Abstract
This manuscript was built on studying the ignition, thermal, mechanical and electrical conductivity properties of the polypropylene by blending with different weight percentages of zinc borate (ZB) and potassium montmorillonite (MM). The tensile strength (TS) and elongation percentage (
Introduction
The polypropylene (PP) is a low-cost polymer 1 dissolved in aromatic solvents such as benzene and toluene at a temperature above 100°C. It is used in lots of fields, such as home textiles, fashions, furniture, electrical appliances and sportswear, but easy ignition combined with shrinking, and dripping due to the chemical structure of the PP. Therefore, it is important to safe PP by improving the fire retardancy and mechanical properties. 2
Zinc borate (ZB) can act as synergistic agents in ethylene-vinyl acetate copolymer/metal hydroxide-based formulations. It is analysed into boric acid and zinc oxide. 3 During the ignition process, ZB starts to degrade at 295°C without the release of toxic elements and forming a thermal insulator layer of char. 4
Montmorillonite (MM) is aluminosilicate layers sitting in clays. The MM dimensions are about 1 nm in thickness and 100–218 nm in length. 5 It is a cheap, a hydrophilic surface, and including nano-mineral which improves the thermal, chemical, physical and mechanical properties of many polymers due to its chemical structure. 6 The MM has adsorption/absorption capacities widely due to their high surface area, cation exchange and swelling capacity. 7 Recently, new composites were synthesized by blending different amounts of MM with polymer to increase mechanical, thermal and ignition properties.8–11 MM has a direct effect on decreasing the heat release rate, peak heat release rate and ash residue during cone calorimeter test.12–17
This study investigated the effect of ZB and MM when blended with PP with different weight percentages. The N3 specimens which synthesized by blending 7.5% of ZB and 2.5% of MM with 90% of PP have the best mechanical properties with the highest electrical resistivity. This may return to form ZnO, boric acid and SiO2 layers which reducing the heat capacity and heat release (oxygen bomb calorimeter (OBC)), with high ash residue, increasing in elongation and tensile strength (TS) values behind improving in the electrical conductivity. The ignition properties enhanced when compared with unblended specimens, since the limiting oxygen index % (LOI%) increased and the rate burning time increased with non-melting.
Material and methods
Materials
The pellets of PP with a density of 0.905 g cm−3 were supplied by Al Waha Petrochemical Company, Saudi Arabia. 18 Table 1 has the competence of PP.19–21 Potassium MM was purchased from Sigma-Aldrich, Germany. ZB was purchased from Sigma-Aldrich, Italy.
Specification of PP.

TS: tensile strength; PP: polypropylene.
Blended polymer preparation
