Abstract
Wearable electronic textiles with high conductivity and excellent antibacterial activity are very desirable to minimize health risks and deteriorated performance of the electronic textiles in application. In this work, conductive fabrics were prepared with chitosan nonwovens with inherent antibacterial activity as the substrate. Silver nanoparticles (AgNPs) were synthesized in situ on the surface of chitosan fabric without any additional reducing agent, and silver nanowires (AgNWs) were adhered to the surface of AgNPs-coated chitosan fabrics by simple dip-coating cycles. The synergy of AgNPs and AgNWs enhances the conductivity of the fabric and the stability of AgNPs on the surface of the fabric. According to the analysis of scanning electron microscope and fourier transform infrared spectroscopy spectra, AgNPs were reduced in situ on the fabric. With synergy of AgNPs and AgNWs, the electrical resistance of the fabric is as low as low as 0.93 Ω/sq and 0.20 Ω/sq after one and four dip-coating cycles of AgNWs respectively. Thermogravimetric analysis and inhibition zone assay showed that combination of AgNPs and AgNWs enhanced the thermal stability and antibacterial activity of chitosan fabrics. The chitosan fabrics with conductivity, thermal stability and antibacterial activity can be used in electronic textiles for different applications.

Introduction
Wearable electronic textiles are emerging as a platform for sensing the changes of strain, touch, and pressure, and converting them into electrical signals. 1 It can also monitor human health by detecting physiological activities, such as heart activity, brain activity, muscle activity. 2 The conductive fabrics can be obtained by the combination of conductive materials and insulated fabrics. They can be used as textile electrodes to detect, collect and transmit the tiny electrical signals. Wearable electronic textiles are body attachable, and there is physical contact between the human body and conductive textile. Therefore, the conductive component should be biocompatible and not irritate the skin in the long-term use. Moreover, antibacterial activity is desirable for wearable electronic textiles because it is easy to breed bacteria when exposed to sweat, which will result in health risks and deteriorated performance of the electronic textiles. 3 Chitosan fabric with inherent antibacterial activity is a good candidate for the substrate of wearable electronic textiles. It is proved that chitosan can lead to the leakage of the intracellular content in the dying process of bacteria. It is realized by the interaction between positive charge of chitosan and the negative charge of cell membrane, which makes the cell membrane malfunction and altered permeability. 4 Silver exhibits the highest electrical and thermal conductivity among all metals, 5 shows antibacterial and antiallergic properties as well. Therefore, it is promising to fabricate electronic textiles with antibacterial activity by combining chitosan fabrics and silver.
Chitosan nonwoven fabrics have attracted enormous attention in wound dressing6–8 for the antibacterial activity, a large number of amine groups in the molecules are active sites to absorb metal ions strongly9,10 due to its nitrogen atoms holding free electron doublets. 11 Silver nanoparticles (AgNPs) can be in situ synthesized on the fiber surface of chitosan nonwoven fabrics by UV-assisted method 12 and electroless plating. 13 The strong adhesion between chitosan and nanosilver can not only increase the durability of the obtained conductive fabrics, but also prevent the firmly anchored AgNPs entry into the human body. The wearable electrodes for electrocardiogram acquisition were fabricated by silver-plated chitosan fabrics in Qin’s work and the electrical resistance can be as low as 0.0332 Ω/sq. 13 However, the fibers were completely coated with AgNPs to form a silver layer in order to obtain high conductivity, it cannot fully make use of the antibacterial activity of chitosan fabrics.
In recent years, silver nanowires (AgNWs) with high aspect ratio are very attracting in application of wearable electronics14–16 and electromagnetic shielding.16–18 A variety of wearable electronics based on AgNWs have been reported including strain sensors,19–21 supercapacitor, 22 and heater. 23 Conductive network is feasible to be formed by interconnection of AgNWs with low content, and the sheet resistance of conductive devices decreased when silver flakes or nanoparticles were replaced by AgNWs. 24 AgNWs can be loaded on the fabrics by a simple dip-coating method, the fabric materials, 25 size of AgNWs 26 and dip-coating times 27 can influence the adsorption of AgNWs on the fabrics and resulting conductivity. The synergy of AgNWs and AgNPs is very promising to improve conductivity, or render conductivity and antibacterial activity simultaneously. AgNPs increase the conductive paths for electrons by filling the gaps between AgNWs, 28 leading to the low resistance and highly sensitive of the wearable strain sensor.29,30 AgNPs could reinforce the weak antibacterial activity that in some cases present the nanowires. 31
In this work, AgNPs were coated on the surface of chitosan fabrics without any additional reductant. Then, AgNWs were coated on the surface of the fabrics through the interfacial interactions between AgNPs and AgNWs. The structure and morphology of the chitosan/Ag nanocomposite fabrics were examined by scanning electron microscope (SEM), X-ray diffraction (XRD) and Fourier transform infrared spectroscopy (FTIR). The electrical conductivity, thermal stability and antibacterial activity of the obtained fabrics were investigated. The nanosilver functionalized chitosan fabrics with synergistic conductivity and antibacterial activity shows good potential application in wearable electronic textiles.
