Abstract
The photodegradation behavior of poly(vinyl chloride) (PVC) nanocomposites containing different amounts of synthesized titanium dioxide (TiO2) nanoparticles and commercial rutile powder is investigated via the formation of carbonyl compounds, peroxides, and polyenes propagation over 5112 h exposure, as a function of irradiation wavelength and intensity. All the PVC/TiO2 nanocomposites are found to exhibit lower concentration of mentioned species compared with the composite samples. The results show that added TiO2 nanoparticles can retard the photodegradation processes, wherein increasing the amounts of these nanoparticles yields more reduction. With an increase in irradiation intensity the rate of formation of investigated groups increases, although in cases with raised temperature range, this pattern is not observed. An evaluation on the irradiation wavelength effect also shows that more photon energy provides more degradation. The loss of weight analysis shows a tremendous and novel result on evaluating a point of meaningful weight loss.
Introduction
Poly(vinyl chloride) (PVC) has become one of the most important and widely used thermoplastics in the lives of human beings for its good properties such as low production cost, good process ability, easy modification, and excellent chemical and fire resistance. 1 –3 With increasing demands for using PVC products in outdoor applications, its long-term weatherability and durability also have become of great importance. By absorbing solar ultraviolet (UV) radiations, PVC can undergo photodegradation reactions, which finally result in its degradation. 4,5 The photodegradation of PVC is complex, but it has been known that irradiation of PVC leads to dehydrochlorination (hydrochloric acid (HCl) elimination) reactions, which result in the formation of conjugated double bonds and chain-scission processes. Furthermore, photooxidation of PVC can lead to the formation of carbonyl and hydroperoxide compounds. 6 –11
The white pigment titanium dioxide (TiO2) is a particularly effective UV absorber because of the specific effects on absorbing UV irradiation, and its influence on the weatherability of different polymer composites (CPs) has been widely studied. 12 –21 Allen et al. 12 studied the influence of coated and uncoated anatase and rutile TiO2 on the thermal and photochemical oxidation of low density polyethylene (LDPE) by Fourier transform infrared (FTIR) spectroscopy and hydroperoxide analysis. Ma et al. 13 investigated the effect of nano TiO2 on the crystalline structure of LDPE. Cho and Choi 14 investigated the solid-phase photocatalytic degradation of PVC/TiO2 CP, whereas Zhao et al. 15 carried out such study on polyethylene (PE)/TiO2 CP films. Gardette and Lemaire 16 studied the photocatalytic influence of zinc oxide and two rutile forms of TiO2 pigments on the changes in the FTIR spectra of irradiated samples where no noticeable photocatalytic effect of the pigments was observed. In general, the way in which polymer matrix is affected varies considerably with the properties of added TiO2 pigments, in particular, crystal type (anatase or rutile), type and level of inorganic coating, the chemistry of the pigment surface, and crystal size. 17 To the best of our knowledge, most investigations have been focused on photocatalytic aspects of TiO2 nanoparticles, wherein little attention has been paid on the photostability behavior of PVC nanocomposites in the presence of TiO2 nanoparticles. Also, most published literatures on the photostability of polymer CPs were carried out in short periods of exposure (<1000 h). Hence, an investigation on the photostability of PVC/TiO2 nanocomposites over long periods of exposure to evaluate a point of meaningful weight loss can be of great importance.
Based on our previous work, 20,21 the photostability behavior of PVC nanocomposites containing different amounts of homemade TiO2 nanostructure materials (nanoparticles and nanorods) as UV absorber was investigated for short periods of exposure. But in this work, to investigate the influence of photo exposure on the structure and chemical properties of the prepared nanocomposites quantitatively and monitoring the effect of wavelength and intensity in a fairly long period of time, the extent of formation of main products of PVC/TiO2 nanocomposites photodegradation was studied. Two different wavelengths and three irradiation intensities were chosen. The degradation mechanism and the structure changes were investigated via FTIR spectroscopy, peroxide analysis, and viscosity measurements. Additionally, the weight loss trend of nanocomposites was monitored. The results of these experiments enable us to assess the effect of wavelength, irradiation intensity, and loading of TiO2 on the photodegradation process of PVC/TiO2 nanocomposites and propose a view on the effect of photodegradation.
Experimental
Materials
PVC (K-value 64–66; Bandar Imam Petrochemical Co., Tehran, Iran) as polymer matrix, two different TiO2, commercial rutile particles (Mapa-Trans Co., Kiev, Ukraine) and homemade rutile nanoparticles as UV absorber, 21 calcium carbonate (Poodrkar Co., Iran) as filler, chlorinated polyethylene (Poodrkar Co., Iran) as impact modifier, calcium stearate and zinc stearate (Poodrkar Co., Iran) as thermal stabilizers, paraffin wax (Poodrkar Co., Iran) as internal lubricant, and stearic acid (EPS IMPEX Co., Kota Kinabalu, Malaysia) as external lubricant were used to prepare PVC/TiO2 nanocomposite compounds.
Preparation of PVC/TiO2 nanocomposites
The codes and composition of prepared PVC/TiO2 nanocomposite samples are described in Table 1. The preparation procedure of the nanocomposites is essentially the same as described in our previous work. 20
Codes and composition of prepared PVC/TiO2 nanocomposites.
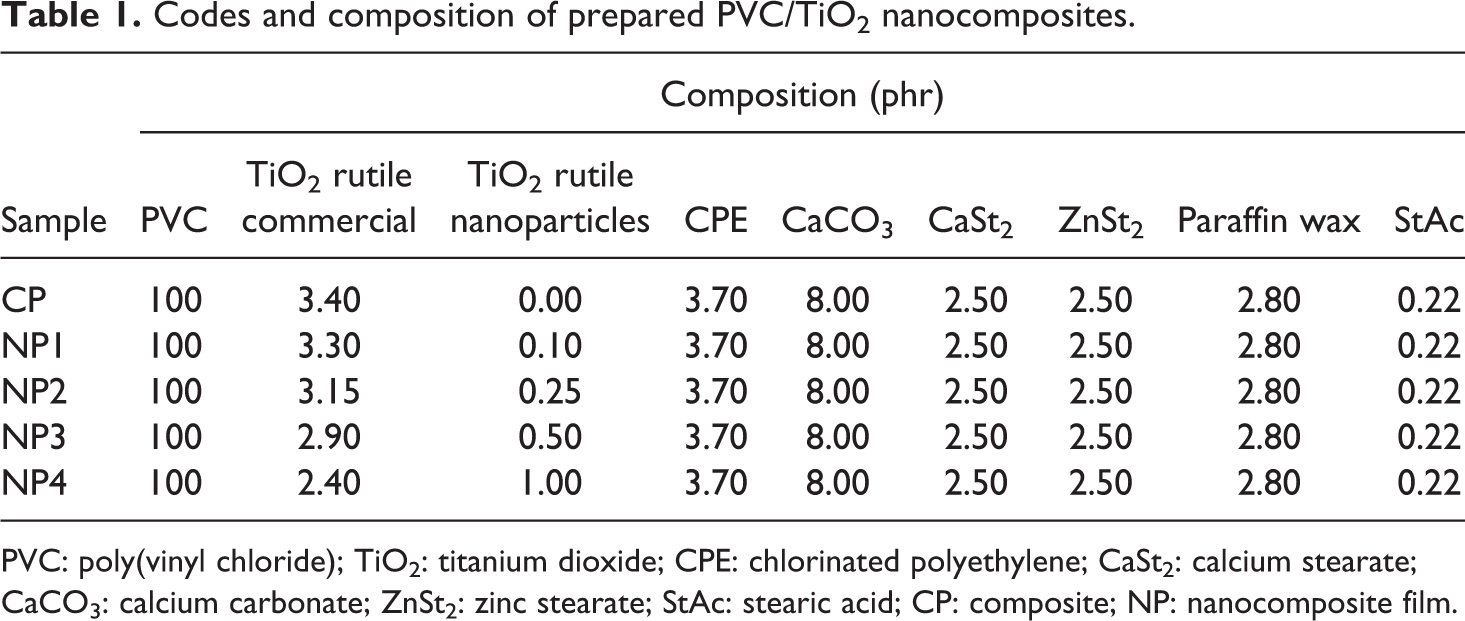
PVC: poly(vinyl chloride); TiO2: titanium dioxide; CPE: chlorinated polyethylene; CaSt2: calcium stearate; CaCO3: calcium carbonate; ZnSt2: zinc stearate; StAc: stearic acid; CP: composite; NP: nanocomposite film.
