Abstract
To improve the flame retardancy of polybutylene terephthalate (PBT), PBT-based flame retardant composites containing antimony trioxide nanoparticles (nano-Sb2O3) and brominated polystyrene (BPS) were investigated. Nano-Sb2O3, BPS, and PBT were dispersed by ball milling method to obtain composite powders, and the nano-Sb2O3/BPS-PBT samples were prepared by melt blending and injection molding methods. The flame retardancy of nano-Sb2O3/BPS-PBT composites was investigated. The results showed that nano-Sbs2O3 can obviously improve the flame retardancy of PBT-based composites. When the nano-Sb2O3/BPS-PBT composite contains nano-Sb2O3 with 5 wt% of mass fraction and BPS with 10 wt% of mass fraction, the nano-Sb2O3/BPS-PBT composite has excellent flame retardancy, in which the UL94 degree of flame retardancy achieves V-0 grade and the limit oxygen index is 28.3%.
Introduction
Polybutylene terephthalate (PBT) is widely used in machinery manufacturing, automotive industry, electronics, and other industries because it has many advantages, such as corrosion resistance, good mechanical properties, low water absorption, easy processing, and so on. Nevertheless, its flammability, lower heat distortion temperature, and serious dripping during combustion limit its wide applications. 1 –3 Thus, improving the flame retardant of PBT plays an important role in expanding its applications field and protecting human life and property security.
In general, incorporating flame retardant additives is an effective method to enhance the fire safety performance of PBT. 4 There are two primary types of flame retardants, namely halogen flame retardants and halogen-free flame retardants. Halogen-free flame retardant is an environmental friendly additive. However, the property of the PBT matrix will be deteriorated when the amount of the flame retardant is too high. Although the combustion products of halogen flame retardant have some effect on the environment, halogen flame retardant is still the main flame retardant additive in PBT industry at present because it has many advantages of less addition, high flame retardancy, and less influences on the comprehensive performance of PBT matrix. 5 Improving the flame retardant of PBT and reducing the amount of the flame retardant additive become important problems for scientists and technicians to solve urgently. Jia et al. added a compound flame retardant containing bromine, phosphorus, and nitrogen into PBT to obtain a kind of PBT-based composites. The UL94 flammability test passed the V-0 grading. But the mechanical property of the composites is lower due to excessive amount of the compound flame retardant. 6 Zuo et al. found that the PBT-based composites containing 30 wt% ammonium polyphosphate (APP) had good flame retardant effect, but the APP with excessive content damaged the mechanical properties of PBT. 7 Gallo et al. found that the PBT-based composites containing TiO2 nanoparticles or Al2O3 nanoparticles, respectively, had lower flame retardancy, but the PBT-based composites containing the combination of metal oxides nanoparticles and aluminum diethlyphosphinate have good flame retardancy. 2 The research of Xu found that the UL94 flammability test of PBT matrix is increased from HB to V-0 grade by adding Sb2O3 with different particle size and brominated epoxy resin. The smaller the particles size of Sb2O3 particles, the better the flame retardancy of PBT-based composites. 8
The halogen-antimony flame retardant system plays an important role in improving the flame retardancy of the PBT matrix. Moreover, due to the special effect of nanoparticles, nano-Sb2O3 can reduce the usage of flame retardant based on improving the flame retardant property of PBT-based composites. 9 In this article, using nano-Sb2O3 as a flame retardant synergist and brominated polystyrene (BPS) as a flame retardant, nano-Sb2O3/BPS-PBT samples were prepared by melt blending and injection molding method, and the flame retardancy of the composites was investigated.
Experimental
Materials
The main materials of the experiment are as follows: PBT with the density of 1.31 g cm−3 was supplied by Jiangsu Chang Chun Chemical Co., Ltd, China. BPS containing bromine with more than 68.7 wt% was purchased from Qingdao Youdian Chemical Co., Ltd, China. Sb2O3 with 38 µm of average particle size was a product of Sinopharm Chemical Reagent Co., Ltd, China
Sample preparation
The Sb2O3 particles with 38 µm of average particle size were grinded by planetary ball mill (QM-3SP04, Nanjing Laibu Science and Technology Industry Co., Ltd, China) for 24 h of milling time under 400 r min−1 of milling speed to obtain nanometer Sb2O3 particles. Then, the nanometer Sb2O3 particles were modified by planetary ball mill using 0.3 g γ-(2,3-epoxypropoxy) propytrimethoxysilane (KH560) and 0.3 g hexadecyl trimethyl ammonium bromide for 6 h of milling time under 400 r min−1 of milling speed to obtain experimental nano-Sb2O3. 10
Table 1 shows the mass fraction composition of the experiment materials. The nano-Sb2O3, BPS, and PBT were blended by the ball mill for 6 h of milling time under 400 r min−1 of milling speed to obtain the experimental composite powder according to the composition ratio of Table 1. The ball milling adopts intermittent ball milling method, namely 6 min of milling and 3 min of stopping in a cycle period. The composite powder was extruded by twin-screw extruders (SJZS-10A, Wuhan Ruiming Machinery Manufacturing Co., Ltd, China) and injection molding machine (SZ-15, Wuhan Ruiming Machinery Manufacturing Co., Ltd) to prepare testing samples. The size of rectangle flame retardancy testing sample is 80 × 10 × 4 mm3 and the size of plate-shape mechanical properties testing sample is 75 × 5 × 2 mm3.
The mass fraction composition of the experimental materials.

