Abstract
Chitosan (Ch) has been incorporated with Tectona grandis leaf extract and stem extract at different compositions (5%, 10% and 15% v/v) to study the physicochemical properties of the biocomposites. The thin-film composites were prepared by the solution casting method to investigate the removal of chromium (VI) from water at varied pH conditions. The results showed that incorporation of the plant extracts improved the mechanical and UV barrier properties of the TFCs. Thermo-gravimetric analysis (TGA) showed that the degradation temperature of the films containing the extracts improved only slightly in comparison to the control film. Adsorption studies showed that for maximum Cr (VI) removal, Ch-L5 and Ch-L10 films were efficient. These eco-friendly prepared films are cost-effective and can also be a suitable material for food packaging.
Introduction
The widespread use of synthetic polymers is creating serious environmental problems due to their non-degradable features leading to many issues and concerns. These polymers are rarely recycled and have simply been thrown away, which may break into smaller pieces within a period, but they remain in our environment as plastic debris. 1 These dumped polymeric debris end up reaching water sources via tributaries or water channels polluting water reservoirs, oceans, etc., causing a serious threat to all living creatures. One of the key focusses to pare down this pollution is to find an alternative by developing biodegradable, biocompatible and edible films from natural resources such as proteins, lipids and polysaccharides.
The production of biopolymers has many beneficial advantages including applications in biomedical, biotechnological, food packaging, industrial and pharmaceutical sectors. Also, the raw materials to produce biopolymers are abundant and easily available. Chitosan, a biopolymer, is a cationic polysaccharide derived from the alkaline or enzymatic deacetylation of chitin, which is the second most abundant natural polymer after cellulose. It can be obtained from natural sources such as exoskeletons of arthropods, insects, shells of crustacean shrimps, prawns, lobsters and crabs.2,3 Chitosan is non-toxic, biocompatible and biodegradable for which it has been widely used in agriculture, cosmetology, food industry, pharmacology, textile industries, wastewater treatment, etc.4–6 Chemically, chitosan is composed of less than 40% of
Tectona grandis, commonly known as teak, is a large deciduous tree 30–35 m tall and belongs to the Lamiaceae family. It is the most widely cultivated, high-value hardwood (HVH) in the entire world and is native to India and Southeast Asian countries. 15 The stem of T. grandis contains phytochemicals such as silica, calcium phosphate, ammonium phosphate, magnesium phosphate, 16 1,5-dihydroxy-2-methyl-9,10-anthraquinone, tecomaquinone-I, tectoquinone, 17 anthraquinone-2-carboxylic acid, anthraquinone-2-carboxaldehyde, 18 lapachol, 5-hydroxy-lapachol, methylquinizarin, squalene 19 and lignins, 20 and the leaves contain tectoleafquinone, 21 anthocyanin which is a natural colouring pigment. 22 Thus, the plant as a whole possesses various medicinal properties such as antibacterial, antioxidant, cytotoxicity, 23 antihemolytic, 24 wound healing, 25 antifungal, anthelmintic, anti-inflammatory, 26 analgesic 27 and diuretics. 28 Traditionally, teak is used against bronchitis, biliousness, hyperacidity, diabetes, leprosy, astringent, burn wound and piles.15,28 Besides its various health benefits, it has also been used as a natural dye15,29 and also as a biosorbent for the removal of heavy metals.30,31 These advantages of the extracts from various parts of T. grandis and its abundance make it a suitable material for the incorporation into the chitosan matrix to widen its application areas.
In this context, the objective of the present work is to produce and characterize chitosan-based thin films incorporated with leaf and wood extracts and also to study the effect of pH on the adsorption behaviour of the films. The produced composite films were characterized using universal testing machine (UTM), Fourier transform infrared spectroscopy (FTIR), scanning electron microscopy (SEM), thermogravimetric analysis (TGA), differential scanning calorimetry (DSC) and UV-Vis spectroscopy. Also, physicochemical properties like moisture content, solubility, swelling degree and light transmission were performed for comparative study.
Materials and methods
Materials
Chitosan (molecular weight 3800–20,000 Da and ≥75% degree of deacetylation, Cas No. 9012-76-4) from shrimp shell was purchased from Hi Media Laboratories, India. Glacial acetic acid was supplied by Merck, Germany. The plant materials (T. grandis) were locally collected from Assam Engineering College Campus, Guwahati, India.