Artificial weathering
The artificially simulated weathering was carried out using two different light source chambers. The first chamber was equipped with an (TL 20 W/05, Philips Co., Hamburg, Germany) UV exposure lamp with a maximum emission at 365 nm. Another one was equipped with a visible (Vis) exposure lamp (HQI-TS 150 W/NDL, Osram Co., Munich, Germany) with a maximum emission at 590 nm and a fan to have the same temperature ranges in both chambers. To investigate the effect of irradiation intensity, irradiations were executed at three different intensities in both set of chambers. The codes and values of selected irradiation intensities and distances between exposure lamps and samples are given in Table 2. The samples were artificially aged in UV and Vis chambers, for 5112 h with continuous irradiation. Three specimens of each sample were placed under irradiation. Irradiations were performed at room temperature (Figure 1) with a relative humidity range of 40–70% during irradiation period.
Selected irradiation intensities with varying distances.
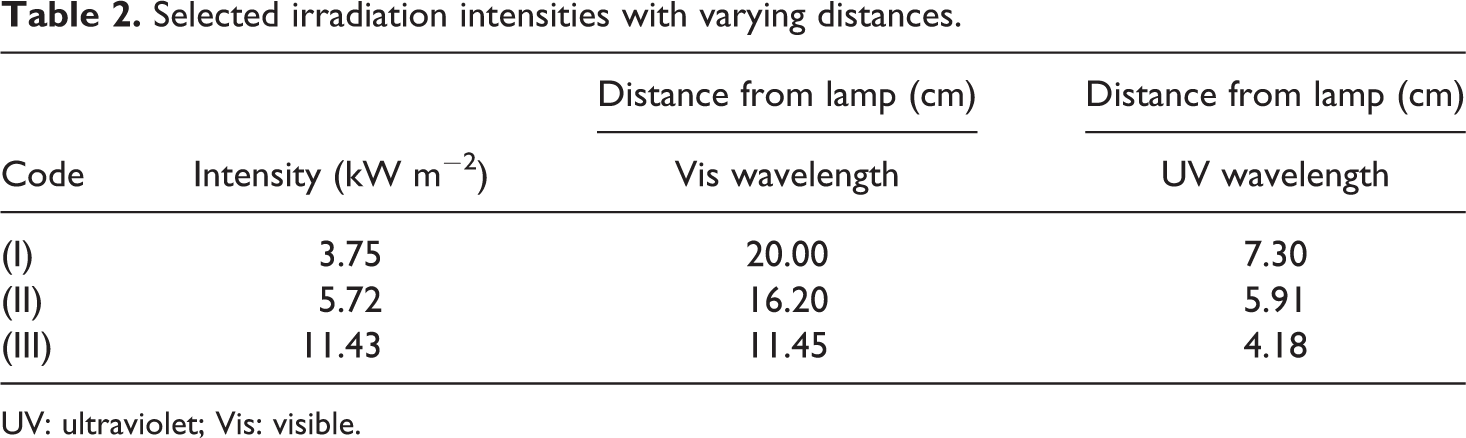
UV: ultraviolet; Vis: visible.
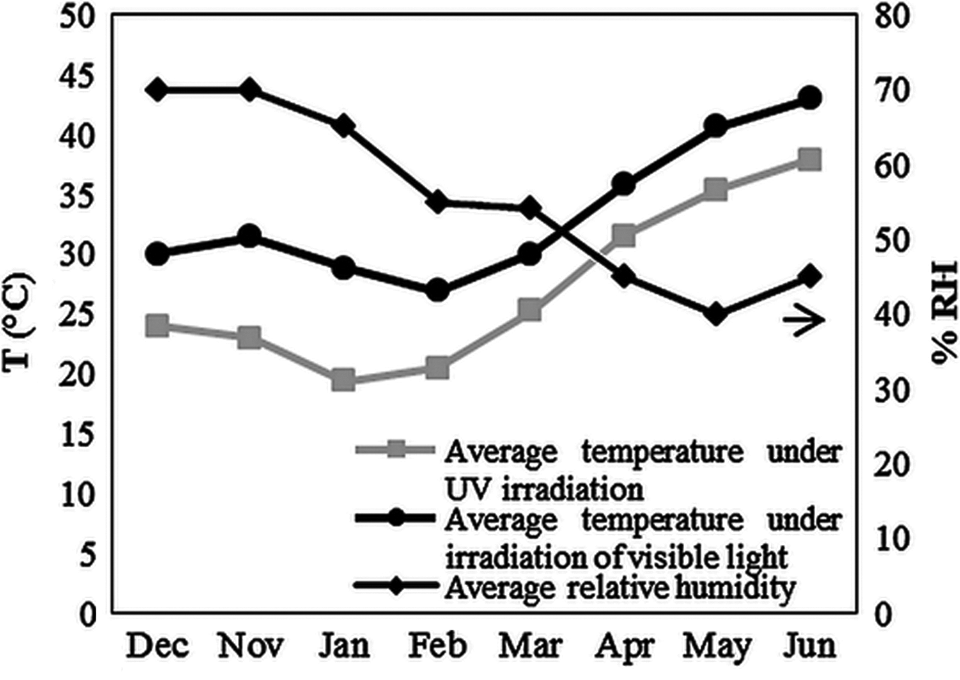
Temperature and relative humidity variation during irradiation period.
Film characterization
FTIR spectroscopy
FTIR spectra were obtained using a TENSOR 27 BRUKER model FTIR spectrometer conducted in attenuated total reflection mode to monitor the structural changes of samples with a thickness of 0.3 ± 0.1 mm before exposure and at periodic intervals after UV- and Vis -accelerated aging. The scanning range was from 400 cm−1 to 4000 cm−1with 4 cm−1 resolution. To obtain a quantitative comparison of the main functional groups formed during the exposure, polyene and carbonyl groups concentration and indices were determined. The infrared absorption bond centered at 1730 cm−1 (ACO) and one at 1650 cm−1 (Au) were used to estimate the carbonyl ([CO]) and polyene ([P
n
]) concentration of the oxidized PVC, respectively. Their concentration (mol L−1) was estimated using following equations
8,22
–24
:

where L is the film thickness (cm) and 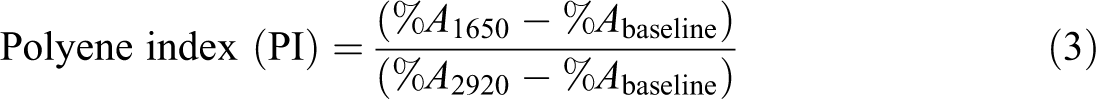
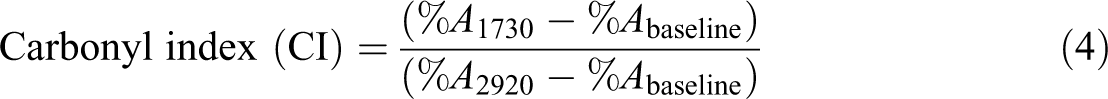
where %Aλ is the value of the integrated peak area of the absorption band at given wave numbers (λ) and %A2920 is taken as the reference value which corresponds to absorption of C–H stretching bands. The peak area of the carbonyl and polyene bands was estimated using the Essential FTIR software (Version 1.1.0.0, Operant LLc, Burke, Virginia, USA).
Peroxide determination
The peroxide (ROOH) content of photooxidized films was determined by iodometric titration as it was done for many different polymers.
25,26
To do so, the method of Wagner, Smith, and Peters was used.