Experimental
Materials
Non-woven chitosan fabrics (100% chitosan, 50 g/m2) were obtained from Hismer Bio-technology Co., Ltd. (Shangdong, China). The fabrics were cleaned via ultrasonication in ethanol and deionized water for 30 min respectively. Silver nitrate (AgNO3, >99.8%) was purchased from Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China). Sodium hydroxide (NaOH) and ethanol were obtained from Tianjin Fuyu Fine Chemical Co., Ltd. (Tianjin, China). All chemical reagents were of analytical grade and used without further purification. AgNWs in ethanol were supplied by Shanghai Macklin Biochemical Co., Ltd. (Shanghai, China). The average diameter and length of the silver nanowires were of 90 nm and 60 mm, respectively.
Preparation of antibacterial and conductive chitosan fabrics
The chitosan fabrics (4 cm × 7 cm) were activated in NaOH solution (20 g/L) at 70°C for 30 min. The fabrics were then rinsed in distilled water to achieve a neutraval pH. The pretreated chitosan fabrics were immersed in 60 mL AgNO3 solution with various concentrations from 0.0005 mol/L to 0.1 mol/L. Silver ions were reduced to AgNPs on the surface of the chitosan fabrics for 120 min in the 80°C water bath. The AgNPs-coated chitosan fabrics were rinsed with distilled water, dried, and then immersed in 50 mL of 0.5 wt.% AgNWs suspension for 3 s as one dip-coating cycle. After drying, the nanosilver functionalized chitosan fabrics were obtained. The process of coating the fibers with AgNWs was optimized by increasing the number of “dip-coating” cycles. The fabric is named AgNPs/AgNWs-coated chitosan fabrics.
Determination of AgNPs content on chitosan fabrics
The AgNPs immobilized on the chitosan fabrics were extracted into solution, and the concentration of AgNPs was measured. Specifically, AgNPs-coated chitosan fabric (0.1 g) was immersed in 20 mL of 15 wt.% nitric acid for 30 min at 80°C to extract silver. Silver concentrations in the nitric acid solution were determined via flame atomic absorption spectroscopy (Atomic Absorption Spectrometer, Z-2000, Hitachi, Japan) equipped with a silver lamp at 328.1 nm wavelength. In addition, the moisture content of the AgNPs-coated chitosan fabric was measured by an infrared moisture meter (MA35, Sartorius, Germany), and the AgNPs content on the surface of chitosan fabrics was calculated by the following equation (1)
Characterization of AgNPs-coated chitosan fabrics
FTIR spectra of chitosan fabrics and AgNPs-coated chitosan fabrics were taken using Fourier transform infrared spectrometer (TENSOR27, Bruker, Germany) at room temperature from 4000 to 500 cm−1.
SEM images were taken with a high-resolution field emission scanning electron microscope (FESEM) (Merlin, ZEISS, Germany). The surfaces of the samples were sputtered with a thin layer of gold prior to each measurement. The SEM measurements were performed at an accelerating voltage of 20 kV. The size of AgNPs was average value of around 50 measured results from SEM images.
The crystalline structure was studied by XRD (Bruker D8 ADVANCE, Bruker, Germany), using Cu Kα radiation at a wavelength of 0.15418 nm, and the diffraction patterns were recorded in the θ range of 10°–90°.
The electrical resistance of the fabric was measured by four-probe square resistance tester (KDB-1, Kunde, Guangzhou, China). The data were processed by averaging 10 measurements per sample.
