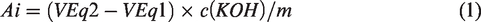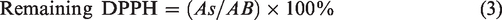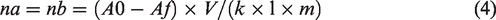Abstract
The present work deals with the preparation of chemically and enzymatically eugenol-modified chitosan and its application as a green coating on viscose for the potential production of medical textiles. The elemental composition of chemically and enzymatically eugenol-modified chitosan was determined by attenuated total reflection Fourier transform infrared (ATR FT-IR) and proton nuclear magnetic resonance (1H NMR) spectroscopy. Moreover, potentiometric titration was used to estimate the number of accessible amino groups. The chitosan–eugenol formulation possesses a high antioxidant capacity level, as shown by the high content of phenolic compounds analyzed by the following methods: (a) Folin–Ciocalteu; (b) reduction of Fe3+ ions; and (c) α, α-diphenyl-β-picrylhydrazyl (DPPH•). Viscose coated with chitosan–eugenol formulations was evaluated by ATR FT-IR spectroscopy and Acid Orange VII spectrophotometric methods, the results of which demonstrated the successful deposition of these formulations on the fibers. Scanning electron microscopy showed the presence of the functional coating on the viscose. Finally, antimicrobial testing was performed to determine inhibition of selected pathogenic micro-organisms using the ASTM E 2149-01 standard. The antioxidant activity of the functionalized viscose was investigated spectrophotometrically by the DPPH• method. It was found that the coatings of eugenol-modified chitosan impart antimicrobial activity to the viscose against Gram-positive and Gram-negative bacteria as well as fungi, and cause inhibition of free radicals by introducing an antioxidant character. The chemical and enzymatic modification of chitosan offers many opportunities for the incorporation of many natural active ingredients into the chitosan system, which provides a good basis for further work related to the functionalization of chitosan for sanitary and medical use.
In recent years, several environmentally and human friendly processes have been developed to produce textile products with antimicrobial effectiveness. As a natural antimicrobial agent, chitosan is often used for this purpose.1–3 Numerous attempts to use chitosan extend to the areas of wound healing, including burns and skin flora protection.4,5 Although chitosan has excellent properties, such as good adhesion, biodegradability and antimicrobial activity, 6 its antioxidant activity is poor.7–9 The antioxidant activity is a valued property in healing processes as it reduces the inflammatory response. 10 Many authors emphasize the importance of modifying chitosan to achieve increased antioxidant activity of this natural polymer.11–13 Furthermore, chitosan also shows selective antimicrobial activity. It works well against Gram-positive bacteria, but it works worse against Gram-negative bacteria. Therefore, the disadvantages of chitosan can be overcome by combining chitosan with one of the natural substrates with antimicrobial and antioxidant activity. The antioxidant values of chitosan can be further improved, for example, by adding natural antioxidants from plant polyphenols.14–16 The polyphenolic compounds in green tea (Camellia sinensis), for example, catechins, theaflavins and arubigens, have good antioxidant activities. Several other publications have documented the efficacy of the antioxidant activity of chitosan films or coatings in combination with many other plant flavonoids, for example, procyanidins from grape seed extract, 17 pomegranate peel extract, 18 apple peel extract 19 and thyme extracts. 20 Essential oil (EO) components are aromatic, bioactive, antioxidant terpenoids derived from plant sources and have been intensively studied as natural food preservatives, but with limited application as their addition may affect the desirable taste of the food 16 and cause an unpleasant odor or color on the fibers. Numerous publications have reported enhanced antioxidant activities of chitosan films or coatings by incorporating terpenoids into EO components that exert radical scavenging, transition metal chelating, singlet oxygen quenching activities and/or act as hydrogen donors.21,22 The combination of chitosan (CH) with EOs of clove, oregano, cinnamon or eucalyptus has been reported to enhance antimicrobial activity in food packaging applications,23,24 but much less attention has been paid to these formulations as coatings for fibers. Incorporation of EOs into fibers through the production of nanofibers by electrospinning has been shown to be more useful than their application in the form of coatings.25,26 In addition, there have been attempts at grafting to achieve antimicrobial efficacy and biocompatibility. 27
For the development of a new generation of textiles for medical use, it is particularly attractive to develop liquid formulations as coatings. Approaches to coating textiles have evolved and diversified considerably in recent decades with the increasing use of medical textiles. These approaches increasingly consider environmental impact, economic considerations and sustainability and efficiency as important benefits of the textile coating. The range of natural antioxidant compounds that can produce such beneficial coatings is diverse, as mentioned earlier, allowing the use of many different plant extracts.28,29 Eugenol seems to be a very interesting natural substance.30–32 Eugenol is the main component of the EO of cloves. It has antiseptic properties, that is, it inhibits bacteria, viruses and fungi effectively. In addition to its antiseptic and antiviral properties, eugenol also acts as an antioxidant, antibiotic, fungicide and bactericide. 32 From the literature review, the combination of chitosan and eugenol is a very attractive approach, and one of the interesting studies showed that grafted eugenol and carvacrol conferred antioxidant activity on chitosan nanoparticles (CHEU NPs, CHCA NPs). These chitosan nanoparticles grafted with an EO component achieved antibacterial activity equivalent or better than that of unmodified chitosan nanoparticles. Cytotoxicity tests with 3T3 mouse fibroblasts showed that the cytotoxicity of CHEU NPs and CHCA NPs was significantly lower than that of pure EOs 33 ; thus, the synergism between chitosan and eugenol showed added value. In a similar study, hydrogels consisting of chitosan and eugenol were prepared to enhance and maintain the antioxidant activity. The vinyl groups of the eugenol monomer were grafted directly to the amino groups of chitosan using cerium ammonium nitrate. Compared to chitosan alone, the chitosan hydrogels grafted with eugenol showed a lower pH sensitivity because the amino groups of chitosan, which were pH sensitive, were grafted with eugenol. The scavenging activity of the tested hydrogels increased with the graft yield of eugenol, since phenolic groups in the eugenol could play an important role as potent free radical terminators, resulting in an improved antioxidant activity in the eugenol-grafted chitosan hydrogel compared to chitosan alone. 34
Enzymatic modification is less common compared to the conventional chemical method, 35 but it is more attractive when speaking of more environmentally friendly techniques. 36 Laccase is known as a green enzyme and can oxidize phenolic compounds into reactive quinone structures in the presence of oxygen and is used as an efficient biotechnology to functionalize chitosan. 37 Chitosan has been functionalized with various phenolic compounds (tannic acid and quercetin, 38 ferulic acid and ethyl ferulate 39 and gallic acid (GA) 40 ) using laccase to improve the antioxidant and/or antimicrobial property.