Preparation of the plant extracts
The leaf extract (LE) and stem extract (SE) were obtained by the decoction method. Plant parts, that is, fresh leaf and stem collected were washed thoroughly with tap water, followed by successive washing with distilled water and oven-dried at 40°C for 24 h. Dried leaves were converted into a fine powder using a stainless steel grinder while the stem was converted to powder using a saw blade. 5 g powder of each leaf and stem were dissolved in 50 ml of distilled water separately and heated at 80°C for 90 min, respectively. Finally, the obtained infusions were filtered through Whatman filter paper, and the extracts were stored for further use.
Preparation of the films
All the chitosan-teak (T. grandis) extract composite films were prepared by the solution casting method. Three different compositions of the chitosan composite films were prepared by incorporating T. grandis (LE and SE) and one controlled chitosan film without any extract. Chitosan solution was prepared by adding 2% (w/v) chitosan in 2% (v/v) glacial acetic acid aqueous solution. The mixture was stirred for 12 h via a magnetic stirrer at 600 r/min to ensure the complete dissolution of the polymer solution. Subsequently, LE and SE in various concentrations (i.e. 5%, 10% and 15% v/v) referred to as Ch-L5, Ch-L10, Ch-L15, Ch-S5, Ch-S10 and Ch-S15, respectively, were mixed to the polymeric solution and stirred for 30 min using a magnetic stirrer at 1000 r/min. The prepared film-forming solutions were centrifuged at 2100 r/min for 5 min to remove bubbles and impurities. Each solution was then cast onto Petri dishes (10 cm diameter) and oven-dried at 60°C for 12 h. The dried films were peeled off and stored at room temperature for further analysis.
Methods
Water content and solubility
The water content and solubility were determined accordingly to Souza et al. and Kaya et al. with minor modifications.32,33 The film samples were cut into square shape (2 × 2 cm) and weighed using an analytical balance (Shimadzu TX323L) for the initial weight (m1). The samples were then dried in a hot air oven at 70°C for 12 h to get the initial dry mass (m2). After that measure, film specimens were placed in vials containing 20 ml of distilled water, covered and stored for 48 h at room temperature (23 ± 2°C). After 48 h, the remaining water (not absorbed by film samples) was discarded off and the specimens were superficially dried with filter paper. The residual film specimens were dried in an oven at 70°C for 12 h to determine the final dry mass (m3). Three measurements were taken for each film sample to calculate the average value of the parameters. Water content and film solubility were calculated by the equations (1) and (2), respectively
Swelling degree
The chitosan blended and controlled films were cut into 2 cm x 2 cm and dried at 60°C under vacuum for 6 h to obtain initial weight (Wi). The films were then immersed in phosphate-based buffer saline (PBS) solution of pH 7.4 at room temperature for 2 h. The swollen films were taken out from the solution and excessive water was gently removed using filter paper. The films were balanced using an analytical balance (Shimadzu TX323L) both before and after immersing in solutions referred to as Wi and Wo, respectively. The swelling degree (Sd) was calculated using equation (3)
Mechanical properties
The mechanical properties of the films such as tensile strength (TS), Young’s modulus (YM) and elongation at break (EB) were analysed by a universal testing machine (Model Z005, Zwick/Roell, Germany) as per ASTM standard method D8802-02. 34 The film strips were cut as per the standard to a length of 80 mm and width of 15 mm. The initial grip separation was maintained at 50 mm, and the crosshead speed was set at 3 mm min−1. The TS, YM and EB values were obtained by using TestXpert II testing software.
Light transmission
The light transmittance of the chitosan control (Ch) film and composite films was analysed in the wavelength range of 210–750 nm using Labtronics LT-2201 UV-Vis spectrophotometer. The films were cut into cuvette shaped rectangular pieces and placed directly into the spectrophotometer test cell. The transmittance of the film samples was recorded at room temperature using an empty cuvette or considering air as a reference.
Fourier transform infrared spectroscopy (FTIR)
Fourier transform infrared spectroscopy was used to study the structural interactions between chitosan and T. grandis extracts in the film. The spectra were recorded using Cary 630 spectrometer in the wavelength range (4000 to 650 cm−1) with a resolution of 4 cm−1 in the transmittance mode for 4–5 scans at room temperature. The samples for FTIR analysis were prepared by grinding the dry films with powdered KBr in the ratio of 1:9 (sample: KBr) and then compressed to form discs.
