Abstract
A novel biocomposite with poly(methyl methacrylate) as resin containing
Introduction
Dental composites have been used as restorative materials for decades. Many studies have focused on reducing breakages by the reinforcement of the acrylic resin. The reinforcement has been previously undertaken by using fibers such as glass, carbon/graphite, calcium oxide nanoparticles, and ultrahigh-modulus polyethylene fibers.1,2 However, only few research works have expounded the antibacterial dental resin-based materials. These materials can efficiently reduce oral biofilm formation and prevent bacterial micro leakage.3,4 In fact, in recent studies, novel polymers containing quaternary ammonium monomer dimethylaminododecyl methacrylate have been synthesized with antibacterial activities.
5
Furthermore, in order to improve the biological activity and antibacterial properties of polyetheretherketone (PK) as bone implants, Tang et al. fabricated nano zinc-magnesium silicate/PK antibacterial biocomposites (nZPC).
6
Furthermore, Tahriri et al. report that the addition of graphene and its derivatives with biocompatible polymers led to obtain an antibacterial biocomposite.
7
In addition, Varoni et al. have investigated the role of the bioactive plant in enhancing the bioactive performance of dental biomaterials.
8
In some works, some natural products, originating from medicinal and food plants, have been reported to have a beneficial role against periodontal disease and in promoting periodontal healing, that is,
In this work, the medical plant
To the best of our knowledge, it is for the first time that the effects of
Thus, given these beneficial therapeutic properties of
Materials and methods
Materials and process
A denture base material that was commercially available, self-cure acrylic resin (Nic Tone; MDC Dental, Gardena, CA, USA), was supplied by MDC Dental (Manufacturera Dental Continental S.A. de C.V.) in the form of powder and liquid. The powder contains approximately 97% of PMMA polymer, while the liquid was MMA mixed with ether glycol as cross-linking agent. The acrylic resin (Nic Tone (R)) Basic Vita Shades 4 × 1 was used as polymer/monomer ratio of 2:1.
The raw
Physical and chemical characteristics of
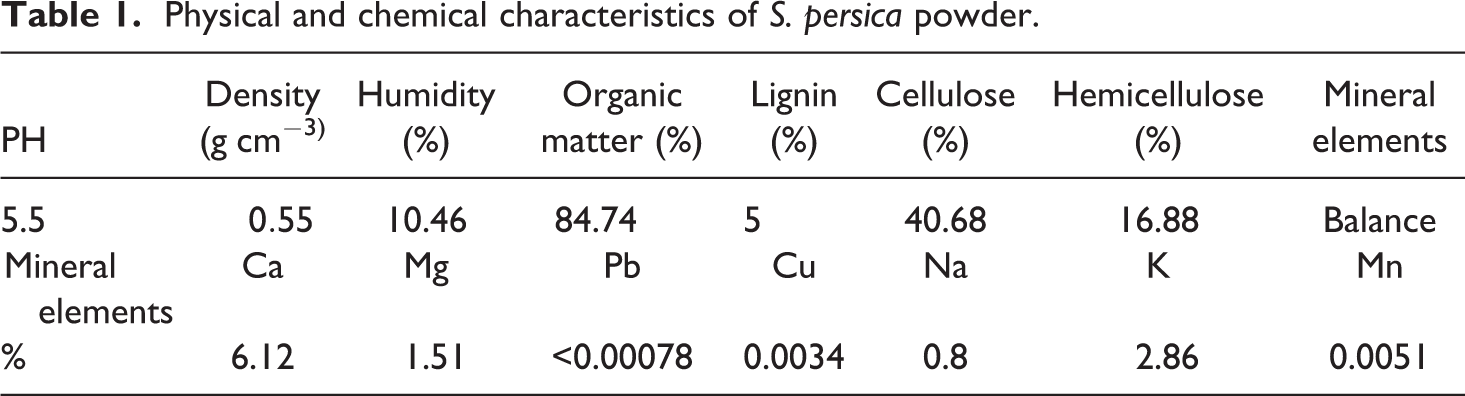
In the present study, only a PMMA composite filled with 30 wt% of untreated
Materials characterization process
The measurement of pH
The pH of
The flame atomic absorption spectroscopy
The flame atomic absorption spectroscopy was also used for the determination of the metal contents in
X-ray diffraction
The X-ray diffractions (XRDs) of samples (composite (30 wt% of
Infrared spectroscopy
The characterization of the chemical structure of the investigated samples (composite (30 wt% of
Scanning electron microscopy
Scanning electronic microscope (SEM) was employed to characterize the morphology of the
Differential scanning calorimetry
Differential scanning calorimetry (DSC) analysis was performed with a DSC 204F1 (DSC 204 F1 Phoenix, NETZSCH, Germany) apparatus equipped with a thermal analysis processing system “Proteus.” A sample of composite (30 wt% of
High-performance liquid chromatography
High-performance liquid chromatography (HPLC) analysis was used to quantity the residual MMA content in the composite (30 wt% of
Antioxidant property analysis
The total antioxidant capacity (TAC) of the extracts of fresh
Composition of artificial saliva.
