Abstract
The incorporation of natural extracts into polymeric films has been a significant advancement as active and eco-friendly food packaging. Chitosan (Ch) based films incorporated with poly(ethylene glycol) (PEG) and
Introduction
Growing awareness for environmental pollution and the demands for quality foods with eco-friendly packaging, researchers have shifted their focus to sustainable and active packaging systems that preserve food quality over some time. Almost all the food items that we purchase, especially processed foods, come packaged in films. These packaging films are prepared from various materials such as paper, plastic, paperboard, cardboard, wax, wood, and many more. But plastic is the most extensively accepted packaging material by both vendors and consumers as it is durable, lightweight, moisture resistant, and relatively inexpensive. 1 These plastic packaging materials are produced from chemicals derived from petroleum, natural gas, and coal—all of which are from non-renewable resources and are specifically meant for single-use only. Single-use plastics are seldom recycled, and thus end up as wastes directly from our households to junkyards, landfills, or our environment. 2 This single-use plastic pollution is becoming a global concern, as they are non-degradable and can remain in the environment for a longer period. To tackle some of the problems arising out of these plastic wastes, substantial research has been undertaken to obtain environment friendly food packaging materials.3,4 Thus, biodegradable packaging materials can be an alternative to conventional plastic-based packaging as they are biodegradable, non-toxic, and versatile in different fields.5,6
To obtain biodegradable packaging films, a range of naturally derived materials including polysaccharides, proteins, and lipids can be modified and altered as per needs. Though biodegradable polymer is considered as an alternative to plastic, its physicochemical properties don’t match that of synthetic polymer and hence researchers are trying to blend natural polymer with various other natural resources to improve its properties.7,8 Florez et al. 9 on his survey found that enriched chitosan-films possess lower mechanical resistance properties, such as lower tensile strength (TS) and elongation at break (EB) values than pure chitosan samples. Similarly, Riaz et al. 10 and Yang et al. 11 reported that the addition of natural extracts in the chitosan decrease the TS and EB of the films. In that context, our study focuses on the improvement of the physicochemical properties of a naturally available biopolymer, chitosan by blending with a mixture.
Chitosan is one such polymer that has been studied extensively as a main structural component in various fields for its wide versatility. 12 Chitosan is a natural biopolymer derived by alkaline deacetylation of chitin or by an enzyme chitinase. 13 It has non-toxic, biodegradable, and antimicrobial characteristics with exceptional physicochemical and film-forming properties which are of great interest for packaging purposes.14,15 Chitosan is bio-adhesive and can readily bind to negatively charged surfaces such as membranes. Chitosan’s positive surface charge and biocompatibility enable it to effectively support the modification of its functional properties. 16 But the major drawback of chitosan is that it is only soluble in an aqueous medium in presence of a small amount of acid such as acetic acid, formic acid, l -glutamic acid, lactic acid, succinic acid, etc. But this drawback can be a favourable property to optimise it for packaging applications. Due to its film-forming properties and antimicrobial character, chitosan is a potential material for packaging films.17,18 However, chitosan films are rigid and need plasticizers to reduce frictional forces between polymer chains, such as hydrogen bonds or ionic forces, to improve mechanical properties. In this study, authors aimed at modifying its mechanical and functional properties by incorporating other polymers and natural substances into it.
Poly(ethylene glycol) is a versatile polyether being utilized in various applications such as biological, commercial, industrial, and medicinal purposes. Poly(ethylene glycol) is also known by its other name as poly(ethylene oxide) (PEO). Ethylene oxide macromolecules with molecular weights less than 20,000 g/mol are called PEG. 19 It is reported that PEG is soluble in water, ethanol, acetonitrile, benzene and dichloromethane. It has low toxicity and is used in a variety of products such as an anti-foaming agent in food and drinks and also as a lubricating coating for various surfaces in aqueous and non-aqueous environments. PEGs also find application as a plasticizer in the polymer industry.
Antioxidant activity is considered as one of the essential functions of active packaging films. Thus, enhancement of the antioxidant activity of chitosan film by incorporating natural substances has been widely studied. 20 Natural antioxidants used for enhancing the antioxidant activity of chitosan films include essential oils,21–23 curcumin, 20 grapefruit extract, 24 plant extracts, 25 blueberry fruit extract, 26 green tea extract, 27 and many more.
Thus, the objective of the present work is to produce and study the antioxidant activity of chitosan-PEG based bio-composites incorporated with different concentrations of TGLE. The produced composite films were characterized using a universal testing machine (UTM), fourier transform infrared spectroscopy (FTIR), and UV-visible spectroscopy. Also, physicochemical properties like water content, solubility, swelling degree, light transmission were performed for comparative study.