Scanning electron microscopy (SEM)
Surface morphologies and texture of the films were analysed using SEM (Zeiss, Germany Model ∑igma VP). The films were coated with Au/Pd sputtering before imaging
Thermogravimetry (TG) analysis
Thermogravimetry analysis was used to determine the thermal stability and decomposition temperature of the film samples. TG analysis was performed in the Perkin Elmer 4000 instrument. Nitrogen was used as a carrier gas at the flow rate of 50 ml min−1. The samples were heated from 30 to 600°C at the rate of 10°C per min to record the TG and differential thermogravimetry (DTG) curves.
Differential scanning calorimetry (DSC)
Differential scanning calorimetry was used to determine the thermal transitions of the films where glass transition temperature (Tg) and melting temperature (Tm) were analysed using a Mettler Toledo DSC3 thermal analyzer. The samples were inserted into the apparatus under a nitrogen atmosphere and scanned in the temperature range between 30 and 420°C at 10°C per min, respectively. The mass of each film sample was 5–6 mg. The Tm of each sample was taken at the initial change in the slope of the heat capacity of the DSC curve.
Adsorption experiments
Adsorption conditions for the batch experiment.

The pH of the solutions was adjusted at 5, 7 and 9, respectively, by using 0.01 

Results and discussion
Water content and Solubility
Water content, solubility and swelling degree of chitosan-based films.

aAll values were mean ± SD of three samples.
High solubility indicates less water resistance; thus, the maximum solubility showed by Ch-S10 and Ch-S5 films indicates faster diffusion or weaker molecular interactions between the chitosan and the stem extract. On the other hand, the decreasing trend in the solubility of Ch-LE films may be due to the interactions between the polyphenolic group of the leaf extract and the chitosan matrix via hydrogen or covalent bonding with hydroxyl (–OH) and amino (–NH2) groups of chitosan. 38 These interactions may limit the availability of the hydrogen groups to form hydrophilic bonding with water, leading to a decrease in solubility. Similar phenomena were reported by Kaya et al. 33 who incorporated chitosan with stem, leaf and seed extracts of Pistacia terebinthus.
Swelling degree
The swelling degree of chitosan films varied according to the type of extract and the composition used, as shown in Table 2. The maximum swelling can be seen in Ch-SE films followed by Ch-LE films with a decreasing trend as the concentration of the extracts is increased. In phosphate buffer saline (pH 7.4), all chitosan films swelled considerably at an initial period till it reaches maximum retention capacity. When the films absorbed water and began to swell, an –NH2 group of chitosan was protonated simultaneously, followed by penetration of the swollen film by phosphate anions in the medium to cross-link at –NH4+ groups of chitosan molecules. 39 As time progressed, the cross-linking process increased, resulting in a denser film while reducing the swelling of the films. Similar phenomena have been reported by Wu et al., 38 who reported that the hydrophobic interaction between the polyphenols and hydrophobic region of the chitosan matrix are known to occur, and also the water in the extracts generates weaker hydrogen bonds than those created between the –OH groups and –NH2 groups of the extract and the chitosan, which results in the swelling reduction.
Mechanical properties
Tensile strength, elongation at break and Young’s modulus value of the prepared film samples.

aAll values were mean ± SD of five samples.
The TS of the chitosan control film is found to be 13.73 ± 1.40 MPa with 4.29 ± 0.89% EB. The TS and EB of Ch-LE films increased when LE concentrations were increased from 5 to 10%, but no significant change was exhibited when the concentration was further raised to 15%. On the other hand, the TS and EB of Ch-SE films were found to be maximum throughout the compositions as compared to that of the control film, but with a gradual decrease from 5 to 15%. The decreasing trend in TS and EB for both the Ch-LE and Ch-SE films may be due to the crystallite formation or heterogeneity between T. grandis components at high concentration and chitosan polymer chain, which leads to hinder the molecular chain mobility and polymer aggregation in chitosan matrix producing brittle film structure with reduced film flexibility and TS. 41
The increase in YM indicates the stiffness of the material. In that context, the higher value of Young’s modulus for Ch-L5 and Ch-S5 was observed comparatively stiffer than the other films. Figure 1 shows that the Ch-S5 and Ch-S10 films exhibited a large EB. Therefore, these composite films had ductile nature. So the Ch-L5 and Ch-S5 films can be suggested as the stiffest among the other considered films. These results exhibited that incorporation of stem extract from T. grandis highly increased the TS and elasticity of the composite films due to the presence of lignin and cellulose as it has the cross-linking ability and adhesive nature.