Although the preparation of chemically and enzymatically eugenol-modified chitosan 41 has received quite a lot of attention, a rather large gap is recognized in its use as a coating for fiber polymers. The surface functionalization of viscose cellulose fibers, as the most representative material for medical textiles, using chitosan chemically and/or enzymatically modified by eugenol has been little researched so far, but, due to the known efficiency of this compound, it is a very interesting possibility to produce green coatings for medical textile products (e.g., bandages, gauze, surgical bedding, etc.) with bioactive, antimicrobial and antioxidant properties. More so, until now, such a consistent comparison of the two modified chitosans and their attachment to fibers has not been studied.
Therefore, the present research work deals with the preparation of eugenol-modified chitosan (i.e., functionalization solution) by chemical and enzymatic methods and the characterization of the two copolymers and their transfer to a viscose substrate as green coatings with the aim of producing antimicrobial and antioxidant active textiles. Both coated viscose fibers were evaluated under physicochemical aspects and with respect to their bioactivity to assess a potential medical application. Antimicrobial and antioxidant activity are the most appreciated properties of medical textiles. Attenuated total reflection Fourier transform infrared (ATR FT-IR) spectroscopy and the Acid Orange VII spectrophotometric methods prove the adsorption success. Scanning electron microscopy (SEM) was performed to study the fiber morphology, showing the presence of copolymers on the fibers. Finally, it was concluded that the fibers show selective antimicrobial activity and antioxidant capacity is introduced. It was concluded that functionalized viscose fibers coated with the developed eugenol-modified chitosan have a high bioactive efficiency and may strengthen the medical textile segment as a potential new product.
Materials and methods
Materials
Non-woven viscose with a surface mass of 185 g/m2 in the form of a strip, 4 cm wide (KEMEX, Netherlands), was used as the cellulose carrier for the adsorption of aqueous solutions of eugenol-modified chitosan. Chitosan, low molecular weight (Mol wt. = 50,000–190,000 Da, Sigma Aldrich), and eugenol (M = 164.21 g/mol, Merck) were used to produce the eugenol-modified chitosan.
Methods
Preparation of eugenol-modified chitosan
Chemical modification
A solution of chitosan with 0.5% by weight was prepared by suspending the required amount of chitosan powder in pure Milli-Q water (18.2 MΩ cm at 25°C). Complete dissolution of chitosan was achieved by adding acetic acid (CH3COOH) at a pH of 3.6. The resulting solution was then transferred to a reaction flask and 60 μL of eugenol (EUG (0.5 × 10−3 M)) was added. This mixture was treated under continuous stirring for 4 hours in an inert nitrogen atmosphere at 40°C. After 4 hours, the initiator of the radical addition polymerization – cerium ammonium (IV) nitrate (CAN, 10−3 M, (Fluka))—was added to the chitosan mixture in combination with EUG. The CAN was previously dissolved in 50 mL 1 N nitric acid (HNO3). 34 After 1 hour, the reaction product was precipitated in acetone, and the resulting precipitate was centrifuged at 5000 rpm for 10 minutes (Universal Centrifuge—Hettich 320 R, Germany). The supernatant was then pipetted, and the precipitate was washed several times with ethanol and water. Any unreacted EUG was removed, which was confirmed by ultraviolet-visible (UV/VIS) spectroscopic analysis; the absorbance value at the maximum of eugenol (287 nm) was zero. The centrifuged product was then dried for 24 hours at 40°C in a vacuum oven (VS-50 SC Kambič, Slovenia). After drying, the samples were transferred into dark bottles and stored in a dark room at room temperature.
Enzymatic modification
A solution of 0.5% (w/v) chitosan (CH) was prepared as with the chemical modification. To maintain the pH value of about 3.6, that is, to dissolve the chitosan completely, acetic acid (CH3COOH) was added dropwise to the solution. At a pH of 3.6, the free amino groups of chitosan are fully protonated (Figure 1(b)), are not susceptible to nucleophilic attack and, as such, cannot react with the o-quinones produced by the enzyme laccase. 42 Therefore, to fulfill the binding mechanism, the pH of the chitosan solution was adjusted to 5.5 with 4 N sodium hydroxide (NaOH). At this higher pH, the amino groups are largely deprotonated (Figure 1(a)) and nucleophilic, so that they can be bound to the electrophilic groups (o-quinones) via covalent bonds.
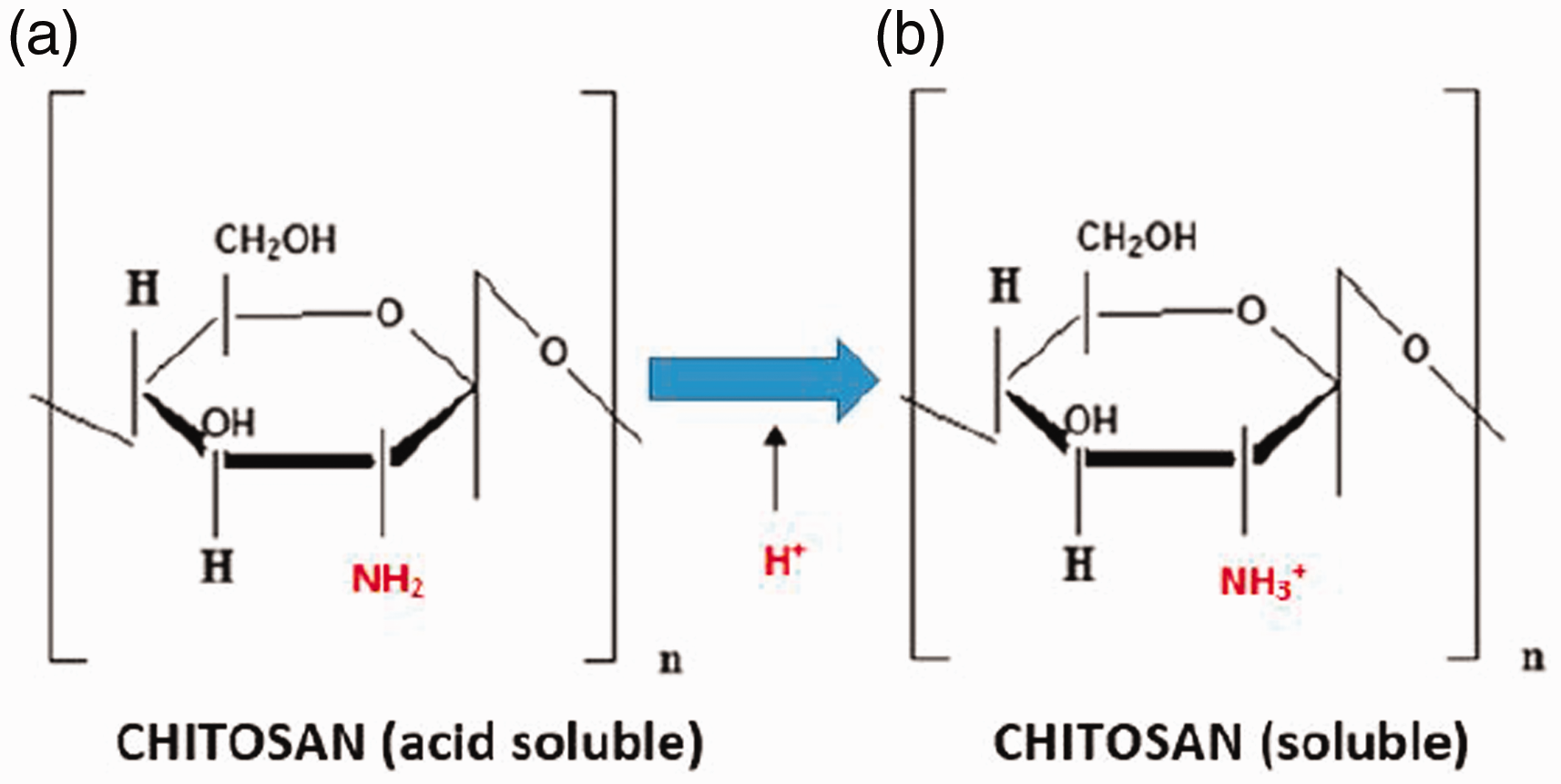
Solubility of chitosan in dependence of pH.