Materials and methods
Materials
Chitosan from shrimp shells (molecular weight 3800–20000 Daltons and ≥75% degree of deacetylation) and poly (ethylene glycol) (PEG) (molecular weight 6000 Daltons) were purchased from HiMedia Laboratories, India. Glacial acetic acid was supplied by Merck, Germany. 2,2-diphenyl-1-picrylhydrazyl (DPPH) and phosphate-buffered saline (PBS) were purchased from Sisco Research Laboratories Pvt. Ltd. (SRL), India. The
Preparation of the plant extracts
The leaf extracts were obtained by the decoction method. Fresh leaves collected were washed thoroughly in tap water, followed by successive washing in distilled water and oven-dried at 40°C for 24 h. Dried leaves were crushed into a fine powder using stainless steel grinder. 7 g powder of leaf was dissolved in 70 mL of distilled water separately and heated at 80°C for 90 min respectively. Finally, the obtained infusion was filtered through Whatman filter paper and the extract was stored for further use.
Preparation of the films
Chitosan (Ch) 2% (w/v) solution was prepared by dissolving 2 g chitosan in 100 mL of 2% (v/v) glacial acetic acid aqueous solution. The mixture was stirred using a magnetic stirrer at 600 rpm for 12 h at 60°C to ensure complete dissolution of the polymer in the solvent. This was followed by the addition of a known quantity of PEG to the chitosan solution and stirred for 2 h at 60°C. The chitosan to PEG mixture ratios were 2:1 and 4:1 by weight. The TGLE was then added to the Ch/PEG solutions at 5% and 10% v/v ratio and stirred using a magnetic stirrer at 1000 rpm for 30 min at 60°C. The prepared film-forming solutions (FFS) were centrifuged at 2100 rpm for 5 min to remove bubbles and impurities. All the Ch/PEG/TGLE composite films were prepared by solution casting method wherein solutions were cast onto a petri dish and dried at 55°C for 18 h. Two controlled Ch/PEG composite films were prepared and identified as CP2:1 and CP4:1. Whereas, four different compositions of the composite films were prepared by the addition of TGLE which were identified as CP2:1-L5, CP2:1-L10, CP4:1-L5, and CP4:1-L10. The dried films were peeled off and stored at room temperature for further analysis.
Water content and solubility
Water content (%) and solubility (%) are essential properties of a food packaging film. These parameters were determined according to Souza et al. and Kaya et al. method with minor modification.35,36 Film samples (2 cm × 2 cm) were weighed in an analytical balance (Shimadzu TX323L) and recorded as initial weight (W1). The samples were then dried in a hot air oven at 60°C for 24 h to get the initial dry mass (W2). The film specimens were placed in vials containing 20 mL of distilled water, covered and stored for 48 h at room temperature (27°C ± 2°C). After 48 h, the insoluble part was filtered out and dried in an oven at 60°C for 12 h to determine the final dry mass (W3). The tests were conducted thrice for each film sample to calculate the average value of the parameters. The water content (%) and solubility (%) parameters were calculated using the following equations (1) and (2), respectively.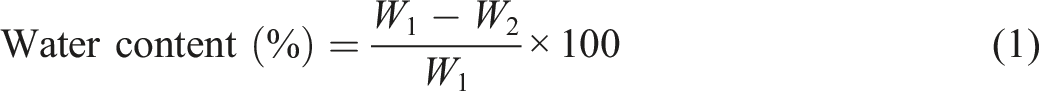
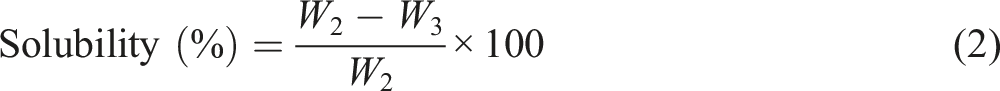
Swelling degree
The film specimens (2 cm × 2 cm) were dried at 60°C under vacuum for 12 h and weighed to obtain the initial weight (Wi). The films were then immersed for 2 h at room temperature in PBS solution of pH 7.4. After 2 h, the swollen films were filtered out from the solution and excessive water was gently removed using filter paper to obtain the final weight (Wf). The tests were performed thrice and the films were balanced using an analytical balance (Shimadzu TX323L) both before and after immersing in solutions referred to as Wi and Wf respectively. The swelling degree (Sd) of the film was calculated using equation (3)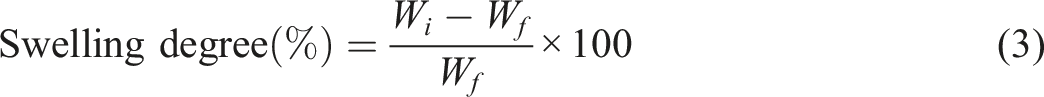
Mechanical properties
ASTM standard method D8802-02 37 was used to measure the TS, Young’s Modulus (YM) and EB of the prepared films. The film specimens were cut as per the standard to a length of 90 mm and width of 15 mm and the tests were carried out in a Universal Testing Machine (Model Z005, Zwick/Roell, Germany). The initial grip separation was maintained at 50 mm and the crosshead speed was set at 3 mm per minute across the tests. The TS, YM, and EB data were recorded by using TestXpert II testing software.