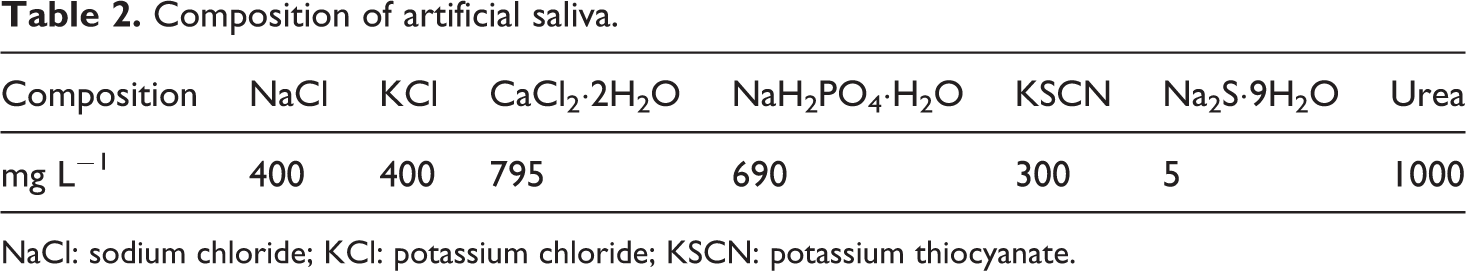
NaCl: sodium chloride; KCl: potassium chloride; KSCN: potassium thiocyanate.
Total polyphenol and flavonoid content
The ethanol extracts (EEs) of
The total polyphenol concentration in the EE was determined according to the Folin–Ciocalteu colorimetric method. 17 For this purpose, 0.5 mL of EE (diluted 2×) was mixed with 2.5 mL of Folin–Ciocalteu reagent (diluted 10×) and 2.0 mL of 7.5% sodium carbonate (sodium carbonate). The absorbance was read at 760 nm after 2-h incubation at room temperature in the dark. Gallic acid (250 mg mL−1) was used as a standard to produce a calibration curve. The average of three readings was used to determine the total polyphenol content, which was expressed as the mg of GAE/g of powder.
The flavonoid content in EEC was determined according to the method described by Meda et al., 17 with minor modifications. For this purpose, 400 µL of EE (diluted 2×) was mixed with 120 µL of NaNO2 (5%). After 5 min, 120 µL of aluminum chloride (10%) was added, and 6 min later, the mixtures were neutralized with 800 µL of 1 M sodium hydroxide