The specimens of PP flame retardant were synthesized by melt blending appropriate amount of PP, ZB and MM on Newplast twin screw extruder (India) at zones temperature 195°C, 195°C and 195°C for zone 1, zone 2 and zone 3, respectively, then blend them together with twin screw speed of 30 r min−1 for 2 min. Standard mould with dimension 7.5 × 7.5 × 0.2 cm3 was used to produce sheets of new composites after heating at 200°C for 5 min, and holding pressure at 10 MPa. Finally, the sheet was obtained after keeping it cool to room temperature at the same pressure. Samples of pure PP were processed in the same way and used for comparison. 1
Fourier transform infrared spectroscopy
The specimen’s spectra were studied by Nicolet 380 Spectrometer (Thermo Scientific, USA) instrument with a wavelength range of 4000–400 cm−1. The Fourier transform infrared (FTIR) analysis was recorded with 4 cm−1 resolution and 32 scans. 22
Electrical conductivity (σ )
LCR Meter instrument (Agilent E4980A, USA) was used to measure the electrical conductivity and resistivity of the tested specimens. Conductivity can be defined as the ability of the material to transfer the electric current when free electron moving.
23
It is the reversal of the resistance (

The best insulator has zero m s−1, while the best conductor reaches to infinity m s−1. To calculate the electrical conductivity of the specimens, these equations were used:



where
Mechanical properties
Both of tensile and elongation % were measured for the specimens using Z010, Zwick, Germany with load cell of 10 kN (USA), according to standard test method. 24 Each specimen with dimension 5 × 15 cm2 was repeated three times to take the average.
Thermal analysis test
Thermogravimetric analysis
Degradation stages and ash residue of the specimen were determined via TGA-50 (Shimadzu, Japan). The test was performed by standard test method25,26 under N2 gas with a flow rate 30 ml min−1 and heating rate 10°C min−1. An open crucible of platinum was used to carry about approximately 7 mg of the tested specimens. The temperatures of the thermogravimetric analysis (TGA) test start from ambient temperature up to 750°C.27–29
Differential scanning calorimetry (DSC)
The enthalpy of unblended and blended specimens was performed using Shimadzu DSC-50 Instrument, Japan. The weight’s specimen was approximately 7 mg in the range of temperature between 25°C and 650°C under nitrogen gas with a flow rate of 30 ml min−1 and heating rate of 10°C min−1. Thermophysical properties such as glass transition (
Flammability estimation
Oxygen bomb calorimeter
The heat capacity/combustion of specimens (polymer, textile, liquid) was calculated according to a standard method; EN ISO 1716 31 using Parr 6200 instruments (USA). The instrument consists of a bomb, bucket, jacket, closed-circuit water and printer (Figure 1). The bomb consists of two valves, one used to fill the bomb with oxygen gas then closed automatically after 1 min and the other give a chance to the gases inside the bomb to release after finishing the test. There is a wire connected to the two electrodes which found at the top of the bomb together. A cotton wire was tied at the wire and the free edge found in the test specimen at the crucible. The specimen’s weight is nearly not more than 1.0 g and but inside the crucible. After closing the bomb, it is filled with oxygen gas then immersed in the bucket has 2 L of water.33,34