This chitosan solution was transferred to the reaction flask and an appropriate amount of EUG (0.5 × 10−3 M) was added, and after 15 minutes 500 mg of the enzyme laccase (Trametes versicolor; 0.5 U/mg, Sigma-Aldrich). The solution was treated under continuous stirring and under light protection for 6 hours under air supply. The reaction product was then precipitated in acetone; the resulting precipitate was centrifuged for 10 minutes at 5000 rpm. The supernatant was then removed, and the precipitate was washed several times with ethanol and, finally, with deionized water. Unreacted EUG and the enzyme used in the experiment were removed, which was confirmed by the UV/VIS spectroscopic analysis of the ethanol washing solution; the absorbance value at the maximum of eugenol (287 nm) was zero. The enzymatic eugenol–chitosan modification was adopted from the literature. 42
The following samples and their notifications are given: CH—chitosan; EUG—eugenol; CH/EUG (CHEM)—chemically eugenol-modified chitosan; CH/EUG (ENZ)—enzymatically eugenol-modified chitosan; VIS CH/EUG (CHEM)—viscose, functionalized with chemically eugenol-modified chitosan; VIS CH/EUG (ENZ)—viscose, functionalized with enzymatically eugenol-modified chitosan.
Evaluation of chemically and enzymatically eugenol-modified chitosan
ATR FT-IR spectroscopy
The ATR FT-IR spectra were recorded with a Perkin Elmer Spectrum GX spectrometer (Perkin Elmer FT-IR, Omega, Ljubljana, Slovenia). The ATR accessories (supplied by Specac Ltd, Orpington, Kent, UK) contained a diamond crystal. For each sample, a total of 16 scans were performed, with a resolution of 4 cm−1. All spectra were recorded at ambient temperature over a wave-number interval between 4000 and 650 cm−1.
Proton nuclear magnetic resonance spectroscopy
Proton nuclear magnetic resonance (1H NMR) spectra were recorded with a 300 MHz spectrometer, and expressed in ppm using tetramethylsilane (TMS) and trimethylsilylpropanoic acid (TMSP) as the internal standard. The following deuterated solvents were used: DCl, D2O, CDCl3. The spectra were processed with the MestReres23 program (Mestrelab Research).
UV/VIS spectroscopy
Spectra were recorded with an Agilent Cary 60 UV/VIS spectrophotometer in the range of 280–780 nm.
Potentiometric titration
The potentiometric acid–base titration was carried out with the automatic titrator MT T70 (Mettler Toledo). Before the neutralization titration, the samples were dissolved in deionized water (R 18.18 MΩ cm) (c = 10 mg/mL) and dialyzed in two steps: (a) aqueous solution of NaOH (c = 1 × 10−6 mol/L, pH = 8)—at this stage the amino groups are deprotonated, thus reducing the solubility of chitosan; (b) by demineralized water, that is, at this stage an excess of hydroxyl and sodium ions were removed, together with possible other ions resulting from the chemical or enzymatic modification. Some 0.1 g of the sample (eugenol has no amino groups and was therefore excluded from the analysis) was placed in a titration beaker and 30 mL of ultrapure boiled milli-Q water was added. To this solution, 1 mL potassium chloride (c(KCl) = 0.1 mol/L) and 3 mL hydrochloric acid (c(HCl) = 0.1 mol/L) were added additionally and titrated in the pH range between 2 and 11. Using the combined glass electrode Mettler Toledo DG-117 (Mettler Toledo, USA), the potential was measured as a function of the titrant volume. During the titration, nitrogen was added to each solution to achieve an inert atmosphere. From the data obtained, the proportion of amino groups was calculated according to the following equation
Folin–Ciocalteu method
The total phenolic compounds (TPCs) was determined as follows: 0.250 mL Folin reagent (w/v, 1:2) was added to 0.4 mL of a sample solution and stirred well. After 5 minutes, 0.250 mL sodium carbonate (Na2CO3, 20% (m/m)) was added, and supplemented with milli-Q water to a final volume of 2 mL. After 40 minutes of incubation at room temperature the absorbance was read off at λ = 765 nm in suitable cuvettes. A standard solution of GA was used to establish the calibration curve.42,43
The mass concentration of the TPCs was calculated using the following equation
Functionalization of viscose
Efficiency of functionalization
Results and discussion
Evaluation of eugenol-modified chitosan
The scheme of the proposed formation of chemically eugenol-modified chitosan (CH/EUG (CHEM)) is shown in Figure 2, and adapted from Jung et al. 34
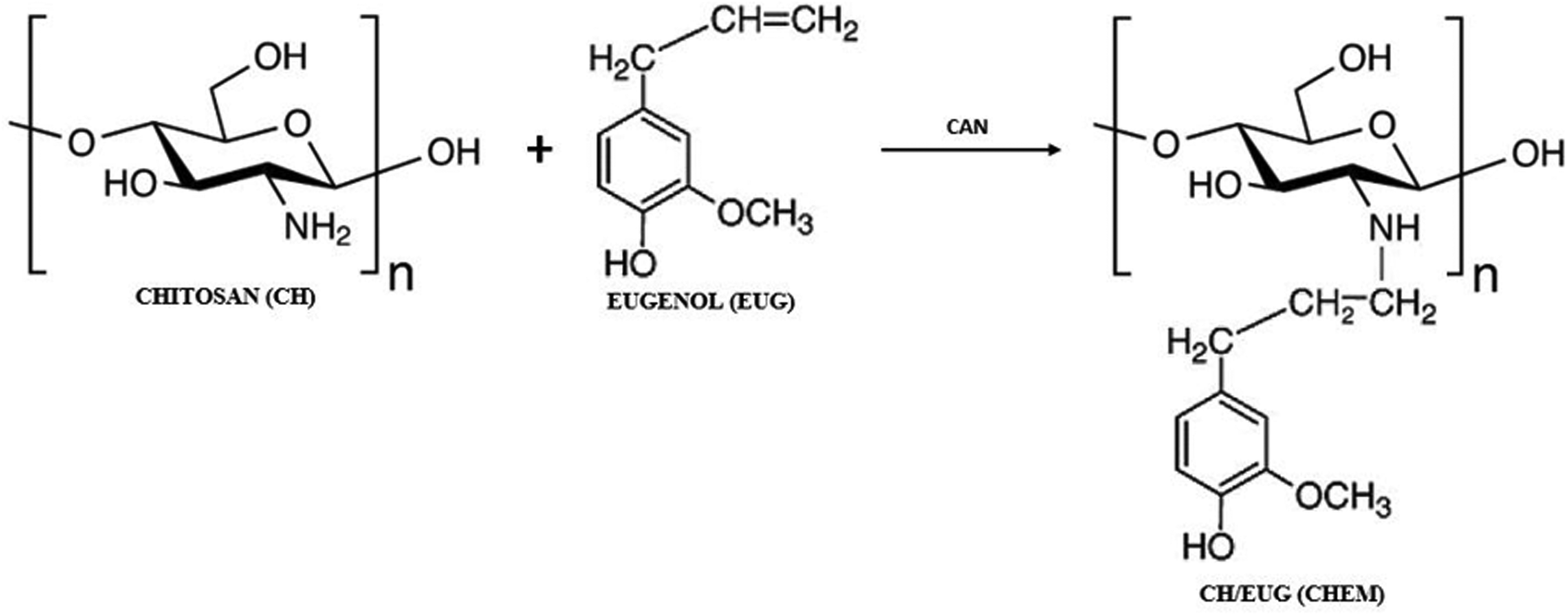
Proposed reaction mechanism of chemically eugenol-modified chitosan (CH/EUG (CHEM)).