Thermogravimetric (TG) analysis was carried out on Simultaneous Thermal Analyzer (STA449 F3, NETZSCH, Germany) at a heat rate of 10°C/min from 30°C to 650°C, with a steady nitrogen flow (25 mL/min).
Antimicrobial properties
The antibacterial activities of the prepared fabrics against Escherichia coli (E. coli.) were assessed using the disc diffusion method. 32 First, 100 μL of E. coli (2.5 × 107 CFU mL−1) suspensions were uniformly spread on an agar disc, and circles of the fabric with diameters of 6 mm were put on the surface of agar disc. After the discs were incubated overnight at 37°C, the bactericidal activity was observed by visual inspection of the bacterial lawn and measurement of the size of the growth inhibition zone by calipers across an average diameter. Each sample was tested at least three times to get the average value of the diameter of inhibition zone.
Results and discussion
In situ coating of chitosan fabrics with AgNPs
The fabrication process of chitosan fabrics with synergy of AgNPs and AgNWs is shown in Scheme 1. After immersion in AgNO3 solution, the amino groups on chitosan fabric can adsorb silver ions and reduce the silver ions to AgNPs in situ. A lone pair of electrons in the nitrogen atom of amino group can be donated to the shared bond between the nitrogen atom and metals, therefore, amine groups of chitosan provide active sites for the formation of complexes with metallic ions.33,34 The complexation interaction between amino groups and silver ions facilitates adsorption of silver ions onto chitosan fabrics. Carbohydrates have been introduced as reducing and/or stabilizing agents for the preparation of metal nanoparticles.
35
AgNPs could be prepared in situ and directly incorporated into viscose fibers and medical cotton.36,37 It also has been reported that chitosan is more than just a stabilizing agent, and that silver ions can be reduced to zero-valent silver nanoparticles using chitosan alone, without any additional reducing agent.
38
Due to the ability of chitosan to reduce silver ions,39,40 the adsorbed silver ions are reduced to AgNPs in situ on the surface of chitosan fabric. AgNPs deposited chitosan fabric is dip-coated in AgNWs suspension, and AgNWs can be coated on the surface of chitosan fabric by the interfacial interactions between AgNWs and AgNPs or chitosan fabric. Preparation process of chitosan fabric with synergy of AgNPs and AgNWs.
Reduction of AgNO3 by chitosan was proved by the FTIR spectra of the chitosan fabric and AgNPs-coated chitosan fabric, as shown in Figure 1. The wavenumber and attribution of main peaks in FTIR spectra of chitosan fabric and AgNPs coated chitosan fabric were summarized in Table 1. The typical absorption peak of chitosan is 3400–3200 cm−1, 2850–2990 cm−1, 1155 cm−1, which comes from O-H and N-H, aliphatic C-H and ether bonds, respectively.
41
In addition, the existence of 1645 cm−1 (amide I band), 1595 cm−1 (amide II band) and 1323 cm−1 (amide III band) proved that there was amide bond on chitosan fabric and it was not completely deacetylated.42–44 After coating AgNPs, the FTIR spectrum of the fabric changed significantly. The disappearance of the peaks at 1595 cm−1 and the change of the peaks at 1323 cm−1 indicate that silver is attached to N atoms; the peaks disappeared at 1421 cm−1 and weakened at 1083 cm−1 and 1028 cm−1,
45
indicating the reaction of -CH2OH. Most importantly, a new peak appeared at 1764 cm−1 (-C=O). Based on the above analysis, it is found that the hydroxyl group on chitosan fabric is oxidized to carbonyl by silver ions, while silver ions are reduced to AgNPs and attached to the fabric surface.
38
Fourier transform infrared spectroscopy spectra of chitosan fabric and AgNPs-coated chitosan fabric. Wavenumber and attribution of main peaks in fourier transform infrared spectroscopy spectra of fabric.

The changes in surface morphology of the pristine chitosan fabrics, compared with the AgNPs-coated fabrics, are shown in Figure 2. From Figure 2(a), the pristine chitosan fabric depicted a smooth surface, without any contaminating particles. After incorporation of AgNPs, a large number of intense and uniform AgNPs with size of 50 nm were coated on the chitosan fabric from Figure 2(c). Field emission scanning electron microscope images of pristine chitosan fabric (a), AgNPs-coated chitosan fabric (b and c).