12,27
Polymer film samples (5–10 mg) were cut into small pieces and refluxed with 7.0 mL of tetrahydrofuran (THF, >99%, Merck, Darmstadt, Germany) solvent, and then the whole solution was refluxed with 2.0 mL solution of sodium iodide (Merck) in isopropyl alcohol (IPA, >99.5%, Merck) for an extra 10 min; the apparent peroxide content did not increase with longer refluxing time. At the end of reflux period, the solution was titrated with a solution of 0.002 N sodium thiosulfate (Na2S2O3, Merck) to a yellow-to-colorless end point. With the above procedure, it is possible to take advantages of the oxidative stability of iodide ions in IPA solutions.
27,28
The peroxide concentration measured with this method is the concentration of total peroxides (hydroperoxides, peracides, and some dialkyl peroxides) in oxidized films.
28,29
With regard to reactions mentioned below, it can be recognized that the consumed Na2S2O3 concentration is proportional to the concentration of ROOH groups formed in the degraded films.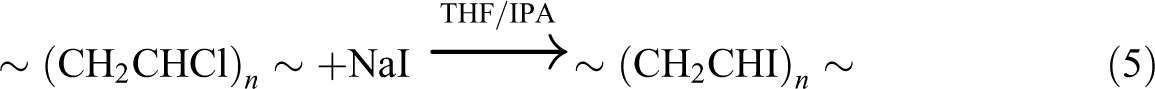
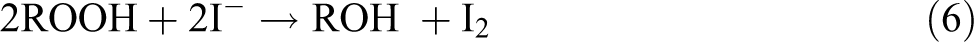

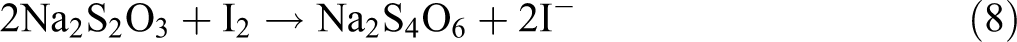
Viscometry
Intrinsic viscosities were determined by dilution viscosity measurements in THF at 25°C by using Huggins equation.
26
Number-average molecular weights 
Each of these mentioned analyses was performed at four periodic intervals, that is, before exposure (0 h) and after 1700, 3400, and 5112 h of irradiation to assess the extent of degradation in the nanocomposites.
Weight loss
To monitor the changes in the mass of nanocomposites over irradiation time, a Shimadzo model AY220 (Japan) balance with an accuracy of 0.1 mg was used. Measurements were carried out every 50 h for samples with the average thickness of 0.71 mm and a surface area of 12 × 25 mm2.
Results and discussion
FTIR analysis of nanocomposites
Degradation is actually due to the attack of highly oxidizing free radicals on the polymer chains with the formation of various end-groups. Figure 2 shows typical spectra of the CP and nanocomposite film (NP2) before exposure and after 3400 h UV irradiation. In general, absorption peaks appeared in the ranges of 500–610, 800–1000, 2500–3100, and 3500–3700 cm−1 and also around 1100, 1252, 1360, 1430, 1733, and 1630 cm−1, respectively. The peak at 1630 cm−1is due to C=C stretching. The peaks around 2500–3000 cm−1 represent C–H vibrations and the one around 1733 cm−1 is indicative of the formation of carbonyl groups as a consequence of degradation, which are in the neighborhood of C–Cl bonds. 5,32 On the other hand, if the possible formed carbonyl groups are classified as γ-lactones, esters, ketones, and carboxylic acids, the appeared peak can be attributed to the esters. 33 The peaks around 500–610 and 800–1000 cm−1are the characteristic of crystalline rutile and its reduction products. The peaks at 3080, 965, and 875 cm−1 can also reveal the presence of unsaturation sites, that is, vinyldenic unsaturation, where their deformation absorption occurs. Unsaturation is believed to be specific to photooxidation where the dominant unsaturation group is vinyl which probably results from the Norrish type II photolysis of backbone ketone groups (Figure 2). 33
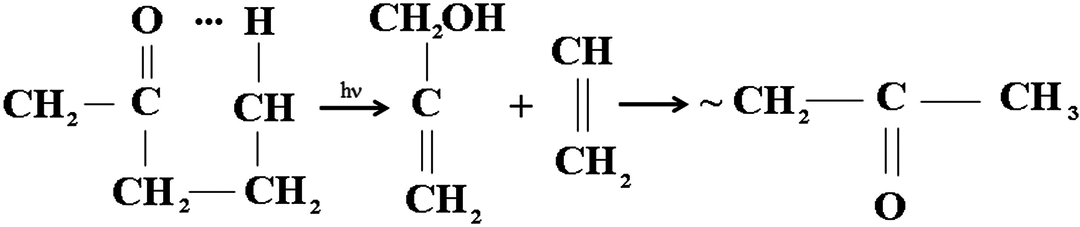
Vinyl formation through the Norrish type II photolysis of backbone ketone groups. 31
The peak at 3560 cm−1can be attributed to non-hydrogen bonded (free) hydroperoxides.
5,33
The peaks appeared around 600–700 cm−1 can also be an indication of C–Cl stretching bonds. The peaks at 1252 cm−1 result from CH2 wagging when the adjacent C atom has a Cl atom attached and those at 1360 cm−1 arise from deformation vibration of H–C–Cl or just a vibration of C–H bonds. The one at 1430 cm−1 represents scissoring C–H bonds and at 1100 cm−1 shows H-C-O bonds. One can also attribute the peaks around 965 and 875 cm−1 to
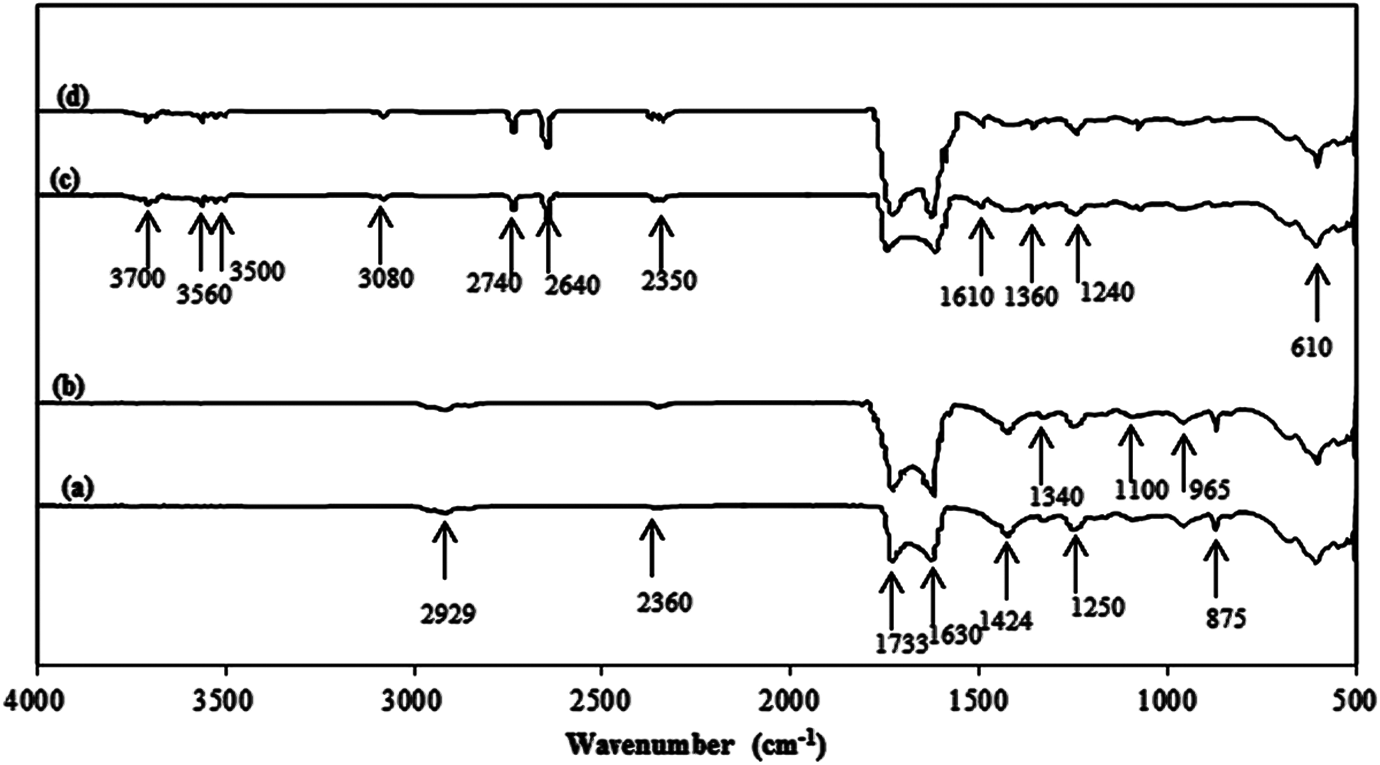
FTIR spectra of NP2 sample (0.25% nano TiO2) (a) before exposure and (b) after 3400 h exposure and CP sample (without nanoparticle) (c) before exposure and (d) after 3400 h exposure of UV irradiation at 11.43 kW m−2. FTIR: Fourier transform infrared; TiO2: titanium dioxide; CP: composite; NP: nanocomposite film.