Figure 3 shows the proposed mechanisms of enzymatically eugenol-modified chitosan (CH in combination with EUG, i.e., (CH/EUG (ENZ)), Schiff base (C=N) and Michael's addition (C-NH). Besides the two predicted products presented in Figure 3 (graft copolymers as a result of Schiff-base and Michael-additional mechanisms), it is also possible that a product is formed based on the electrostatic interaction between chitosan and eugenol (Coulumb interactions). 46
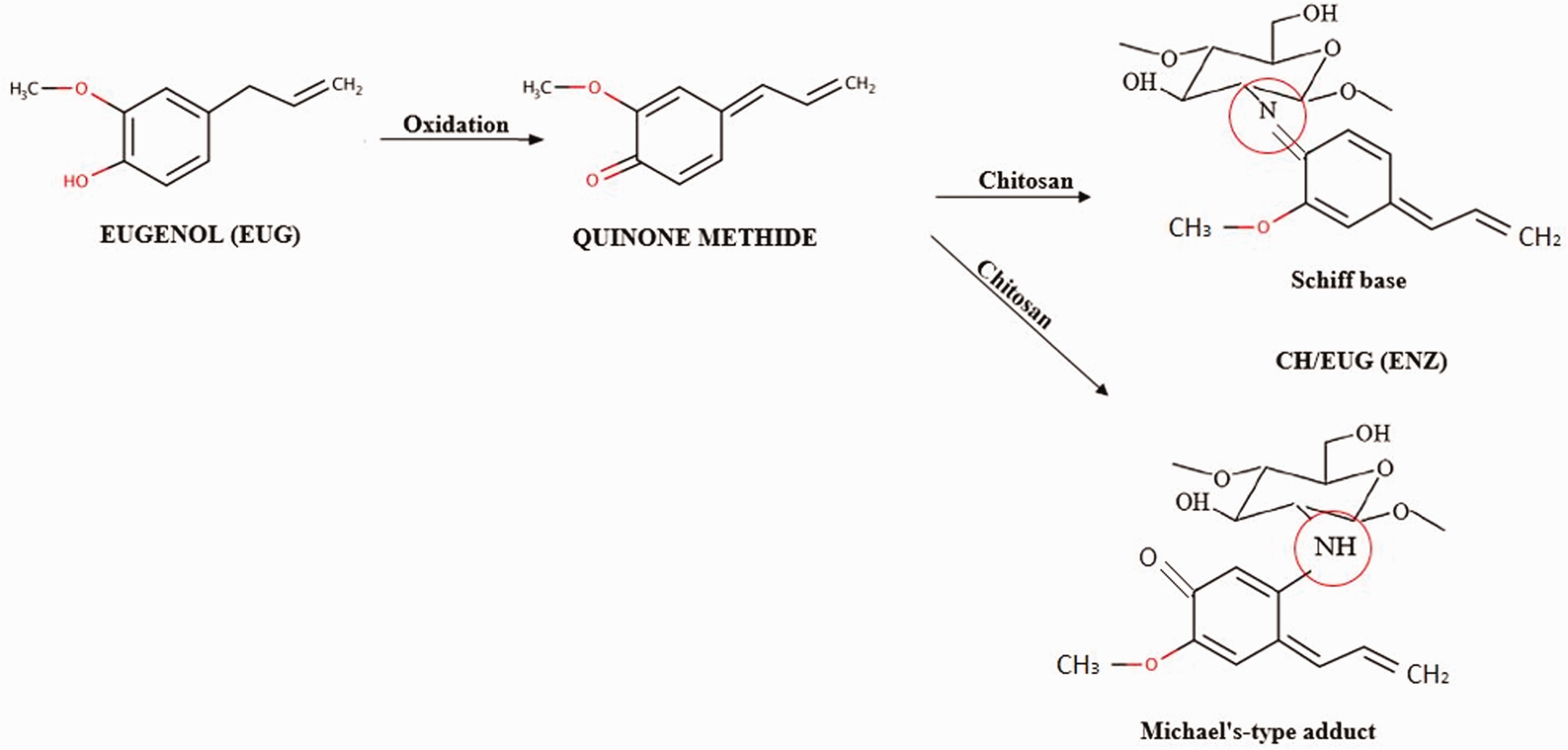
Proposed reaction mechanism of enzymatically eugenol-modified chitosan (CH/EUG (ENZ)).
