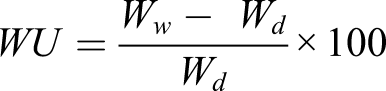Abstract
Valorization of environmental wastes into beneficial products is a global direction that is recently adopted. Potato peels are one of these wastes that are daily released to the environment in tremendous amounts. The current research is concerning by the extraction of starch polymer from potato peels waste and depending on its water uptake capacity to be used as a blending material in the currently applied diapers. Starch has been blended in different percentages with poly acrylamide (PAAM) and chitosan (CTS) polymers to increase their overall water capacity. The blended polymers were characterized by Fourier-transform infrared and Raman spectroscopy which confirmed the corresponding interaction among the three polymers depending on the detected functional groups. The thermal gravimetric analysis showed that the addition of starch to PAAM-CTS membrane increased the thermal stability and reduces the weight loss from 53.86% to 7.9%. The results of surface roughness were increased from 0.19 to 0.28 μm after the addition of starch which were highly matched with SEM images and suggesting the increasing of the swelling kinetics of the prepared membranes. Moreover, water uptake test as a physicochemical characterization revealed that the swelling ratio of PAAM-CTS membrane was around 48% which has been elevated to 92% after starch amendment. These results indicate that the addition of starch to polyacrylamide and chitosan blend can approximately duplicate their water-holding capacity which can repurpose it it to be widely applied in diapers applications.
Introduction
As a disadvantage of modern life, the amounts of disposables items are increasing every day. These disposables include shopping bags, towels, wipes, vessel washing scrub, tablecloths, diapers, and many more. 1 In addition to its economic disadvantage, it has also environmental disadvantage. Although, diapers are categorized as “unallowable expenses” by public food programs, 2 the production of diapers worldwide increases every year. In order to manage the diapers wastes, it is important to manage its main non-degradable component, the polymeric adsorbent materials.
Polyacrylamide, abbreviated as (PAAM), is a polymer (-CH2CHCONH2-) formed from acrylamide subunits. It can be synthesized as a simple linear-chain structure or cross-linked, typically using N,N'-methylenebisacrylamide. They are water-soluble synthetic linear polymers made of acrylamide or the combination of acrylamide and acrylic acid. Polyacrylamide finds applications in pulp and paper production, food processing, agriculture and mining, as a flocculant in wastewater treatment, and as super-adsorbent in agriculture 3 and diapers production. On the other hand, biodegradable polymers, such as starch and cellulose, gain much attention due to their environmental benefits.4,5
The exploitation of bioactive components from the agro-industrial wastes has gained much attention in recent years. These wastes are highly rich with essential bioactive compounds which can be used for many valuable purposes. Potato peel waste is one of these wastes. 6 It contains 25% of starch (St) in its raw form which is increased to 52% starch in its dry weight.7,8
Starch as a natural polymer has many advantages, including biodegradability, low cost and availability for large area production, 4 and promising water absorbing capacity. 9 The starch-based bio-polymers are the future promising materials which will be used in various applications, 10 such as food packaging industry, 11 water treatment, 12 and biomedical applications. 13 Herein, this research aims to prepare new combined water-absorbing polymers including starch waste in order to increase the water absorbing capacity in diapers in addition to improve the recycling process of potato peels wast. In addition, in this research, we provided a disposable diaper using chitosan, which has excellent anti-bacterial, deodorizing and aeration properties, prevents skin irritations, adsorbs harmful materials, and alleviates atopic dermatitis.
Experimental Section
Materials
Chitosan (CTS) with medium molecular weight (MW) was obtained from Acros Organics (Mw: 100,000–300,000 Da, and Deacetylation %: 65–75%), Belgium. Polyacrylamide (PAAM) was purchased from Fischer. Glacial acetic acid was obtained from Acros Organics, Belgium.
Starch Waste Collection and Extraction
The starch waste of the current study was extracted from the potato peels according to the following method. At first, the potato peels waste was collected from the domestic municipal solid waste at Borg El Arab city. The collected peels were extensively washed with tap water and left air dried for 24 h. The dried peels were cut into small pieces using sharp scissor. The pieces were packed to an electrical bender and filled with tap water with subsequent blending for 5 min. The formed mixture was filtered through stainless steel mesh in order to separate the liquid part into a new clean container. The container was then left to dry at 50oC for 48 h. The precipitated white powder was collected, grinded into smaller particles, and preserved in a dry container till use.