The oxidation of PVC can be followed by FTIR through the absorbance variations in the range of 1600–1900 cm−1 which corresponds to the main formed groups: double bonds (between approximately 1600 and 1680 cm−1) and carbonyl groups (approximately 1650 to 1800 cm−1). The absorbance intensity variation in this range results as a consequence of PVC degradation (dehydrochlorination with the consequent formation of carbonyl groups) and also from the consumption of stabilizers during the exposure process. Figure 4 shows the typical FTIR spectra of samples in the range of 1500–1900 cm−1 irradiated for 0 and 5112 h.
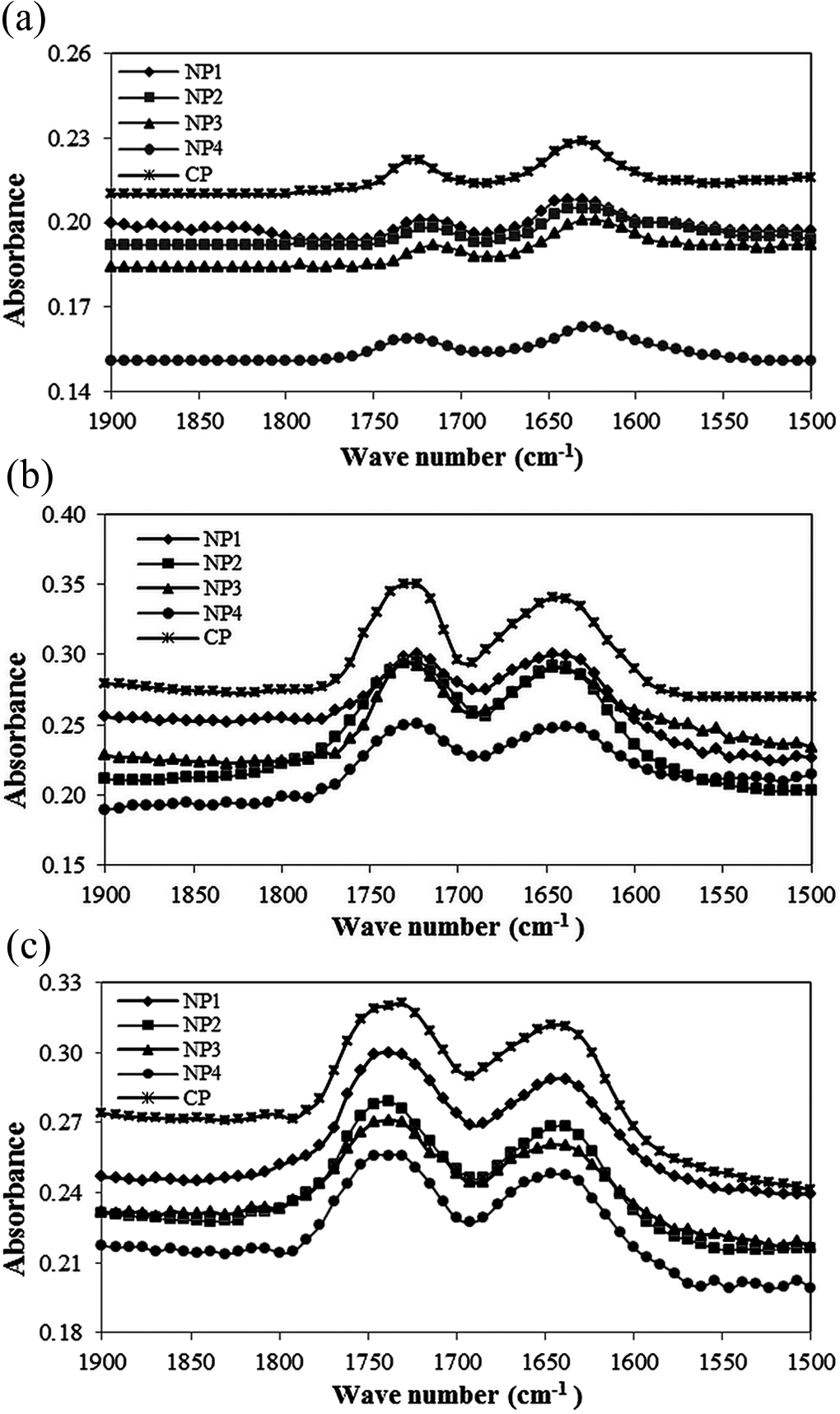
FTIR spectra in the range of 1500–1900 cm−1 for nanocomposites and commercial samples (a) before exposure and after 5112 h exposure under irradiation of (b) UV light at 5.72 kW m−2 and (c) Vis light at 11.43 kW m−2, respectively. FTIR: Fourier transform infrared; UV: ultraviolet; Vis: visible.
As shown in Figure 4, the significant changes in polyene and carbonyl intensities were observed. The absorption intensity pattern of polyenes, in all samples, followed an increasing trend till 3400 h of exposure and then decreased; while for carbonyls, an increasing pattern up to 5112 h irradiation was observed. This pattern can be obviously followed through the concentration and indices graphs of these groups calculated based on equations (1) to (4), versus irradiation time (Figures 5 and 6). All samples before exposure exhibited low values of CI and PI. With increasing exposure time, the indices values rose significantly. This indicated the incidence of HCl elimination in the PVC matrix resulting into double bonds formation, suggesting the degradation of all samples during exposure in the UV and Vis light region. This pattern in the case of carbonyl groups also suggested that the photooxidation reaction had taken place in the films. In the composite films, TiO2 nanoparticles can be excited by the influence of light and generate active electron–hole pairs, and then by reacting with the surface-absorbed molecules form active oxygen species. These active oxygen species oxidize C–H groups to form carbonyl ones. 34 After irradiated for 5112 h, the indices values of samples containing TiO2 nanoparticles were lower than that of commercial CP. Also with increasing the amount of these nanoparticles in the compound structure, the indices values showed a decreasing trend. This observed pattern could be attributed to light absorbing property of the synthesized TiO2 nanoparticles. These nanoparticles with absorbing more light quanta lead to less diffusion of light into the nanocomposites, which results in less dehydrochlorination and less formation of consequent products. So, this means that the photostability of PVC in the presence of synthesized TiO2 nanoparticles increases.