UV-visible light transmission
The UV-visible light-blocking property of the prepared films was analysed using a UV-Vis spectrophotometer (Labtronics LT-2201) in the wavelength range of 210–750 nm. The transmittance of the film samples placed directly into the spectrophotometer test cell was recorded at room temperature considering air as a reference.
Fourier transform infrared spectroscopy
Fourier transform infrared spectroscopy was used to study the structural interactions between chitosan, PEG, and
2,2-diphenyl-1-picrylhydrazyl free radical scavenging activity
The antioxidant activity of the prepared bio-composite films was analysed by using DPPH (2,2-diphenyl-1-picrylhydrazyl) free radical scavenging assay based on bleaching of bluish-red or purple colour of DPPH solution as a reagent following the method used by Wang et al.
38
with slight modification. All the film samples (50 mg) were dissolved in 6 mL of distilled water and then 3 mL of film extract were mixed with 1 mL of 1 mM methanolic solution of DPPH. UV absorbance at 517 nm was measured after the solution was allowed to stand at dark, ambient temperature for 30 min. The percentage of scavenging was calculated using equation (4).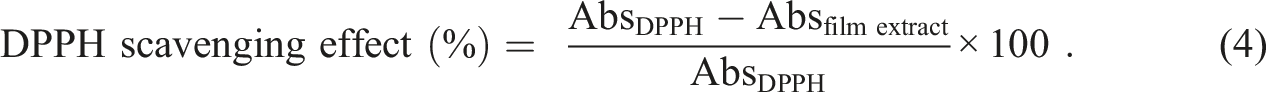
Results and discussion
Film formability and physical observation
The physical appearance and colour of the films are considered to be important factors in the packaging industry. As shown in Figure 1, Ch/PEG control films looked opaque and immiscible which is clear in CP2:1 film, but on increasing the weight ratio of chitosan to PEG, the film becomes homogeneous (CP4:1). The immiscibility or phase separation in the Ch/PEG This immiscibility or phase separation in the Ch/PEG blend films as explained by Chen et al. and Bairamov et al.39,40 may be due to the difference in the molecular weight between the two polymers or diffusion of PEG during the film formation process. When film formation starts with the evaporation of the solvent, viscosity of the FFS increases and the PEG rich droplets precipitate due to gravity. With further evaporation of the solvent, crystallization of PEG occurs on the surface of the film without fusion and coalescence, resulting in an opaque film at high content of PEG i.e. CP2:1 film.
39
This layering of the PEG on chitosan surface caused difficulty in peeling off the film from the petri dish in case of CP2:1 film. However, this difficulty was reduced to a great extent in CP4:1 film due to the increasing weight ratio of chitosan to poly(ethylene glycol). It was further reduced after incorporation of the leaf extract supporting the fact that the leaf extract helped chitosan and the PEG to bind with each other giving rise to a homogeneous film. The colouration of the Ch/PEG films changed with the addition of leaf extract from light brown to dark brown due to the presence of the colouring pigment “anthocyanin” in TGLE.
41
Visual appearance of Ch/PEG control and composite films incorporated with TGLE.
Water content and solubility
Water content, solubility and swelling degree of chitosan-based films.
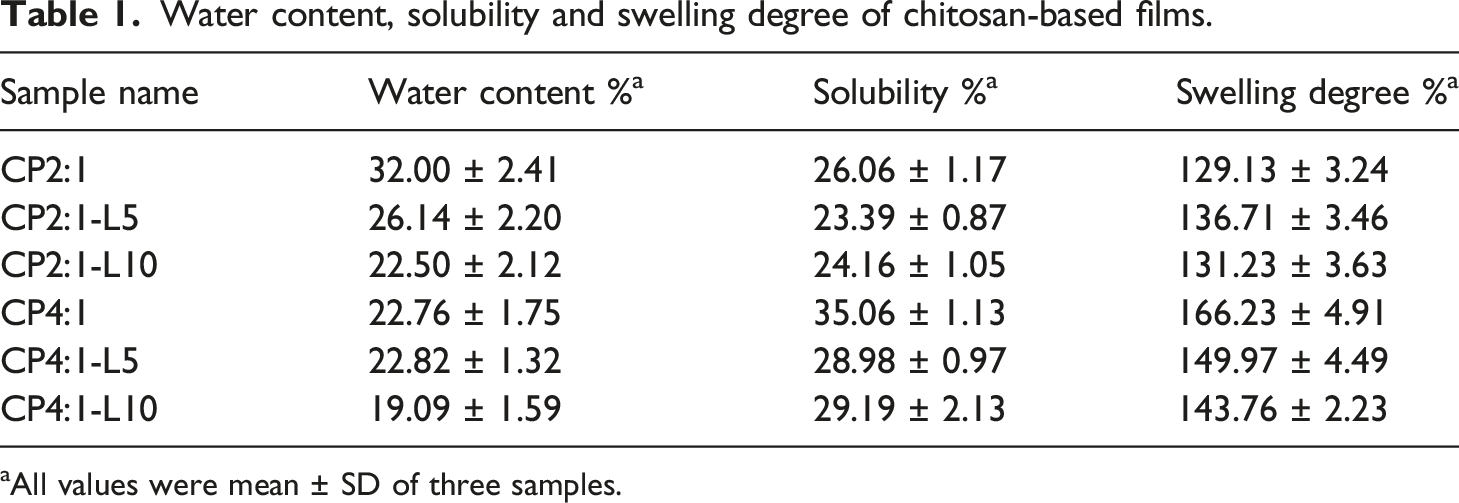
aAll values were mean ± SD of three samples.
