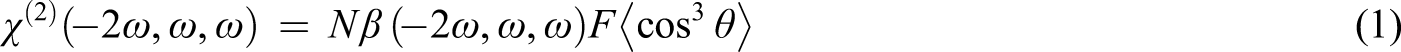Abstract
Cross-linked polymers with chromophore fragments built-in main chains can, depending on their structure and synthesis conditions, keep during a long time the orientation of chromophores that is necessary to create new organic polymer materials demonstrating nonlinear optical properties. The present work is devoted to cross-linked polymers on the basis of di-, tri-, and tetraglycidyl ethers of quercetin-8-sulfonic acid sodium salt (3,5,7,3′,4′-penthahydroxyflavon-8-sulfonic acid sodium salt) which were synthesized, and then, poled in electrical field of corona discharge. Study of thermal, optical, and nonlinear optical parameters of the polymer films was carried out. It was found that the polymers obtained from di- and triglycidyl ethers of quercetin-8-sulfonic acid sodium salt are characterized by the higher values of macroscopic quadratic susceptibilities comparing with such a value for polymer obtained from tetraglycidyl ether of quercetin-8-sulfonic acid sodium salt. However, the latter polymer demonstrates a minimal intensity of relaxation processes that evidence its highest long-term stability, and thus, could be used as a nonlinear optical material. It is supposed that the difference of the nonlinear optical properties is due to peculiarities of intermolecular interactions in physical network of the polymers, namely, to the difference between numbers of hydrogen bonds formed by hydroxyl groups of chromophore fragments and number of hydrogen bonds linked of inter fragmental parts of the polymeric chains.
Introduction
Wide usage of polymer materials with nonlinear optical (NLO) properties in photonics and microelectronics is due to a number of advantages compared to similar inorganic materials.1–5 The use of the latter in NLO applications is limited by the need to grow large-size crystals of a special shape, while NLO polymer materials are more technologically and economically effective, have faster NLO response, and their NLO properties can be optimized by modifying the chemical structure of the materials.6,7
Currently, there are various approaches to obtaining the functional polymer materials with NLO properties. 8 One of the pathways for obtaining such polymer systems, devoid of the disadvantages of other methods, is the synthesis of polymers with covalently linked chromophore fragments.
The condition necessary for such polymers is the presence of a rigid structure, that hinders shifting light-absorbing fragments—chromophores. This condition is usually realized in cross-linked polymers where moving chain fragments are more or less hindered. It is known that some cross-linked polymers with chromophore fragments built-in can, depending on their structure and synthesis conditions, keep during a long time the orientation of chromophores that is necessary to create new organic polymer materials demonstrating nonlinear optical properties.
There are two well-known methods of the synthesis of such polymers. The first method involves the synthesis of polymers with side chromophore fragments. The disadvantage of this method is that the obtained polymers have lack of long-term stability of the NLO properties. Another method is the synthesis of polymers, where the chromophore fragments are incorporated in main chains.
It is necessary to note that in the both mentioned cases the macroscopic second-order polarizabilities of synthesized polymers are defined by the hyperpolarizabilities on the molecular level (β), that is, by NLO activities of the chromophore fragments.9–13 Therefore, the high-priority problem in the NLO polymers obtaining is the increase and optimization of NLO activity of the chromophores. In this regard, the search for new chromophores or chemical modification of the known ones is of a great interest. The chromophores should contain reactive functional groups providing the possibility to obtain chromophore-containing polymers under standard conditions.
In our previous publications,14,15 new NLO active organic polymers containing chromophore fragments based on widespread natural compounds as 3,5,7,3′,4′- penthahydroxyflavone (quercetin) and its derivatives were obtained. These chromophores were not used as dopants but were inserted in the main polymer chains that would significantly increase the relaxation time of chromophores. As the result, NLO polymers based on glycidyl ethers of quercetin were obtained and studied. The synthesis of such polymers was based on obtaining a monomer—an epoxy derivative of quercetin, by glycidylation of hydroxyl groups of the chromophore.
The present work is aimed at expanding a number of chromophores and developing methods for the synthesis of polymers with covalently attached chromophore fragments. The synthesis and data about studied NLO properties of quercetine-8-sulfonic acid (hereinafter-called 8-sulfoquercetin) chromophore-containing polymers are described in the paper.