42
Thus, it can be assumed that the judicial use of both extracts can improve the mechanical properties of chitosan films significantly. Variation of tensile strength with elongation at break of the prepared film samples.
Light transmission
The light transmissions of the films were studied by scanning the films at a wavelength between 190 and 750 nm, as shown in Figure 2. Chitosan control film showed poor light-blocking properties; the light transmittance at 315 nm and 400 nm was 40.50% and 76.19%, respectively. But the film with LE showed good light-blocking ability than the other films as the amount of light transmitted was much lower than that of the chitosan control films as shown in Table 4. This may be due to the presence of the natural colouring pigment anthocyanin present in the leaves of the T. grandis.22,43 On the other hand, the light transmission of Ch-SE films was higher than the Ch-LE films and marginally lower than that of the control film, thereby showing poorer light-blocking property as compared to that of the Ch-LE films. UV-Vis spectroscopy of Ch, Ch-L5, Ch-L10, Ch-L15, Ch-S5, Ch-S10 and Ch-S15 films. Percentage transmittance of chitosan control, Ch-LE and Ch-SE films.

UV light plays a significant role in the oxidation process of various chemical reactions which can accelerate the deterioration of food surfaces. Hence, the UV light-blocking capacity is considered a desirable characteristic for the packaging materials as it can extend the shelf life of products. Thus, the Ch-LE composite films can be a promising material for packaging industries due to the higher blocking capacity than all the films at both 315 nm and 400 nm range, that is, in the UV range.
Fourier transform infrared spectroscopy (FTIR)
FTIR spectra have been used to analyse the functional groups and structural bonding between Ch-LE and Ch-SE composite films as displayed in Figure 3. The strong and broad band centred at 3426 cm−1 is due to the axial O–H and N–H stretching.
44
The peak at 2929 cm−1 can be attributed to C–H symmetric stretching. The structural region between 1700 and 1600 cm−1 (amide I band) is mainly due to the C=O stretching vibrations of the peptide linkage present in acetamido groups (amide I) of chitosan.45,46 The peak at 1654 cm−1 suggests the stretching vibration of C=O group due to the characteristic of the amides (amide I) and other compounds containing the same in the film.
47
The characteristic peak at 1560 cm−1 can be assigned to the angular deformation of N–H bond of the amine group (amide II). The band at around 1420 cm−1 shows the presence of carboxylate groups which is due to the leftover acetic acid used for the preparation of the chitosan film-forming solution. The bands in the fingerprint region between 1154 and 1073 cm−1 are due to the vibrations of glycosidic bonds, C–O and C–O–C of the polymeric chain.
48
After the incorporation of LE and SE with chitosan the bands in the wavelength region between 1735 and 901 cm−1 seems to be broad which may be due to the inter and intramolecular hydrogen bonding with the –OH and amine group of chitosan. FTIR spectra of chitosan biocomposites incorporated with leaf extract and stem extract.
Scanning electron microscopy (SEM)
SEM has been used to study the surface characteristics of chitosan control (Chi), Ch-LE and Ch-SE films of different proportions. Figure 4 represents the SEM micrographs of all the composite films and it can be seen that the films are clear and smooth without any fractures or pores. The micrographs show that both the LE and SE are uniformly incorporated in the chitosan matrix. The results indicate that the excellent compatibility between the two components of the films resulted in a homogeneously dispersed film. SEM micrographs of (a) Ch, (b) Ch-L5, (c) Ch-L10, (d) Ch-L15, (e) Ch-S5, (f) Ch-S10 and (g) Ch-S15.