The water solubility of a film is defined as the percentage of solute dry matter after 48 h of immersion in distilled water. High solubility indicates less water resistance, thus, the maximum solubility showed by CP4:1 film indicates faster diffusion or weaker molecular interactions between chitosan and PEG. Also, PEG has hydrophilic property and it may provide too many hydroxyl (–OH) groups to the film matrix. These extra groups could also interact with –OH groups of chitosan that would reduce the attractive force.
On the other hand, the decreasing trend in the solubility of CP2:1 and CP4:1 bio-composite films may be due to the interactions between the polyphenolic group of the leaf extract and the chitosan matrix via hydrogen or covalent bonding with –OH and amino (–NH2) groups of chitosan. 44 These interactions may limit the availability of the hydrogen groups to form hydrophilic bonding with water leading to a decrease in solubility.
Swelling degree
The swelling degree of chitosan films varied according to the type of extract and the composition used, as shown in Table 1. A higher swelling degree can be seen in CP4:1 composite film than that of the CP2:1 film, but as the concentration of the extract is increased the swelling degree tends to be decreased. In PBS (pH 7.4), all Ch/PEG/TGLE composite films swelled considerably at an initial period till it reaches maximum retention capacity. When the films absorbed water and began to swell, –NH2 group of chitosan was protonated simultaneously, followed by penetration of the phosphate anions in the medium into the swollen films to cross-link at –NH4+ groups of chitosan molecules. 45 The hydrophilicity of Ch/PEG/TGLE composite films can be attributed to the immobilization of PEG chains on the material surfaces. PEG has hydrophilic polymer chains which improve the swelling property when immobilized on the film surface due to the presence of the terminal –OH groups in it.
Mechanical properties
Tensile strength, elongation at break and Young’s modulus value of the prepared film samples.
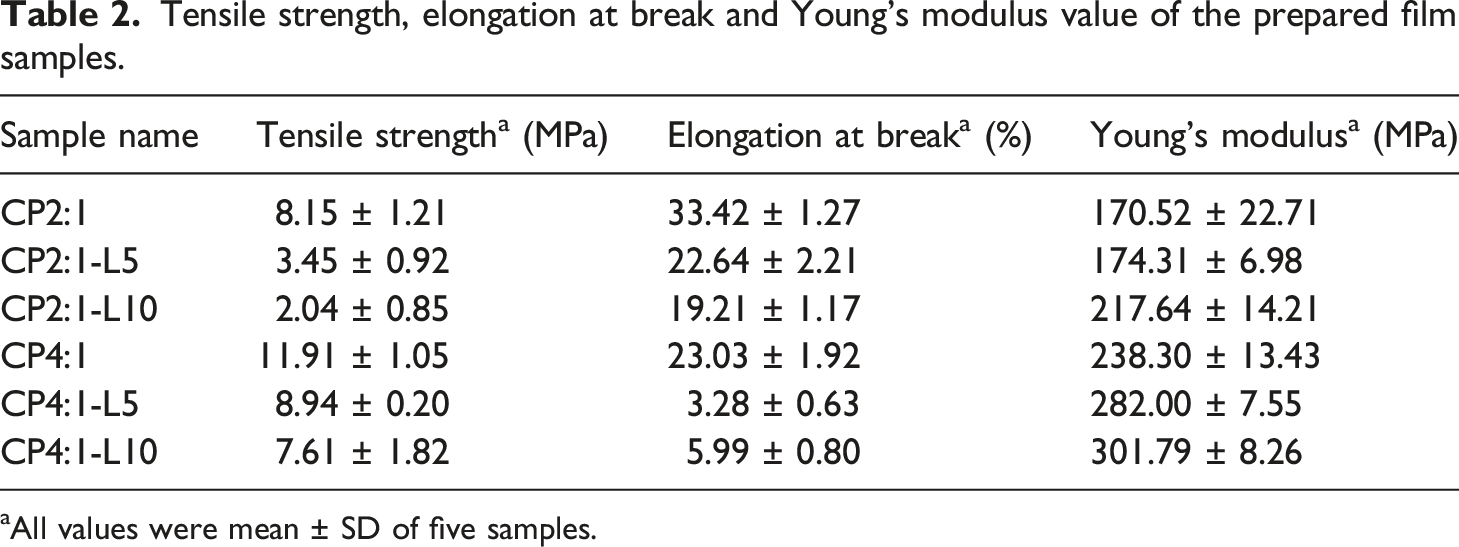
aAll values were mean ± SD of five samples.