BPS: brominated polystyrene; PBT: polybutylene terephthalate.
Characterization
Fire tests and mechanical test
The flame retardancy of testing samples was measured by limiting oxygen index (LOI) according to GB/T 2406.2-2009 and vertical burning test (flame retardancy grade of UL94) according to UL94-2009. The LOI measurements were performed for six times by a limited oxygen index tester (PX-01-005, Suzhou Phinix Quality Inspection Instrument Co., Ltd, China). The higher the LOI, the better is the flame retardancy. The flame retardancy grade of UL94 was performed for five times by vertical burning test instrument (SDT-94, Shanghai Qinsun Precision Electromechanical Technology Co., Ltd, China).
The tensile strength of sample was performed for five times by universal mechanical tensile testing machine (HS-100KN, Yangzhou Huahui Inspection Instrument Co., Ltd, China) at 20 mm min−1 of tensile rate according to the GB/T1040.1-2006 and took the average of the results.
Scanning electron microscopy
The testing samples (4 ± 2 g) were put into ceramic crucible with 20 mm of diameter, and then, the samples were heated from room temperature to 500°C at a heating rate of 10°C min−1 and kept for 5 min in the muffle furnace (SX-5-12, Beijing Kewei-Yongxing Instrument Co., Ltd, China). The carbon residue on the surface of the combustion product was selected, and the surface morphology of the residue was observed by scanning electron microscope (SEM, JSM-6700F, JEOL Co., Ltd, Japan). All samples were sputter-coated with gold prior to observation.
Fourier transform infrared spectroscopy
The sample mixed with KBr powder was performed by using Fourier transform infrared spectrometer (FTIR-650, Tianjin Gangdong Science & Technology Co., Ltd, China) with the scanning range from 400 cm−1 to 4000 cm−1.
Thermal gravimetric analysis
Thermal gravimetric analysis (TGA) was carried out using a thermal gravimetric analyzer instrument (TGA 4000, PerkinElmer Co., Ltd., America) at a linear heating rate of 10°C min−1 from room temperature to 750°C under nitrogen environment.
Results and discussion
Flame retardancy and tensile strength
Our research found that the use of nano-Sb2O3 alone has little effect on the flame retardancy of PBT. Adding BPS to PBT can improve the flame retardant efficiency of BPS-PBT composites to some degree with increasing of BPS content. When nano-Sb2O3 and BPS were used at the same time, the flame retardancy of PBT was significantly improved, indicating that there was a good synergistic flame retardant effect between nano-Sb2O3 and BPS. When the added BPS content is less than 10 wt%, the flame retardant efficiency of BPS-PBT composites and nano-Sb2O3/BPS-PBT composites is low, which cannot make PBT reach the flame retardant grade. When the content of added BPS is 10 wt%, the flame retardancy of PBT has been significantly improved with increasing of nano-Sb2O3 content. Table 2 shows the LOI values, UL94 degree, and tensile strength of the experimental materials. Pure PBT (sample 1#) exhibits an LOI value of 21.0% and no grade in the UL94 flammability test, which shows poorer flame retardant of pure PBT. In the UL94 testing of pure PBT, the residual flame remained for a long time, and it could still burn after producing the melting dripping. In addition, the melting dripping could ignite the absorbent cotton. When PBT contains BPS with 10 wt% of mass fraction (sample 2#), its LOI is increased to 22.9% and the UL94 degree is V-2 grade. In the UL94 testing of sample 2#, the testing sample will be extinguished when melting dripping drops onto skimmed cotton, which can ignite the skimmed cotton. The result indicates that the BPS can improve the flame retardancy of the PBT-based composites to a certain extent, but the effect of the promotion is not obvious. When the nano-Sb2O3 with 1 wt% of a mass fraction was added into the BPS-PBT matrix (sample 3#), the LOI of the nano-Sb2O3/BPS-PBT composites is significantly increased to 25.4% and the UL94 degree is V-1 grade. In the vertical combustion experiment, the sample extinguished rapidly at dropping of melting dripping, which could not ignite the skimmed cotton. This result indicates that nano-Sb2O3 has an excellent synergistic effect in improving the flame retardancy of PBT-based composites. With increasing of mass fraction of nano-Sb2O3, the flame retardant performance of nano-Sb2O3/BPS-PBT composites improves. When the mass fraction of nano-Sb2O3 is 5 wt%, the LOI value of nano-Sb2O3/BPS-PBT composites (sample 5#) reached 28.3% and the UL94 degree was V-0 grade, which achieved fire retardant grade.
Flame retardancy properties and tensile strength of experimental materials.