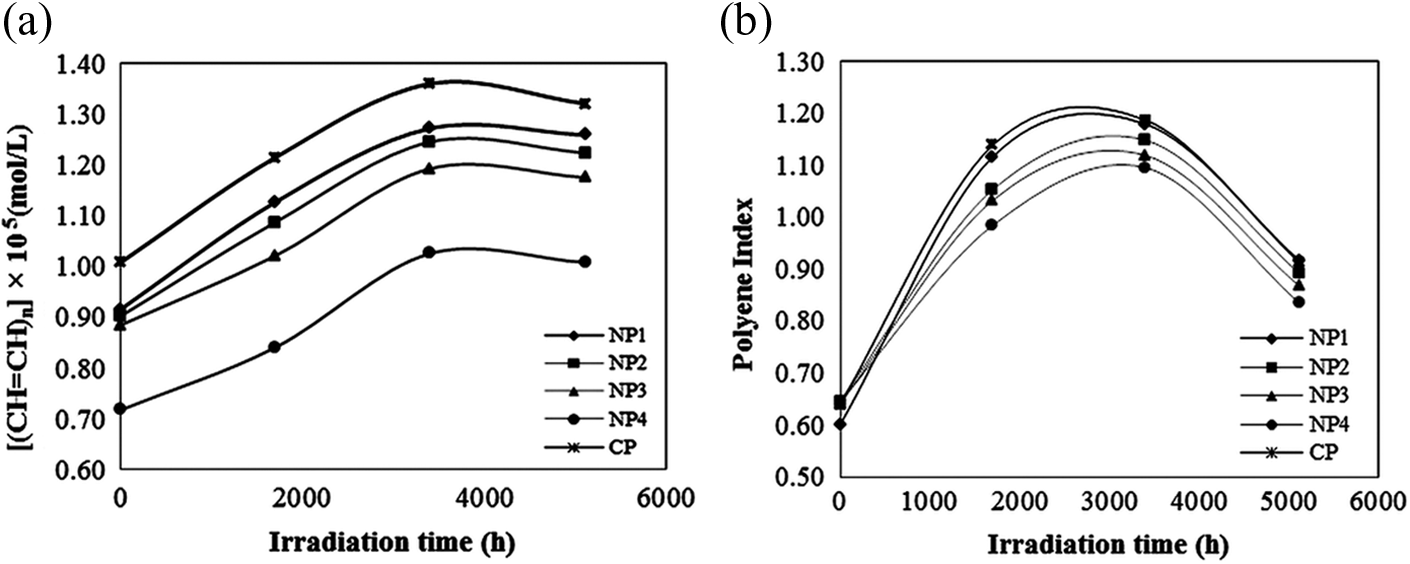
Typical polyene (a) concentration and (b) index variation versus irradiation time under UV exposure at 3.75 kW m−2. UV: ultraviolet.
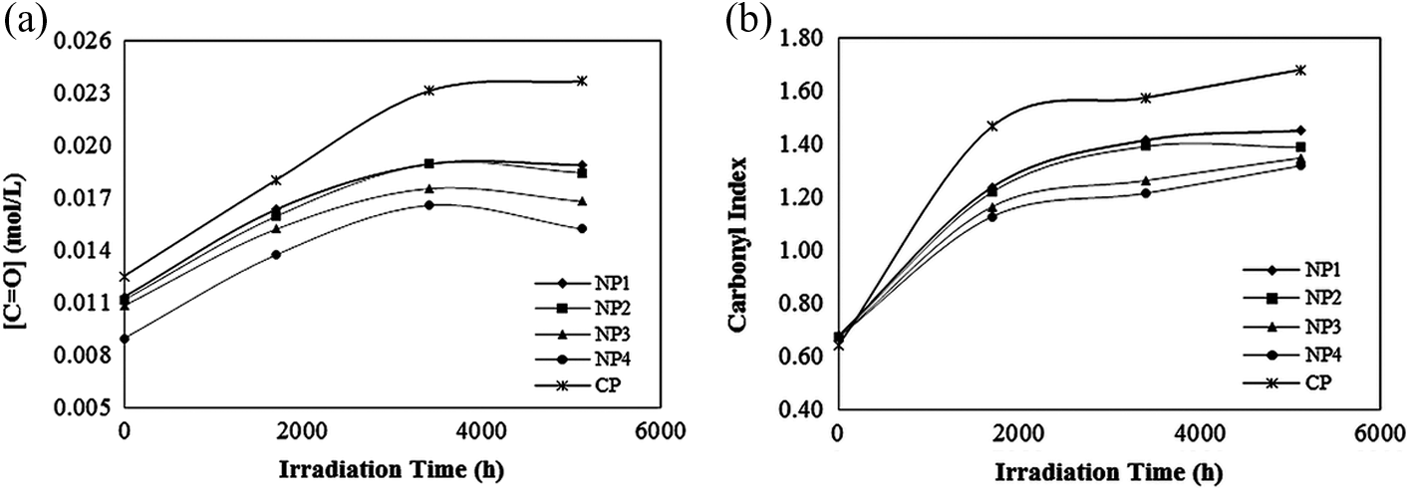
Typical carbonyl (a) concentration and (b) index graph versus irradiation time under UV exposure at 11.43 kW m−2. UV: ultraviolet.
Peroxides determination
Figure 7 shows a typical concentration graph of formed peroxide groups in the degraded samples, although for a convenient comparison relative concentrations were considered (Figure 8). As seen in Figure 7, all samples have an initial concentration of peroxide groups, which is due to the history of processing procedure of samples till placing under irradiation in chambers, though samples with TiO2 nanoparticles show less peroxide content compare to commercial ones.
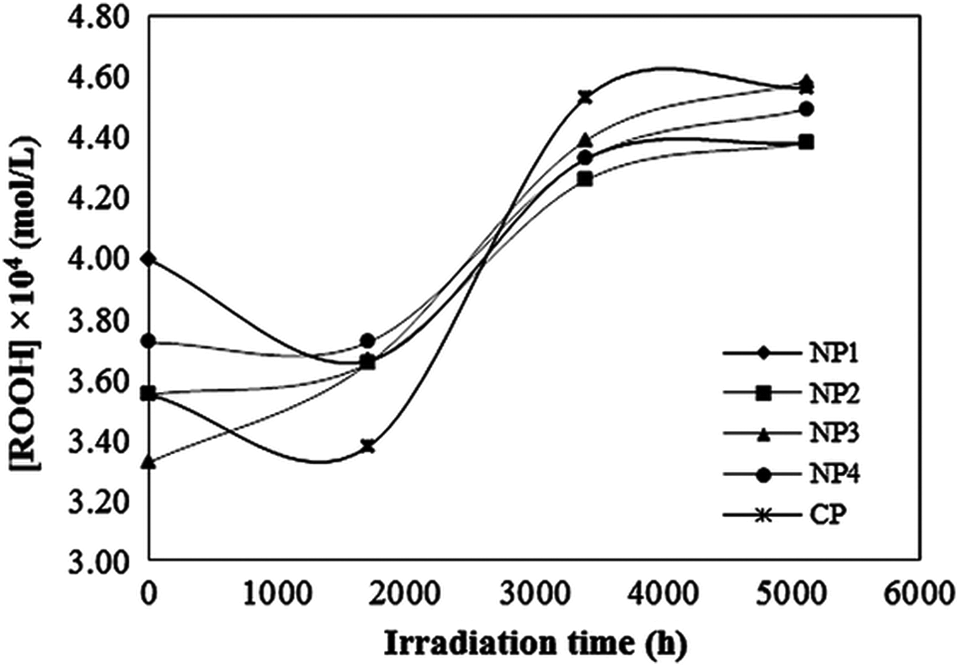
Typical concentration of peroxide species versus exposure time under UV irradiation at 5.72 kW m−2. UV: ultraviolet
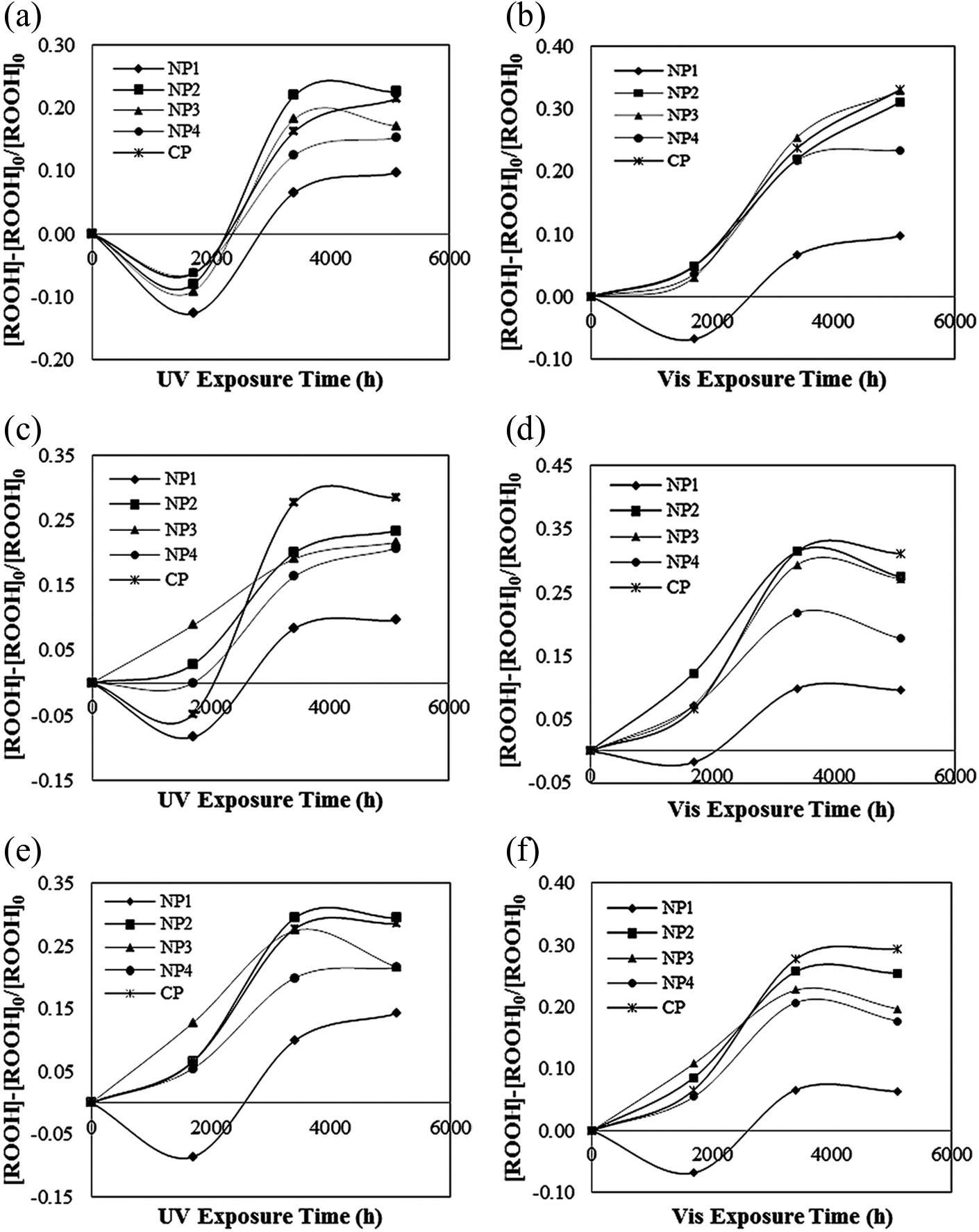
Relative concentration of peroxides under UV and Vis light irradiations at (a) and (b) 3.75 kW m−2, (c) and (d) 5.75 kW m−2, and (e) and (f) 11.43 kW m−2. UV: ultraviolet; Vis: visible.
In Figure 8, an S-shaped kinetic curve of formed peroxides is observed. These S-shaped kinetic curves that were reported by other authors too can be explained as follows.
23,33,35
In the early stages of degradation, irradiation causes a decline in –OOH level to accelerate oxidation, which can be observed in times between 0 h and 1700 h. Three reasons may explain this behavior: (i) with regard to reactions represented in literatures for PVC photodegradation, it is known that –OOH groups are formed as a consequence of polyenes photocleavage. These formed –OOH groups by absorbing light quanta convert to
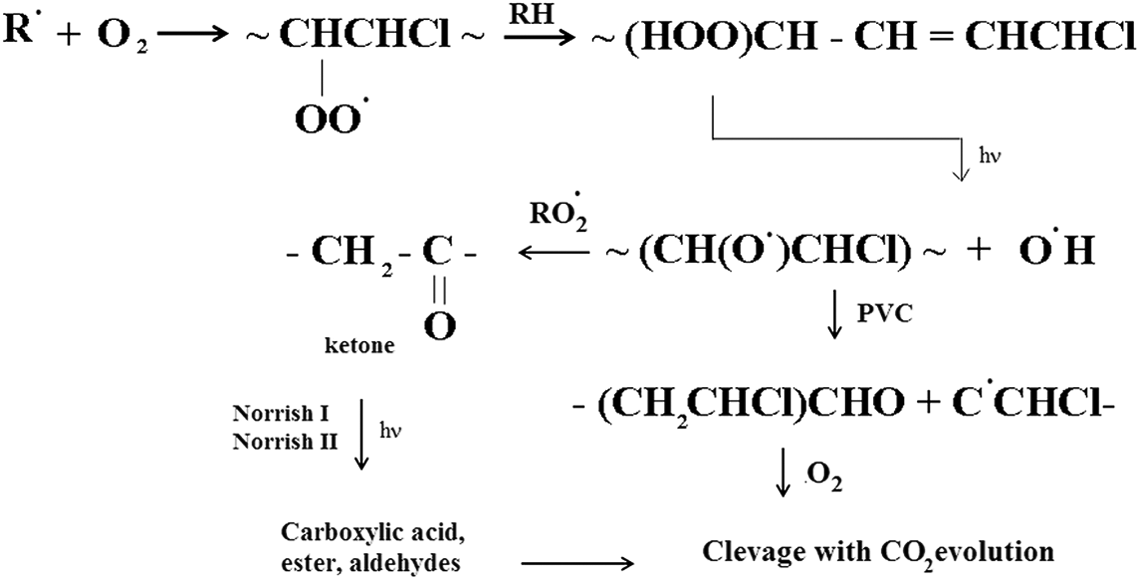
Photooxidation mechanism of peroxides.
When polyenes begin to build up, with increasing the irradiation time, the incident photons are increasingly absorbed by the top layer of film which leads to an auto-accelerated degradation and accordingly the concentration of peroxides increase. This behavior can be observed in times between 1700 h and 3400 h after exposure. After this time, with more irradiation, the concentration pattern slowed down and reached to a plateau value. The plateau value of peroxides after 3400 h irradiation may indicate that this group reached a photostationary level. The presence of the photostationary conditions may support all investigations on different polymers which report such data and explain in part why –OOH groups are not photoinitiators in PVC/TiO2 samples too, although they are clearly present in PVC. 30 In the later stages, the observed slowing down is assumed to be a result of the formation of a thin layer of totally degraded PVC on the irradiated side. This colored top layer by absorbing the entire incident light, converted it into heat then by acting as an efficient UV screen prevented the underlying layers from further degradation, as explained by Decker and Balandier. 