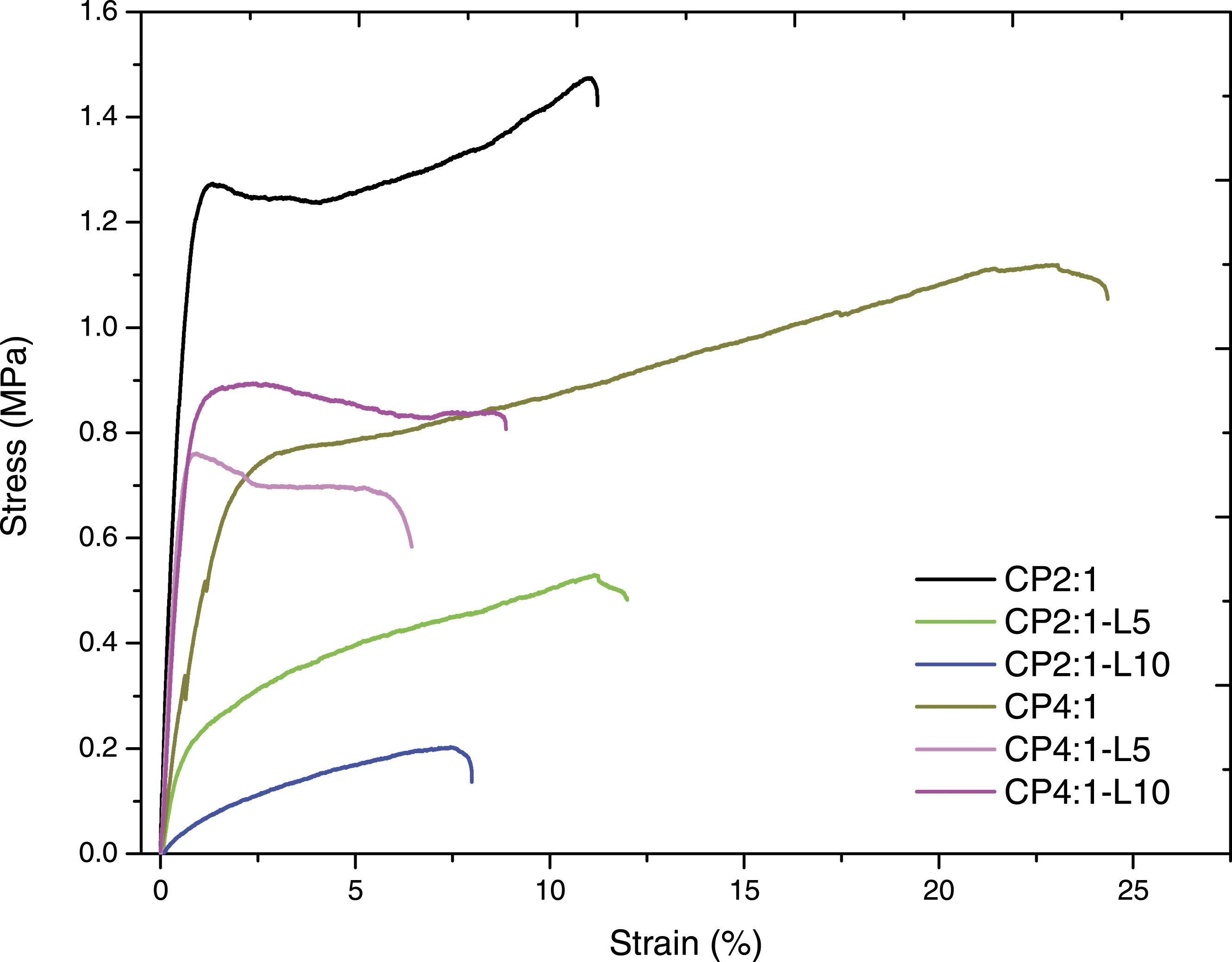
Tensile stress-strain curves of the prepared film samples.