Thermogravimetry (TG) analysis
Figure 5(a) and (b) show the TG and DTG curves corresponding to thermal decomposition and weight loss of all the prepared films. It can be seen that the TG curves are smooth with two degradation patterns except for the Ch-S5 film with three degradation patterns. The first degradation starts at 55°C to about 150°C which may be due to dehydration of the film or the removal of residual solvent. The second degradation starts from 160°C due to the decomposition of the main polymeric chains of chitosan. DTG curve shows the major weight loss of up to 43% during the second degradation. The third weight loss for Ch-S5 film showed at 560 to 600°C. However, at 600°C, the weight loss of chitosan film is higher in comparison to the other composite films. Thus, the incorporation of both the LE and SE with chitosan enhanced the thermal stability and reduced the weight loss of the film among which Ch-S10 and Ch-S15 showed maximum stability. (a) TG and (b) DTG curves of Ch, Ch-L5, Ch-L10, Ch-L15, Ch-S5, Ch-S10 and Ch-S15 films.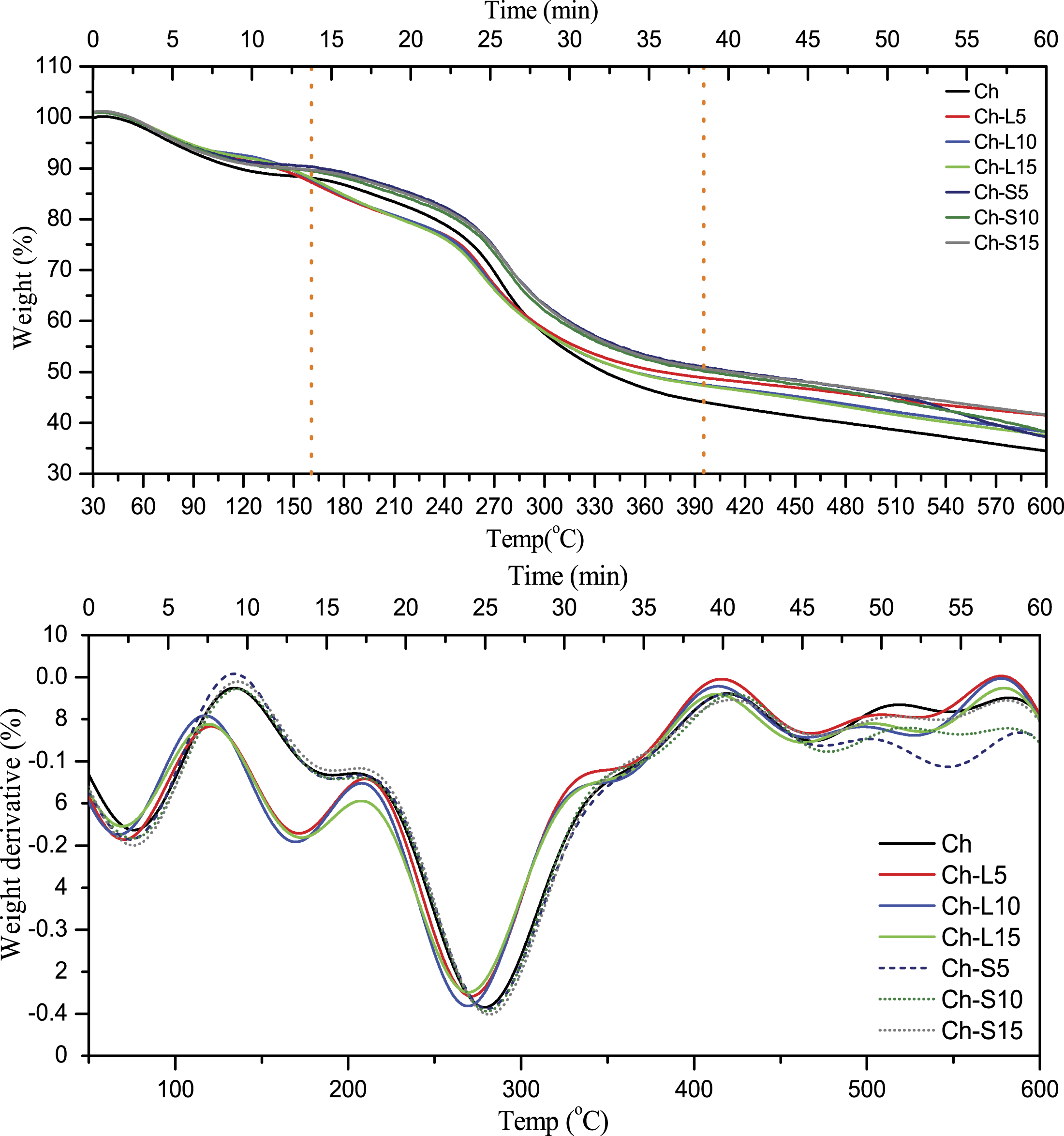
Differential scanning calorimetry (DSC)
The DSC thermogram of the chitosan and chitosan-based films is shown in Figure 6. The thermogram shows one broad endothermic peak around 70–80°C, which is due to the evaporation of residual solvents used in the film preparation or moisture absorbed from the atmosphere. Sakurai et al. reported that the Tg of chitosan is around 203°C.
