Abstract
The aim of the present study was to investigate and compare the quantity of residual monomers leached from the bulk-fill composites with different compositions polymerized at varying layer thickness. Three bulk-fill (X-tra-fil, Beautifil Bulk Restorative, Fill-Up) and a nanohybrid composite (Filtek Z550) were used for the study. The composite resin samples were prepared with a stainless steel mold. For each composite, two groups were constructed. The samples in the first group were prepared using the 2 + 2 mm layering technique. In the second group, the composite samples were applied as a 4 mm-thick one layer and polymerized. Then, each composite samples were kept in a 75% ethanol solution and residual monomers released from composite resins were analyzed with an HPLC device after 24hour and 1 month. The data were analyzed using Kruskal-Wallis and Mann-Whitney U tests. Except the Fill-Up, all of residual monomer elution from the bulk-fill composites was significantly affected by the layer thickness (p < 0.05). The greatest monomer release was detected at 1 month after polymerization as a single 4 mm layer for Beautifil Bulk Restorative. Fill-Up composite showed similar residual monomer release in polymerization at different layer thicknesses compared to other composite resins. In the 2 + 2 mm layering technique, the least monomer elution was detected in the Filtek Z550 composite group. While Bis-GMA was the most released monomer in X-tra fil composite, UDMA was the most released monomer in all other composite resins. During polymerization of the bulk-fill composite, the layer thickness of the composite applied may affect the amount of residual monomers released from the composite resins. Conventional composites may release less monomer than bulk-fill composites when used with layering.
Introduction
Light-cured composite resins are used increasingly in today’s dental practice due to its superior aesthetics, easy application and high bonding capacity to dental tissues. 1 The safe and long-lasting use of these materials is directly related to their chemical and physical properties as well as the dentist’s experience and application technique. 2 The primary disadvantage of the composite resins is insufficient polymerization. In clinical procedures, not all of the dimethacrylate monomers contained in the composite are turned into a polymer, and considerable quantity of residual monomer may release from resins. 3 Different types of monomers that are part of the composite formulations are responsible for various properties of the composite such as viscosity, reactivity and polymerization shrinkage. 4 Therefore, elution of these monomers or formation of degradation products may affect the biocompatibility of the composites. 5
There are many toxicity and biocompatibility studies which demonstrated estrogenic, mutagenic, teratogenic and genotoxic effects of monomers, comonomers and additives released from the composite materials.6,7 The monomers most commonly leaching from the composite and adhesive systems include urethane dimethacrylate (UDMA), bisphenol A glycidyl dimethacrylate (Bis-GMA), triethylene glycol dimethacrylate (TEGDMA), ethoxylated bisphenol A dimethacrylate (Bis-EMA) and 2-hydroxyethyl methacrylate (HEMA). 8 One study concluded that released residual monomers such as Bis-GMA, TEGDMA and HEMA are metabolically turned into epoxy compounds which are both mutagenic and carcinogenic in hepatic microsomes. 9 The amount of monomer elution from the composite resins depends on factors related to the material itself including chemical structure and size of the monomers and filler particles, filler ratio, and concentration and type of photoinitiator and external factors such as polymerization mode and conditions and increment thickness.10-13
For the restoration of deep cavities, application of conventional composite resins in several layers is necessary during placement and light curing. 14 Oblique or horizontal placement of traditional composite resins in 2 mm layers reduce polymerization shrinkage stress by increasing the polymerization of the composite. 15 In recent years, the bulk-fill composites were introduced to the market which enable placement of the resin in layers of 4 mm or more as opposed to placing conventional composites in 2 mm increments. 16 The application of bulk-fill composites in a single layer of 4 mm reduces the chair-time spent by the dentist and reduces the risk of contamination and air entrapment between the layers formed in the layering technique.17,18 Greater polymerization depth achieved with these composites has been attributed to their improved formulation through incorporation of more reactive photoinitiators, photosensitive materials and different types of fillers such as prepolymer particles and fiberglass rod segments as well as higher translucency of the bulk-fill composites.19,20