Materials and methods
Materials and instruments
Synthesis of glycidyl derivatives of 3,5,7,3′,4′-penthahydroxyflavon-8-sulfonic acid and their salts was carried out using commercial 3,5,7,3′,4′-pentahydroxyflavone (Quercetin, Mw = 302.24, Sigma-Aldrich Chemicals), sulfuric acid (ρ = 1.84 g × cm−3), and epichlorohydrin (Mw = 92.52, Sigma-Aldrich Chemicals). Acetone used as solvent was purified and dried before usage.
1H-NMR spectra were measured with a Bruker Avance III NMR Spectrometer (500 MHz) using DMSO-d6. The presence of residual solvent in the polymer films were monitored by FTIR-ATR (attenuated total reflection) spectroscopy detecting a stretching vibration absorption band of acetone C=O group at about 1710 cm–1. FTIR-ATR spectra were recorded by an ALPHA FT-IR Spectrometer with a diamond crystal in the range of 400–4000 cm−1. Resolution of 4 cm−1 was used, and 32 scans were averaged. UV-visible spectra of the polymer films were obtained using UV-Vis spectrophotometer Hitachi-U3210 in the range of 210–500 nm. Mathematical treatment of the spectra was performed using Spectra Data Lab software package. 16 Glass transition temperature (Tg) of the polymers were obtained by differential scanning calorimetry (DSC). DSC analysis was carried out with a DSC1 (Mettler Toledo) thermal analyzer with sample weights of 9–6 mg over the temperature range from 0 to 180°C and with the heating rate of 10°C/min. The thickness of the studied polymer films was measured using a Linnik-type interference microscope MII-4. The Linnik interferometer configuration (a kind of Michelson configuration) was described in Mishurov et al.’s study 17 Calculations of the Mulliken charges on quercetin-8-sulfonic acid sodium salt chromophore fragments were carried out according to DFT theory 18 using the B3LYP functional 19 and cc-pVDZ basis sets20,21 implemented in the GAUSSIAN 09 program package. 22
Synthesis of (3,5,7,3′,4′-penthahydroxyflavon-8-sulfonic acid) sodium salt (NaQSA-8)
The synthesis of NaQSA-8 was carried out by the method reported previously in Mishurov et al.’s study. 23 2 g (6.62 mmol) of previously dried (at 125oC) quercetin was stirred with 8 cm3 of concentrated sulfuric acid (ρ = 1.84 g cm−3) for 5 h at 20oC in 100 cm3 round-bottom flask. Then, 20 cm3 of very cold water was added into the reaction mixture. Obtained orange–red precipitate was filtered at a reduced pressure and recrystallized twice from the hot saturated water solution. Next, the orange–red sediment was dried in air at room temperature. The synthesis yield was 40%.
Sodium salt (NaQSA-8) was obtained by neutralizing supernatant fluid with 20% solution of NaOH up to pH 4. The yellow precipitate was filtered off and crystallized twice from a hot saturated aqueous solution. The precipitate was dried in air at room temperature. The synthesis yield was 44%.
Synthesis of NaQSA-8 glycidyl derivatives
NaQSA-8 glycidation was carried out by the method reported previously in Woznicka et al.’s study.
24
Monoglycidyl ethers of quercetin-8-sulfonic acid sodium salt were not obtained. Reaction of NaQSA-8and epichlorohydrin in molar ratio 1:1 resulted in a mixture of starting reagents and diglycidyl ether. Using higher epichlorohydrin concentrations, diglycidyl (I), triglycidyl (II), and tetraglycidyl (III) ethers of quercetin-8-sulfonic acid sodium salt were obtained. Scheme of synthesis of the latter ether is presented in Figure 1. Scheme of sulfoquercetin glycidyl ethers synthesis.
Synthesis of (7,4′-diglycidyloxy-3,5,3′-trihydroxyflavon-8-sulfonic acid) sodium salt (2GESQ, I)
Mixture of NaQSA-8 (2.00 g, 6.6 mmol), epichlorohydrin (1.034 mL, 13.2 mmol), was heated at 80°C for 30 min in argon atmosphere. Then, solution of sodium hydroxide (2.65 g, 66.2 mmol) in water (9.0 mL) was added, and the mixture was heated at 80°C for 4 h. After that, the mixture was concentrated and cooled; obtained precipitation was re-crystallized from acetone. Finally, 2GESQ was purified by column chromatography (silica gel; chloroform/acetone = 10:1 v/v). Yield was 65%.