ATR FT-IR spectroscopy
The ATR FT-IR spectrum of chitosan (powder) as a reference (CH) (Figure 4) shows characteristic signals at wave numbers: 3289, 1647, 1587 and 1419 cm−1. The intense signal at 3289 cm−1 is attributed to the O–H stretching vibrations of the hydroxyl (–OH) groups, superimposed with the symmetrical and asymmetrical stretching vibrations of the N–H bonds. The characteristic signals at 1647 and 1587 cm−1 correspond to the carbonyl (C=O) group (amide I) and the bending of the N–H bond (amide II) of the primary amino group of chitosan. At 1419 cm−1 a characteristic signal of the CH–OH bond is seen, and a CH2–OH bond appears at 1375 cm−1. The signal at 1151 cm−1 corresponds to the asymmetric stretching of the C–O–C bond, and the signals at 1060 and 1027 cm−1 are due to the presence of the C–O bond. Typical signals of eugenol (EUG) as a reference are seen at wave numbers of 3505, 1637, 1613, 1514 and 1432 cm−1. The signal at a wave number of 3505 cm−1 corresponds to the O–H stretching vibrations of the hydroxyl groups (–OH) bound to the benzene ring. The signals at wave numbers 1637 and 1514 cm−1 represent the aromatic content, and the signal at 1432 cm−1 belongs to the CH2 = vinyl group of eugenol. In the ATR FT-IR spectrum of the CH/EUG (CHEM), the disappearance of the signal at wave number 1647 cm−1 (associated with the free amino groups (–NH2) of chitosan) and the signal at 1432 cm−1 (characteristic of the vinyl group of eugenol) confirmed the covalent bonding of the vinyl group of eugenol to the amino group of chitosan (as shown in Figure 4). The ATR FT-IR spectra of eugenol-modified chitosan obtained by the enzymatic method, CH/EUG (ENZ), shows a broad signal at the wave number of 3342 cm−1, which corresponds to the stretching vibrations of the O–H bonds, and overlaps with the oscillation of the N–H bonds. The signals at wave numbers of 1643 and 1375 cm−1 are assigned to the vibrations of the carbonyl (C=O) groups (amide I) and CH2–OH bonds. Furthermore, a signal at a wave number of 1149 cm−1 can be seen, which is due to the asymmetric stretching of the C–O–C glucoside bonds. The ATR FT-IR spectra of enzymatically eugenol-modified chitosan (CH/EUG (ENZ)) do not show structural elements showing the formation of a Schiff base. At a wave number of about 1650 cm−1 there is an overlap of the signal characteristic of the vibrations of the C=N (imino) bond, and the signal characteristic of amide I in the chitosan molecule. However, the shift of the amide II signal in the chitosan molecule to the wave number of 1561 cm−1 is also an indication of oscillations of the N-H bond (Figure 4). The shift of the primary amine to the secondary coincides with the predicted reaction mechanism of Michael addition.
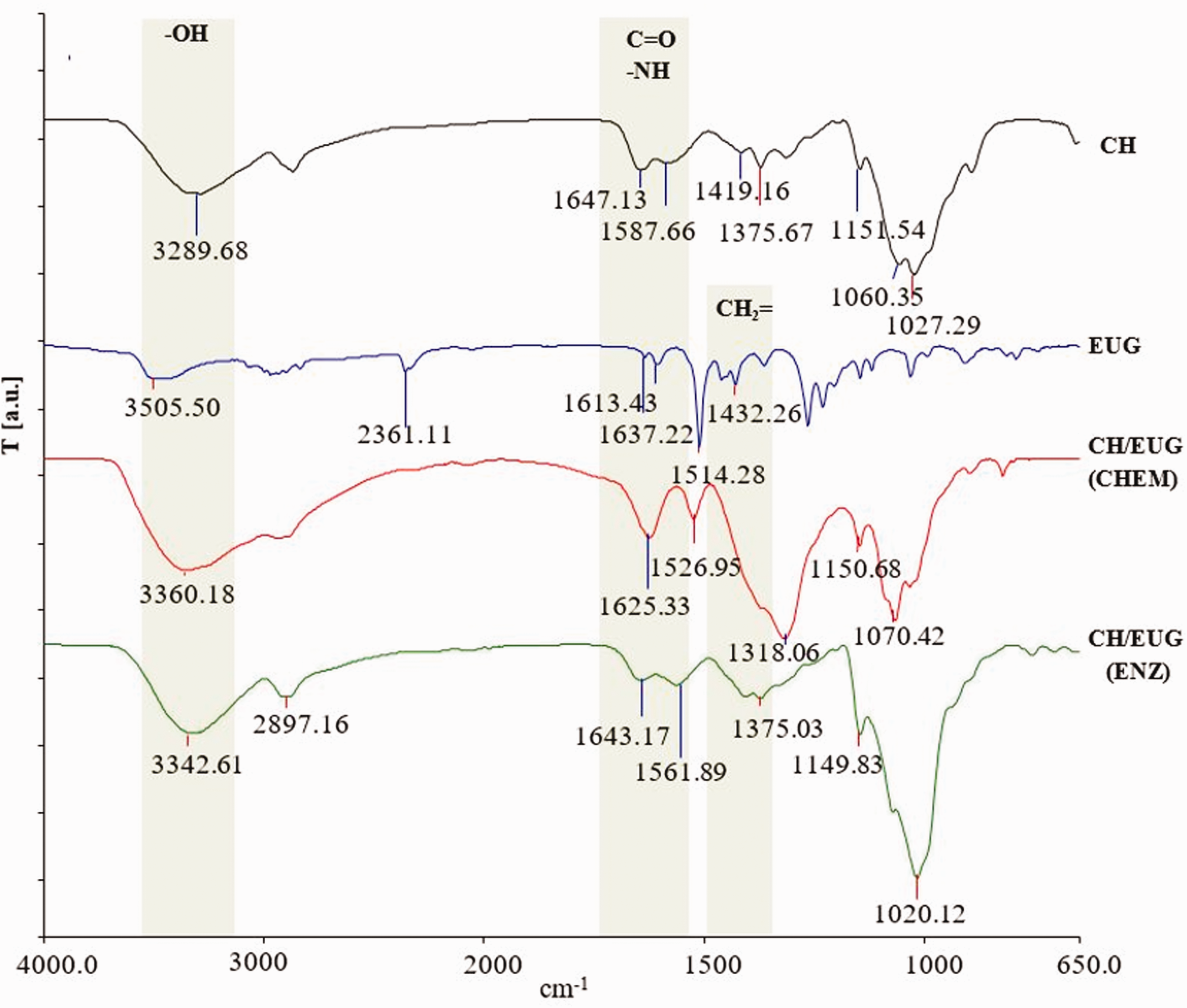
Attenuated total reflection Fourier transform infrared spectra of chitosan (CH) and eugenol (EUG) as references and spectrum of the chemically (CH/EUG (CHEM)) and enzymatically eugenol-modified chitosan (CH/EUG (ENZ)).
1H NMR spectroscopy
1H NMR spectra were recorded to confirm the chemical structures of the newly formed modified chitosan further. The 1H NMR spectra of CH and EUG are shown in Figure 5. The arrows show the signals for hydrogen atoms bound to the carbon atoms of the functional groups that are important for the binding mechanism. For chitosan, this is the –CHNH2 group at the resonance peak of 3.3 ppm, and for eugenol it is the protons of the hydroxyl (at resonance peak of 5.5. ppm) and vinyl groups (5.1 and 5.9 ppm).