solution. The absorbance was immediately read at 510 nm. Quercetin (120 mg mL−1) was used as a standard to produce a calibration curve. The average of three readings was used to determine the flavonoid content, which was expressed as the mg of quercetin equivalents (QE)/g of powder.
Antibacterial activity of elaborated composite
The following oral cavity pathogen strains were selected for antimicrobial activity testing of the composite:
The antibacterial activity of
Statistical analysis
Data are expressed as mean ± standard deviation (
Results and discussions
Materials characterization
The beneficial effects of
X-ray diffraction
The composition of the samples was identified using the XRD. Figure 1(inset d) shows the X-ray diffractograms of PMMA. A characteristic broad amorphous hump was observed around 2
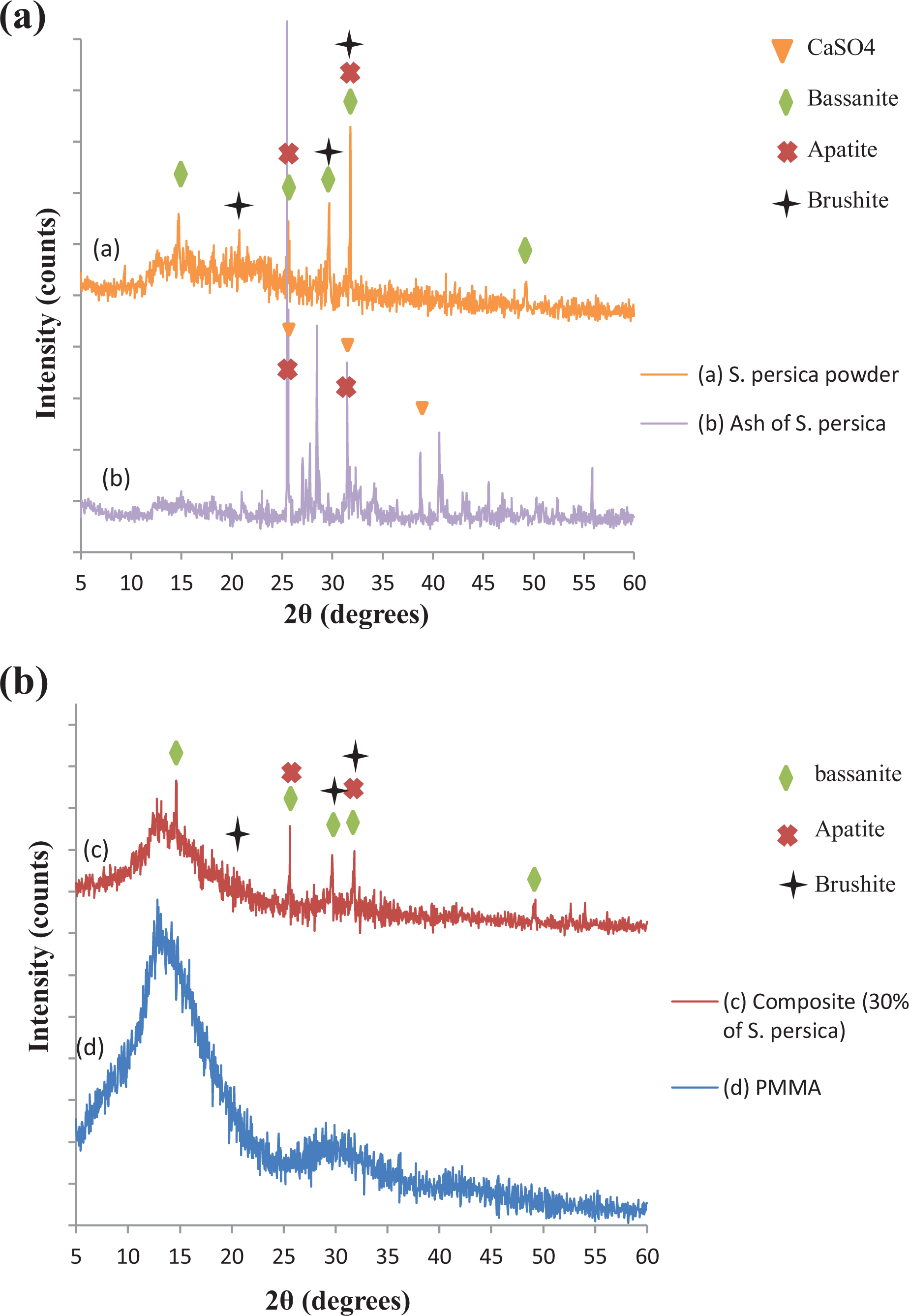
XRD patterns: (a) (inset a)
The powder of
Figure 1(inset a) shows the peaks of the
For the mineral compounds, the scan can prove the presence of apatite, brushite, and bassanite. The characteristic peaks of bassanite (CaSO4·0.5H2O) were 2
Moreover, the XRD of