LOI: limiting oxygen index.
As shown in Table 2, the tensile strength of pure PBT (sample 1#) is 55.3 MPa. When PBT contains BPS with 10 wt% of mass fraction (sample 2#), the tensile strength of the PBT-based composites is 59.4 MPa, which is 7.4% higher than that of PBT. With increasing of mass fraction of nano-Sb2O3 in BPS-PBT matrix, the tensile strength of nano-Sb3O2/BPS-PBT composites increases firstly and then decreased. And when the mass fraction of nano-Sb2O3 is 3 wt% (sample 4#), the tensile strength of the nano-Sb2O3/BPS-PBT composites reached the maximum of 62.5 MPa, which is 13.0% higher than that of pure PBT. Due to the physical–chemical effect between modified nano-Sb3O2 and polymers, the interface bonding between nano-Sb3O2 and BPS-PBT matrix is better. Under external load, nano-Sb3O2 can absorb a large amount of interaction energy and enhance the ability of exterior force resistance of nano-Sb3O2/BPS-PBT composites, thereby improving the tensile strength of the composites. However, when the mass fraction of nano-Sb2O3 is excessive (e.g. sample 5# and sample 6#), the tensile strength of the composites is gradually reduced because of the agglomeration effect of nano-Sb3O2. 11
In summary, nano-Sb2O3 improves not only the flame retardancy of nano-Sb2O3/BPS-PBT composites but also the mechanical property within a certain range. When the mass fraction of nano-Sb2O3 is 5%, nano-Sb2O3/BPS-PBT composites has the best flame retardancy and better tensile strength.
Morphology of char residues
Figure 1 shows the SEM photographs of the charred layers of PBT and PBT-based composites. In Figure 1(a), the microstructures of the char residue of the pure PBT look like foamy with many holes, which cannot protect the internal material against flame. Incorporating of 10 wt% BPS, there are complete charred layers at the partial surface of the PBT-based composites, as shown in Figure 2(b). Complete charred layers can isolate the internal material from air and high temperature, which also can protect the internal material. It indicates that BPS can enhance char yield. Compared with PBT and BPS-PBT experimental materials, the charred layers are more smooth and compact with increasing of the mass fraction of nano-Sb2O3, 12 as shown in Figure 1(c) to (f). It can be explained that the addition of nano-Sb2O3 is beneficial to form compact residual layers due to its catalytic esterification cross-linking reaction between the part of organic molecule. 13 In addition, Br· and HBr from the BPS-splitting reaction would react with the chain link of polymer molecular to form a double bond. The reaction is as follows:


SEM micrographs of char layer of experimental materials: (a) sample 1#, (b) sample 2#, (c) sample 3#, (d) sample 4#, (e) sample 5#, and (f) sample 6#. SEM: scanning electron microscope.

