Abstract
Sb2O3 nanoparticles (nano-Sb2O3), montmorillonite (MMT) and brominated polystyrene (BPS) were used to enhance flame retardancy of polypropylene (PP) in the work, in which nano-Sb2O3 was modified by cetyl trimethyl ammonium bromide and polyethylene glycol and MMT was modified by silane coupling agent. PP matrix flame retardant composites were prepared by melt blending after pre-mixing by high energy ball milling technique. Then, the flame retardancy, thermal stability behaviour, crystallization performance and tensile strength of PP matrix composites (nano-Sb2O3/MMT/BPS-PP, nano-Sb2O3/BPS-PP) were investigated. The results show that MMT can significantly improve the thermal stability and flame retardancy of PP matrix due to improving the strength of char layer and forming barrier effect. Compared with 3.5%nano-Sb2O3/8%BPS-PP composites, the limiting oxygen index of 3.5%nano-Sb2O3/3%MMT/8%BPS-PP composites was increased from 26.9% to 29.0% and its UL94 grade was achieved to V-0 level. Moreover, MMT and nano-Sb2O3 can increase the crystallinity and tensile strength of PP matrix composites because of the heterogeneous nucleation effect of MMT and nano-Sb2O3 particles on the crystallization of PP matrix.
Introduction
Polypropylene (PP) is widely used in automobiles, textiles, electrical insulation, architectural materials, interior decorations and so on with high-cost performance, excellent mechanical and physicochemical properties.1,2 However, PP is inflammable and may be dangerous closing to fair. It may also enhance fair propagation due to its dripping tendency during combustion. The application of PP in many fields is severely limited due to its flammability and dripping behaviours. 3 Thus, the flame retardancy of PP has to be improved. A lot of work has been done in the past several decades to overcome the shortcoming of PP and some progresses have been achieved. The flame retardancy of PP is usually improved by adding flame retardants into PP or grafting flame retardant elements onto PP. The flame retardants include halogen, phosphorus, silicon, etc. As the synergistic system of brominated flame retardants and Sb2O3 (Br-Sb system) is considered as a more effective flame retardant system, its use in polymer materials can prevent oxygen, heat, flammable materials and thermal degradation reactions, thus achieving flame retardant effect.4,5 For instance, The poly (pentabromobenzyl acrylate), Sb2O3 and modified magnesium hydroxide are compounded with PP to prepare flame retardant PP matrix composites. The results show that the PP matrix composites had excellent thermal stability and flame retardancy. 6 In the Br-Sb system, the dispersion of Sb2O3 particles has a significant effect on the flame retardancy of polymer materials. Some research results show that the surface modification of Sb2O3 particles can effectively improve its dispersion in polymer materials and improve flame retardancy.7,8
Brominated polystyrene (BPS) is a new high-temperature brominated flame retardant with higher bromine content, higher flame-retardant efficiency and higher molecular weight. In particular, BPS is non-toxic and is difficultly absorbed by the organism. Materials containing BPS does not produce dioxins during burning, which complies with EU RoHS directive and EPA regulations. The compounded flame retardant composed of BPS and Sb2O3 has good flame retardant efficiency due to the synergistic effect of Br-Sb system. The compounded flame retardant has been well applied in PBT and PET and has broad application prospects. 9
The flame retardancy of PP is still realized by adding convention halogen flame retardant and Sb2O3. However, the kind of flame retardant will release a lot of black smoke, toxic gases and corrosive smoke during combustion, which will cause serious environmental pollution. 10 In order to improve the environmental friendliness of flame retardant PP, nano-Sb2O3/MMT/BPS-PP and nano-Sb2O3/BPS-PP composites were prepared, respectively, by using Sb2O3 nanoparticles (nano-Sb2O3), BPS and montmorillonite (MMT) as flame retardant additives in the paper. And the effects of Br-Sb and MMT on flame retardancy, thermal stability behaviour, crystallization performance and tensile strength of PP matrix composites were studied.