23 Such a light-screening effect by photo products is a usual process in photooxidation of polymers where highly absorbing chromophores are formed. This in the case of PVC is due to both large amounts of produced conjugated polyenes and their high absorption coefficients.
Molecular weight changes
Cleavages of the polymer chain and cross-linking are two important features that are directly responsible for the embrittlement, loss of mechanical properties of PVC, and also variation in molecular weight distribution of photodegraded films. 22 Figure 10 shows a typical relative molecular weight variation of samples, calculated based on equation (9), versus irradiation time under UV exposure at the irradiation intensity of 5.72 kW m−2 (intensity (II)). In the initial stages of irradiation (0–1700 h), the intrinsic viscosity of the PVC/TiO2 films significantly decreased, indicating that chain scission is predominated. Such a result was also reported by Decker and Balandier for UV-degraded PVC. 23 Chain scission is often accompanied by carbonyl formation, because these groups are produced essentially through the β-scission of alkoxy radicals or ketone photolysis. The main chain scission of polymer backbone can be due to both photochemical and thermal reactions of radicals. So, these results can be another explanation why the concentration of carbonyl groups increased in 0–1700 h of exposure. With increasing irradiation time, between 1700 h and 3400 h of exposure, the molecular weight of all samples increases, indicating that cross-linking is taking place. The possibility of such behavior was reported for photodegraded PVC films in literatures. 23 –25 Then in the later stages of exposure (3400–5112 h), the cross-linking again shifted in favor of chain scission. It is known that chain scission is associated with oxidation, while cross-linking is believed to be happened as a consequence of polyene formation and also of the intermolecular peroxide bridges formed by termination reactions of macroperoxy radicals. 22,31,36 The above illustrated relations between chain scission and cross-linking with the formation of investigated groups and the observed pattern in the content of polyenes, peroxides, and also in that of carbonyls will properly explain the observed trend in molecular weight changes.