FTIR spectra of experimental materials and their charred residue: (a) sample 1# charred residue, (b) sample 2# charred residue, (c) sample 5# composites, and (d) sample 5# charred residue. FTIR: Fourier transform infrared.
In the reaction, the double bond is easy to be cross-linking or aromatization and further forms a char layer. Meanwhile, HBr can react with hydrogen of aromatic ring to form bromobenzene compound, and then, the bromobenzene compound can further react with aromatic compounds to form polycyclic aromatic hydrocarbons. The polycyclic aromatic hydrocarbons are precursors of graphitic carbon. 14 The above results demonstrated that nano-Sb2O3 in combination with BPS had efficient synergism in forming compact char layer, and the effect of nano-Sb2O3 is more obvious. The compact char layer not only prevents the heat and combustible gas from flame but also suppresses the heat transfer into the burning materials, ultimately improving the flame retardancy of the PBT-based composites. 15
FTIR analysis
Figure 2 shows the FTIR spectra of experimental materials and their charred residue. The curve (a) in Figure 2 is the FTIR spectrum of the charred layers of pure PBT (sample 1#). There is a characteristic absorption peak at 1631 cm−1 corresponding to C=O bending vibration, which demonstrates that all groups of PBT are destroyed in the combustion process and the pure PBT fully burns. The curve (b) in Figure 2 is the FTIR spectrum of the charred layers of PBT-based composites containing BPS with 10 wt% of mass fraction (sample 2#). The peak at 2369 cm−1 is assigned to the stretching vibration absorption peak of C–H, which indicates that the combustion product of the material has some residual saturated hydrocarbon groups and the combustion of the material is incomplete. The curve (c) in Figure 2 is the FTIR spectrum of the nano-Sb2O3/BPS-PBT composites (sample 5#) containing nano-Sb3O2 with 5 wt% of mass fraction and BPS with 10 wt% of mass fraction. There are 10 characteristic absorption peaks. The peak at 741 cm−1 is assigned to the Sb–O vibration of the cross-linked structure. The peak at 1110 cm−1 is attributed to the stretching vibration of the C–O. The peaks at 1448, 1509, and 1577 cm−1 are assigned to characteristic absorption peaks of the benzene skeleton vibration. The peaks at 1401, 1631, and 1716 cm−1 are attributed to C–H in-plane bending vibration, C=O bending vibration of the ketones, and C=O bending vibration in aldehyde group of PBT, respectively. The peak at 2369 cm−1 is assigned to the characteristic absorption peak of C–H stretching vibration and the peak at 2948 cm−1 is attributed to asymmetric stretching vibration of −CH3 group in BPS. 16 The curve (c) shows that the composites consist of PBT, BPS, and Sb3O2, which are consistent with the material composition.
The FTIR spectrum of residual charred layers of the composites (sample 5#) after burning is shown in curve (d). There is a peak at 2369 cm−1 attributed to the stretching vibration of C–H. It demonstrates that residual charred layers still have some residual saturated hydrocarbon groups and the burning of the composites is still inadequate. Comparing curve (c) and curve (d), it can be seen that nano-Sb2O3 and BPS participated in the combustion and then impeded full combustion of PBT, which nano-Sb2O3 and BPS did not survive. Comparing curve (a), curve (b), and curve (d), it can be seen that the intensity of C–H absorption peak of curve (d) is higher than that of curve (b). The intensity enhancement of C–H absorption peak of curve (d) shows that the content of residual saturated hydrocarbon increases in combustion products of PBT-based composites. This is a result of incomplete combustion of the composites to retain more saturated hydrocarbon organics. The compared results not only reflect that nano-Sb2O3 has better flame retardancy and BPS has a certain flame retardancy but also show that nano-Sb2O3 synergistic with BPS can effectively improve the flame retardancy of nano-Sb2O3/BPS-PBT composites.
Thermal gravimetric analysis
Figure 3 shows the thermogravimetry and differential thermogravimetry curves of the experimental materials. Table 3 lists the corresponding thermal degradation data based on Figure 3. In Table 3,

TGA and DTG curves of experimental materials: (a) TGA curves and (b) DTG curves. TGA: thermogravimetric analysis; DTG: differential thermogravimetry.
The corresponding thermal degradation data of experimental materials.