The TS of CP2:1 control film is found to be 8.15 ± 1.21 MPa with 33.42 ± 1.27% elongation at break and that of CP4:1 film is 11.91 ± 1.05 MPa with 23.03 ± 1.92% elongation at break. The TS and EB of Ch-PEG composite films decreased when leaf extract concentrations were increased from 5% to 10%. The decreasing trend in TS and EB for both the CP2:1 and CP4:1 variant films may also be attributed to the crystallite formation or heterogeneity between PEG components at high concentration and chitosan polymer chain, which leads to hinder the molecular chain mobility and polymer aggregation in chitosan matrix producing brittle film structure with reduced film flexibility and TS. 47
The increase in Young’s modulus indicates the stiffness of the material. In that context, the higher value of Young’s modulus for CP4:1-L10 film was observed comparatively stiffer than the other films. Figure 2 shows that CP2:1 and CP4:1 film exhibited a large EB. So, the CP4:1-L10 film can be suggested as the stiffest among the other considered films. These results exhibited that incorporation of both PEG and leaf extract decreased the TS and elasticity of the composite films.
UV-Visible light transmission of the films
Percentage transmittance of Ch/PEG based bio-composite films.
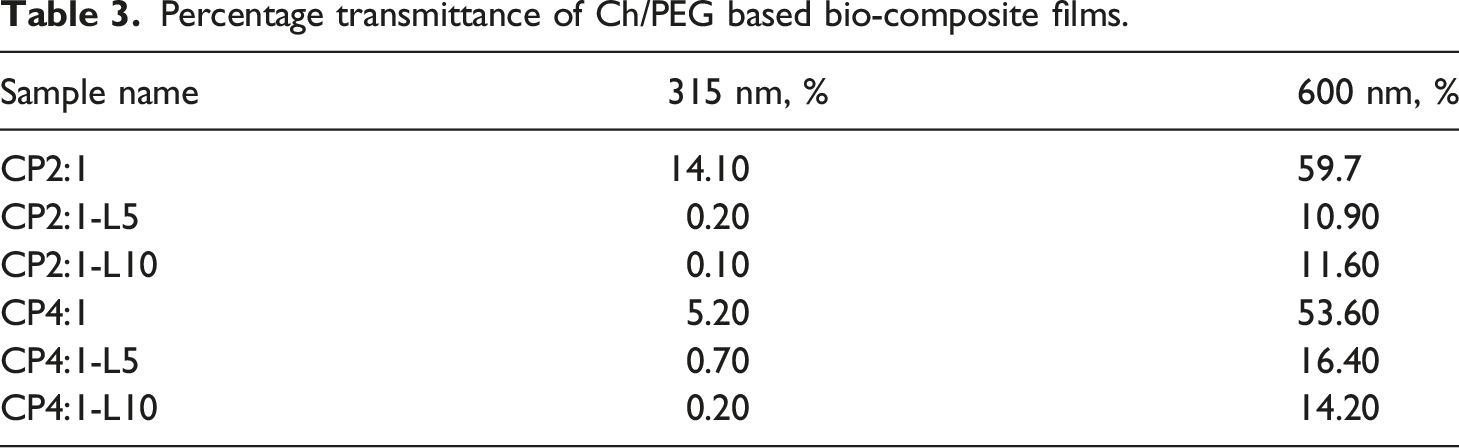
Visible and UV light plays a significant role in the oxidation process of various chemical reactions which can accelerate the deterioration of food surfaces. Hence, the UV light blocking capacity is considered a desirable characteristic for the packaging materials since it can extend the shelf life of products. Thus, the Ch/PEG composite films incorporated with TGLE can be a promising material for packaging industries due to the higher blocking capacity than all the films at both 315 nm and 600 nm range i.e. in the UV and visible range.
Fourier transform infrared spectroscopy
Figure 3 displays the FTIR spectra of all the prepared bio-composite films. The strong and broad transmittance band centred around 3475–3500 cm−1 is due to the axial O–H and N–H stretching.
50
The peak at 2866 cm−1 can be attributed to C–H symmetric stretching. The structural region between 1700–1600 cm−1 (Amide I band) is mainly due to the C=O stretching vibrations of the peptide linkage present in acetamido groups (Amide I) of chitosan.51,52 The peak at 1650 cm−1 suggests the stretching vibration of the C=O group due to the characteristic of the amides (Amide I) and other compounds containing the same in the film.
53
The band at around 1385 cm−1 indicates that a strong interaction occurred between chitosan and PEG components as both chitosan and PEG possess abundant OH and C-O groups, which could form hydrogen bonding and improve the compatibility between them.
50
The band at 1100 cm−1 is due to the vibrations of glycosidic bonds, C–O and C–O–C of the polymeric chain.