Bulk-fill composite resins can be classified in two different ways according to their viscosity as flowable and high-viscosity bulk-fill composite. 21 While the entire cavity can be filled with high-viscosity bulk-fill composite resins, flowable bulk-fill composite resins are used as base material and must be covered with a conventional composite resin. 22 Bulk-fill composite resins posses less post-gel and polymerization shrinkage property than conventional composites.23,24 However, there are conflicting studies regarding the layer thickness required for adequate polymerization of bulk-fill composites. While some studies report that 4 mm is an acceptable thickness for sufficient polymerization of bulk-fill composites, other studies claim that bulk-fill composites are sufficiently polymerized at 2–3 mm thickness.25-28 Although the bulk-fill composites are considered as being superior to conventional composites in terms of their certain mechanical features, the lower degree of conversion of the bulk-fill composite resins still represents a significant challenge for such materials.29-31
Recently, there have been rapid developments in the bulk-fill composite resins as a result of increased use of these materials. Currently, dual cure bulk-fill composites which provide better polymerization and giomer-based bulk-fill composites releasing fluoride ions are used in the clinical practice. Although there are studies that examined the effect of the layer thickness on the elution of residual monomers these materials have been used in a layer thickness of only 2 mm and 4 mm.15,32,33 This might also affect overall monomer elution as a result of reduced material volume when 2 mm-thick composite is used in comparison to 4 mm. Unlike former studies, the composites tested in the study were applied as 2 + 2 mm layers and a 4 mm single layer and also bulk-fill composites with different characteristics (e.g. dual cure, giomer-based) were used. The purpose of the present study was to investigate the effect of application of the composite in two different layer thickness (2 + 2 mm and 4 mm) on the quantity of residual monomers leached from the bulk-fill composites with different compositions in comparison to a conventional composite resin. The null hypothesis of the study was that the application of the composite at varying layer thickness would not affect the amount of residual monomers released from tested bulk-fill composites.
Materials and methods
A bulk-fill composite [X-tra Fill (VOCO, Cuxhaven, Germany)], a giomer-based bulk-fill composite [Beautifil Bulk Restorative (Shofu Dental Corporation, San Marcos CA, USA)], a dual cure bulk-fill composite [Fill Up (Coltène/Whaledent AG, Altstätten, Switzerland)] and a nanohybrid composite [Filtek Z550 (3 M ESPE, St. Paul, MN, USA)] were used for the study. Detailed information on the study composites including their composition and manufacturers is provided in Table 1. A second generation LED (Light Emitting Diode) light curing device [Elipar DeepCure–(3 M ESPE, Seefeld, Germany)] with a light intensity of 1470 mW/cm2 and a wavelength of 430–480 nm was used for polymerization of the samples.
Composite resins used in this study and their composition.

PEGDMA: Polyethylene glycol dimethacrylate; Bis-MPEPP: 2,2-bis(4-methacryloxypolyethoxyphenyl) propane; S-PRG: Surface pre-reacted glass-ionomer; TMPTMA: trimethylol propane trimethacrylate.
Sample preparation
A stainless steel mold with grooves of 5 mm diameter and 4 mm thickness was used in the preparation of composite samples. The composite samples were divided into two groups with six samples (n = 6) each according to the thickness of the composite layer. The samples in the first group were prepared using the 2 + 2 mm layering technique and each layer was polymerized with the LED light curing device for 20 seconds. The composite samples in the second group were applied as a 4 mm-thick single layer and polymerized with the LED light curing device for 20 seconds. The intensity of the curing light was checked using a radiometer (Peng Lim Enterprise Co., Ltd., Taiwan) at periodic intervals. Subsequently, a total of 48 samples prepared like that were removed from the mold and each sample was immediately placed to amber colored vials containing 1.5 mL of 75% ethanol solution for extraction. The ratio of samples to solution in the vials was approximately 1:20 and the samples was immersed in solution.
HPLC analysis
At the end of 24 hours and 1 month, 1 mL ethanol solution was obtained from the samples kept at 37C and analyzed by HPLC device. An HPLC device (UFLC-XR, Shimadzu, Japan) and C18 reversed-phase HPLC columns (250 × 4,6 mm CS-2546-C185, Higgins, USA) were used for the residual monomer analysis. In the calibration of the HPLC device, firstly, standard solutions of 0.1, 1, 10, 100, 500, 1000 ppm were prepared with pure monomers [HEMA, BİS-GMA, TEGDMA and UDMA (Sigma Aldrich, St Louis, MO, USA)], which are used as standards and injected into the HPLC system. Thus, retention times, linear calibration graphs and peak areas of each monomer were determined. Table 2 shows the data for the aforementioned monomers. A mixture of 80% acetonitrile (MERCK, Darmstadt, Germany) and 20% water was used as the mobile phase in the analysis.
Linearity results and chromatogram values of monomers.

RT: retention time; R2: correlation coefficient; x: concentration; y: peak area.
