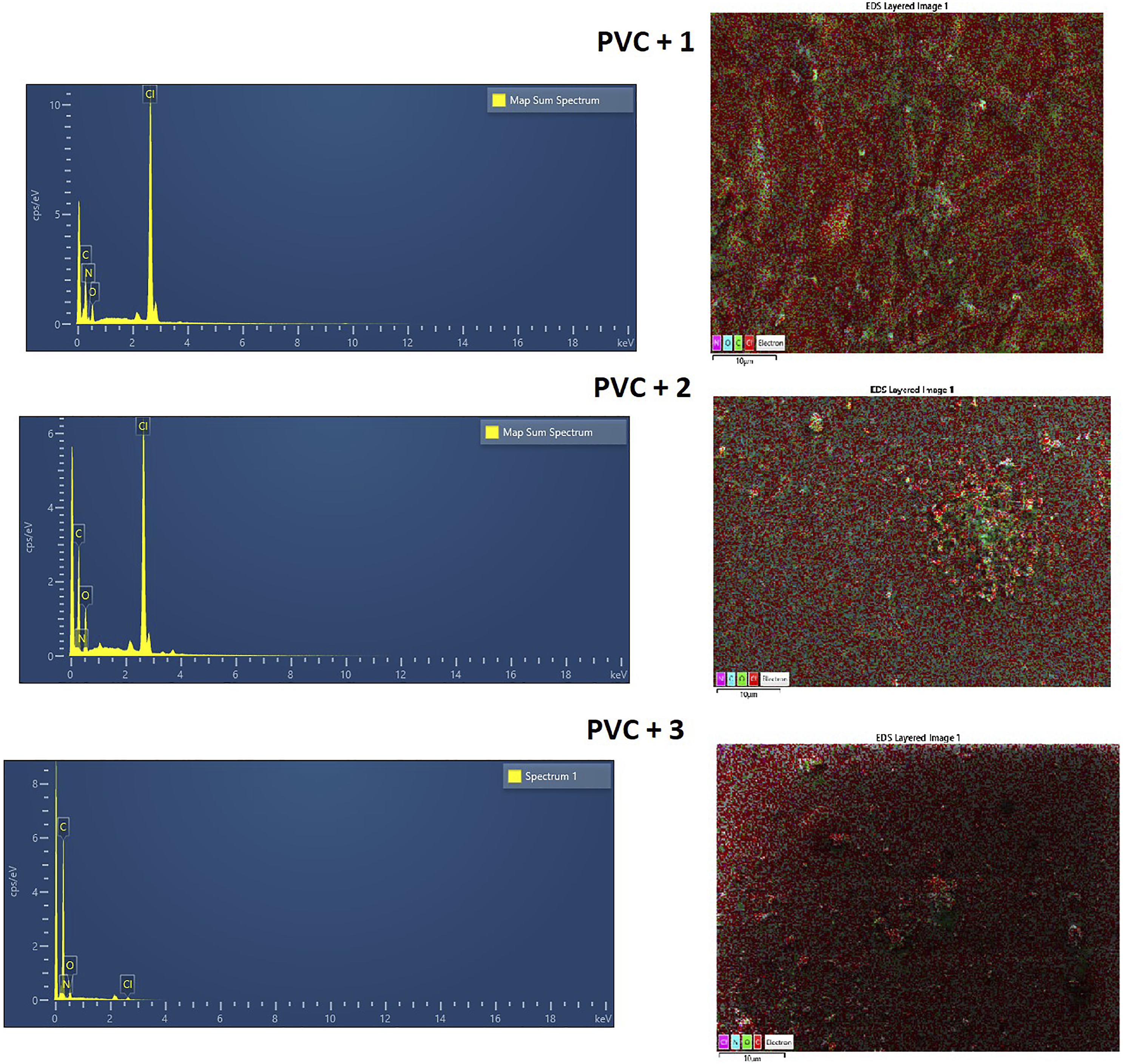
The energy-dispersive X-ray technique was also implemented to analyze the chemical composition of the blank and the blend PVC films before and after UV light exposure. The EDX mapping pattern indicates that the prepared Schiff bases were distributed homogenously throughout the films.
36
The mapping spectra of the PVC blank (Figure 11), and for those containing Schiff bases (Figure 12), revealed bands corresponding to nitrogen, oxygen, carbon as well as chlorine atoms in high percent. Post-irradiation the carbon content of the PVC films increased, with an equivalent reduction in chlorine content. The decreases in the chlorine content are ascribed to elimination of HCl due to photo-degradation and photo-oxidation of the PVC. Likewise, the PVC films containing additives possess a higher percentage of chlorine than that of the pure PVC Figure 12. These observations agreed with previous findings.
37
Energy-dispersive X-ray (EDX) mapping of a pure PVC film post-irradiation. Energy-dispersive X-ray (EDX) mapping of the PVC films post-300 h of irradiation.
PVC photo-stabilization proposed mechanisms
The most common industrial UV absorbers are titanium oxide, carbon black, benzophenones, and triazoles (e.g., hydroxylbenzophenone and hydroxyphenylbenzotriazole). While, the most common additives used recently in research include Schiff bases.13,31,33,38 In general, Schiff bases 1–3 were found to be effective photo-stabilizing additives for PVC against photodecomposition and photo-oxidation. In addition, the low concentration of these additives within the polymeric matrix minimizes any harmful effect at the time of irradiation. However, Schiff base 1 was the most effective as photo-stabilizer for PVC as has been established in Figures 4 to 6. These beneficial properties arise due to the high aromaticity of Schiff base 1, as it contains three phenyl rings, in addition to the azomethine bond within its structure. The additives, specifically additive 1
Conclusions
Successfully, three new highly aromatic Schiff bases containing trimethoprim-based moieties were established to be an excellent photo-stabilizers and antioxidants to reduce the PVC photo-degradation. Interestingly, comparing the weight loss and carbonyl indices of the blank PVC film with those containing the new Schiff bases has demonstrated that the latter have much lower weight loss and carbonyl indices. The surface morphological study of the prepared films by microscope images, field emission scanning electron micrographs and AFM indicate that poly(vinyl chloride)/stabilizer films exhibited much smoother surface and fewer cracks than the blank one after exposure to ultraviolet light.
Footnotes
Acknowledgements
This work is dedicated to Prof. N. C. Billingham, in gratitude for his friendly advice and encouragement at Sussex University, U.K.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.