54
FTIR spectra of Ch/PEG based composite films incorporated with leaf extract.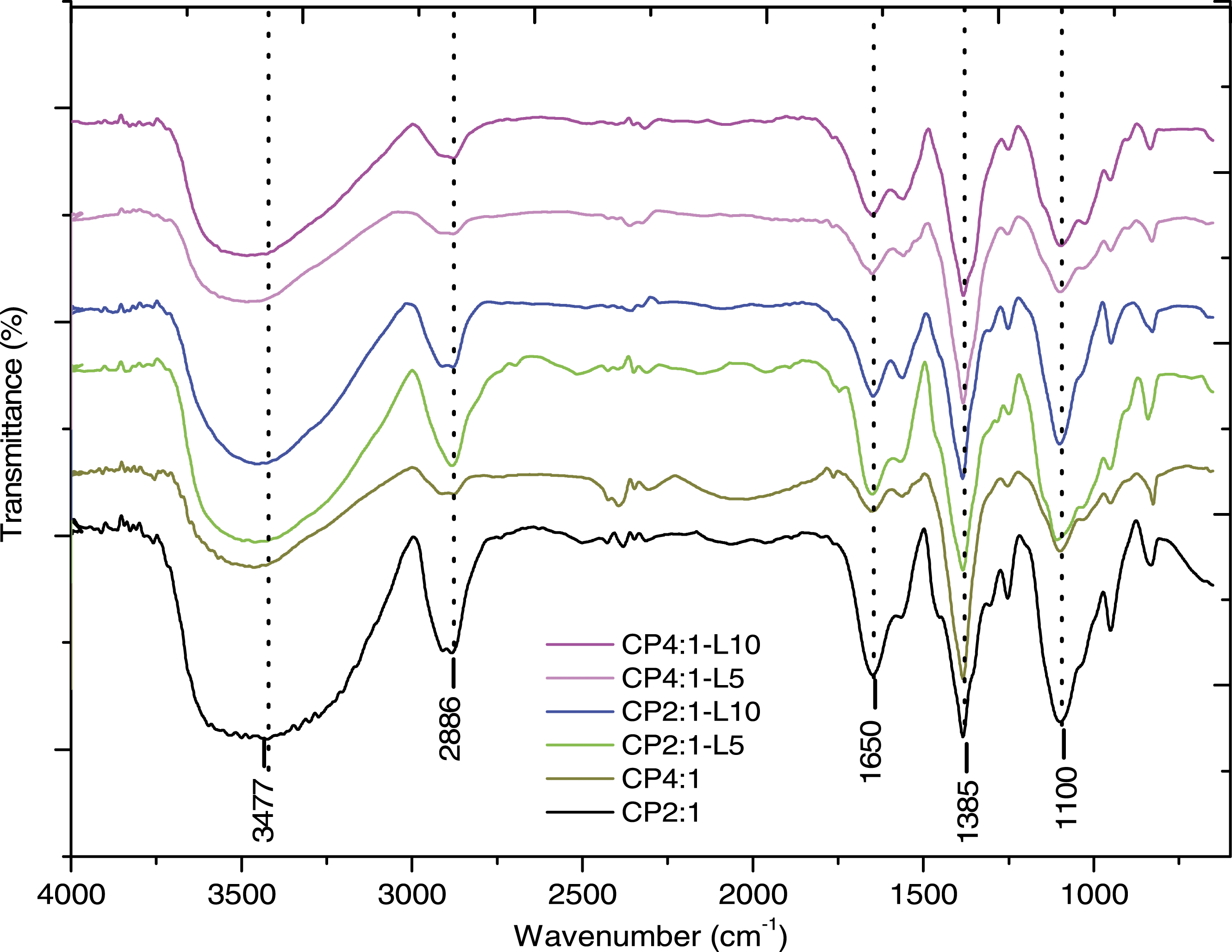
2,2-diphenyl-1-picrylhydrazyl free radical scavenging activity
The antioxidant activity of the films has been expressed as a percentage of free radical-scavenging activity (DPPH method) based on the reduction in the concentration of the DPPH radical in the presence of an antioxidant molecule that captures the odd electron becoming paired with hydrogen. As a result, colourfastness of the solution occurs and the degree of this discolouration, corresponding to a decrease or loss of absorbance, indicates the scavenging efficiency of the tested samples. Figure 4 shows the DPPH radical-scavenging capacity of Ch/PEG composite films. The Ch/PEG control films demonstrated significantly strong scavenging activity alone (74.32 and 73.49%) but the addition of an adequate concentration of TGLE increased it to 87.52%. The results revealed that CP2:1-L5 and, to a greater extent, CP4:1-L5 contain more phenolics capable of quenching free radicals to give rise to more stable products. This might be due to the effect of higher intermolecular interaction, mainly by weak interactions such as hydrogen bonding, between phenolic compounds of TGLE, PEG and chitosan. Also, the antioxidant mechanisms of the natural antioxidants may be attributed to their hydrogen donating ability and their effectiveness as scaven=gers of hydrogen peroxide, superoxide and free radicals.