Considering the above results, the
This finding is in agreement with previous research works. Ahmad and Rajagopal have proved the presence of flavonoids, the organic sulfur compounds, and carbohydrates in
All these compounds, of very different classes, can be present in
Ultimately, the major peaks of the composite (30 wt% of
Infrared spectroscopy
It should be noted, firstly, that FTIR spectra are used not only to indicate the details of functional groups present in materials but also to investigate the incorporation of
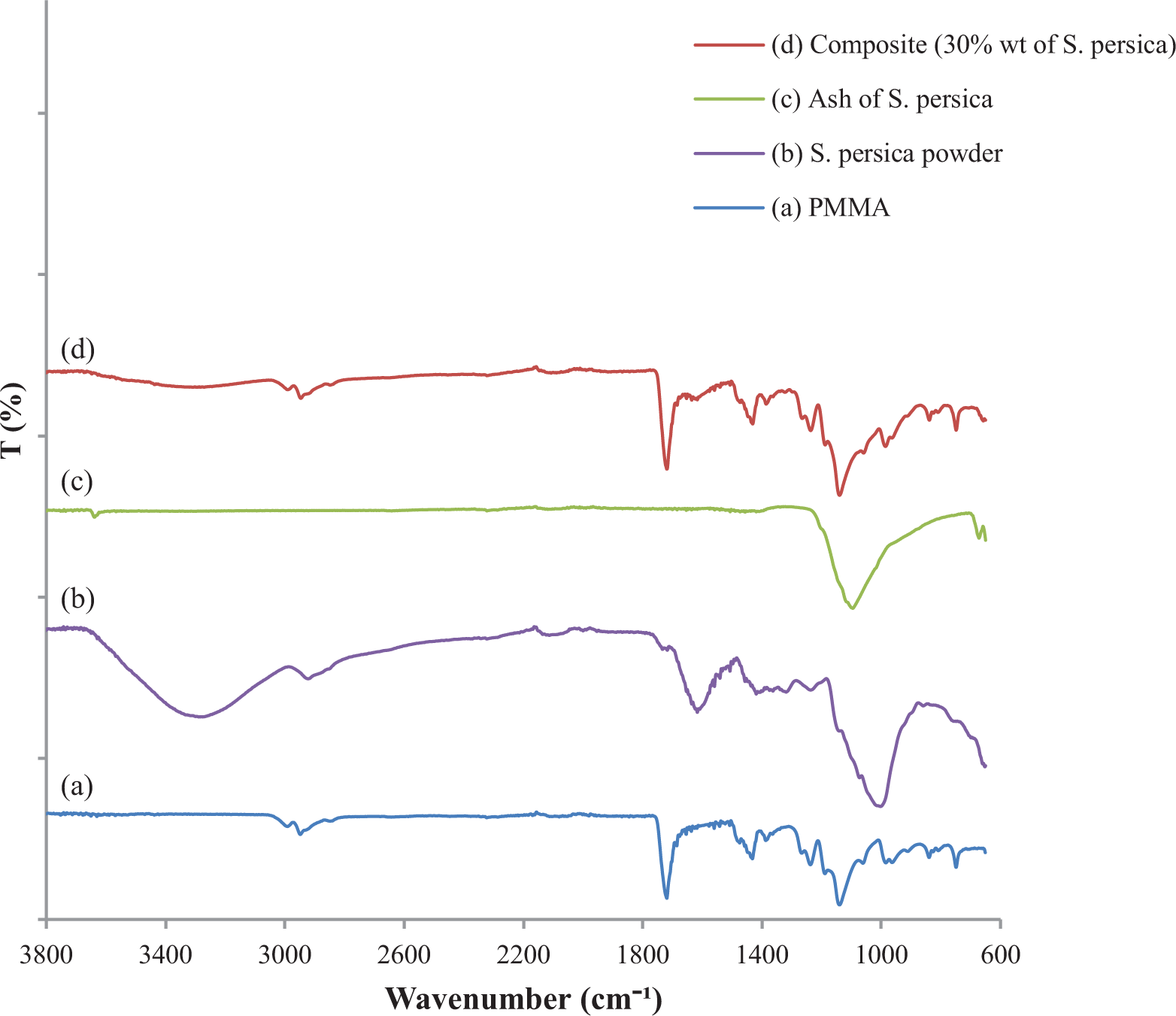
The FTIR spectra of (a) PMMA, (b)
The FTIR spectrum of
The IR spectrum of the ash of
As shown in Figure 2(d), compared with the spectra of PMMA, the spectra of the obtained composite (30 wt% of
Finally, we can conclude from Figure 2 that the spectra of pure PMMA and the composites exhibit some difference, indicating that the inclusion of
Scanning electron microscopy
Figure 3 shows the SEM images of the morphology of

SEM images of the
EDS of the
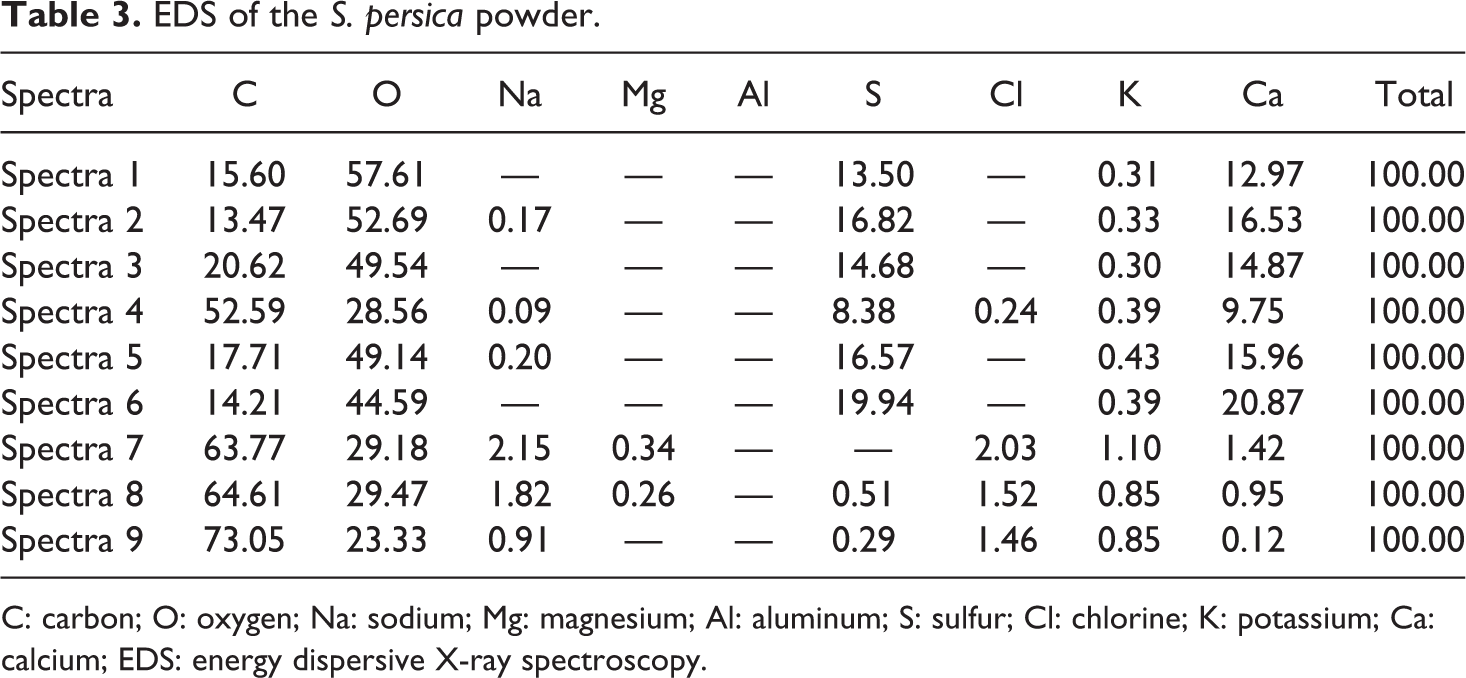