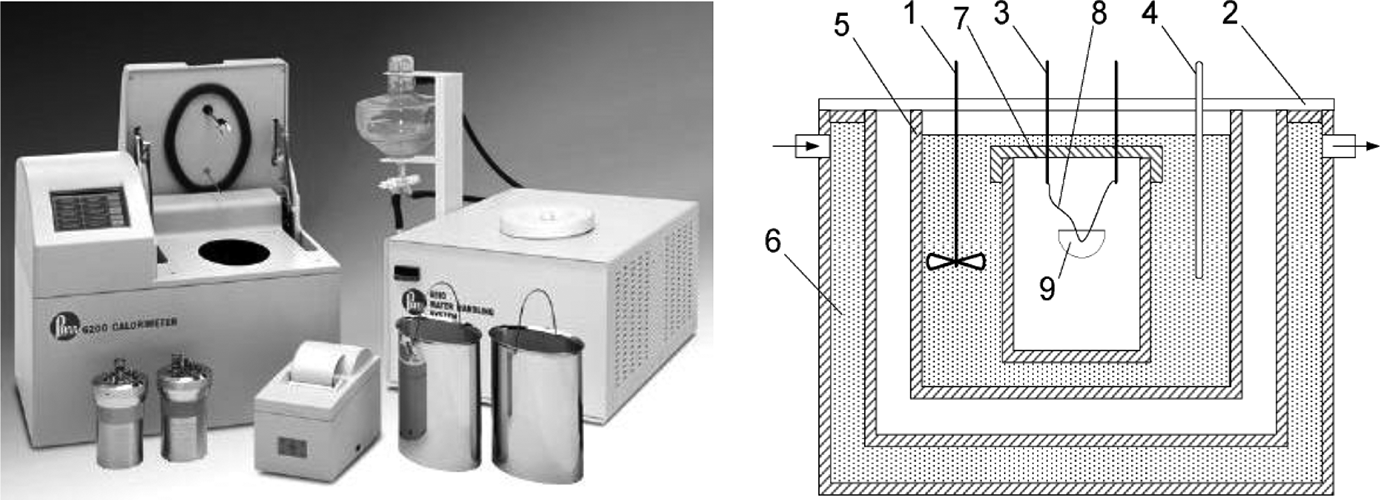
Calorimeter parts: 1 – mixer for uniform mixing of water, 2 – jacket cover, 3 – fuse cables, 4 – water thermometer, 5 – calorimeter vessel, 6 – water in the jacket, 7 – calorimetric bomb, 8 – fuse wire and 9 – crucible containing the sample. 32
Limiting oxygen index
The lowest percentage of oxygen required to ignite the specimen continuously in mixture of nitrogen and oxygen gases was measured on sheets with dimension 10 × 1 × 0.2 cm3 according to the standard test method EN ISO 4589. 2
UL-94 horizontal burning test
This test was used to classify the tested specimens according to the standard test method, 35 by measuring the length and ignition time of test specimens to calculate burn rate (BR). The instrument has a tendency to measure the BR of the specimen at horizontal and vertical position. In this test, the specimen was tested horizontally using U holder. A composite specimen with dimension length = 10 cm, width = 1 cm and thickness: 0.2 cm. A small flame of methane with length 3.8 cm was used to ignite the test specimen for 30 s. After this time, the flame removed and notified the distance and the time of burning to calculate the BR by equation (5):
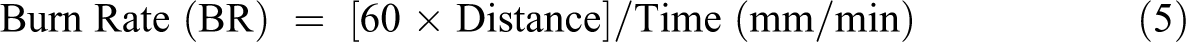
During the test, a piece of cotton placed under the tested specimen, if the specimen ignited with dripping this lead to ignite the cotton piece and has to be mentioned in the comments.
Results and discussion
FTIR analysis
The IR spectrum of MM and ZB was illustrated in Figure 2. At wavenumber 3622 cm−1, the peak of the hydroxyl linkage (–OH) group appeared. The presence of –OH groups was clarified by broad band at 3448 cm−1. The peak at 1638 cm−1 refers to –OH bending mode of adsorbed water. The band at 1050 cm−1 was assigned to stretching vibration for silicate layers (Si–O). The Si–O–Al stretching mode for MM was appeared at 798 cm−1. Finally, the sharp peaks at 523 and 467 cm−1 return to presence of Si–O–Al and Si–O–Si, respectively. 36
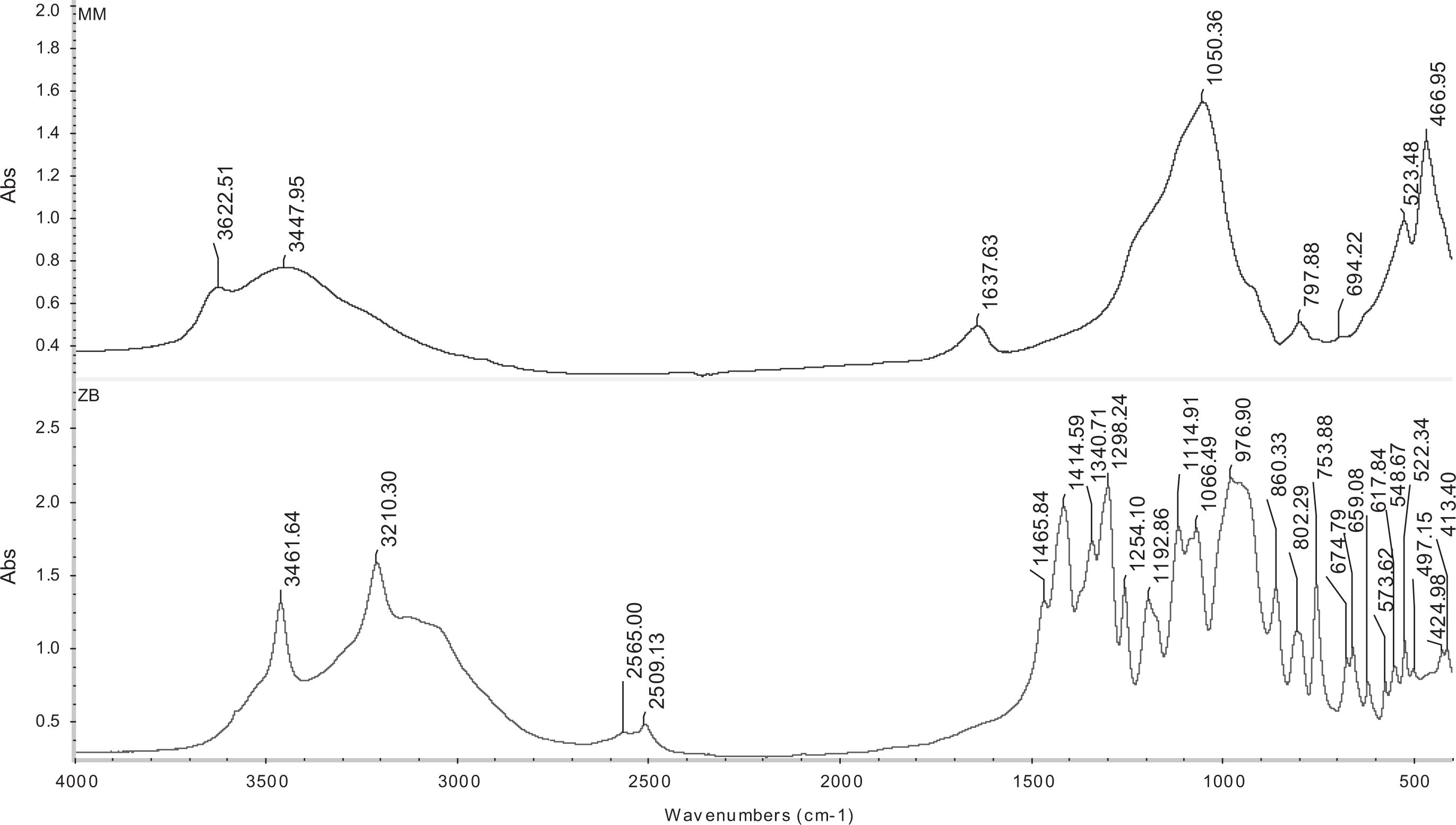
FTIR spectrums of MM and ZB.
In case of ZB, the stretching vibration of O–H was observed in 3461 cm−1, while asymmetric vibration of trihedral borate (BO3) groups appeared in 1340 and 675 cm−1. In 1066 and 976 cm−1, the asymmetric and symmetric stretching vibrations were observed due to the presence of tetrahedral borate (BO4) groups. 37
The IR spectra of the unblended and blended specimens were shown in Figure 3. Figure 3-N1 illustrates the IR spectra of the PP. The bending vibration of C–H was observed at 3210, 1458, and 1377 and 895 cm−1. The broad peak between 2950 and 2960 cm−1 reveres to symmetrical vibrations C–H. 38
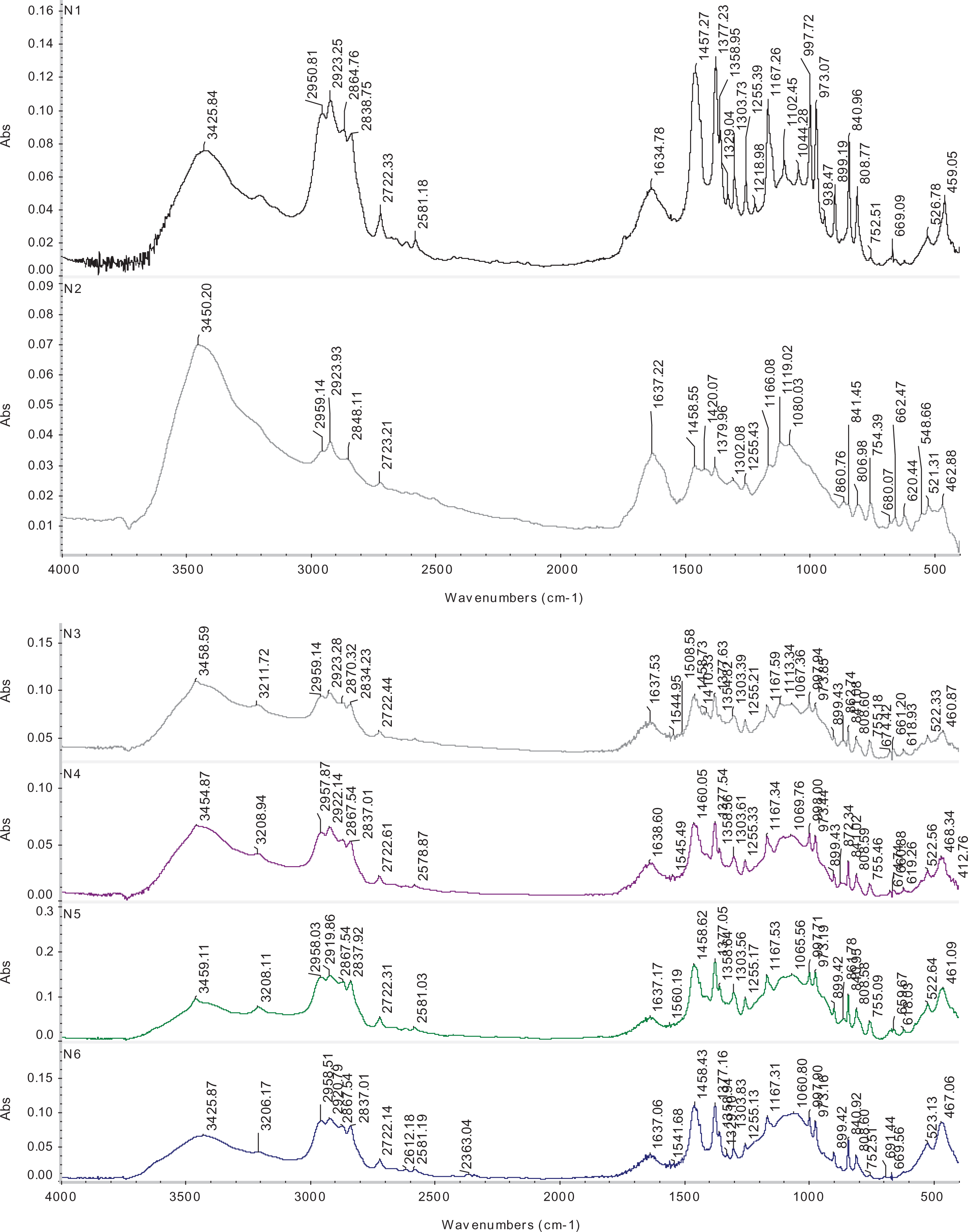
FTIR spectrums of unblended (N1) and blended specimen (N2–6). Whereas N1: PP (100% by weight), N2: PP (90%) blended with 10% of ZB, N3: PP (90%) blended with 7.5% of ZB and 2.5% of MM, N4: PP (90%) blended with 5.0% of ZB and 5.0% of MM, N5: PP (90%) blended with 2.5% of ZB and 7.5% of MM, N6: PP (90%) blended with 10% of MM.