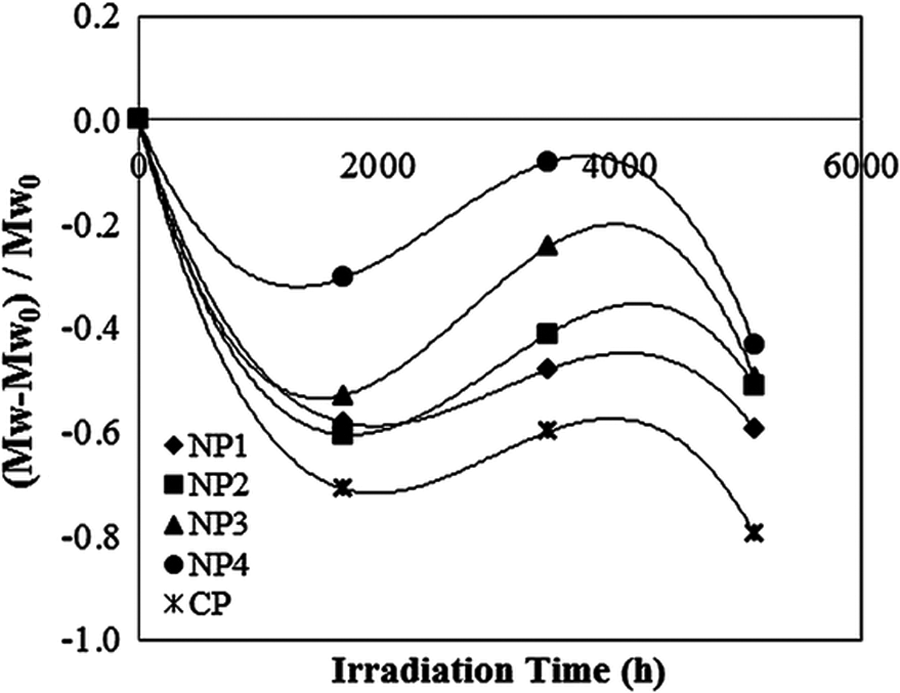
Typical relative molecular weight variations of samples versus irradiation time under UV exposure at 5.72 kW m−2. UV: ultraviolet.
The essential point to state is that CP samples in early stages of exposure go through chain-scission processes much more than samples containing TiO2 nanoparticles, which is an indication of more influence of irradiations on the former samples and more photostability of the nanocomposites. Also with increasing the concentration of nanoparticles in compound structure the molecular weight showed less variation, indicating that the photostability of nanocomposites increases with increasing TiO2 nanoparticles.
Effect of irradiation intensity
Photon energy from the irradiation source is an important factor in photolytic reactions. So, for clarifying the effect of irradiation intensity on the photodegradation of PVC/TiO2 (nano)composites, the polyene and carbonyl indices and peroxides concentration at the same wavelengths over irradiation period were studied (Figure 11). As expected, under UV irradiation with increasing the intensity the extent of these groups formation increase. But at irradiations under Vis light an unexpected result was yielded. Intensity (II) > (I) > (III) showed more degradation, respectively (intensities I, II, and III denote for 3.75, 5.72, and 11.43 kW m−2, respectively). In regard to the temperature variation graph over irradiation period (Figure 1), it can be obviously seen that the average temperature of chambers under irradiation of Vis light was higher than chambers under UV irradiation. This observation was due to the higher temperature of boxes at intensities (I) and (II) which had an average higher temperature of 5°C and 10°C, respectively, compared with the box at intensity (III). The high temperature increases the rate of oxygen diffusion into the surface. Also, oxygen by reducing the activation energy increases the rate of dehydrochlorination. On the other hand, for all polymers, an increase in surface temperature increases the rate of photooxidation as well as the rate of dehydrochlorination, and this thermo-oxidation process creates oxidation products, such as hydroperoxides and carbonyls. 37
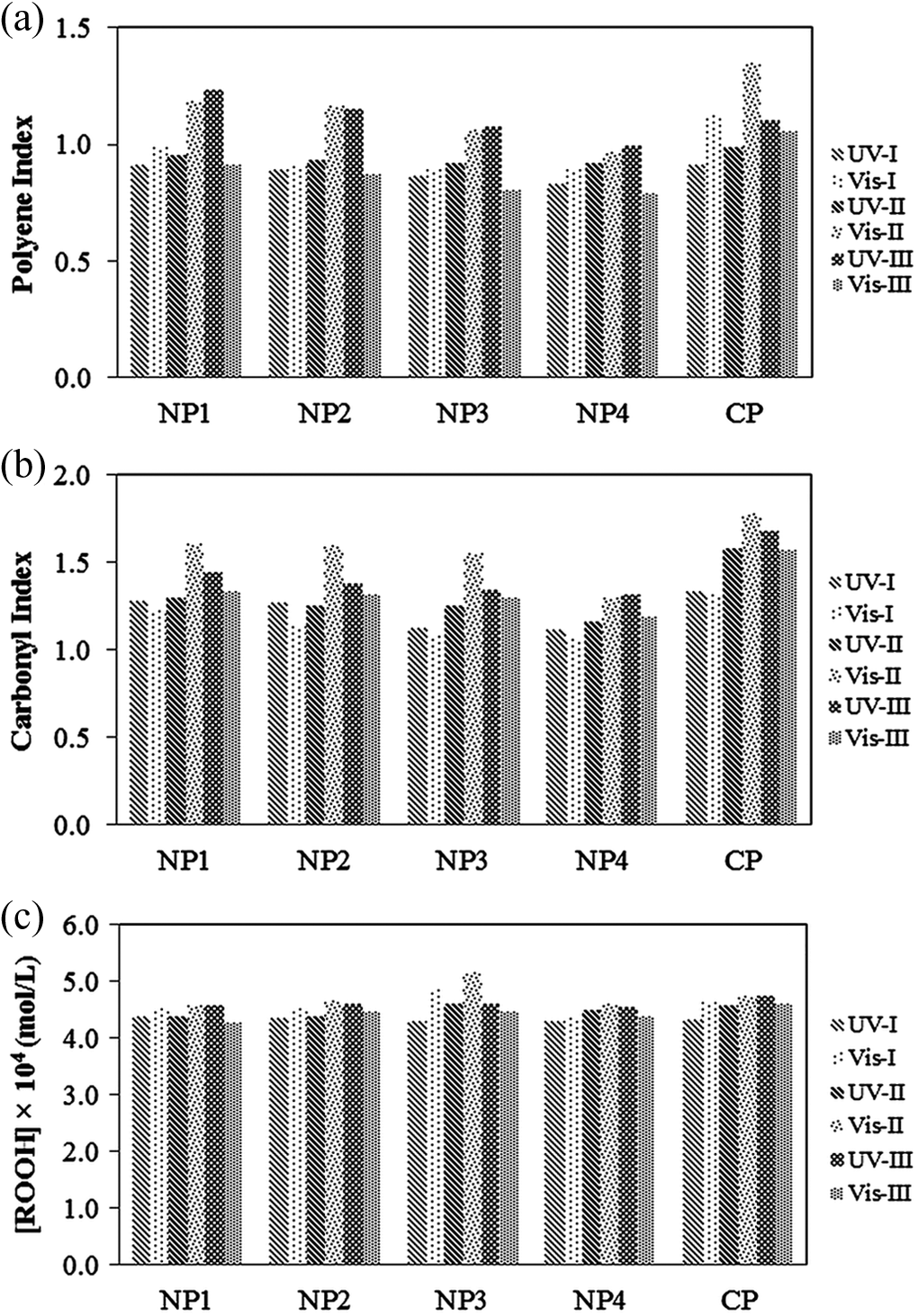
Variation of samples (a) polyene, (b) carbonyl index, and (c) peroxide concentration at all investigated conditions after 5112 h exposure; UV-I, -II, and -III denote the UV exposure at intensities 3.75, 5.75, and 11.43 kW m-2, respectively. Similarly Vis-I, -II, and -III indicate the Vis exposure at the same intensities as UVs. UV: ultraviolet; Vis: visible.