Experimental
Materials
Silane-modified MMT with 15 μm of average particle size was supplied by Sunda Chemical Co. Ltd. of China. Nano-Sb2O3 modified by Polyethylene glycol (PEG-4000) and cetyl trimethyl ammonium bromide (CTAB) with 50 nm of average particle size was made by our laboratory. 11 The main reagents used in the preparation process were PEG-4000, CTAB, hydrogen peroxide (H2O2, 30% of concentration) and distilled water that were all analytically pure. PP was purchased from Shihua Petrochemical Co. Ltd. of China. BPS containing more than 68.7% of bromine content was supplied by Qingdao Uchem Co. Ltd. of China.
Sample preparation
The composition of experimental materials.

Characterization
Flame retardancy
The limiting oxygen index (LOI) was analyzed by PX-01-005 limited oxygen index tester (Suzhou Phinix Quality Inspection Instrument Co. Ltd. of China) according to GB/T2406.2-2009. The UL94 grade was performed by STD-94 vertical burning tester (Shanghai Pan Standard Textile Testing Technology Co. Ltd. of China) according to GB/T2408-2008. The Catalytic Effectivity (CAT-EFF) of nano-Sb2O3 for PP matrix composites is defined as a variation of LOI with 1% mass fraction of metal ion at the optimal concentration of the catalyst.
12
The value is calculated by equation (1)
The flame retardant efficiency (EFF) and the synergistic efficiency (SE) are calculated by the following equations
13


Thermal gravimetric analysis
Thermal decomposition of PP matrix composites was investigated by HS-TGA-101 thermal gravimetric analyzer (Shanghai Heson Instrument Co. Ltd. of China). The sample mass was approximately 10 mg. Thermal gravimetric analysis (TGA) testing was performed at 10 °C/min of heating rate in the temperature range of 20 ∼ 600°C in nitrogen gas.
Scanning electron microscopy
The surface morphology of the char layers of experimental materials after vertically burning was observed by JSM-6700F SEM (Japan Electron Optics Laboratory Co. Ltd.).
X-ray diffraction
X-ray diffraction (XRD) patterns of samples were recorded at room temperature by Ultima IV XRD (Rigaku Corporation of Japan) with 2θ diffraction angle range from 2° to 10° at 2°/min of scanning speed. The value of interplanar distance was calculated by Bragg’s law
14

The crystallite sizes of nano-Sb2O3 and MMT were calculated from full width at the half-maximum (FWHM) by Scherrer’s formula
15

Differential scanning calorimetry
The crystallization and melting behaviours of samples were investigated by using a differential scanning calorimetry (QT-DSC-500C, Shanghai Qiantong Instruments Co. Ltd. of China). Samples of about 15 mg were encapsulated in aluminium pans and heated from room temperature to 200°C, and kept isothermal for 5 min to erase the thermal history, then cooled rapidly to room temperature and subsequently heated again to 200°C. Heating rate is 10 °C/min as well as cooling rate.
The melting temperature (Tm) of the samples was determined from the maxima of the fusion peaks. The crystallinity (Xc) of experiment materials was calculated by equation (6)
16

Mechanical properties
The tensile tests of samples were carried out by tensile tester (HS-100KN, Yangzhou Huahui Testing Instrument Co. Ltd. of China) with a crosshead speed of 20 mm/min. The results were the average values of at least five specimens.
Results and discussion
LOI and UL94 analysis
Flame retardancy of the experimental materials.