In case of N2–5 specimens, the characteristic peaks of ZB (BO3 and BO4) were observed. The existence of the band in the range between 1425 and 1408 cm−1 is shown to the asymmetric stretching vibrations of trihedral (B–O) in (BO3), whereas the symmetric stretching band of the (B–O) in (BO3) appeared in the range between 1057 and 1085 cm−1. The presence of ZB can be fixed either by the presence of (B–O) in tetrahedral (BO4) as asymmetric stretching vibrations in the range between 1665–1170 cm−1 and the symmetric stretching band was observed between 860 and 857cm−1. Appearance of clay at composite can be proved by presence of stretching vibration of Si–O (between 1050 and 1060 cm−1), Al–O (between 522 and 525 cm−1) and Mg–O (between 460 and 467 cm−1). This result confirms that there was an interaction between the MM and ZB with PP. The previous groups appeared in the spectrums of the N3–6 different in intensity of the vibration bonds.
Electrical properties
The tabulated data in Table 2 illustrate that the electrical resistivity (
The electrical conductivity and resistivity of unblended (N1) and blended specimens (N2–6).
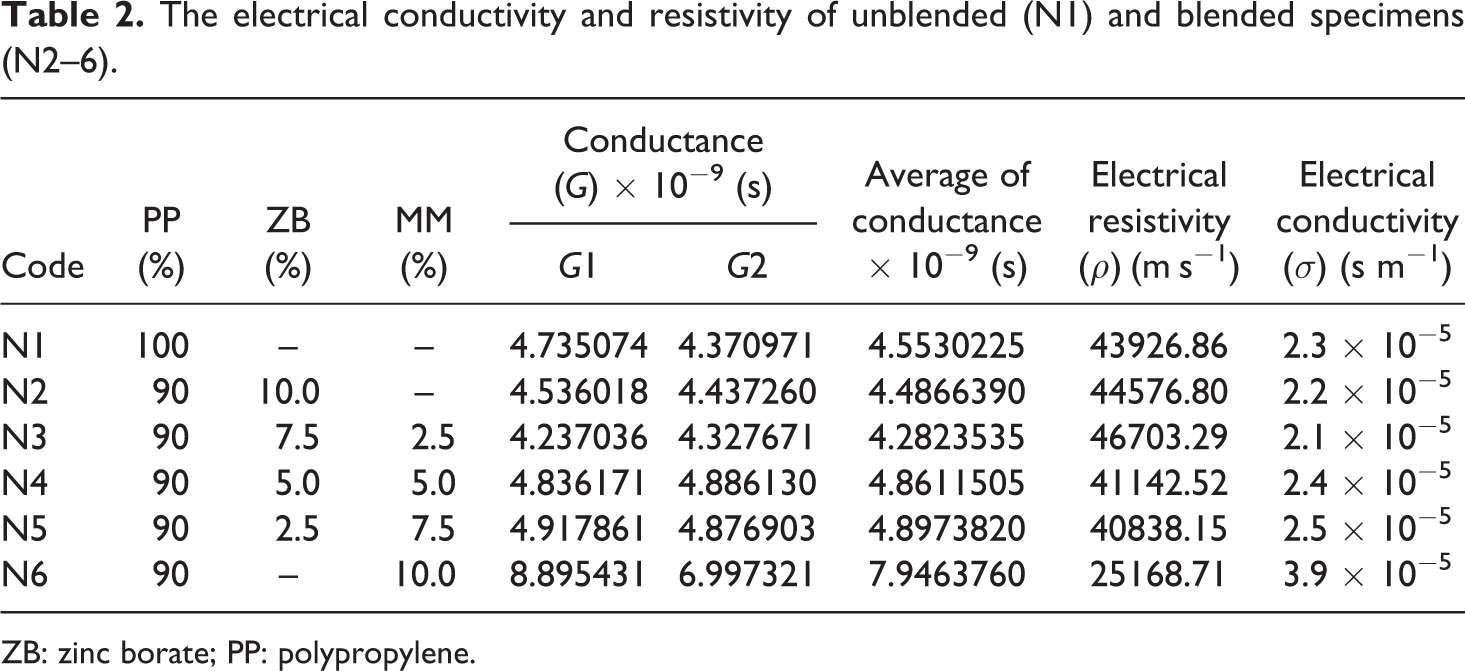
ZB: zinc borate; PP: polypropylene.
Tensile breaking strength and elongation
The mechanical properties of the unblended and blended specimens have been tabulated in Table 3. The TS and
Mechanical properties of the unblended and blended specimens.