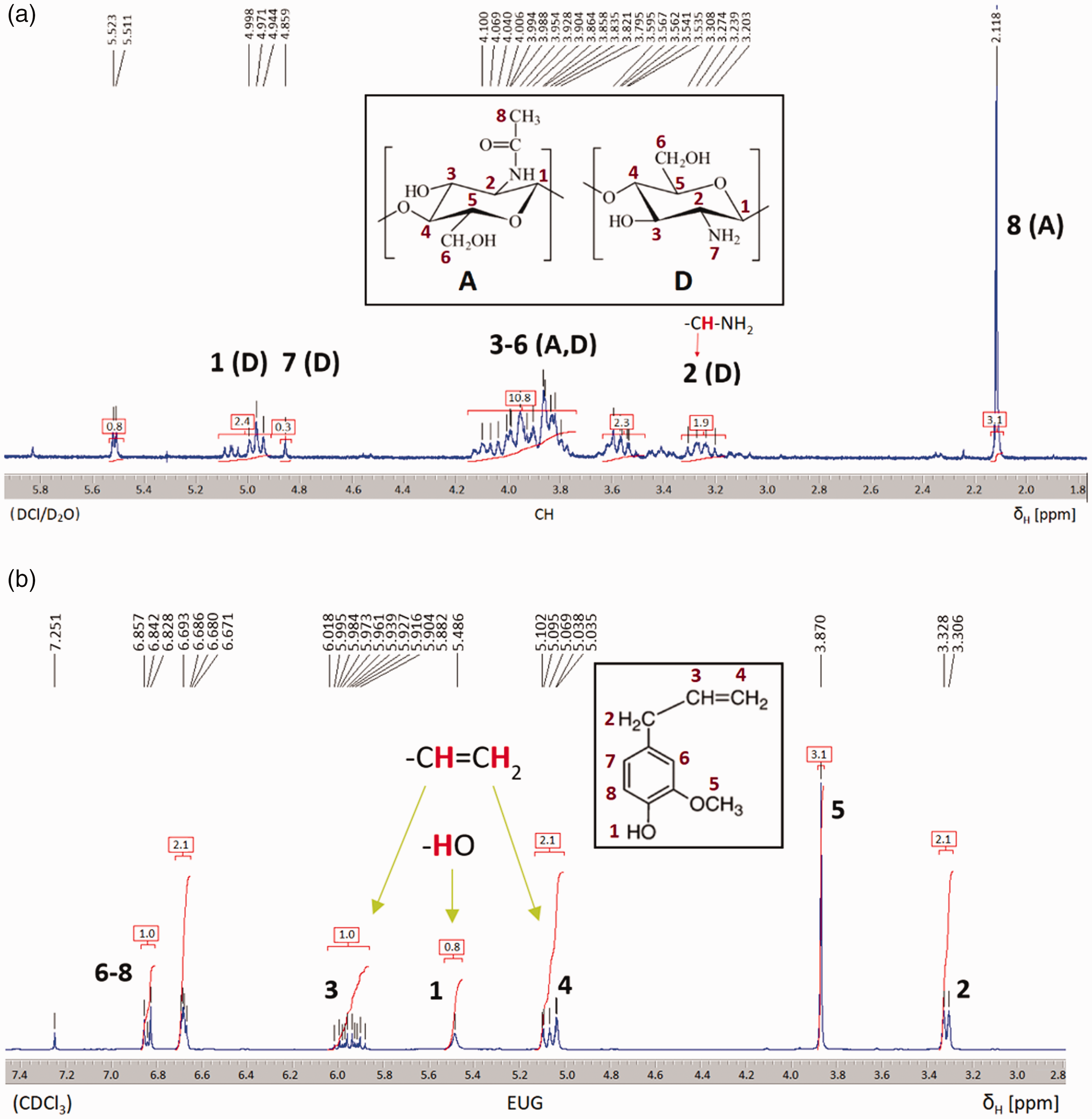
Proton nuclear magnetic resonance spectra of chitosan (CH) (a) and eugenol (EUG) (b).
The 1H NMR spectra of the chemically and enzymatically eugenol-modified chitosan are shown in Figure 6. The 1H NMR spectra of the chemically eugenol-modified chitosan CH/EUG (CHEM) show a shift in the signals (H1, H2) of chitosan to lower ppm values, a decrease in the signal at 4.8 ppm belonging to the -NH2 groups of chitosan and the disappearance of the signals belonging to the eugenol–vinyl group at 5.0 and 6.0 ppm. The results are consistent with the results of the ATR FT-IR spectroscopy, and further confirm the success of EUG binding to the chitosan polysaccharide chain. The 1H NMR spectra of the enzymatically eugenol-modified chitosan CH/EUG (ENZ) show the disappearance of the signal belonging to the –NH2 groups of chitosan (4.8 ppm), and the appearance of weak signals due to the vinyl group of eugenol at 5.0 and 6.0 ppm.