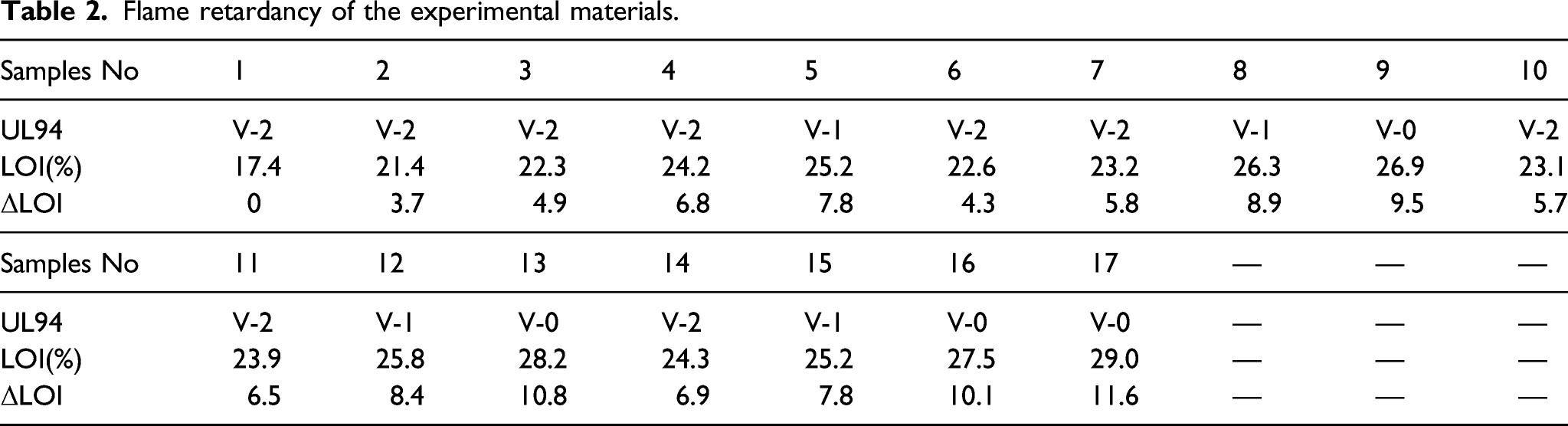
Synergistic flame retardant efficiency between MMT and Br-Sb.

The synergistic flame retardancy effect of Br-Sb system is attributed to the chemical reaction of Sb2O3 and gas phase HBr, which can product SbBr3 and water. The reaction is as follows

Thermal gravimetric analysis
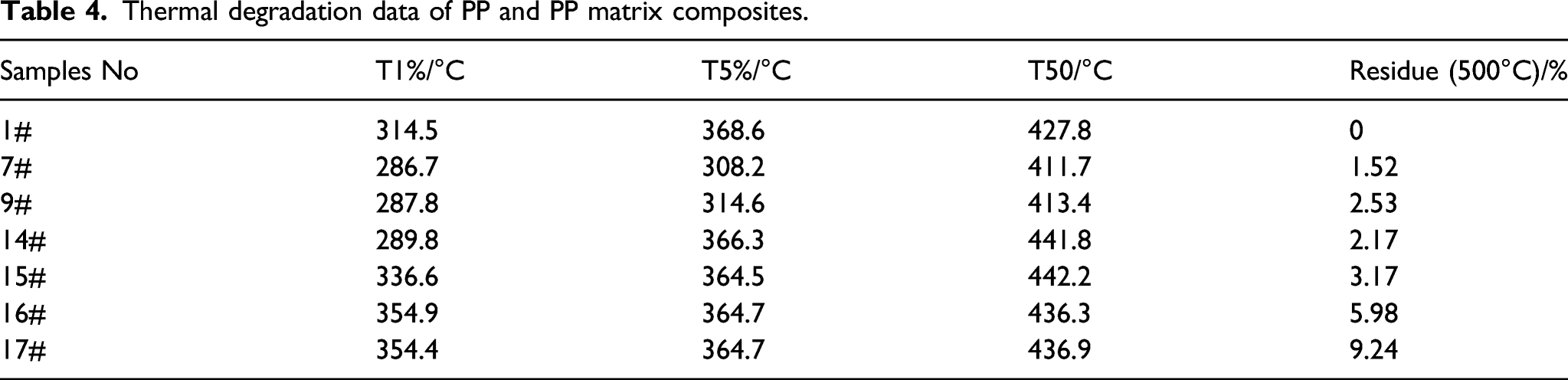
The effects of nano-Sb2O3 and MMT on the thermal stability of the experimental materials were studied by TGA under nitrogen atmosphere. Figure 1 shows the TGA curves of the experimental PP and PP matrix composites and Table 4 lists the characteristic parameters of theses TGA curves. In Table 4, T1%, T5% and T50% correspond to the temperatures when thermal mass loss is 1 wt%, 2 wt% and 3 wt%, respectively. The thermal degradation of PP takes place through one-step process corresponding to random C-C bond scission mechanism.