After calibration, a total of 48 solutions taken from each sample were kept in the refrigerator at +4 degrees until analysis. 1 mL of solutions were taken from the solutions with the help of eppendorf pipettes and injected into the HPLC device. The detector was set at 254 nm. Since the longest retention time was 5 minutes, chromatograms were obtained by performing each running process for 7 minutes. In the chromatograms obtained, measurements were performed on the peaks predetermined for each monomer. In this study, the amount of each monomer released was determined in ppm using the measurement of peak areas. Some sections of the study’s stages are shown in Figure 1.

Systematic view of study phases. (a) sample preparation, (b) light polymerization of samples, (c) composite resin samples, (d) vials containing ethanol solution for extraction, (e) taking sample solution from the solutions with the help of eppendorf pipettes for HPLC analysis, (f) placing sample solutions in the HPLC device, (g) HPLC analysis of solutions, (h) a chromatogram image of FU composite.
Statistical analysis
The SPSS Version 22.0 (IBM, NY, USA) software and non-parametric Kruskal-Wallis test was used for the analysis of study data. Comparisons among the groups were performed using the Mann-Whitney U and Wilcoxon Signed Rank test. The significance level was set at α = 0.05.
Results
The mean (±standard deviation) quantities of HEMA, Bis-GMA, TEGDMA, UDMA (in ppm) eluted from four different composite resins polymerized through varying layer thickness at 1 and 30 days of the study are shown in Tables 3 to 6 and Figure 2. In addition, the correlation between the total quantity of monomers released and layering thickness is shown in Figure 3.
Mean and (standard deviations) concentrations (ppm) of eluted HEMA from composite resins polymerized in different layer thickness.

Mean and (standard deviations) concentrations (ppm) of eluted Bis-GMA from composite resins polymerized in different layer thickness.

* Same uppercase superscript letters indicate statistically homogenous groups.(Mann-Whitney U Test).
Mean and (standard deviations) concentrations (ppm) of eluted TEGDMA from composite resins polymerized in different layer thickness.
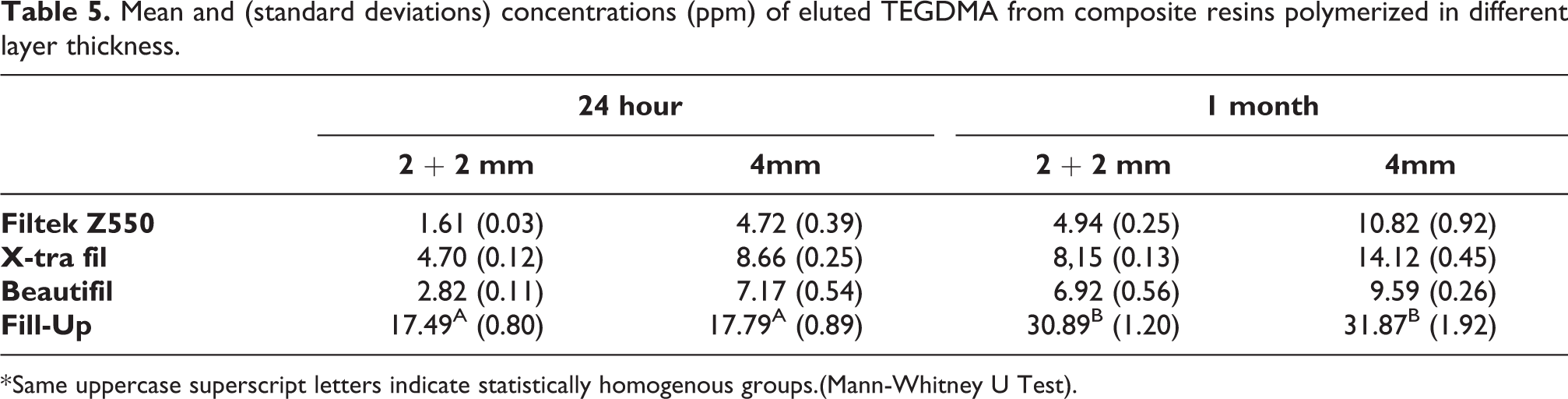
* Same uppercase superscript letters indicate statistically homogenous groups.(Mann-Whitney U Test).
Mean and (standard deviations) concentrations (ppm) of eluted UDMA from composite resins polymerized in different layer thickness.

* Same uppercase superscript letters indicate statistically homogenous groups.(Mann-Whitney U Test).

The amount of HEMA, Bis-GMA, TEGDMA, UDMA released from composite resins used in the study at different layer thickness and time periods.

Total residual monomer quantities released from composites depending on layer thickness at different time periods.