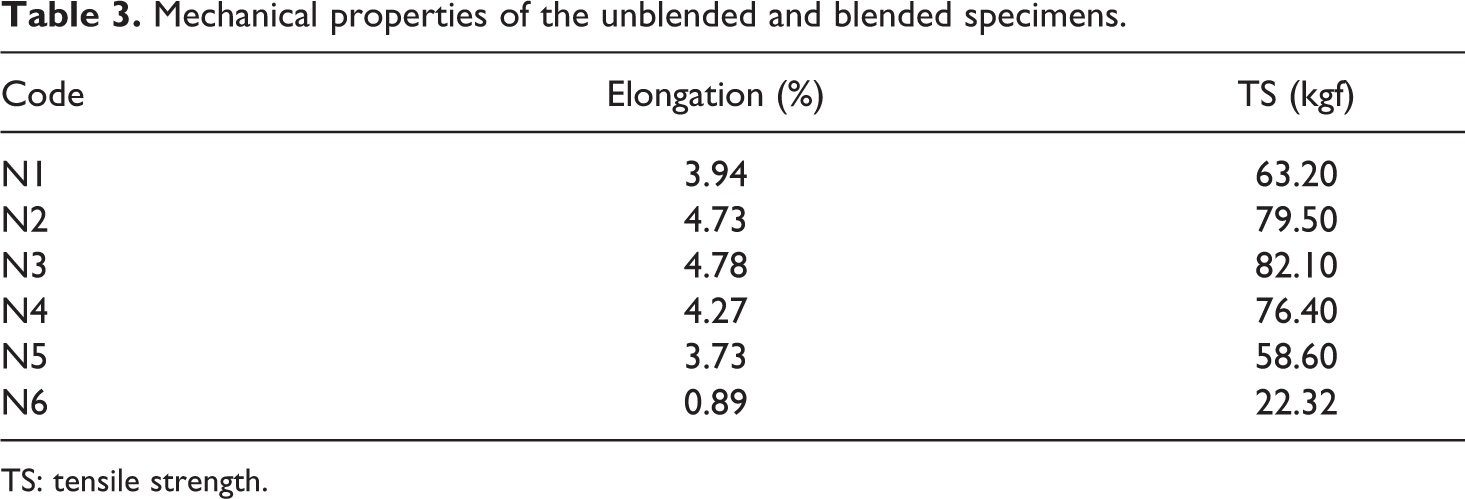
TS: tensile strength.
Thermal analysis results
The thermograms of TGA
The thermal properties of the unblended and blended specimen were studied by TGA instrument under nitrogen gas. Figure 4 illustrates the degradation curves for N1-6, ZB and MM, and the results are summarized in Table 4. The ZB specimen has two stages of degradation. Firstly, up to 109°C the dehydration occurred, then the zinc oxide and boric acid formed in the second stage between 206°C and 443°C with weight loss 10%. 39 It is clear from this figure that MM has three stages of degradation. Firstly, up to 100°C desorption of the water from the MM occurred. The dehydration of the hydrated cation (in the interlayer) achieved in the second stages (305°C) of the degradation. Finally, the dehydroxylation of the MM has been achieved between 495 and 750°C. 40
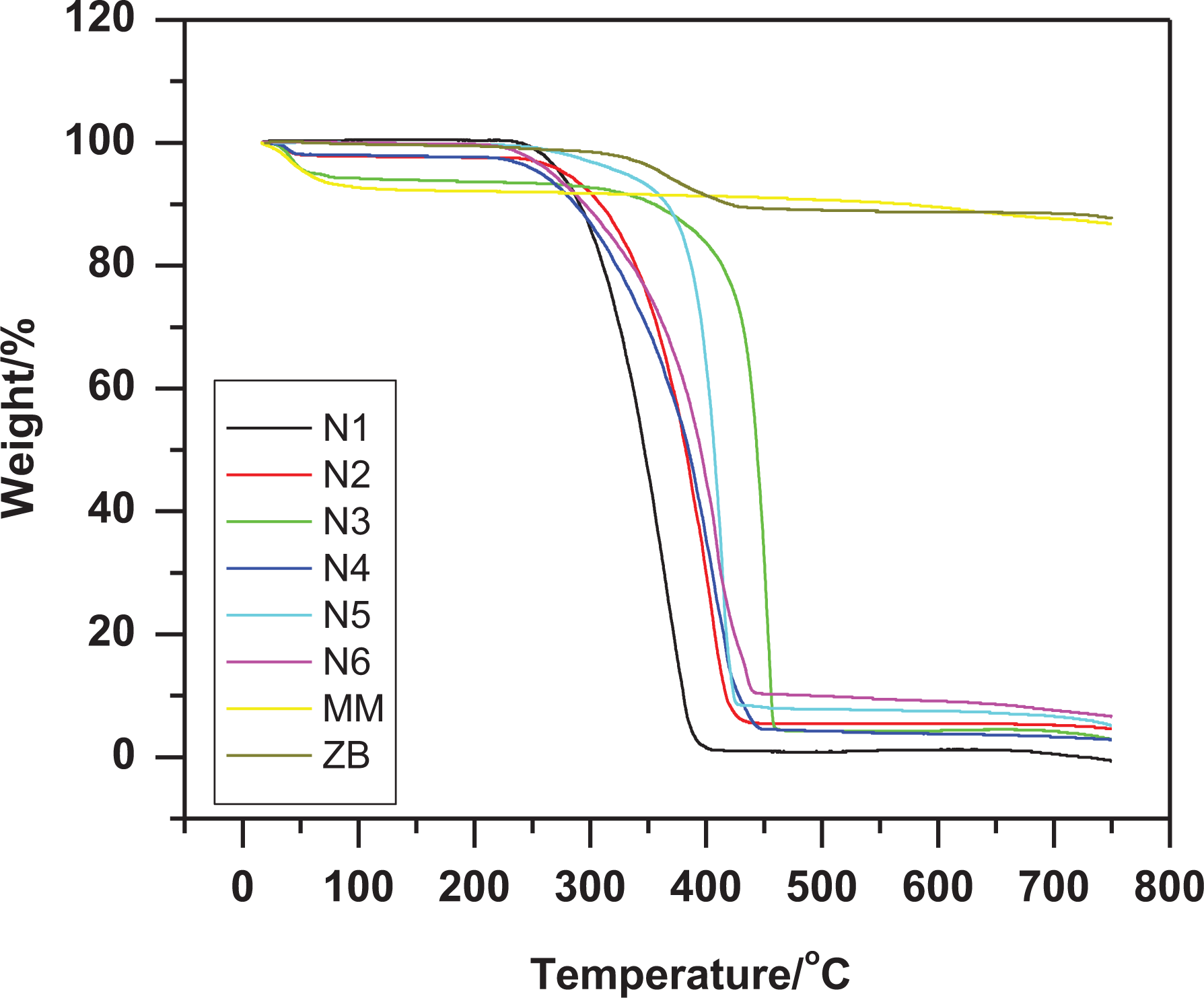
TGA of N1: PP (100% by weight), N2: PP (90%) blended with 10% of ZB, N3: PP (90%) blended with 7.5% of ZB and 2.5% of MM, N4: PP (90%) blended with 5.0% of ZB and 5.0% of MM, N5: PP (90%) blended with 2.5% of ZB and 7.5% of MM, N6: PP (90%) blended with 10% of MM.