20
The decomposition temperature of the experimental nano-Sb2O3/BPS-PP composites is lower than that of pure PP due to advance decomposition of BPS. Adding MMT particles, the decomposition temperature of the experimental nano-Sb2O3/MMT/BPS-PP composites is increased, which is assigned to tight combining between PP and MMT and thereby limiting the mobility of polymer chains.
21
In addition, MMT can increase T50% of decomposition temperature of PP because MMT can promote the formation of a complete and solid char residue layer on the surface of PP matrix composites, which the amount of char residue of the experimental composites increases with increasing of content of MMT. In Table 4, the char residue of 3.5%nano-Sb2O3/3%MMT/8%BPS-PP composites (sample 17#) is 9.24 wt%, 2%nano-Sb2O3/8%BPS-PP composites (sample 7#) is 1.52 wt% and 1.5%nano-Sb2O3/3%MMT/8%BPS-PP composites (sample 14#) is 2.17 wt%. It shows that not only MMT can increase char residue, but also nano-Sb2O3 can improve thermal stability of PP. Thermal gravimetric analysis curves of PP and PP matrix composites. Thermal degradation data of PP and PP matrix composites.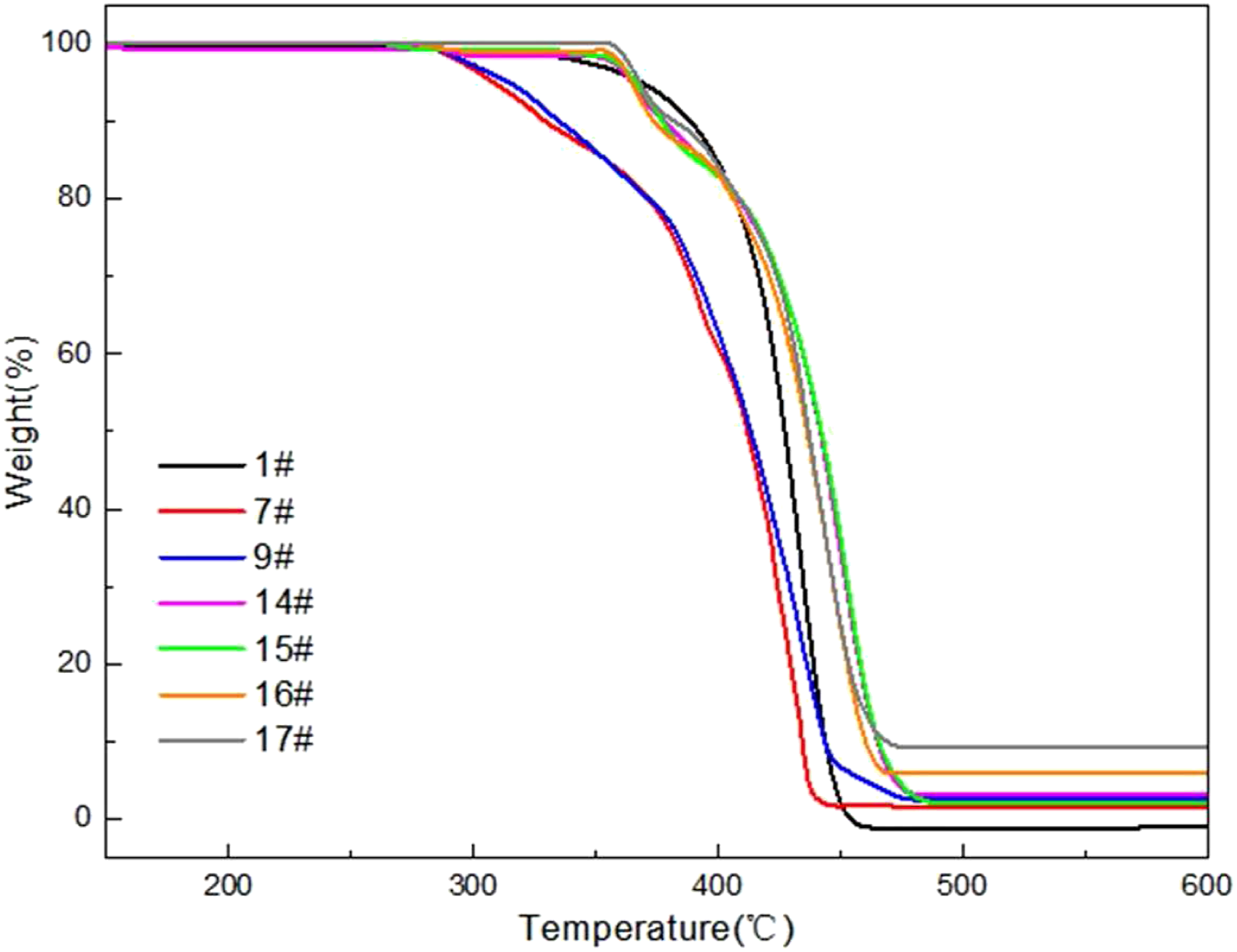
Morphology of char residues
The Scanning electron microscopy (SEM) images of the char residues of the experimental PP matrix composites under vertical burning are presented in Figure 2. Figures 2(a) and (b) show SEM images of the char residues of sample 7# and sample 9#, respectively. It can be seen from Figure 2(a) that there are lots of holes and voids on the surface of char residues, which means that the volatilization rate of the surface of PP matrix in combustion is very fast. What’s more, the surface of the char residues of sample 7# easily damaged due to their weak loose structure, which could not effectively protect the PP matrix against fire. Figure 2(b) shows that the surface of the char residues of sample 9# is continuous and compact. When the content of nano-Sb2O3 is higher, nano-Sb2O3 can react with BPS to form a more complete and firm char layer. Nano-Sb2O3 can promote the char layer formation and the layer can insulate the heat transfer, thus improving the flame retardancy of the experimental composites and reducing the heat release rate of the matrix. Scanning electron microscopy images of char layers after vertical burning (a)7#, (b)9#, (c)15#, (d)17#, (e)15#.