C: carbon; O: oxygen; Na: sodium; Mg: magnesium; Al: aluminum; S: sulfur; Cl: chlorine; K: potassium; Ca: calcium; EDS: energy dispersive X-ray spectroscopy.
Figure 3(b) demonstrates a good adhesion between PMMA and
The contents of residual monomers in the elaborated composite
The reaction of polymerization between the polymer and the monomer is more or less incomplete, leaving part of the free monomers able to diffuse in the oral medium. These residual monomers are regarded as poison when its proportion is more than 0.4% of the finished product. Residual monomers in polymerized denture base polymers cause the oral mucosa inflammatory symptoms and the irritations of the mucous membranes. 35 Therefore, it is necessary to determine the percentage of the residual monomers using DSC and HPLC.
Thermal analysis
Figure 4 shows typical DSC thermograms of the composite. The thermogram obtained on the scan (heating curve) reveals no exothermic peak but only a glass transition pattern (
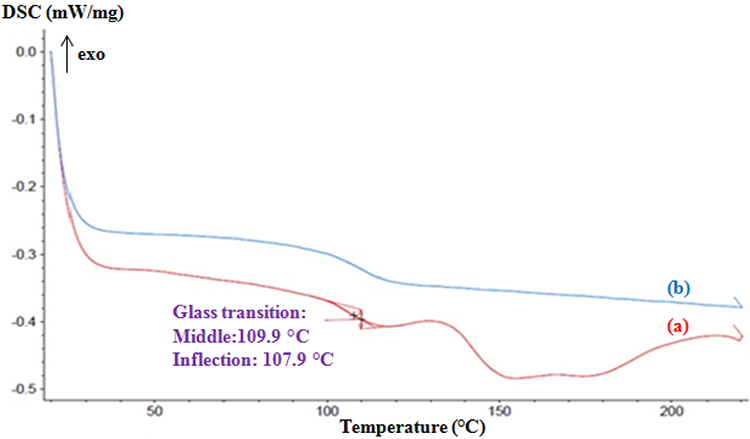
DSC thermograms of the resulting composite (30 wt% of
To confirm this hypothesis, we try to determine the percentage of residual monomers using HPLC.