49
But in our study, it is observed that after incorporation of LE and SE at different compositions into the chitosan matrix leads to the disappearance of Tg. This may be due to the compact structure of the composite film in comparison to chitosan.
50
Also, it has already been reported that Tg varies accordingly and the reason behind that has been attributed to the fact that natural polymers’ properties like crystallinity, MW and DDA can present wide variations according to the source and methods of extraction.51,52 The exothermic peak at around 275–280°C is due to the melting of the chitosan polymer chain. The proper analysis shows that the exothermic peaks of Ch-SE films are at higher temperature than both the Ch control and Ch-LE films which indicates an enhancement of the films’ thermal stability. DSC thermogram of Ch, Ch-L5, Ch-L10, Ch-L15, Ch-S5, Ch-S10 and Ch-S15 films.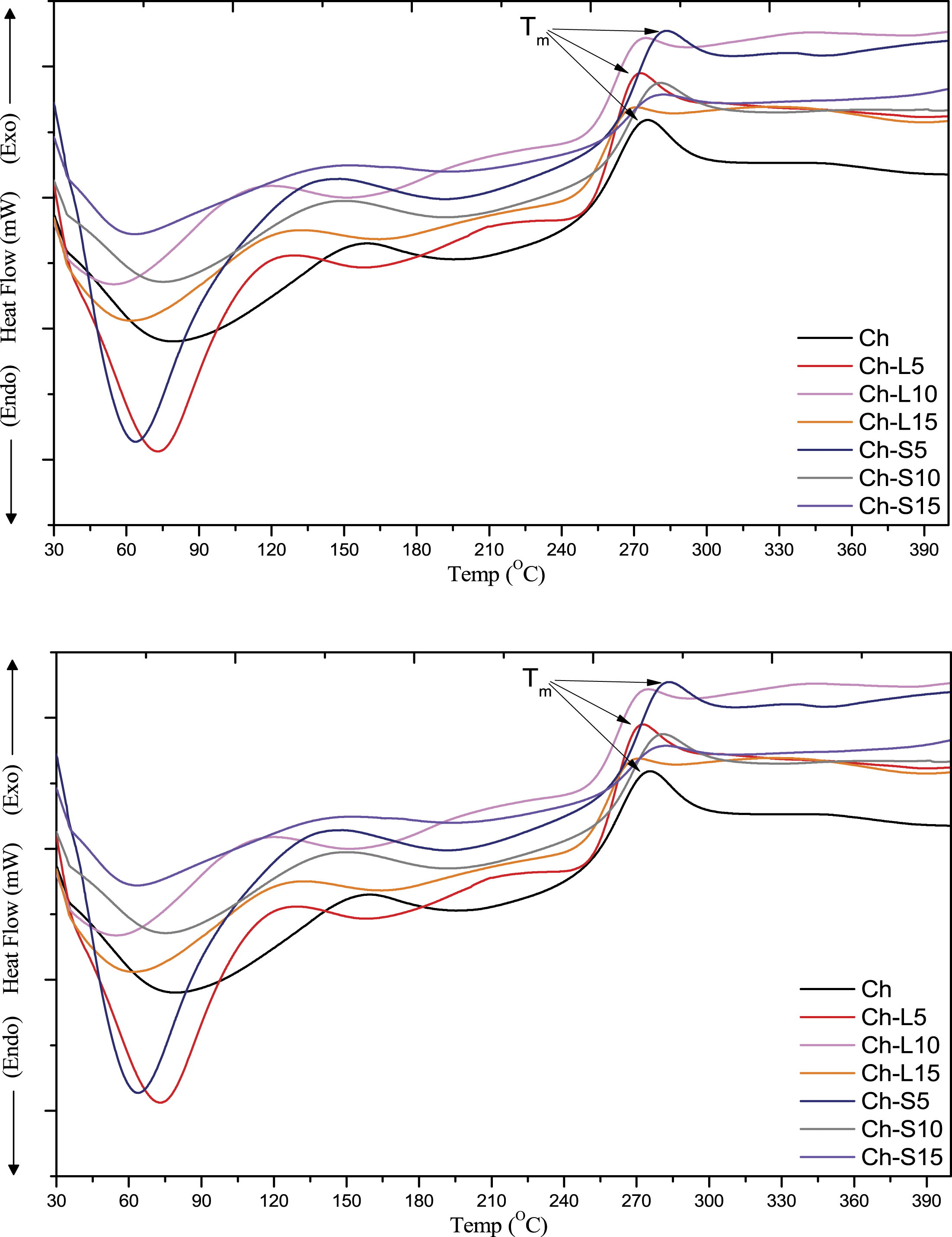
Adsorption experiment
The adsorption of metal ions (adsorbate) on Ch-control, Ch-LE and Ch-SE films (adsorbent) was studied under varied pH conditions as shown in Table 1. Figure 7 shows that the adsorption of Cr (VI) by the prepared films is highly pH-dependent. The percentage removal efficiency of Cr (VI) decreased with an increase in the pH for all the films as the pH of the solution largely affects the adsorbate and adsorbent behaviour under aqueous conditions. However, the adsorption trend for the Ch-SE films varied slightly from the Ch-LE films as in Ch-SE films maximum adsorption can be seen at pH 7. This may be due to the fact that at low pH, the Ch-SE films start getting dissolved in the aqueous solution causing desorption of the metal ion. Effect of pH on adsorption of Cr (VI) by Ch-control, Ch-LE and Ch-SE films under optimum condition.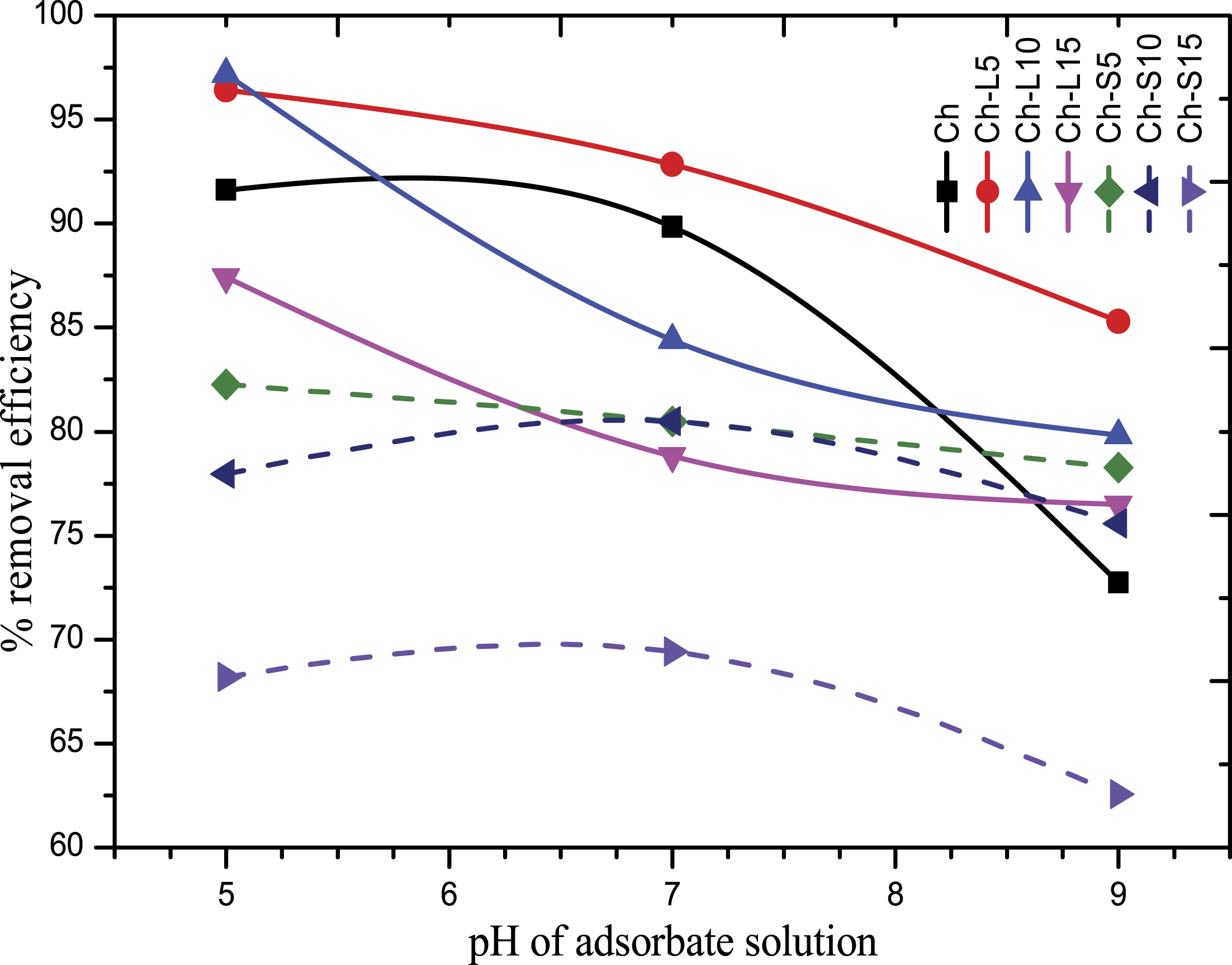