Scanning electron microscopy images of the char residues of sample 15# and sample 17# are presented in Figures 2(c) and (d), respectively. Figure 2(e) is a magnified image of C1 region of sample 15#. MMT can easily form a peculiar structure of char layer during burning, namely, cortex-honeycomb structure of the layer. 22 The upper layer of the char layer is a relative dense cortex. And the lower layer is a honeycomb layer, which is char residue structure with more gaps accumulated by some aggregates of large size of layered char residues. It can be seen from Figures 2 (c) and (d) that the lamellar aggregates of MMT in the cortex presents a three-dimensional network structure with higher mechanical strength, which can protect the inner materials and avoid the damage of char residue under thermal action. At the same time, the cortex has better heat insulation and gas-resisting, and then effectively relieves the decomposition of inner material. In Figure 2(e), the wall of the honeycomb layer is formed by the aggregation of MMT, resulting in forming a honeycomb char layer as a whole. It can improve the thickness of char layer and then effectively play the role of flame retardant layer. Therefore, MMT can improve flame retardancy of nano-Sb2O3/MMT/BPS-PP composites by means of char residue layer.
Melting and crystallization behaviour
X-ray diffraction analysis
Figure 3 shows the XRD patterns of pure PP, MMT and PP matrix composites. The grain size of nano-Sb2O3 was calculated by Scherrer’s formula, expressed as equation (5). The grain size of nano-Sb2O3 on (222) crystal face is about 46.7 nm and that of MMT on (001) crystal face is about 3.7 nm. The interplanar distance of MMT is about 2.1 nm at 4.22° of 2θ calculated by Bragg’s equation. For 3%nano-Sb2O3/3%MMT/8%BPS-PP composites, there is not a diffraction peak of MMT in range of 2θ from 2° to 35°, which indicates that the intercalation structure of MMT exfoliates and inserts into PP matrix. It also shows that the MMT presents exfoliated structure in nano-Sb2O3/MMT/BPS-PP system composites prepared by mechanical mixing. In the XRD patterns of nano-Sb2O3/BPS-PP composites, it is clearly seen that the pattern still has both of diffraction peaks of pure PP and Sb2O3 nanoparticles. It indicates that the crystal structure of PP and Sb2O3 nanoparticles do not change after being prepared into the experimental composites. X-ray diffraction patterns of PP, MMT and PP matrix composites.