The silver contents in AgNPs-coated chitosan fabrics were measured by nitric acid extraction and atomic absorption spectroscopy. From Figure 3, AgNPs content in the fabrics increased with increasing silver nitrate concentration, especially for chitosan fabrics treated with 0.5–10 mmol/L. Silver contents were reordered to be increased from 144.85 mg/g at 10 mmol/L AgNO3 to 175.38 mg/g at 100 mmol/L AgNO3. However, the loading efficiency decreased with increasing silver nitrate concentration. The possible reason was that with the increase of the concentration of silver nitrate, the more silver particles produced by the reduction of chitosan, until the surface of the chitosan fiber was covered by the silver nanoparticles, and the loading amount on the surface of fabrics was gradually approaching the maximum, while the load efficiency decreased to the lowest. It can be seen from Table 2 that the amount of silver reduced by chitosan fabric was much higher than those reduced by viscose fibers and medical cotton.36,37 The effect of AgNO3 concentration on the AgNPs content and loading efficiency. The comparison of reduced silver content by viscose fiber, medical cotton and chitosan fabric.

AgNPs/AgNWs-coated chitosan fabrics
In order to further enhance the antibacterial properties, electrical conductivity and stability of the fabrics, the silver nanowires were adhered to the surface of AgNPs-coated chitosan fabrics by the dip-coating method. The durability of AgNWs on chitosan fabrics is poor because there is no interaction between the metal Ag and the polymer.46,47 By first coating the chitosan fabric with AgNPs, AgNWs could be successfully bound to the fabric due to the interfacial interactions between AgNWs and AgNPs due to their high surface energies.
In XRD spectrum of chitosan fabric, the peak around 20° was attributed to diffraction from chitosan (Figure 4(a)).
48
Upon oxidation, the diffraction peak intensity of chitosan become weaker than that of pristine chitosan, which was due to the degradation of the crystallinity of chitosan. Compared to the pristine chitosan fabric, the XRD pattern of the conductive fabric (Figure 4(c)) included five peaks at 38.32°, 44.42°, 64.65°, 77.46°, and 81.64°, corresponding to the (111), (200), (220), (311), and (222) planes of AgNWs, respectively.48,49 The high-intensity diffraction peak at the 2θ value of 38.32° indicated that AgNWs might have a preferred orientation along the (111) crystalline plane. X-ray diffraction patterns of pristine chitosan fabric (a) and AgNPs-coated chitosan fabric (b) and AgNPs/AgNWs-coated chitosan fabric (c).
The dip-coating process was repeated several cycles to increase the amount of AgNWs content on the chitosan fabrics. The amount of AgNWs adsorbed on the surface of the fabric increased with the number of dip-coating cycles, as depicted in the FESEM images in Figure 5. After one dip-coating cycles, AgNWs adhered to the fabric were significantly less, and there were a large number of blank areas. After two dip-coating cycles, the blank area on the fabric was significantly reduced. Until ten dip-coating cycles, a large number of AgNWs were coated on the fabric. The yellow AgNPs-chitosan fabrics changed to gray after 10 dip-coating cycles of 0.5 wt.% AgNWs in ethanol. Field emission scanning electron microscope images of AgNWs-coated chitosan fabrics synthesized by dip-coating 0.5 wt.% AgNWs in ethanol for one (a), two (b), four (c) and ten (d) cycles, respectively.