The adsorption of Cr (VI) on the adsorbents can be explained by the nature of the ionic charges present in the adsorbate and the adsorbent. At pH <6, the –NH2 groups in chitosan gets protonated to –NH3+and create a network of polycations on the surface of the adsorbent, which acts as an active binding site for the metal ions. However, as the pH increases above 6.5, the protonated species of the adsorbent starts getting deprotonated, that is, –NH3+ to –NH2, reducing the metal-binding sites. On the other hand, Cr (VI) exists as a negatively charged species, that is, HCrO4− ion at pH between 2 and 8 and Cr2O7−/CrO42−ion at pH 4–12, respectively.53,54 These negatively charged metal ions get attached to the –NH3+ group of the adsorbents at low pH. But as the pH gets increased, the adsorbent with reduced metal-binding sites starts repelling the negatively charged metal ions. As a result, at high pH, the adsorption capacity of the adsorbent decreases. 35
Conclusion
The current investigation aimed at producing an active biodegradable composite film from chitosan incorporated with T. grandis LE and SE with enhanced physicochemical and thermal properties to study the effect of pH on the adsorption behaviour of heavy metal. With the incorporation of LE and SE, the functional properties of the films improved significantly. The solubility of the films has been reduced to a great extent by the addition of the extracts. The Ch-S5 film showed maximum TS and EB with 29.57 ± 3.20 MPa and 37.84 ± 3.75%, respectively. Also, the light transmission rate was reduced from 76.19% for chitosan control film to 14.95% for Ch-L15 film at 400 nm. The thermal properties of the films were slightly improved in comparison to the chitosan control film. The batch adsorption experiments carried out at different pH showed maximum adsorption capacities for Ch-L5 and Ch-L10 at pH 9 and 5, respectively. Thus, the Ch-L5 and Ch-L10 films can be potential materials for the removal of Cr (VI) from various water sources. Moreover, as chitosan is recognized to be non-toxic, the prepared LE and SE incorporated films can be considered as good adsorbents for removing heavy metals and also as a promising UV-resistant packaging material for the preservation and storage of different food materials.
Footnotes
Acknowledgements
This work was supported by the Assam Science and Technology University (ASTU) under the Collaborative Research Project (CRS/2019/CRSAEC16) of the TEQIP-III scheme. The authors acknowledge the Assam Science and Technology University (ASTU), Guwahati, India; Central Instruments Facility (CIF), IIT Guwahati, India; Department of Chemistry, B. Borooah College, Guwahati, India and Central Instruments Facility (CIF) for the support to carry out the analytical characterizations.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Assam Science and Technology University/TEQIP-III/Collaborative Research/2019/ Award No: CRSAEC16.