Differential scanning calorimetry analysis
Figure 4 shows the Differential scanning calorimetry (DSC) crystallization and melting curves of pure PP and the experimental PP matrix composites and Table 5 lists the corresponding DSC characteristic data. It can be seen that the Tm,Tc and Xc of PP matrix composites are higher than those of pure PP. It shows that the nano-Sb2O3 particles with high dispersibility act as nucleating agents. However, the dispersed inorganic particles with high concentration will prevent to form large crystalline region due to a large number of nucleation points and result in limiting the movement of polymer chains.
23
For the sample 8# and sample 9#, the crystallinity of PP matrix of the composites decreases sharply with increasing of the content of nano-Sb2O3. When the content of nano-Sb2O3 is higher, nano-Sb2O3 particles can easily agglomerate, which can reduce the number of nucleation points. So, the crystallinity increases firstly and then decreases with increasing of the content of nano-Sb2O3. For nano-Sb2O3/MMT/BPS-PP system composites, MMT with 3 wt% of content can increase sharply the crystallinity of PP matrix of the composites because MMT causes an increase of its nucleation substrate. Differential scanning calorimetry of pure PP and PP matrix composites (a) Crystallization curve, (b) Melting curves. DSC data of PP and PP matrix composites. DSC: Differential scanning calorimetry.

Tensile strength
Tensile strength of PP and PP matrix composites.

Flame retardant mechanism
Figure 5 shows the flame retardant mechanism of the experimental flame retardant PP matrix composites. The Br-Sb system and MMT can play roles of flame retardant effects. For Br-Sb system, BPS plays two roles in flame retardancy of the experimental composites. Firstly, BPS can quench the active free-radicals. In the early stage of combustion, PP can decompose via β-scission and generate alkyl free-radicals under heat and oxygen.
25
Simultaneously, BPS can generate bromine radicals through thermal degradation and oxidizing reaction. The bromine radicals can react with H• and OH• free radicals, which can block the chain scission reaction and then prevent the heat feeding back to PP matrix.
26