1Н NMR (DMSO–d6, J = 500 МHz): 3,65-4,45 (m, 10Н, –СН2СНСН2О-); 6.38 (d, 1Н, С6-Н); 7.67 (d, 1H, С2’-Н); 7.04 (d, 1H, С5’-Н); 7.53 (dd 1H, С6’-Н); 9,27 (d, 1Н, С3- ОН); 9,56 (s, 1Н, С3'-ОН);12,54 (s, 1Н, С5-ОН).
Elemental Analysis: Calc. (wt%): C—60.87; H—4.38; O—34.75. Found (wt%): C—59.70; H—4.55; O—35.75.
Synthesis of (7,4′,3′-triglycidyloxy-3,5-dihydroxyflavon-8-sulfonic acid) sodium salt (3GESQ, II)
Synthesis of II was carried out as described above at molar ratio of NaQSA-8/epichlorohydrin (1:3). Yield was 61%.
1Н NMR (DMSO–d6, J = 500 МHz): 3,65-4,45 (m, 15Н –СН2СНСН2О-); 6.38 (d, 1Н, С6-Н); 6.86 (d, 1H, С8-Н); 7.67 (d, 1H, С2’-Н); 7.04 (d, 1H, С5’-Н); 7.53 (dd, 1H, С6’-Н); 9,27 (d, 1Н, С3- ОН); 12,54 (s, 1Н, С5-ОН).
Elemental Analysis: Calc. (wt%): C—61.28; H—4.71; O—34.01. Found (wt%): C—60.34; H—4.68; O—34.98.
Synthesis of (7,3,4′,3′- tetraglycidyloxy -5- hydroxyflavon -8- sulfonic acid) sodium salt (4GESQ, III)
Synthesis of III was carried out as described above at molar ratio of NaQSA-8/epichlorohydrin (1:4). Yield was 56%.
1Н NMR (DMSO–d6, J = 500 МHz): 3,65-4,45 (m, 20Н–СН2СНСН2О-); 6.38 (d, 1Н, С6-Н); 6.86 (d, 1H, С8-Н); 7.67 (d, 1H, С2’-Н); 7.04 (d, 1H, С5’-Н); 7.53 (dd, 1H, С6’-Н)); 12,54 (s, 1Н, С5-ОН).
Elemental Analysis: Calc. (wt%): C—61.59; H—4.98; O—33.43. Found (wt%): C—61.28; H—5.01; O—33.71.
Preparation of polymer films
Glycidyl ethers of NaQSA-8 were dissolved in acetone at a concentration of about 0.1 g/mL. DETA was added in stoichiometric quantities proportionally to number of glycidyl groups in the corresponding NaQSA-8derivatives. Thereafter, obtained thin films were spin-coated onto pre-cleaned glass microscope cover slides of 170 μm at 1000 rpm for 0.5 min and cured at the room temperature for 24 h in vacuum. The thickness of the obtained films was 1.0 μm.
The used glass microscope cover slides were ultrasonically cleaned in detergent solution for about 10 min, followed by rinsing it in deionized water. Then the slides were then boiled in ethanol and dried at 90oC for 10 min.
The network segments of polymers poly (2GESQ), poly (3GESQ) and poly (4GESQ) obtained correspondingly from di-, tri-, and tetraglycidyl derivatives of sulfoquercetin are depicted in Figure 1.
Polymer films poling
The polymer films were poled by the corona poling technique. Poling was done simultaneously with curing reaction. Temperature of the film was increased to 100oC then the corona field was applied, and poling was carried out for 1 h. The corona current was held at 2.5 μA with an electrode potential of 6 kV. Finally, the films were cooled to the room temperature, while the electric field was kept on. To remove residual solvent, the polymer films were then annealed for 3 h at 100oC.