High-performance liquid chromatography
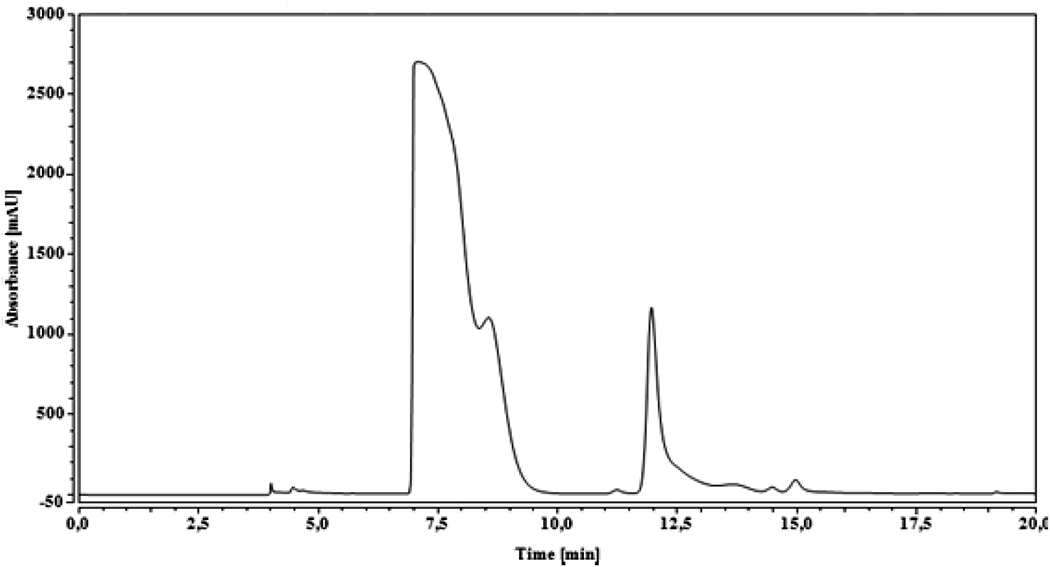
Figure 5 presents the chromatogram of the reference which contains the MMA and a low percentage of glycol ether. Two peaks have been distinguished. As for the first, it is between 7 min and 8 min, which is a very important peak, and the second is between 11.5 min and 12 min, which is a low peak. According to Viljanena et al., 36 the peak of MMA was among 7–8 min. However, the chromatogram obtained on the second scan (Figure 6) shows no significant peak between 7 min and 8 min. Therefore, we can conclude that the composite analyzed in the present study does not present residual monomers. This reveals a positive effect on the biocompatibility of the composite as reported by Viljanena et al. 36 The migration of residual monomers, which are toxic, was avoided in the body and the oral environment.
Chromatogram of the sample “Reference.”
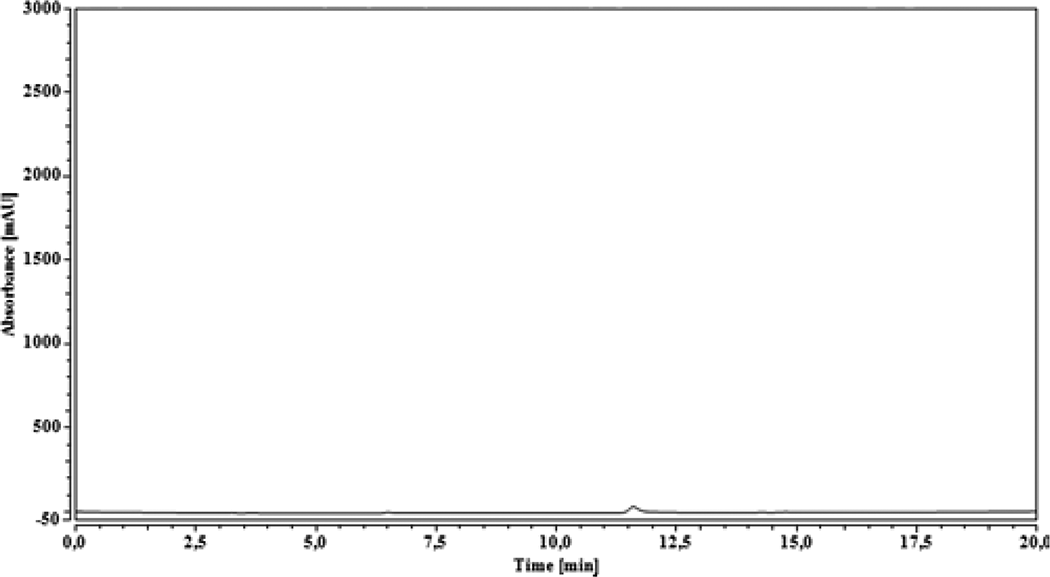
Chromatogram of the resulting composite (30 wt% of
Antioxidant activity of elaborated composite
Dental amalgam, which contains mercury, has an oxidative action on brain neurons. This phenomenon is an etiological factor of Alzheimer’s disease. 37 For that, the development of dental material, which has antioxidant activity, has a positive effect on oral hygiene and helps the body to protect itself against various types of oxidative damage.