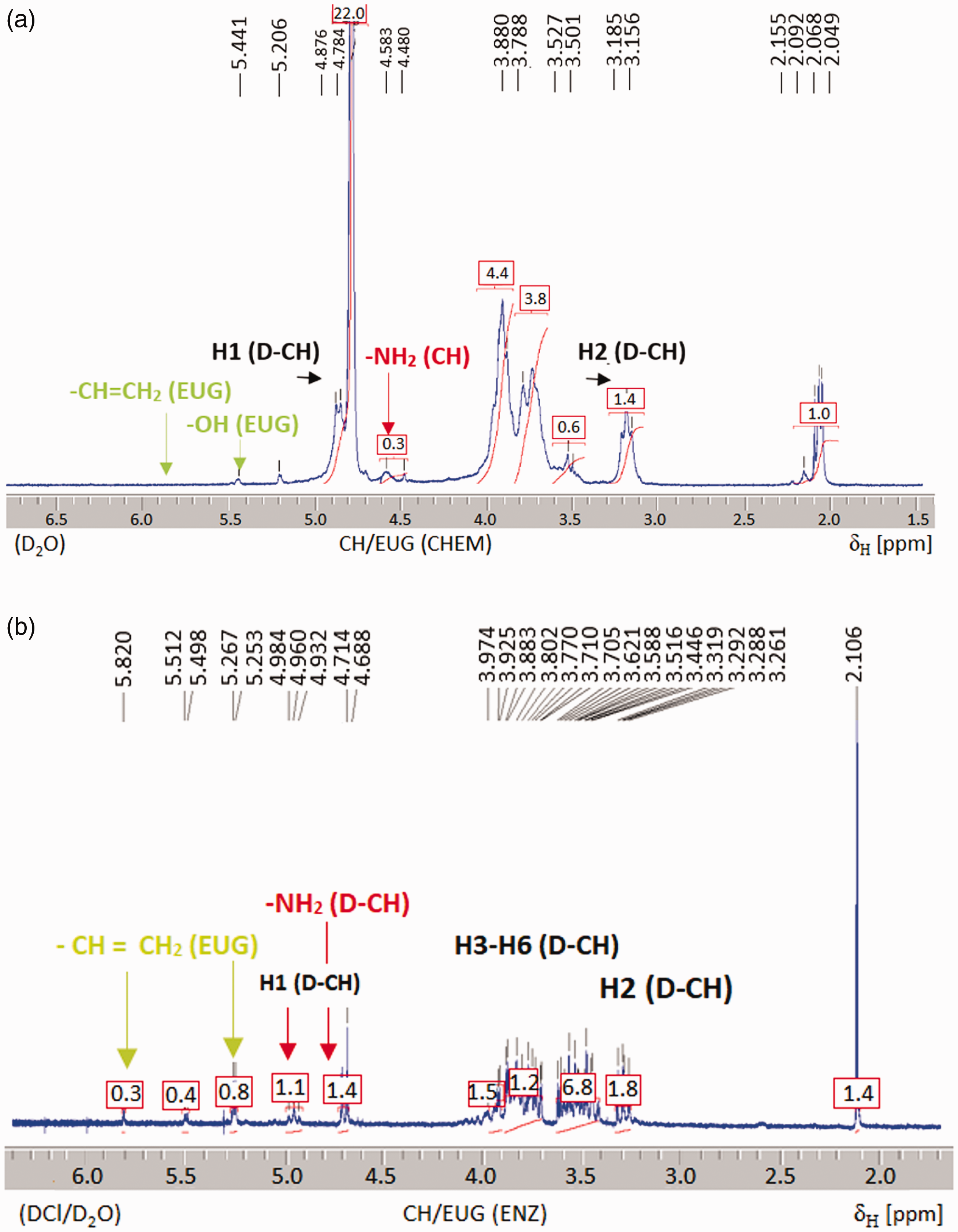
Proton nuclear magnetic resonance spectra of the chemically (CH/EUG (CHEM)) (a) and enzymatically (CH/EUG (ENZ)) (b) eugenol-modified chitosan.
UV/VIS absorption spectroscopy
The enzymatic oxidation of phenolic compounds (EUG) to reactive quinones and their subsequent non-enzymatic reaction with the primary amino groups of chitosan were monitored by UV/VIS absorption spectroscopy. As shown in Figure 7, significant changes can be seen, based on the UV/VIS spectra of the solutions taken during the synthesis process of enzymatic modification.
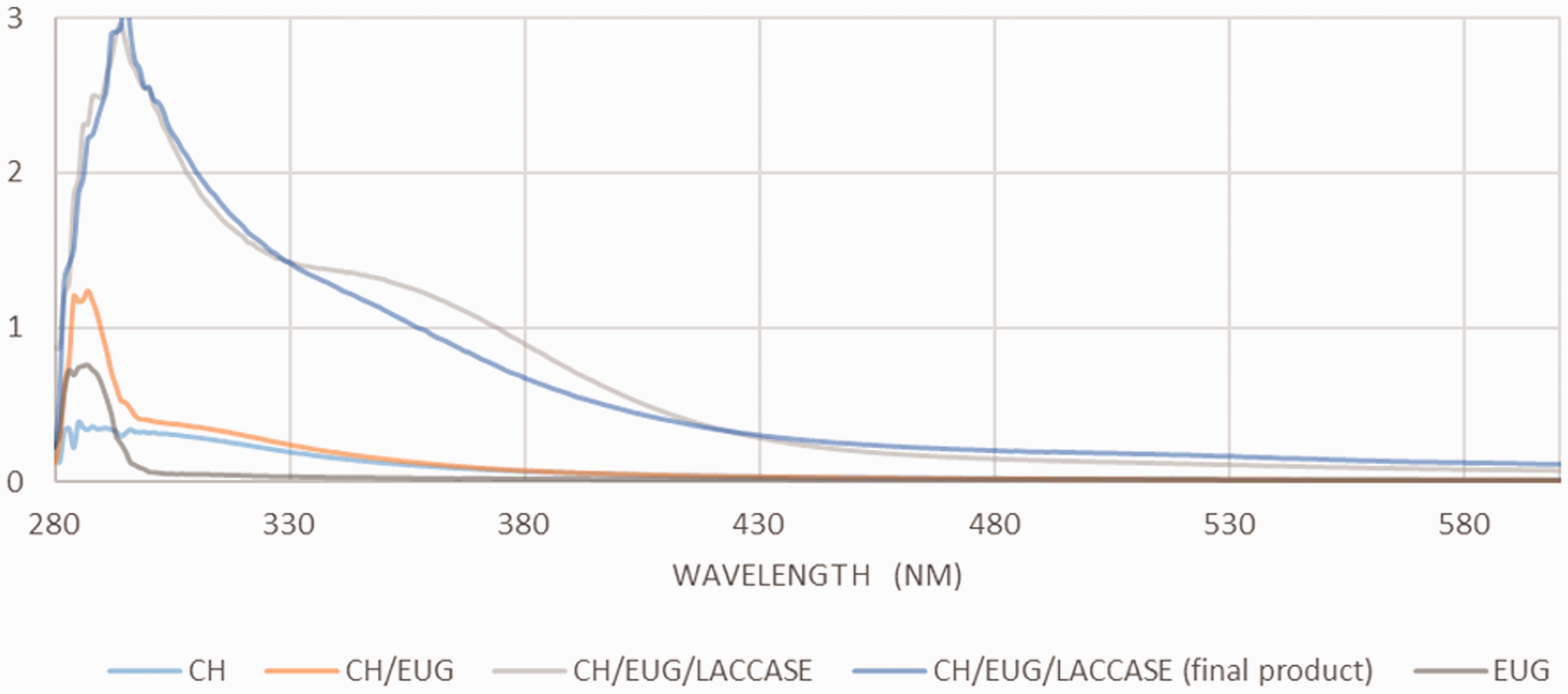
Ultraviolet-visible spectrum of enzymatic oxidation of eugenol and subsequent non-enzymatic reactions with the primary amino groups of chitosan.
Initially, the reference solutions of CH and EUG were monitored time-dependently. In this way, a possible spontaneous hydrolysis was verified, and the possibility was excluded of an increase in absorption in the further synthesis process. Subsequently, the chitosan solution was mixed with a corresponding amount of the phenolic EUG substance, and the absorption spectrum was recorded again. No changes were observed with the reference solutions alone, or with the chitosan solution in combination with the phenolic substances. Subsequently, a corresponding amount of the enzyme laccase was added; in this way, the activity of laccase could be confirmed for the oxidative polymerization of the phenolic compounds. In the case of CH/EUG (ENZ), a shift of the peak at 284 nm to a wavelength of 290 nm was observed (Figure 7). In addition, a new peak at 350 nm appeared immediately after the addition of the enzyme laccase. As the reaction progressed, this peak disappeared, and the color of the solution changed from colorless to dark yellow. The UV/VIS absorption peaks of the phenolic reference compounds (EUG) were assigned to the π benzene ring system. 47 The position of these peaks depends on the ionization state and the type of ionization present, that is, electron donation or deprivation. The shift and decrease of the peaks were attributed to the lower amount of energy required for the transition of π-π* and n(unbound)-π * electrons in the process of the coupling reaction, that is, the integration of phenolic compounds into the chitosan polysaccharide chain.