In all groups, HEMA release at 1 and 30 days of the study was found to be affected by the thickness of the polymerized layer. The greatest HEMA monomer elution was observed for the FU composite resin polymerized as a single 4 mm layer and the least HEMA elution was seen in the XF group at 1 and 30 days. HEMA elution was significantly increased at 30 days in all groups. Among all monomers, HEMA was the least eluted monomer in all composite and layer thickness groups.
Except for the 30-day results of the FU group, Bis-GMA elution at 1 and 30 days of the study were affected by the thickness of the polymerized layer. In the FU group, the thickness of the polymerized layer did not affect the amount of Bis-GMA released over 30 days. The greatest Bis-GMA release was detected for the BBR composite resin polymerized as a single 4 mm layer at 1 and 30 days of the study, whereas the lowest Bis-GMA release was seen in the 2 + 2 mm layer group of the Z550 composite resin. Among the groups, the greatest increase in Bis-GMA elution rate was detected for the Z550 composite resin as a result of increased layer thickness. The amount of Bis-GMA leached from the composites showed a significant increase after 30 days in all groups. Bis-GMA was the most released monomer from XF composite resin.
Aside from the FU composite resin, TEGDMA release was affected by the layer thickness in all groups at 1 and 30 days of the study. In the FU group, the thickness of the polymerized layer did not have a significant effect on the amount of TEGDMA eluted. At 1 and 30 days of the study, the highest concentration of TEGDMA monomer was seen for FU composite resin which was polymerized as a single 4 mm layer and the least TEGDMA release was detected in the 2 + 2 mm layer group of the Z550 composite resin. The quantity of TEGDMA released showed a significant increase in all groups after 30 days.
UDMA release was affected by the thickness of the polymerized layer in all groups apart from the FU composite resin at 1 and 30 days of the study. The thickness of the polymerized layer did not have a significant impact on the amount of UDMA released in the FU group. At 1 and 30 days after polymerization, the greatest UDMA monomer leaching was seen in the BBR composite resin polymerized through a single 4 mm layer and the lowest concentration of UDMA monomer was detected in the group in which the Z550 composite resin was applied in 2 + 2 mm layers. However, in the groups where a single 4 mm layer was used, the least UDMA leaching was observed with the XF composite resin. After 30 days, UDMA concentration was significantly increased in all groups. The application of the composite as a single 4 mm layer was associated with a highly significant increase in the UDMA release in the Z550 group. Except for the XF composite resin, UDMA was the most released residual monomer type from other composites at all layer thickness.
Discussion
The current study investigated the impact of the thickness of the layer applied during polymerization on the concentrations of residual monomers leached from the bulk-fill composites. Since the HPLC method is the most widely used technique for assessment of residual monomers released from the composite resins, it was chosen for quantitation of residual monomers in our study. Additionally, in line with the previous studies, 75% ethanol solution was used as the storage medium. 34 This solution is recommended by the United States Food and Drug Administration (FDA) guidelines as a food-simulating liquid. 35 Former studies have reported that the greatest monomer release occurs within the first few days after polymerization, and continues with gradual decline for weeks or months.36,37 Consistently, samples were obtained from the stock solutions at 1 and 30 days after polymerization.
Up to date, the use of the layering technique has been recommended for direct restoration procedures in order to prevent polymerization shrinkage and to control residual monomer elution by achieving adequate polymerization depth. 38 However, recently introduced bulk-fill composites were reported to provide an adequate polymerization depth of 4 mm or greater due to their improved translucency through changes in filler particles.22,24 In a meta-analysis study, it was reported that bulk-fill and conventional composites exhibit clinically similar properties and performance and are preferred by dentists for ease of use. 39 A monomer conversion degree of more than 55% falls within clinically acceptable range but is lower when compared to conventional composite resins. 22 Lower degree of monomer conversion causes greater monomer release from the composite resin. 40 Potential negative effects of residual monomers on the tooth, human body and mechanical properties of the filling may be mitigated by increased polymerization. While the bulk-fill composites were designed for application at a layer thickness of 4 mm or greater, our findings show that the use of the incremental technique had a considerable effect on the concentration of residual monomers in most of the groups. Therefore, our null hypothesis was rejected. In one study, it was reported that restorations made with the incremental technique and conventional composite resulted in less post-operative sensitivity than restorations made with bulk-fill composites and the bulk technique. 39
Z550 is a conventional nanohybrid composite resin which is recommended by its manufacturer to be applied in 2 mm-thick layers. For comparison with bulk-fill composites, Z550 was applied both in two increments (2 + 2 mm) and as a single 4 mm layer. However, it showed less monomer elution at a layer thickness of 4 mm as compared with some of the bulk-fill composites. This might be explained by the differences in its composition, filler particles and monomer structure and ratio. 22 Yet, the application of this composite at a 4 mm thickness instead of layering led to an increased rate of residual monomer leaching in comparison to other composite resins.