TGA analysis for ZB, MM, unblended and blended specimens under nitrogen gas.
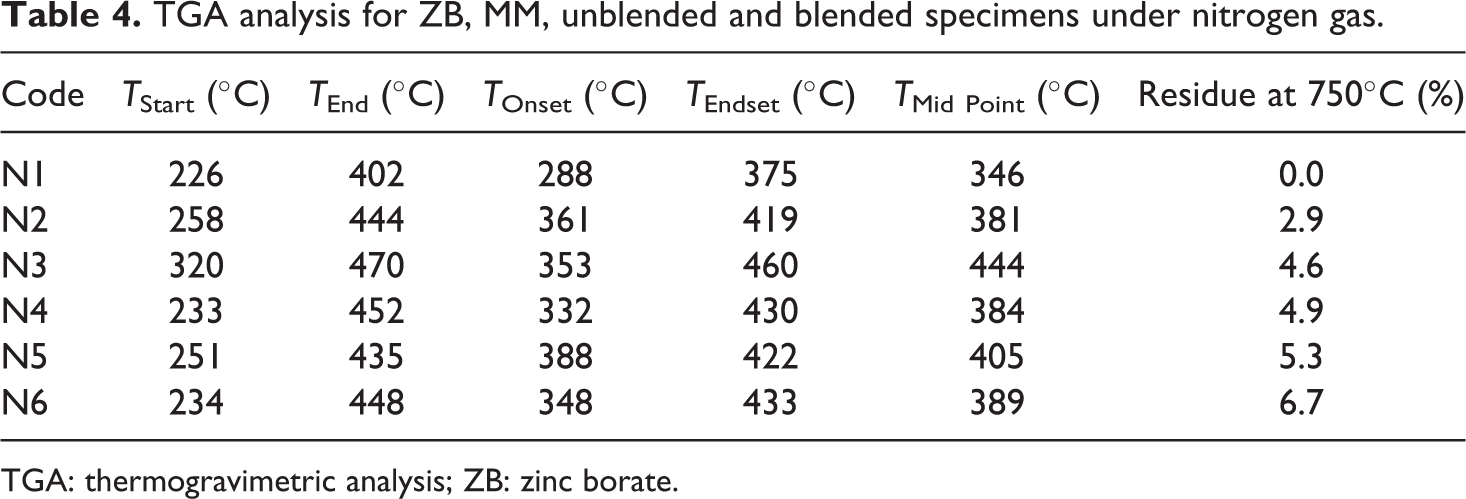
TGA: thermogravimetric analysis; ZB: zinc borate.
The N1 specimen has no char residue after one degradation stage between 226 and 402°C, since 99.9% of weight loss. At this stage, more gases have been emitted as carbon monoxide and carbon dioxide.
29
The tabulated data illustrate the main role of ZB and MM at improving the onset temperature, mid point temperature and the ash residue % at 750°C. The blend specimens N2–6 have one degradation stage either, but different in
The N3 specimen has a higher initial temperature (320°C) and the highest
DSC analysis
The glass transition (
UL-94 and LOI results of the unblended and blended specimens at ambient temperature.
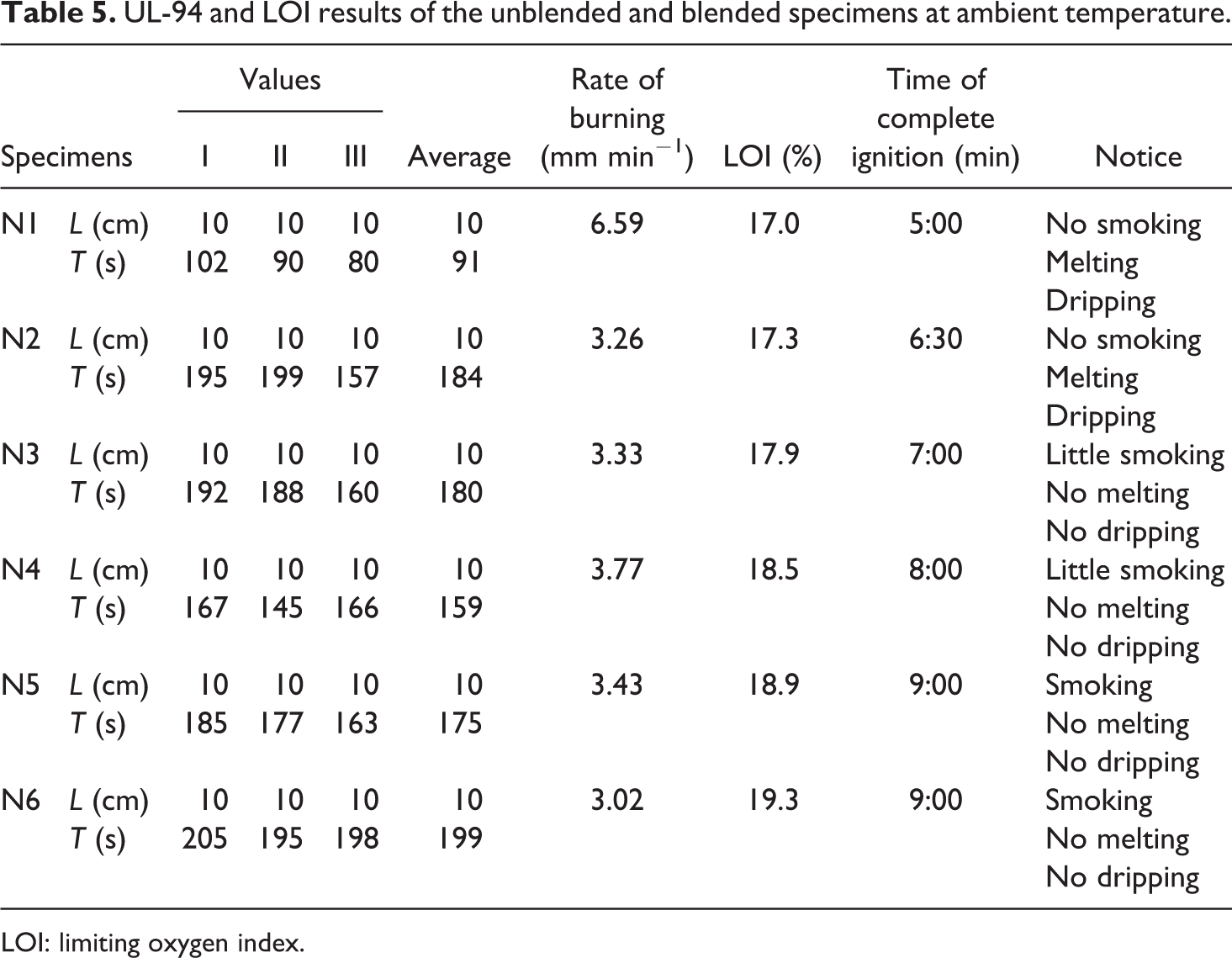
LOI: limiting oxygen index.