Results and discussion
Optical properties
The absorption spectra of polymer films poly (2GESQ), poly (3GESQ), and poly (4GESQ) are showed in Figure 2. It can be seen, there is a small hypsochromic shift on going from poly (2GESQ) (396 nm) to poly (4GESQ) (380 nm), that is, from less to more substituted quercetin-8-sulfonic acid sodium salt chromophore small shift indicates that the replacement of end-donor OH groups on glycidyl groups have no significant effect on the conjugated polymer structures. Uv-Vis spectra of polymers based on sulfoquercetin glycydyl ethers.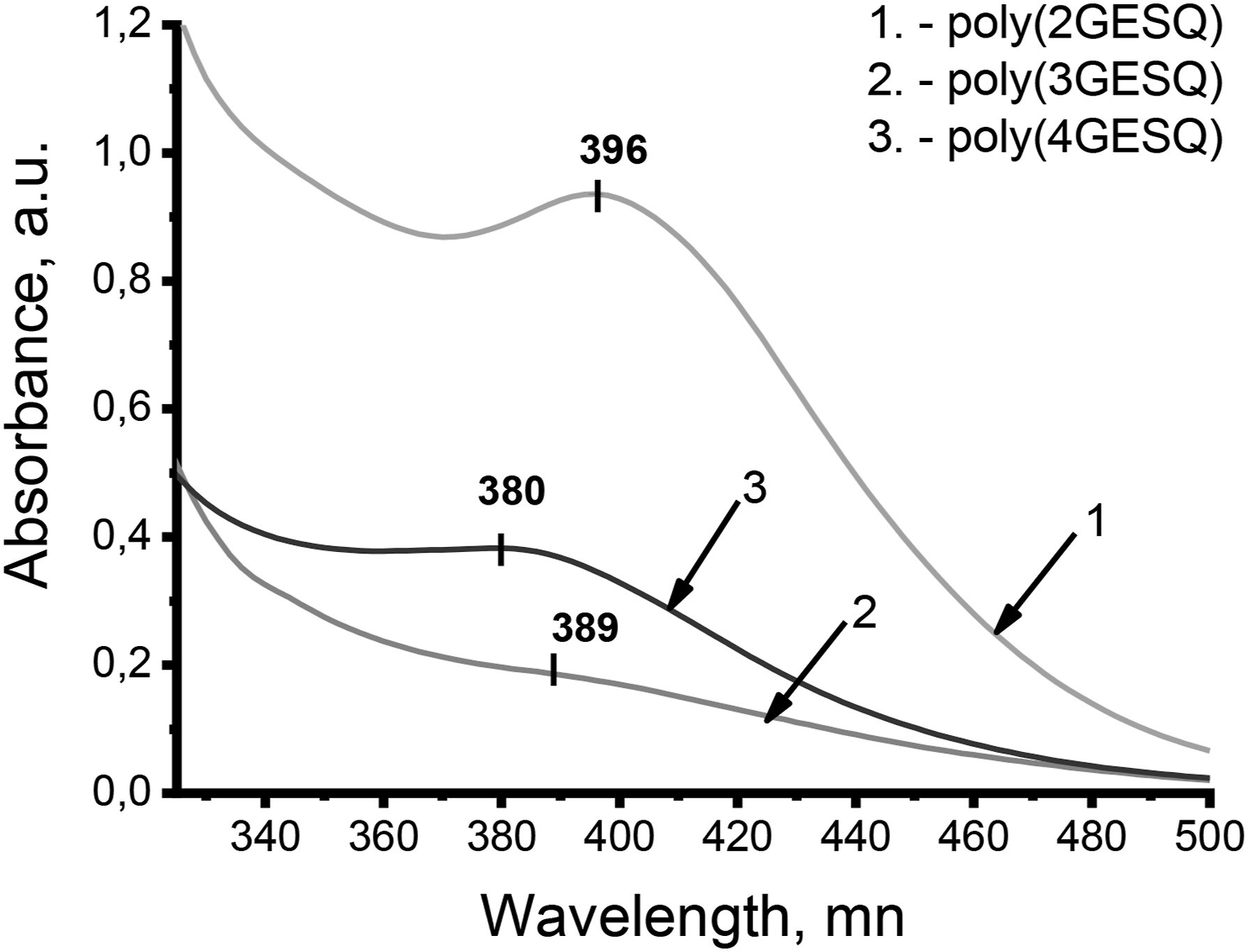
Absorption bands of previously investigated polymer films obtained from quercetin glycidyl ethers are centered at about 362 nm. Thus, the introduction of sulfonic group into quercetin molecule results in the bathochromic shift of corresponding absorption band in the polymer films. It should be noted that the bathochromic shift of an intense long-wavelength band, that is, decrease in energy of corresponding electronic transition leads to increase in values of the first-order hyperpolarizability.25,26 The obtained results also show that no other obvious absorption shoulders or peaks are observed in the investigated polymer films. This indicates the absence of some intense dipole–dipole interaction between chromophore fragments or formation of their aggregates in the polymer molecules. In this case, we can assume that the alignment of the chromophore fragments in investigated NLO polymer films could be easier to implement, and perhaps, higher poling efficiency and greater macro second-order polarizabilities in these materials could be achieved.27,28
Thermal analysis
Thermal analysis of the polymer films based on glycidyl derivatives of quercetin was carried out in our previous work. 14 It was showed that the increase in number of glycidyl substituents in the quercetin moiety results in the decrease of Tg, which could be explained by formation of the polymer networks with greater free volume. The growth of the free volume can be due to formation of a greater number of disordered regions in the polymer network of higher mobility of the polymer chains. In contrast with diglycidated quercetin, the use of tri- and tetra-glycidated monomers results in formation of the polymer networks having higher branching degree that sterically hinders a denser packing of the chain segments. This leads to amorphization of the obtained polymers and to the decrease of their Tg.