Conductivity and thermal stability
Figure 6 showed the relationship between electrical resistance of AgNWs-coated chitosan fabric and the number of dip-coating cycles. The resistances of chitosan fabric and AgNPs-chitosan fabric are infinite. As shown in Figure 6, the electrical resistance of the chitosan fabric was 200 mΩ/sq after four dip-coating cycles and less than 100 mΩ/sq after eight dip-coating cycles, which was due to the increasing mass content of AgNWs on the chitosan fabrics shown in Figure 5. After one dip-coating of AgNWs, the electrical resistance of fabric containing AgNPs contents of 108.25 mg/g, 144.85 mg/g and 166.00 mg/g AgNPs was 17.9 Ω/sq, 1.21 Ω/sq and 0.93 Ω/sq, respectively. Therefore, the conductivity of the obtained fabrics is related to the AgNPs content of the chitosan fabric, Ag NPs can interconnect AgNWs by filling the gaps between AgNWs and increase the conductive paths for electrons. The relationship between electrical resistance of AgNWs-coated chitosan fabric and the number of dip-coating cycles.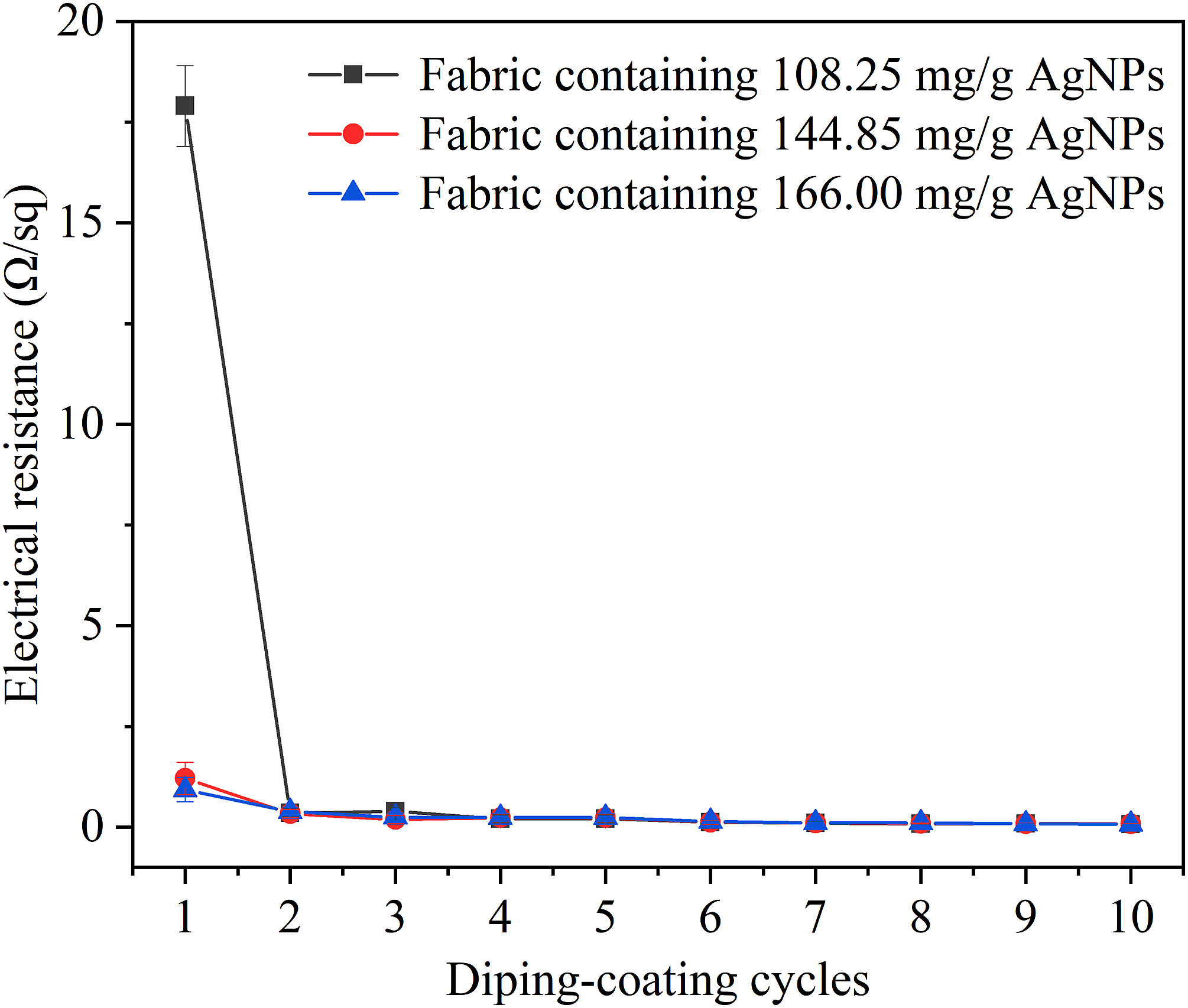
TG analysis was performed to investigate the thermal stability of chitosan fabrics with various silver content. As presented in Figure 7(a), all samples had two stages of mass loss, before 100°C and 200–350°C. The mass loss before 100°C was due to the loss of moisture. Degradation of the chitosan structure was between 200°C and 350°C. It was clear from Figure 7 that the original chitosan fabric began to undergo a thermal decomposition at 284°C, while after loading AgNPs, the initial decomposition temperature is reduced to 245°C. It seems to be related to the damage of crystalline structure of chitosan in alkaline pre-treatments. In addition, the deposited AgNPs is not dense enough to cover the surface of fibers completely. The thermal decomposition of AgNPs/AgNWs-coated chitosan fabric increased to 299°C. This is due to the existence of dense AgNWs layer on the fabric, which increases the thermal decomposition temperature. The mass loss of pristine chitosan fabric was nearly 90% for temperature up to 650°C, while 67% of chitosan fabric was thermally degraded after loading AgNPs. This shows that the existence of AgNPs can improve the thermal stability of fabrics, which is conducive to the application of electronic textiles. The mass loss of AgNPs/AgNWs-coated chitosan fabric is similar to that of AgNPs-coated one, which indicated that the further AgNWs coating cannot improve the thermal stability of the fabrics due to the gaps between AgNWs. Thermogravimetric analysis of chitosan fabric, AgNPs-coated chitosan fabric and AgNPs/AgNWs-coated chitosan fabric.