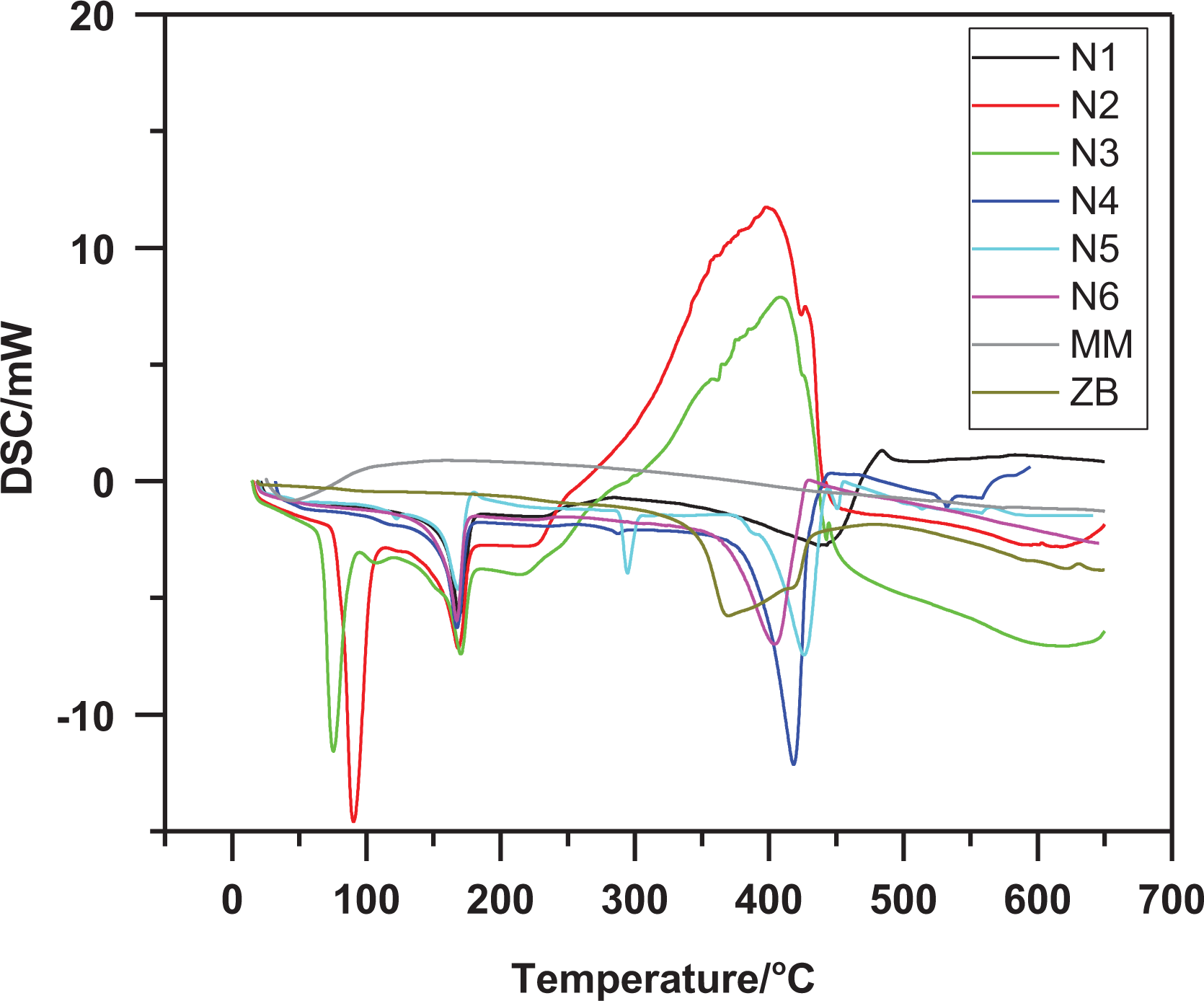
DSC plot of ZB, MM, unblended and blended specimens.
The N1 specimen has a melting temperature (
MM has a direct effect to change the exothermic peak to endothermic peak when blending with PP with different weight %. It is clear from the figure that N4 specimen has an endothermic peak at (417°C) which characteristic by the depth peak, then N5 (425°C) and N6 (402°C).
Combustion test
Calorimeter bomb
Figure 6 illustrates the heat combustion of the unblended and blended specimens. It shows that the PP has been burned completely recording heat combustion with value 47.01 MJ kg−1. Blending PP with 10% of ZB only N2 achieved heat combustion (45.56 MJ kg−1) higher than N3 (44.32 MJ kg−1) and N4 (43.77 MJ kg−1). By increasing the weight % of MM, it is noticed that the heat combustion decreased to record (42.02 MJ kg−1) in case of N6 which synthesized by blending PP with MM only. This proved that blending ZB with MM has a positive effect in enhancing the ignition properties of PP and go with the results of UL-94 and LOI.