In addition, it can be seen from Table 3 that the maximum thermal mass loss rate of the pure PBT was 2.52% min−1. When nano-Sb2O3 synergistic with BPS is added into PBT matrix, the maximum thermal mass loss rate of nano-Sb2O3/BPS-PBT composites reduced. Meanwhile, the maximum thermal mass loss rate of PBT-based composites decreased with increasing of the content of nano-Sb2O3. The reason is that nano-Sb2O3 and BPS captured high-energy free radicals, then converted high-energy free radicals into nonflammable water, and diluted flammability. When nano-Sb2O3 with 7 wt% of mass fraction was added into PBT matrix, the maximum weight loss rate of the composites is higher 34.65% than that of PBT-based composite containing 5 wt% nano-Sb2O3, which may be due to agglomerates of excessive nano-Sb2O3 in the matrix and worse synergistic flame retardant effect between nano-Sb2O3 and BPS. As can be seen from Table 3, the residual char rate of the nano-Sb2O3/BPS-PBT composites was higher than that of pure PBT, and the rate of the nano-Sb2O3/BPS-PBT composites increased with increasing of the content of nano-Sb2O3. It indicated that nano-Sb2O3 favored the formation of charred residue, consistent with the results of infrared analysis of charred residue. 18
Flame retardant mechanism
When the nano-Sb2O3/BPS-PBT composites encounter flame, BPS can release HBr under heating, and then HBr reacts with Sb2O3 to form SbBr3 and Sb
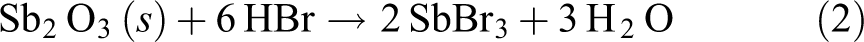

The resulting SbBr3 and Sb
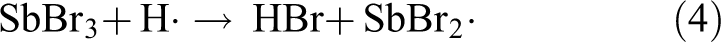

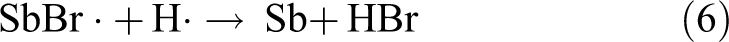





SbOBr can also decompose at high temperatures and release some bromine–antimony compounds slowly. The reaction formulas are as follows:



The resulting bromine–antimony compounds can again participate in the process of capturing high-energy free radicals, such as ·OH, H·, and so on. These compounds can maintain the effect of quenching flame for a relatively long time that it is equivalent to prolonging the lifetime of free radical collecting agents of SbBr3 and Sb
Figure 4 shows a schematic diagram of the flame retardant mechanism of nano-Sb2O3/BPS-PBT composites, which is mainly composed of physical and chemical flame retardant. The chemical flame retardant presents that the free radical collecting agents of SbBr3 and Sb

A schematic diagram of the flame retardant mechanism of nano-Sb2O3/BPS-PBT composites. BPS: brominated polystyrene; PBT: polybutylene terephthalate.
Conclusions
Adding modified Sb2O3 nanoparticles (nano-Sb2O3) and BPS into PBT, the synergistic effect of nano-Sb2O3 and BPS can effectively improve the flame retardancy of nano-Sb2O3/BPS-PBT composites. When the mass fraction of nano-Sb2O3 is 5 wt% and the mass fraction of BPS is 10 wt%, the LOI of nano-Sb2O3/BPS-PBT composites is 28.3% and UL94 degree of flame retardancy achieves V-0 grade, which the composites reaches the flame retardant level. Nano-Sb2O3 and BPS mainly play a flame retardant role in the gas phase, which can effectively reduce the heat release rate of PBT-based composites and slow down the combustion process. Within a certain range, the flame retardant efficiency of PBT-based composites increases with increasing of nano-Sb2O3 content. Nano-Sb2O3 and BPS have both physical and chemical flame retardancy in the process of flame retardancy. The chemical flame retardancy is mainly achieved by capturing ·OH, H·, and other free radicals by SbBr3 and HBr. The physical flame retardation is mainly the formation of compact charred layers in combustion process that it can isolate oxygen and heat to achieve the flame retardant effect. In addition, nano-Sb2O3 could improve the mechanical properties of the PBT material. Incorporating of nano-Sb2O3 with 5 wt% of mass fraction and BPS with 10 wt% of mass fraction, the tensile strength of nano-Sb2O3/BPS-PBT composites reaches 61.1 MPa and the composites has excellent flame-retardant properties and mechanical properties.
Footnotes
Authors’ note
The manuscript is an original article. It has not been previously published, is not currently submitted for review to any other journal, and will not be submitted elsewhere before a decision is made by this journal.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by the National Natural Science Foundation of China (no. 51761025) and the Science and Technology Program Project Funds of Gansu Province of China (no. 17CX2JD075).