Antioxidant property analysis
The purpose of this test is to evaluate the antioxidant activity of
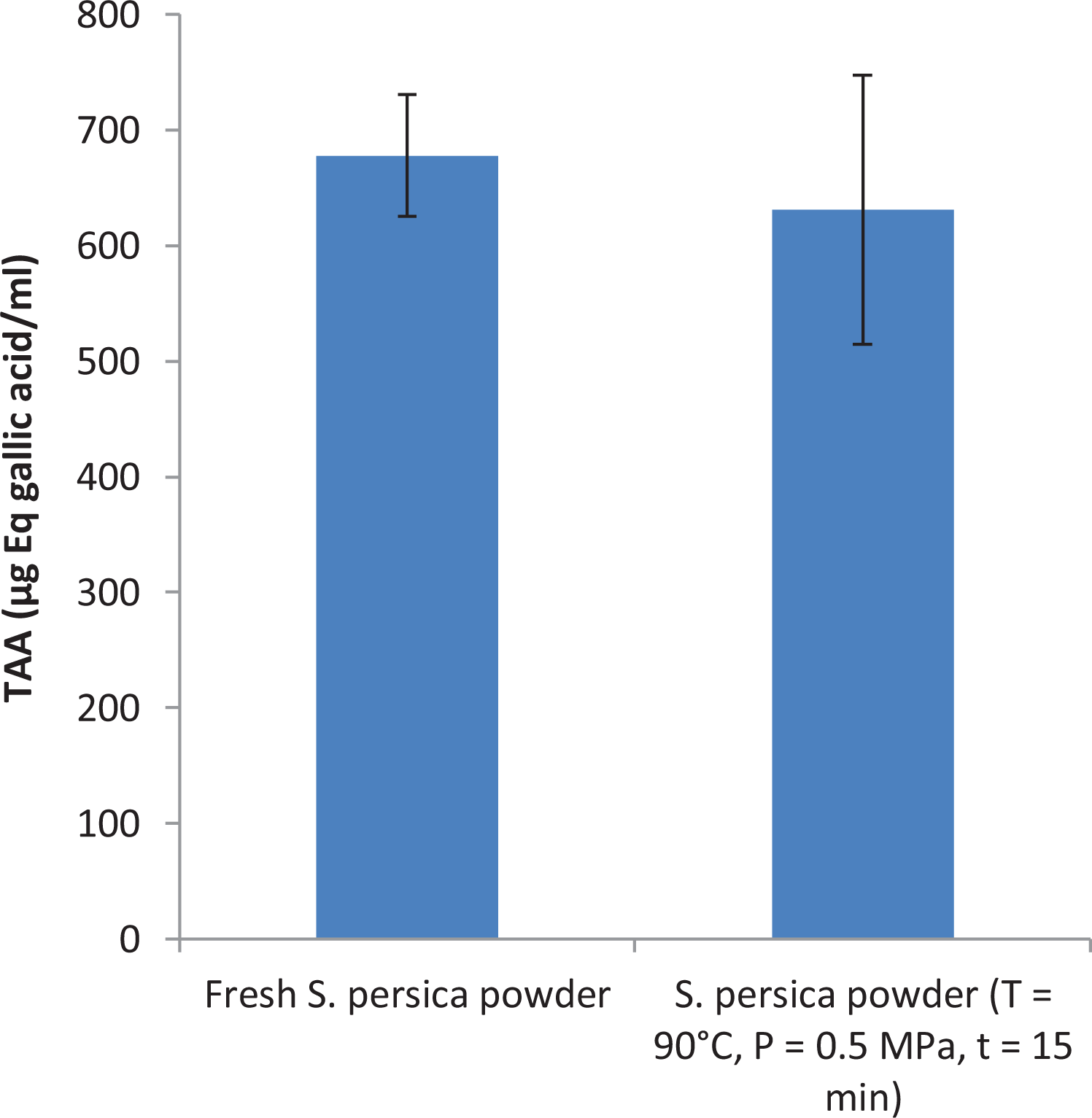
Total antioxidant activity of
Total polyphenol and flavonoid content
The total polyphenol concentrations present in the EES and EEC were 87.79 ± 1.32 mg of GAE/g of
Antibacterial activity of elaborated composite
The development of bacteria on the surface of the dental prosthesis, producing acid layer, accelerates the damage and the failure of dental composite prosthesis. To overcome this problem, we aimed to develop a bioactive material that can limit the prosthesis aggression caused by bacteria. Therefore, we elaborated a biocomposite reinforced with 30% of
Figure 8 shows the antibacterial activity of
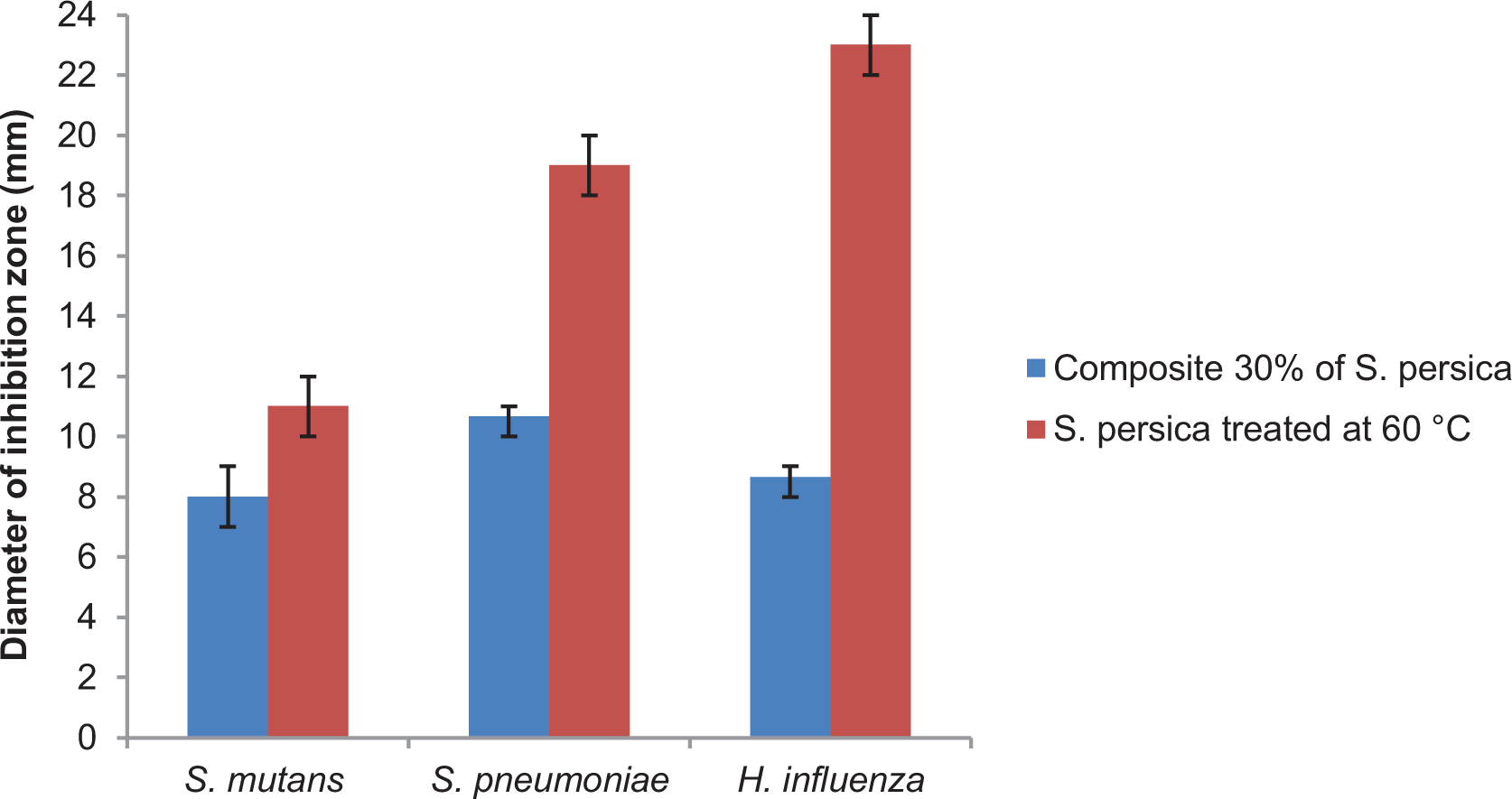
Antibacterial activity of
Conclusion
The work presented in this article focused on the elaboration and characterization of a biocomposite material based on PMMA/
XRD revealed the presence of the hydrated calcium sulfate and the calcium phosphate compounds in The FTIR analysis of SEM images and EDS analysis show the presence of bassanite crystals of biogenic origin in the The obtained composite did not present residual monomers that cause the oral mucosa inflammatory symptoms and the irritations of the mucous membranes. The antioxidant and the antibacterial tests prove the antioxidant and antibacterial activities of
Footnotes
Acknowledgements
The authors would like to express our special thanks of gratitude to Pr. Emna Ammar (National Engineering School in Sfax, University of Sfax) as well as Mme. Wafa Gargouri (Research Group of Agri-Food Processing Engineering, National School of Engineers of Sfax, University of Sfax) for their assistance in laboratory analyses and Mme. Leila Mahfoudhi (Faculty of Sciences, Sfax) for proof-reading our manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