Preparation of (PAAM-CTS/St) Based Membranes
Poly acrylamide-chitosan (PAAM-CTS 1:1) solution (2% w/v) was prepared by dissolving in 2% acetic acid (solution A). Starch solution was prepared by dispersing varying proportions (4%) of starch in distilled water (solution B). Blended membrane-forming solutions were prepared by mixing their pre-prepared solutions (A and B) with stirring to obtain PAAM-CTS/St blends at different ratios of 1:0.5, 1:1, and 1:2. The prepared blend solutions were cast in plastic Petri dishes and the membranes were mounted between two cellophane sheets after drying at room temperature for 24 h (Scheme 1). Preparation of (PAAM-CTS/St) based membranes.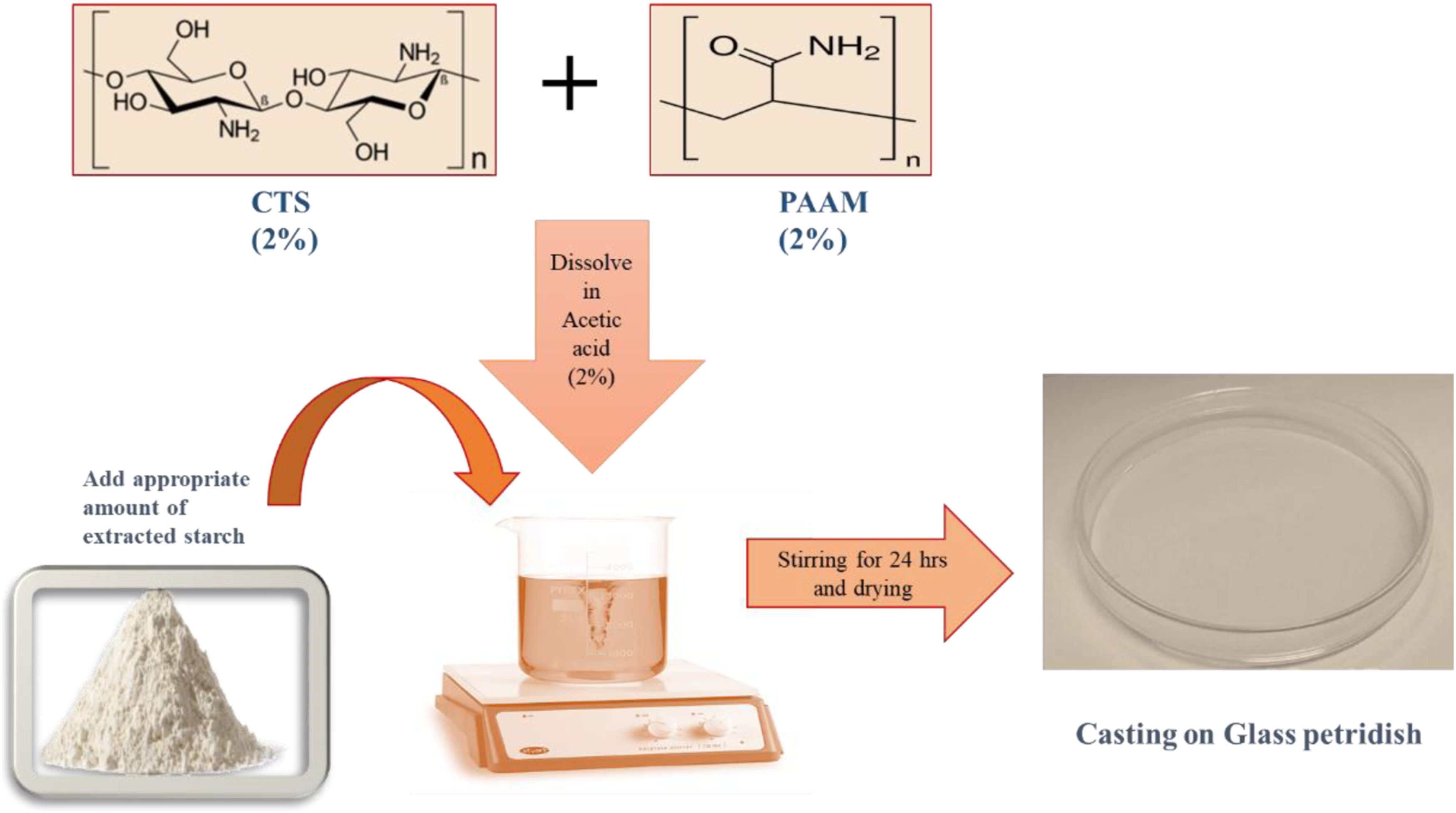
Characterization
The presence of the functional groups of both mixed and individual polymers was verified by Fourier-transform infrared (FTIR) technique (Shimadzu FTIR-8400 S, Japan)14–16 and Raman scattering spectrometer (SENTERRA-Bruker, Germany). The surface morphology of the prepared membranes was observed by using scanning electron microscope (SEM, JEOL JSM-6360LA, Japan). Specimens of the obtained membranes were examined at an acceleration voltage of 10 KV. 17 Thermal stability of these membranes was investigated using TGA analysis (Shimadzu TGA-50, Japan). The measurements were performed on TA instruments, at a heating rate of 10°C min-1, under nitrogen atmosphere from 30 to 800°C. The surface roughness of all prepared membranes was determined using surface roughness meter (SJ- 201P, Japan).18-20
Water uptake and water contact angle
Water uptake of the prepared membranes was obtained by the change in the weight and area between the wet (Ww) and dry (Wd) samples as shown in the following equation21–23
The hydrophilicity of the prepared membranes was evaluated from contact angle (CA) measurements. For each sample, CA was measured at room temperature using an optical system comprised of a zoom video lens (Ramé-Hart Instrument Goniometer, model 500-F1, France) connected to a charge-coupled device (CCD) camera operated via software. Contact angle was determined using Image J software (1.47v) with the plug-in drop shape analysis. Small drops (∼2 μL) were manually deposited on each membrane using a precision micropipette. 18
Results and Discussion
FTIR Spectra