Figure 3 shows thermograms of the polymer films based on glycidyl ethers of quercetin-8-sulfonic acid sodium salt. It can be seen that the glass transition temperatures of the latter are lower than those of the polymer films based on quercetin glycidyl ethers studied previously. It is evident that substitution of a hydrogen atom by more voluminous sulfonic group results in changes of the supramolecular structure of the three-dimensional polymer and, consequently, in changes in orientational ordering of the chromophore fragments in the macromolecules. The additional effect leading to the increase in distance between chromophores is the negative charge of sulfonic groups, which is not completely neutralized by the counter ions. Thus, the additional amorphizarion of the obtained polymers and, as a result, the decrease in the glass transition temperature, in our opinion, could be explained by sterical hindrances and electrostatic interactions due to the presence of sulfonic substituents. DSC curves of polymers based on glycidyl ethers of sulfoquercetin.
Nonlinear optical activity and long-term stability of the polymer films
The macroscopic second-order susceptibilities (χ(2)) (at 1064 nm) in the polymer films were obtained. The former ones were calculated according to the one-dimensional rigid oriented gas model
29
The parameter β for each monomer unit was determined by the solvatochromic method. The order parameter Φ was calculated from absorbance values of the films after (А1) and before (А0) the corona poling according to the equation
30
It can be seen (see Figure 2) that the absorption spectra of investigated polymers consist of several bands. To determine exact intensities of the long-wavelength band used for determination of Φ value, the absorption spectra were previously deconvoluted. The absorption bands deconvoluted from the polymer films spectra before and after poling procedure are presented in Figure 4. Uv-Vis spectra of polymers based on sulfoquercetin glycydyl ethers before and after poling (a) poly(2GESQ), (b) poly(3GESQ), (c) poly(4GESQ).
Dipole moments and molecular second-order susceptibilities (β) of monomers.

aβesu = 2.368 × 109βSI.
Nonlinear parameters of the polymers based on glycidyl ethers of quercetin-8-sulfonic acid sodium salt.

aThe second-order molecular susceptibilities of the corresponding monomers in electrostatic(esu) units.
bχ(2) (SI) = (4π/3) × 10−4χ(2)(esu).
It is known that a chromophore fragment responsible for spectral properties of flavonol molecules (to whom quercetine is related) is the chromone one. The side phenyl ring of flavonols almost always plays the role of an auxochrome fragment which affects the charge redistribution between itself and the chromone moiety depending on number and electron-releasing ability of the substituents in both fragments. Usually, substituent-controlled interfragmental charge redistribution leads to not very remarkable changing of the charge on the chromon fragment that explains not very substantial changes in the spectral properties of flavonols, particularly, the second-order susceptibilities.
The chromone fragment in 2GESQ, 3GESQ, and 4GESQ has substantial negative charge due to the presence of sulfonic group. In this case, the role of the side phenyl fragment increases substantially, and in particular, the degree of the interfragmental electronic density redistribution much drastically depends on the electron-releasing ability of the substituents in the side ring. Hydroxyl groups having high electron-releasing ability prevent the charge transfer onto phenyl ring, and the negative charge on the chromone fragment remains significant. Substitution of hydroxyl groups by glycidyloxy ones, having weaker electron-releasing ability, results in more intensive charge transfer onto the phenyl ring and in lower negative charge on the chromone moiety, correspondingly.
The estimations of negative charge on the chromone moiety made by the DFT method show that the glycidylation of quercetine up to 3,7,3′,4′-tetraglycidylquercetin (5-hydroxy group cannot glycidylated due to its very low acidity) results in unsignificative decrease of the negative charge on the chromone moiety—from −0.046ē to −0.044ē (Δq = −0.002ē). In the case of sulfonated quercetin similar glycidylation leads to changing of the chromone charge from −0,413ē to −0.365ē (Δq = −0.048ē).