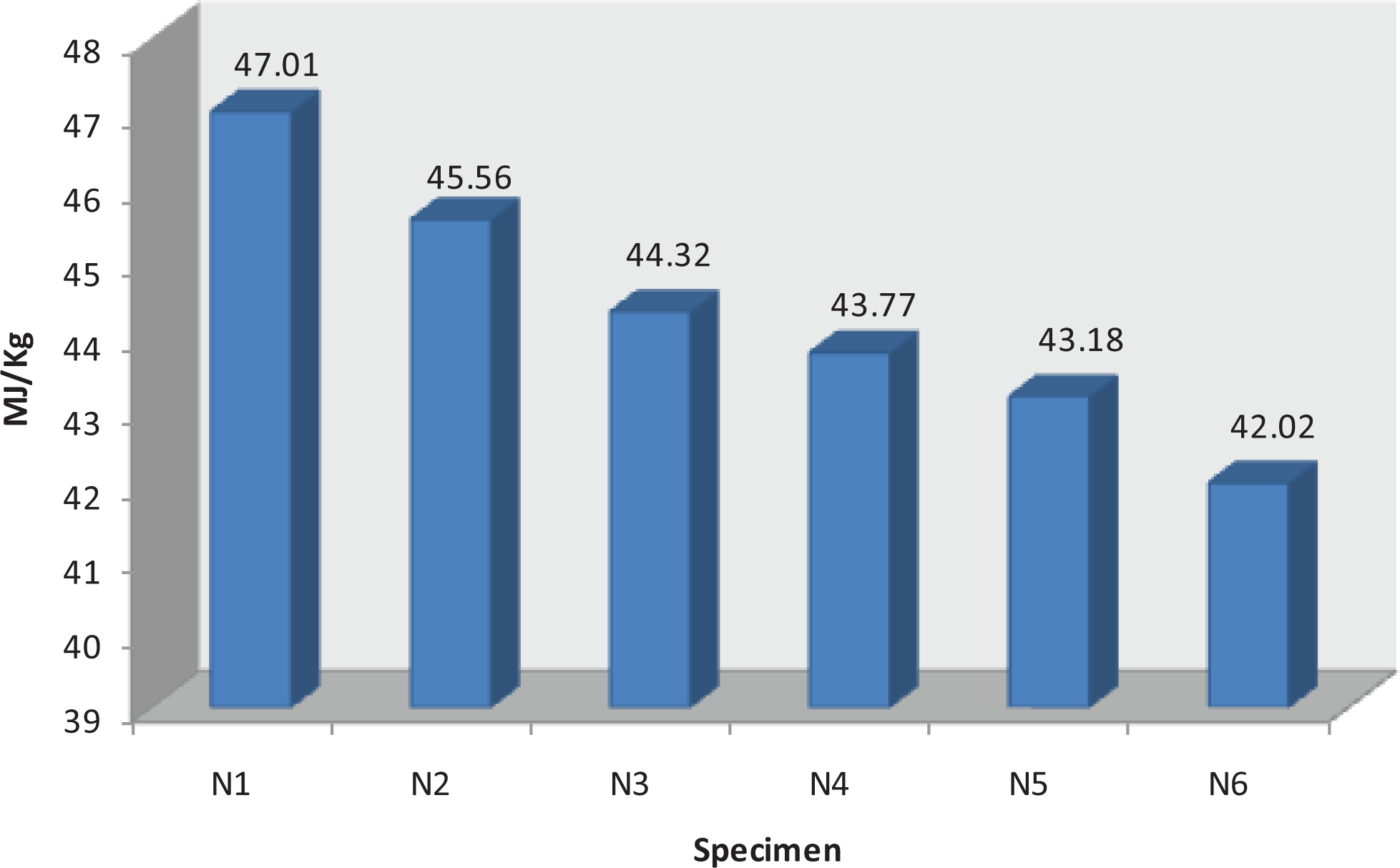
The heat combustion of unblended and blended specimens.
Oxygen index
The test was performed according to standard test method and the results were tabulated in Table 5. The blended specimens record improvement in LOI% compared to unblended one N1. Firstly, N1 specimens easy burning at LOI = 17% showing high flammability with dripping and no smoking. Adding ZB to PP (specimen-N2) increased the LOI = 17.3% with the same physical characteristics of the N1 specimens.
By blending a small weight % of the MM with PP and ZB (specimen-N3) leads to raise in the LOI value up to 17.9%, which leads to stop melting and dripping, but emitting a little smoke. When the weight percentage of the MM increases, the LOI% increase recording the highest value LOI = 19.3% in the case of (specimen-N6). The specimen was consumed 9 min to burn completely, which gives a chance for people to escape from the combustion place.
UL-94H test
Underwrites Laboratory (UL-94) is a test method used to measure the burning rate of the specimens in horizontal (H) burning test. The measuring test results were performed at ambient temperature and the results were tabulated in Table 5. The rate of burning decreased for all blended specimens by rate 48–50% compared to unblended, which burns very fast after ignition at LOI = 17.0%.
The physical properties changed in case of N3 specimen, return to presence of MM, since no melting and dripping noticed. The N6 specimen has the lowest value of burning rate return to blend PP with the highest percentage (10%) of MM. This revelled that presence of MM has the main role to increase the complete ignition time of the tested specimen by 50%. These results were supported by TGA and OBC either. 41
Conclusions
Combination of different flame retardant (ZB and MM) by dissimilar weight percentages with PP leads to improvement in the mechanical, flame retardant and electrical properties of PP. Blending 7.5% of ZB and 2.5% of MM with 90% of PP leads to enhanced mechanical properties. The MM has a direct effect in fighting the rising in the temperature by improving the LOI (19.3%), OB (42.02 MJ Kg−1), ash residue (6.7%) of PP after blending with MM. By increasing the weight % of MM lead to break the test specimen easily, decrease the electrical conductivity due to the hole between molecules, and stop dripping by aggregate over the test specimen. This aggregate is responsible for rising the % of the LOI and ash residue.
Footnotes
Acknowledgements
The author wishes to express his thanks to Prof. Dr Abdel-Rahman M Naser, Professor at Chemical Department, Al-Azhar University, Egypt, for supporting and helping him.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