Presence of the functional groups in the prepared hydrogel samples was confirmed by FT-IR spectra as shown in Figure 1. It showed the characteristic peaks of poly acrylamide (PAAM) and chitosan (CTS). The combined band at 3331 cm-1 could be assigned to O–H and N–H stretching group, and the band at 1663 cm-1 is ascribed to the ν C=O group of the amide. The band at 2881 cm-1 is ascribed to aliphatic CH stretching vibration. Additionally, the bands at 1584, 1148, and 1022 cm-1 are assigned to δN–H of NH2, –CH2 bending, anti-symmetric stretching of the C–O–C, respectively. Appearance of a band at 1663 cm-1 could be assigned to the stretching vibration of the amide carbonyl group which is a confirmation for the interaction between PAAM and CTS.24,25 Meanwhile, the spectrum of the PAAM-CTS/St blend membranes showed stretching bands at 851, 967, 1146, and 1331 cm-1 which are characteristic bands of saccharide structure. There is also a band at 1339 cm-1 for (–C–N–) group.
26
In addition, there is also a distinct absorption band at 3400 cm-1 corresponding to the hydroxyl group. It is clear from the spectra that the PAAM-CTS/St superabsorbent membrane had both characteristic peaks of PAAM-CTS and the saccharide unit of starch, providing evidence of the formation of PAAM-CTS/St hydrophilic network that can be used in baby diapers as it can absorb and retain large amounts of water and aqueous solutions. Fourier-transform infrared spectra of PAAM-CTS and PAAM-CTS/St blend-based membranes.
Raman scattering spectra
Raman spectra of blending PAAM-CTS and PAAM-CTS/St are presented in Figure 2. As it can be seen, mainly, bands around 3300 cm-1 are due to stretching vibrations of N–H. The bands at 1645, 1600, and 1414 cm-1 are ascribed to C=O (amide I), N–H (amide II), and C-N (amide III), respectively. The bands observed at 1158 cm-1 and in 1190–1000 cm-1 region are attributed to C–O stretching vibrations. Therefore, the presence of PAAM and CTS together in the network suggests the interaction of PAAM and CTS backbones.
27
The blending of starch with PAAM-CTS backbones show a characteristic absorption band at 2931 cm-1 due to stretching band of methylene group. The spectrum also exhibited absorption band at 1647 cm-1, which could be assigned to the C=O group, as well as the wave number 1076.3 cm-1 indicates the presence of a C–O group on the starch. The presence of these key functional groups in the spectrum has indicated the successful formation of hydrophilic blending PAAM-CTS/St membrane.
28
Therefore, in this study, we developed a biodegradable superabsorbent polymer using chitosan, poly acrylamide, and starch to be used as diapers. Raman spectra of PAAM-CTS and PAAM-CTS/St blend-based membranes.