In other studies looking at the concentrations of residual monomers leached from composite resins containing Bis-GMA, UDMA and TEGDMA it was observed that UDMA was released more than the other two monomers, similar to our study.13,41 In the present study, although UDMA monomer showed the highest elution among all composites, Bis-GMA released more in the XF group only. Contrary to the study conducted by Van Landuyt et al., 42 the release rate of the monomers investigated in our study were in the following order: UDMA > Bis-GMA > TEGDMA > HEMA. This may be due to the difference in the materials or method used and the higher proportion of UDMA and Bis-GMA in the composite content compared to TEGDMA. The fact that the main monomer in the content of the XF composite is Bis-GMA may also explain the greater elution of this monomer. Although it is not in the structure of the composites that HEMA monomer shows little release as a result of the study. It can be explained by the formation of trace amounts as a degradation product of the UDMA monomer. 43 This finding also supports the finding that the FU composite showed the greatest HEMA elution due to its greater content of UDMA.
The chemistry and the structure of polymer matrix were the most important factors influencing sorption and solubility of dental composites. 44 The use of the UDMA monomer in the composite resins increases the degree of monomer conversion and the depth of polymerization as compared to Bis-GMA. 45 However, flexible monomers such as UDMA and TEGDMA provide a more heterogeneous polymer network which is less resistant to degradation in comparison to rigid monomers such as Bis-GMA. 46 This might explain why the UDMA monomer was released at the highest concentration from all composites in the current study. In the current study, TEGDMA showed the lowest level of elution after HEMA and the greatest TEGDMA release was found in the FU group. This may be explained by the lower content of TEGDMA in the tested composites than other monomers, with the greatest content in the FU composite.
Among the bulk-fill composites tested in the study, the greatest Bis-GMA and UDMA monomer elution was observed in the groups in which giomer-based bulk-fill (BBR) was used. Relative to other composite resins, much higher concentrations of these monomers were detected in the BBR composite at the end of 1 month. In line with our findings, the lowest degree of monomer conversion was reported with BBR in two different studies comparing the degree of conversion of the BBR composite versus other bulk-fill composites.47,48 In addition, fluoride ions released from S-PRG (Surface Pre-Reacted Glass) filler particles might have led to degradation of the material structure over time, affecting monomer elution. A single 4 mm-thick layer was found to have an impact on the residual monomer elution in all composite groups except the FU composite. Some of the residual monomers released from the FU composite was not affected by layer thickness possibly due to the dual cure feature of the FU composite which provided a homogeneous polymerization and the use of a different initiator system in the FU composites.
Although bulk-fill composites are intended for use as a single layer of 4 mm or greater, the amount of residual monomers was significantly affected by layer thickness, supporting the findings of previous studies.15,32 Higher levels of eluted monomers as a result of polymerization of the bulk-fill composites as a single 4 mm-thick layer might cause cytotoxic effects on certain cell types of the organism by diffusing into the pulp through dentin tubules or leaking into the gastrointestinal tract. 49 This in vitro study gives limited information on the amount of residual monomers and other components in the composite resins used that can penetrate through to the dental pulp. Only the analysis of elution of some residual monomers (HEMA, Bis-GMA, TEGDMA, UDMA) will not provide an absolute measurement of the amount of released components. Also, while only ethanol was used as food-simulating agent in the study, composite restorations are exposed to many chemical and mechanical effects in the mouth. These are limitations of this study. Although the bulk-fill composite materials are widely used in current dental practice, further studies are needed to better understand their material formulation and polymerization characteristics.
Conclusion
In the present study, increased layer thickness during polymerization resulted in greater elution of residual monomers from the bulk-fill composites, except for certain monomers contained in the FU composite. FU composite showed a close amount of monomer release at 2 + 2 mm and 4 mm layer thickness polymerization. The greatest monomer release was seen in the BBR composite at 4 mm single layer thickness polymerization. The least monomer elution was observed FZ550 (conventional composite) at 2 + 2 layer thickness polymerization. While HEMA was the least released monomer type in all groups; except for the XF group, the most released monomer type was found to be UDMA. The amount of residual monomer released at the end of 1 month has significantly increased compared to the amount of residual monomer released in 24 hours. Application of the bulk-fill composites as a single layer and the type of the composite were found to affect the amount of residual monomers released from the composite resins.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