So, we suggest that the decreases in the molecular second-order susceptibilities is connected with the decrease of negative charge on the chromone fragment when all hydroxyl groups of the side phenyl cycle are substituted by glycidyloxy groups of the low electron-releasing ability.
Values of the macroscopic second-order susceptibilities of corresponding polymers are listed in Table 2.
It can be seen that values β and χ(2) decrease on going from poly (2GESQ) to poly (4GESQ). This phenomenon can be explained by the rigidity of the chromophore environment. Poly (2GESQ) has linear polymer chains, and it is of a “softest” structure, and in its case, the poling procedure results in a maximal re-orientation of the chromophore fragments and a maximal anisotropy effect achievement. Poly (3GESQ) and poly (4GESQ) are network polymers, the later one has higher crosslink degree, and its polymer network is more rigid. In the case of these polymers, the poling does not allow us to achieve the maximal re-orientation of the chromophore fragments, and anisotropy of the after-poling samples is lower for poly (3GESQ) and much lower for poly (4GESQ).
One of the most important parameters of NLO polymer films is the long-term stability of their nonlinear optical properties after turning off the applied electric field. It was previously shown that the rate of the second-harmonic intensity decreases after turning off the applied electric field depends on chemical structure of a polymer, the nature of non-specific and specific interactions between fragments of chains and free volume of the polymer network. 14
The relaxation behavior of the NLO properties of investigated polymer films was studied by measuring the dependence of χ(2)t/χ(2)0 ratio on time at ambient temperature during 28 days. Figure 5 shows that the polymers obtained from 4GESQ demonstrate the higher long-term stability of the NLO properties than polymers obtained from 2GESQ and 3GESQ monomers. The relaxation processes in poly4GESQ take place during 3–5 days, and lead to decrease of χ(2)t/χ(2)0 ratio by about 30%. In the case of poly2GESQ and poly3GESQ, relaxation processes top in 25–28 days, and χ(2)t/χ(2)0 values decrease twice times approximately. Long-term stability of the NLO polymer films at 25oC for 28 days.
Changing of χ(2)t/χ(2)0 values can occur by two simultaneous pathways. First, the decrease in χ(2)t/χ(2)0 can be due to the post-curing process, which is characteristic of cross-linked polymers, and especially, for epoxy polymers. 31 It is additional crosslinking of the polymer network, which occurs during 2–5 days and results in disordering of the chromophore fragments and in fall of the NLO properties of the polymer films. The second and, probably, main pathway is the thermal motion of the chromophore fragments and the chain fragments connecting them. The most rigid network of poly4GESQ prevents free rotation of the chromophore fragments in the polymer chains and motion of the chains leading to changing of the chromophore positions. Thus, the ordered orientation of chromophores obtained due to the poling process does not change significantly over time. In the case of polymers poly2GESQ and poly3GESQ of the lower rigidity, the thermal motion of the chromophore fragments and polymer chains is more intensive that explains longer relaxation process over time and greater decrease in χ(2)t/χ(2)0 values. However, it is worth to note a factor limiting the relaxation processes in poly3GESQ and especially in poly2GESQ polymers. The chromophore fragments of poly3GESQ and poly2GESQ have one and two free hydroxyl groups, respectively, which form a strong “physical” network due to hydrogen bonds between the chromophores and hydroxyl groups in the polymer chains. 32 The presence of such a network limits free motion of the polymer chains and chromophore fragments and thus prevents the NLO property losses.
Conclusions
New linear (2GESQ) and cross-linked polymers (3GESQ and 4GESQ) based on glycidyl ethers of quercetin sulfonic salt were synthesized. All obtained polymers are characterized by the high refractive indices and the great thermal stability. It was found that the polymers obtained from 2GESQ and 3GESQ have higher values of macroscopic quadratic susceptibility, than 4GESQ-based polymer. However, poly4GESQ polymers show a long-term stability of the NLO properties, while the 2GESQ and 3GESQ polymers undergo the relaxation process resulting in decrease of the NLO effect.
The obtained results allow us to conclude that the 2GESQ- and 3GESQ-based polymers could be used as NLO media in various photonics and optoelectronic applications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the financial support of this research by the Ministry of Education and Science of Ukraine (Project No. 0119U002536).