Effect of irradiation wavelength
In order to investigate the wavelength effect on the formation of polyenes, carbonyl groups, and peroxides, their indices and concentrations versus UV and Vis wavelengths at the same intensities are shown in Figure 11. As the photon energy is proportional to reciprocity of wavelength, UV radiation has more energy than Vis light. So, it is expected that irradiation under UV exposure causes more degradation than Vis light. But as observed in Figure 11, at intensities (II) and (I), a contrary result has been obtained and irradiation under Vis light causes more formation of polyenes, carbonyls, and peroxide groups which can be due to higher temperature of these intensities compared with UV chamber.
Effect of photodegradation on weight loss
Figure 12 displays the photoinduced weight loss of all investigated samples. The kinetic curves of weight loss show an overall induction time of nearly 3400 h irradiation and then follow a decreasing pattern. But as observed in Figure 12, the induction time for samples with TiO2 nanoparticles is more than CP one, confirming more durability of samples including the synthesized TiO2 nanoparticles. Also, the durability of samples increases with the increase of nanoparticles concentration. The above weight loss data also revealed that the photodegradation of PVC/TiO2 film led to the bond scission and formation of a mass of volatile intermediates.
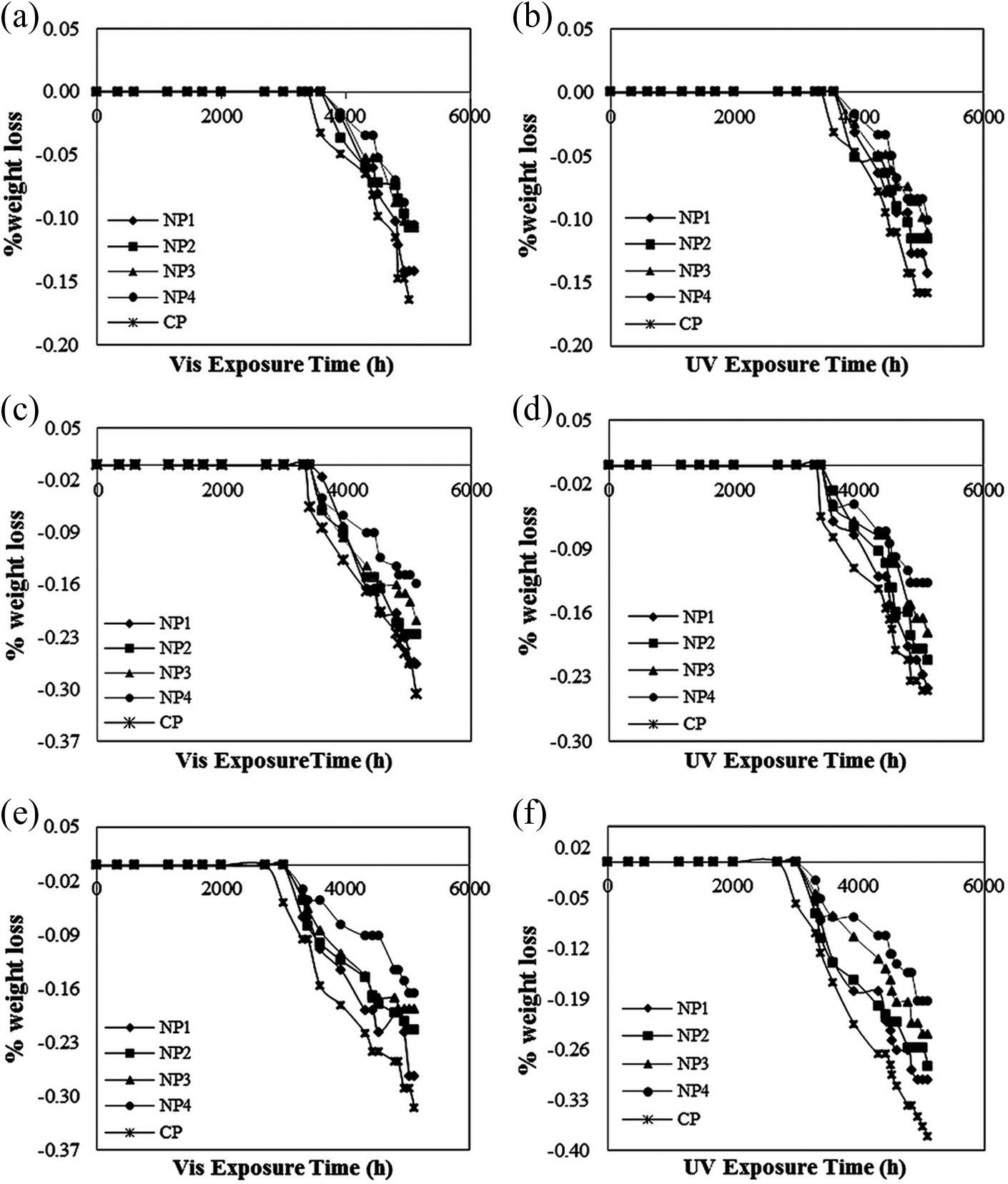
Kinetic curves of weight loss along Vis light and UV exposure time at intensities (a) and (b) 3.75 kW m−2, (c) and (d) 5.75 kW m−2, and (e) and (f) 11.43 kW m−2. UV: ultraviolet; Vis: visible.
Here the most interesting observation to discuss is the induction time. The reason for such an unexpected long induction time can be identified with the formation of polyenes, peroxides, and carbonyl groups which were discussed in FTIR analysis of nanocomposites and peroxides determination. The increasing pattern that was observed in the formation of these mentioned groups concealed the influence of evolved chlorine on the weight loss. After this time, with more irradiation (3400–5112 h), decrease in the concentration of polyenes and initiation of a photostationary level for peroxides and also considering the formation of volatile intermediates through peroxides and carbonyls photocleavage caused the weight loss to be observable.
Conclusion
The artificial UV- and Vis-accelerated exposure tests of PVC/TiO2 composites and nanocomposites were carried out to investigate the chemical properties of the (nano)composites over long periods of exposure and evaluate the effect of irradiation intensity and wavelength on the photodegradation behavior. The experimental results showed that the concentration of polyenes decreased after an increasing pattern up to 3400 h exposure, while for peroxides, an S-shaped kinetic curve was observed. In the case of carbonyl groups, an increasing behavior till the end of irradiation period was obtained. With increasing the irradiation time, competitive chain scission and cross-linking processes were also developed in the photooxidized nanocomposites. As for the effect of degradation on mass, a decreasing trend was observed after an induction time. This induction time was really unexpected because all the performed analyses supported the occurrence of photodegradation but with no meaningful variation on mass; noting that this novel result was reported for the first time by this research team. The variation trend of polyenes and peroxides with respect to weight loss results reveals that these groups did not influence the rate of photodegradation of nanocomposites. According to investigated analyses, the presence of TiO2 nanoparticles revealed a decreasing pattern of degradation due to their light-absorbing properties. An increase in the irradiation intensity increased the rate of degradation, although in cases with increasing of temperature this pattern may act in contrary. An evaluation on the irradiation wavelength effect also showed that with more photon energy more formation of degraded groups can be obtained.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