SEM Micrographs
Apart from the observed functional groups in the FTIR and Raman spectra, the successful blending of the prepared membranes was also observed by the SEM micrographs which compares PAAM-CTS and PAAM-CTS/St membranes with different ratios (1:0.5, 1:1, and 1:2). Figure 3a and b shows that the surface morphology of PAAM-CTS membrane is homogenous with no pore cracks or gross defects. On the other hand, the presence of rough surface topology after blending of starch and PAAM-CTS with the appearance of small pores 29,30 indicate the presence of starch granules and the interaction of starch and PAAM-CTS. In addition, the membrane roughness and thickness were proportionally increasing as the concentration of starch was increased. Existence of these pores in the membrane’s surface strongly suggests the increase in the swelling kinetics of the prepared membranes. The scanning electron microscopy was used to confirm the presence of macro pores in the prepared films, which supposed that these pores are the regions of the water permeation and interaction sites of water molecules with hydrophilic groups of the prepared membranes to be easily used as diapers. (a) Scanning electron microscope micrographs of the surfaces of PAAM-CTS and PAAM-CTS/St blend-based membranes at magnification power of 500 X. (b) Scanning electron microscope micrographs of the cross-sections of PAAM-CTS and PAAM-CTS/St blend-based membranes at magnification power of 500 X.
Thermal analysis
TGA of PAAM-CTS membrane (Figure 4) shows a weight loss in three stages. The first stage occurs in the range of 100–200°C due to the desorption of water. The second stage ranges between 210 and 310°C shows about 20.21% loss in the weight. This may be corresponding to the decomposition of the amide side groups in the form of ammonia. The third stage of weight loss starts at 350 °C and continues to 500°C during which there was 53.86% of weight loss due to the degradation of the main backbone. The TGA of the PAAM-CTS/St blended membrane supports the formation of the blending process. It is observed in Figure 4 that the prepared membranes began to lose the adsorbed water at 110°C. The main decomposition stage of the product occurred in the temperature range from 190 to 250°C with a weight loss of 71.19% which is corresponding to random chain breaking, deacetylation of chitosan, and decomposition of the amide groups. In the third stage, a weight loss of 27.9% occurred in the range of 371–433 °C which is due to the decomposition of the main backbone.31,32 According to these results, the addition of starch to PAAM-CTS membrane increased the thermal stability of the prepared membranes. TGA of PAAM-CTS and PAAM-CTS/St blend-based membranes.
Surface Roughness
Surface roughness values.

Water uptake and contact angle
Water uptake and contact angle values.

Suggested mechanism of water uptake by the polymeric membranes
In this study, immersion of the chitosan-acrylamide blend membranes in water for 24 h caused an increase in water absorption. The revealed water absorption values were due to the weight gaining that likely occurred in relation to diffusion during immersion. We propose a diffusion mechanism in the presence of water molecules, with regards to being ionized into H+ and OH–, and were considered polar. They were trapped at the absorption sites, that is, –OH, –COC, and –NH functional groups in the chitosan polymer and –OH and –COOH in the acrylamide polymer. This absorption might encompass the hydrogen-bonded and/or hydrogen-unbounded state of the polar groups. The uneven water absorption of some membranes might be due to the uneven behavior of the fractional absorbed volume. This effect may also be explained by water transfer depending on the surface diffusion and, consequently, the sensitivity of the diffusion coefficient to the activation energy.
Different water absorption was seen in different membranes. This is due to the increase in starch percentage concentration, which increased the polarity nature of the membranes. Thus, an increase in the number of polar groups cause increase in the number of water absorption sites, resulting in increasing the water uptake percentage.
Conclusion
Blended membranes of polyacrylamide/chitosan and starch membranes were prepared in ratios of 1:0.5, 1:1, and 1:2. These membranes were characterized using FTIR, Raman, TGA, SEM, surface roughness, contact angle, and water uptake. The performed characterization techniques confirmed the proper blending process of the three polymers in all prepared concentrations. It would be concluded that the addition of starch to polyacrylamide/chitosan blend can improve its water holding capacity to an elevated range and recommend its usage in water storage depending applications such as diapers industry. So, in this research, we provided a disposable diaper using chitosan, which has excellent anti-bacterial, deodorizing and aeration properties, prevents skin irritations, adsorbs harmful materials, and alleviates atopic dermatitis.
Footnotes
Acknowledgments
Authors thank the Deanship of Scientific Research at King Khalid University for funding under Grant no. (RGP-1/244/42) and thank the City of Scientific Research and Technological Applications and its central laboratories for providing all the tools, facilities, and instruments required for the preparation and characterization of the prepared materials.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported byKing Khalid University (RGP.-1/244/42).