Meanwhile, the incombustible gases (H2O) can dilute the combustible gas and reduce the flame heat. Secondly, BPS and nano-Sb2O3 show good synergistic flame retardant effect in PP matrix composites during combustion. Nano-Sb2O3 reacts with the HBr generated by the degradation of BPS to form SbBr3 and H2O. Combining with the dilution effect of SbBr3, the bromine radicals can effectively suppress the flame and even extinguish it. For MMT, it plays a role of condensed phase flame retardant in the experimental composites. MMT can improve the density of the char residues on the combustion surface, which can increase the isolation effect of the char layer and prevent the transmission of combustible gas, oxygen and heat. The formation of char layer makes the protective effect more effective and enhances the flame retardant effect of the condensed phase. Flame-retardant mechanism of PP matrix composites.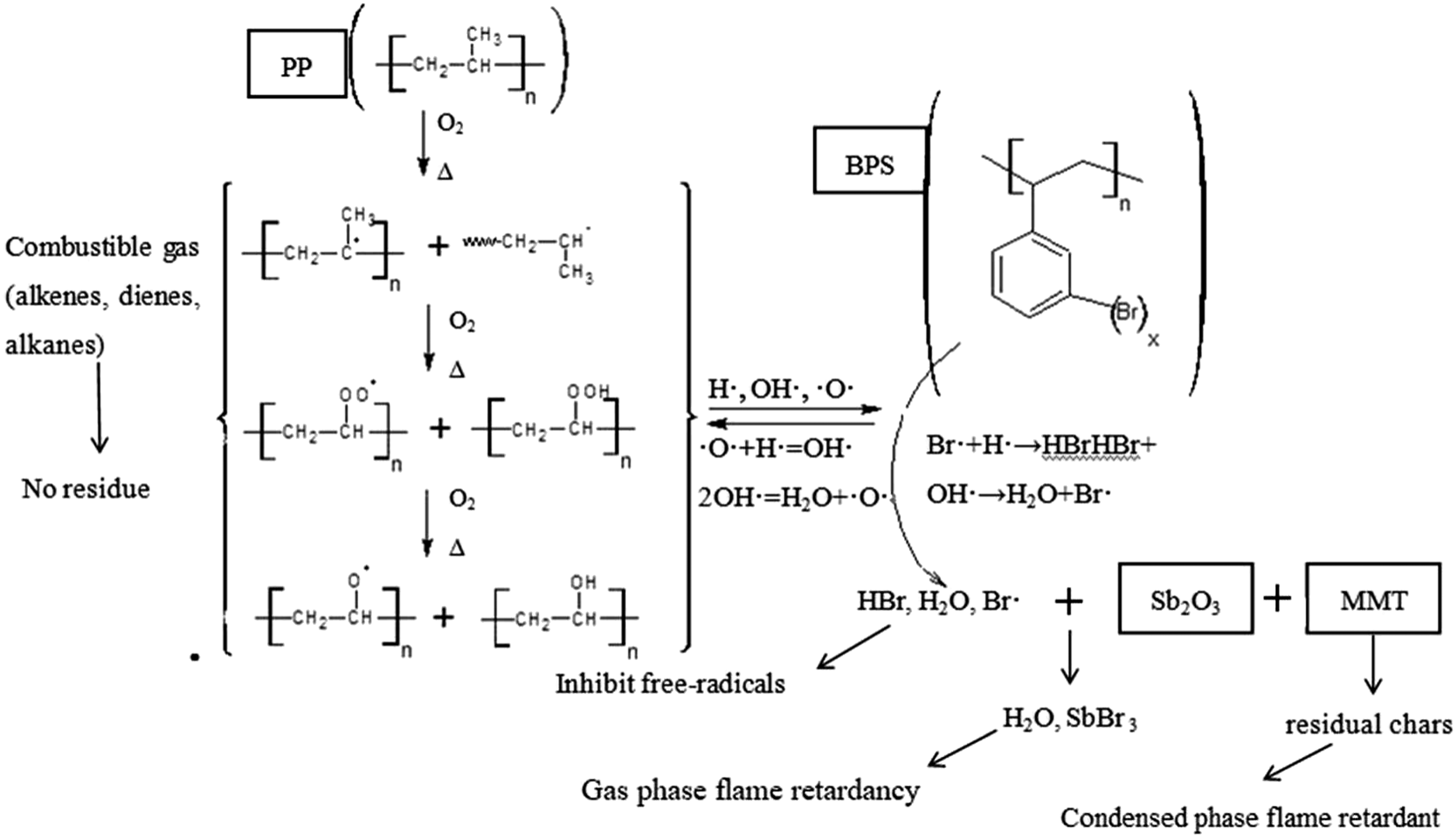
Conclusion
(1) Sb2O3 nanoparticles (nano-Sb2O3) and BPS have good synergistic flame retardant effect, which the maximum of SE is 1.82. Not only nano-Sb2O3 and BPS have gas phase flame retardant effect via reaction, but also the reaction product SbBr3 has condensed phase flame retardant effect. (2) Incorporation of MMT, nano-Sb2O3 and BPS within PP matrix has good flame retardant effect, which the maximum of flame retardant efficiency EFF of experimental materials achieves 2.11. Montmorillonite can not only improve the thermal stability of PP matrix composites, but also increase the density of char layer on the burning surface of the composites, so as to improve the flame retardant effect of condensed phase. (3) Nano-Sb2O3 and MMT can improve the crystallinity of PP matrix composites due to their heterogeneous nucleation. And they can improve tensile strength of PP matrix composites to some extent because of dispersion strengthening effect of nano-Sb2O3 and intercalation structure formed by MMT and PP matrix. (4) The 3.5%nano-Sb2O3/3%MMT/8%BPS-PP composites has excellent flame retardant properties and tensile strength, which its LOI is 29% and its UL94 grade reaches V-0 level and its tensile strength is 39.15 Mpa.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 51761025).