38
DPPH scavenging activity of Ch/PEG based bio-composites.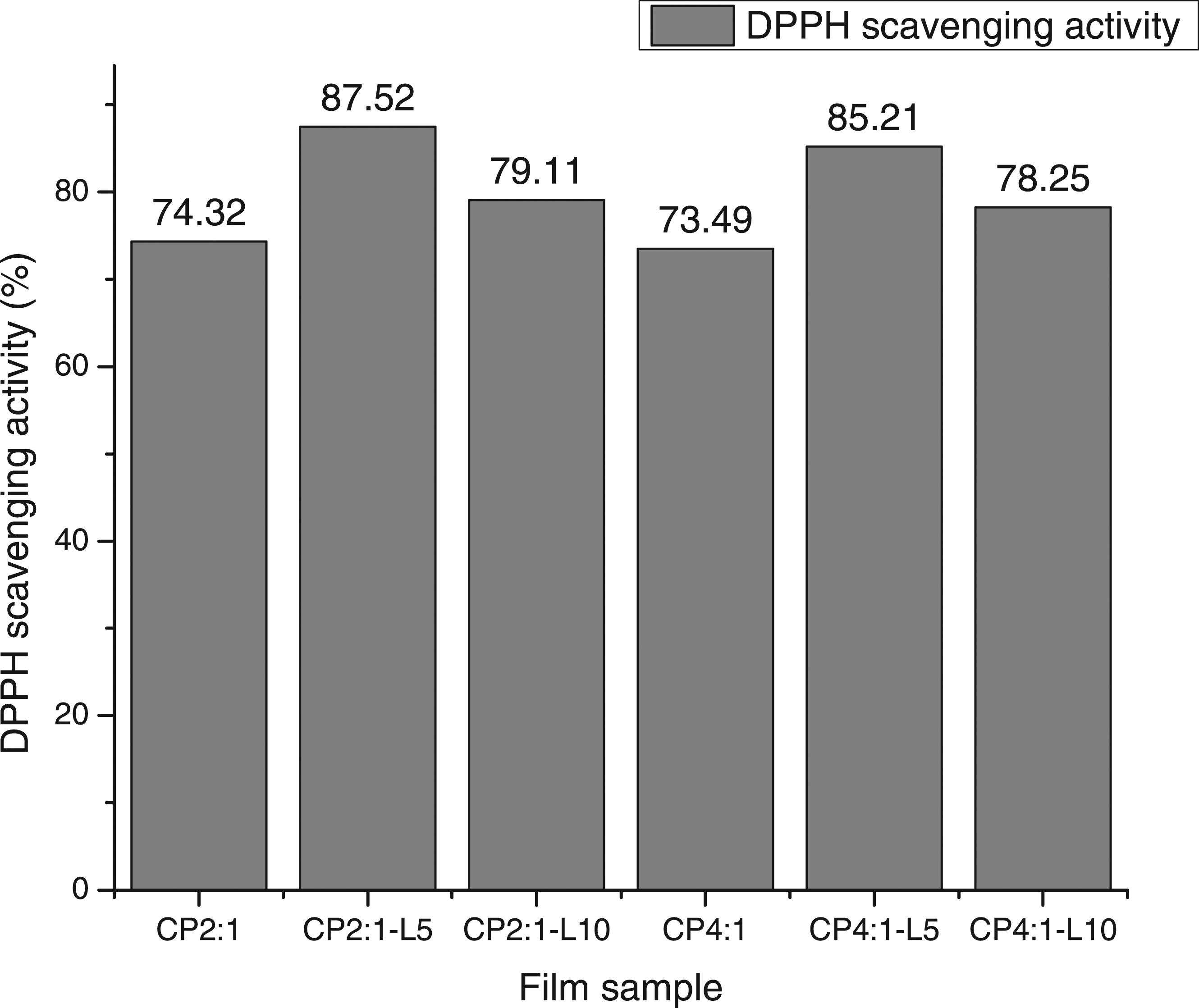
Conclusion
The results show that chitosan can be easily combined with different support materials, but combining chitosan with natural extracts can be a great alternative due to its abundance in nature and favourable properties for packaging applications. Both PEG and TGLE led to a considerable change in the physicochemical properties of the bio-composite films, but this addition showed a certain decrease in the mechanical properties of the films. The water content, solubility and swelling of the films have reduced to a great extent with the incorporation of the leaf extract and is considered to be a suitable property for packaging films to increase the shelf life of food products. The incorporation of the leaf extract also played a major role in increasing the antioxidant activity of the films. All films showed good UV-visible light-blocking property with CP2:1-L5 composition resulting in the maximum. Altogether, the Ch/PEG film properties were affected favourably due to the incorporation of the leaf extract and indicate their potential commercial application as an active food packaging material.
Footnotes
Acknowledgements
The authors acknowledge Assam Science and Technology University (ASTU), Guwahati, India; Central Instruments Facility (CIF), IIT Guwahati, India and Department of Chemistry, B. Borooah College, Guwahati, India for the support to carry out the analytical characterizations.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Assam Science and Technology University (ASTU) under the Collaborative Research Project (CRS/2019/CRSAEC36) of the TEQIP-III scheme.