Antimicrobial activity
It was proved that AgNPs and AgNWs showed effective antibacterial activity on both Gram negative and Gram positive bacteria.14,38,50–54 Moreover, in the presence of AgNPs, the bacterial activity of E. coli is usually higher than that of Staphylococcus aureus.
52
Therefore, E. coli was selected to determine the antibacterial activity based on the diameter of the growth inhibition zone in this wok. Photographs of antibacterial fabric and the inhibition zone measurements were shown in Figure 8. To confirm the antimicrobial activity of Ag-coated chitosan fabric, the antimicrobial activities of chitosan fabrics containing different amounts of AgNPs and AgNWs were tested against E. coli. A distinct growth inhibition zone was observed around the pristine chitosan fabric sheets (Figure 8(a)). The antibacterial effect of chitosan is mainly related to the leakage of intracellular substances, which is caused by the change of permeability of negatively charged cell membrane adsorbed chitosan.
4
The chitosan fabric containing AgNPs showed better antibacterial activity than pristine chitosan fabric (Figure 8(b), (d) and (f)). Studies have proved that the antibacterial effect of AgNPs is related to the particle size.
55
The antibacterial effect of AgNPs with particle size greater than 10 nm mainly depends on the contact with cells rather than the release of silver ions.56,57 The antibacterial activity of AgNWs is due to the impalement of bacterial cells by the sharp tips of AgNWs except for the release of silver ions.
58
Therefore, compared with the chitosan fabric containing only AgNPs, the chitosan fabric containing both AgNPs and AgNWs exhibited a better antibacterial activity (Figure 8(c), (e) and (g)). Moreover, the diameter of the growth inhibition zone increased with increasing silver content in the chitosan fabric. The antimicrobial activities and diameter of the growth inhibition zone of pristine chitosan fabric against E. coli (a); chitosan fabric containing 21 mg/g AgNPs (b) and AgNWs (c); chitosan fabric containing 144.85 mg/g AgNPs (d) and AgNWs (e); and chitosan fabric containing 166.00 mg/g AgNPs (f) and AgNWs (g) against E. coli.
Conclusion
Nanosilver functionalized chitosan fabrics with conductivity and antibacterial activity were fabricated by immobilizing both silver nanoparticles (AgNPs) and silver nanowires (AgNWs) onto chitosan fabrics. For deposition of AgNPs, silver nitrate was reduced by the chitosan fabric itself, without any additional reductant agents, and AgNPs were successfully coated on the chitosan fabrics, as determined by SEM and FTIR. The conductivity of the fabrics is related to the AgNPs content of the chitosan fabric, AgNPs can increase the conductive paths for electrons by interconnecting AgNWs. With more dip-coating cycles, the mass content of AgNWs increased and the electrical resistance of the chitosan fabric decreased. After four dip-coating cycles, the electrical resistance of fabric was as low as 0.20 Ω/sq. The chitosan fabrics coated with AgNPs and AgNWs exhibited higher antibacterial activity and thermal stability than pristine chitosan fabric. The chitosan fabric with synergy of AgNPs and AgNWs shows good potential application in wearable electronic textiles. It provides a new strategy for preparation of conductive fabrics with excellent conductivity and antibacterial activity.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Guangdong Province, China (2018A0303130100) and the Opening Foundation of State Key Laboratory of Advanced Materials and Electronic Components (FHR-JS-201909006).