Potentiometric titration
In addition to the techniques already used (ATR FT-IR, 1H NMR), pH potentiometric titration was used to determine the proportion of the remaining accessible amino groups due to the modification of CH with EUG. The lower proportion of accessible amino groups of chemically and enzymatically eugenol-modified chitosan (CH/EUG (CHEM), CH/EUG (ENZ)) was to be expected, considering the binding success, since amino groups are assumed to be potential binding sites for phenolic compounds. The results (average of three measurements) of the proportion of amino groups (mmol/g) for CH and for the chemically and enzymatically eugenol-modified chitosan (CH/EUG (CHEM), CH/EUG (ENZ)) are shown in Figure 8.
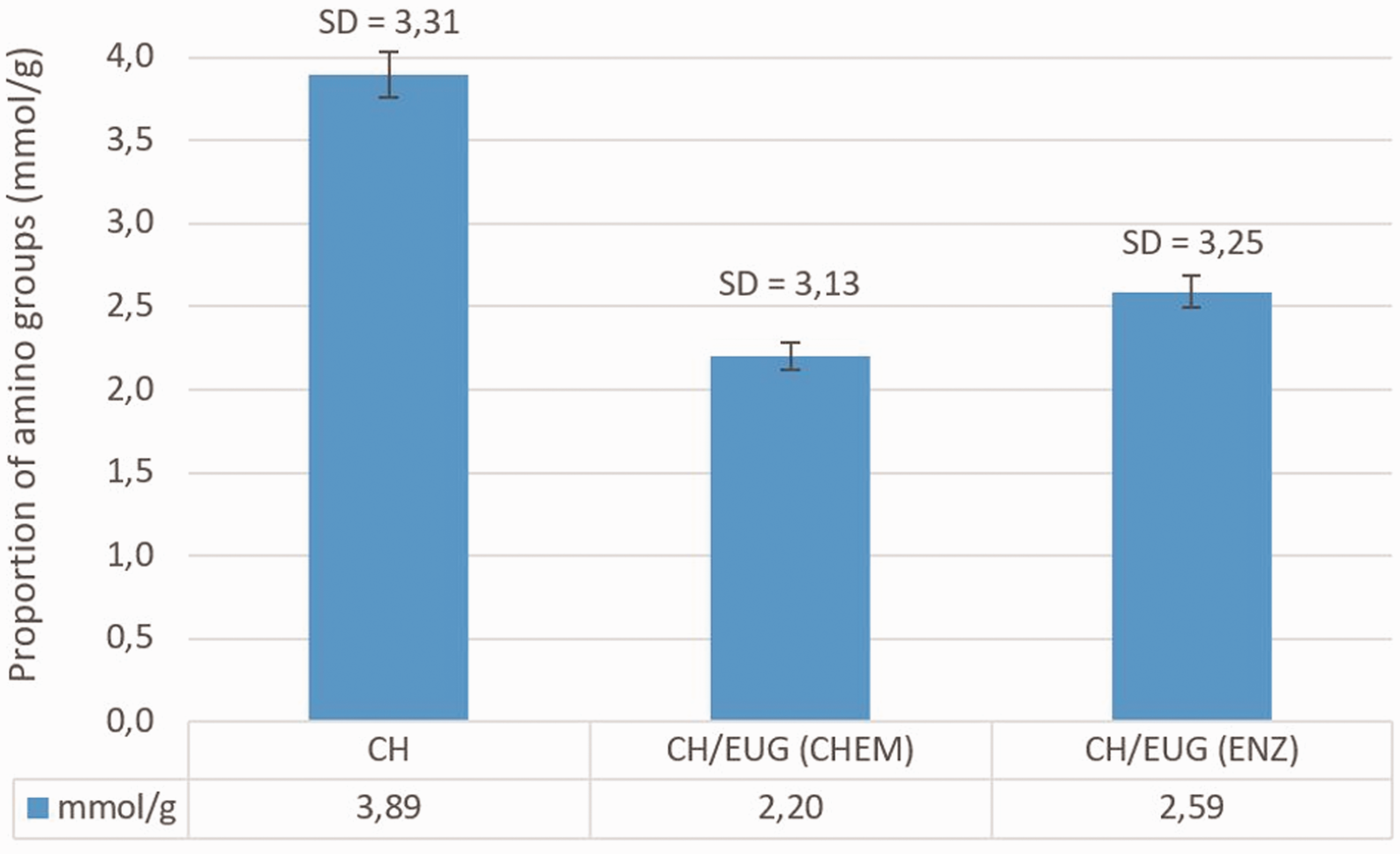
Proportion of amino groups of chitosan (CH) and of chemically and enzymatically eugenol-modified chitosan (CH/EUG (CHEM), CH/EUG (ENZ)) determined by pH potentiometric titration. The values are average values of three measurements and the standard deviation is in the range of 3.1–3.3 mmol/g.
Figure 8 shows the highest content of amino groups in the case of chitosan (CH), followed by a reduction of these groups in CH/EUG (ENZ), obtained by the enzymatic method, and a further reduction of these groups by CH/EUG(CHEM), obtained by the chemical method. The results of the potentiometric titration confirm the reduction of the number of available primary chitosan amino groups when modified chemically and enzymatically, thus proving the chemical binding or Coulomb interactions (possibility by enzymatically grafting) of the phenolic compounds to the primary chitosan amines.
Antioxidant efficacy
Folin–Ciocalteu
The Folin–Ciocalteu (FC) method refers to the content of TPCs, expressed in µg/mL of GA, and states that a higher content of TPCs increases the antioxidant activity. Considering the results of the enzymatic and chemical modification, the highest antioxidant activity, associated with a higher content of total phenols, was found in the chemically eugenol-modified chitosan (CH/EUG (CHEM)) (4.35 µg/mL TPC), where, after modification, a phenyl was still attached to a hydroxyl group, which was no longer the case with enzymatic modification (Figures 2 and 3). The available number of phenolic compounds in the chitosan modified enzymatically by eugenol clearly indicated the possibility of an electrostatic interaction between the chitosan and eugenol. Otherwise, eugenol (52.61 µg/mL TPC) itself has the best antioxidant activity. Chitosan shows no noticeable antioxidant activity (0.64 µg/mL TPC), as emphasized previously (Figure 9).
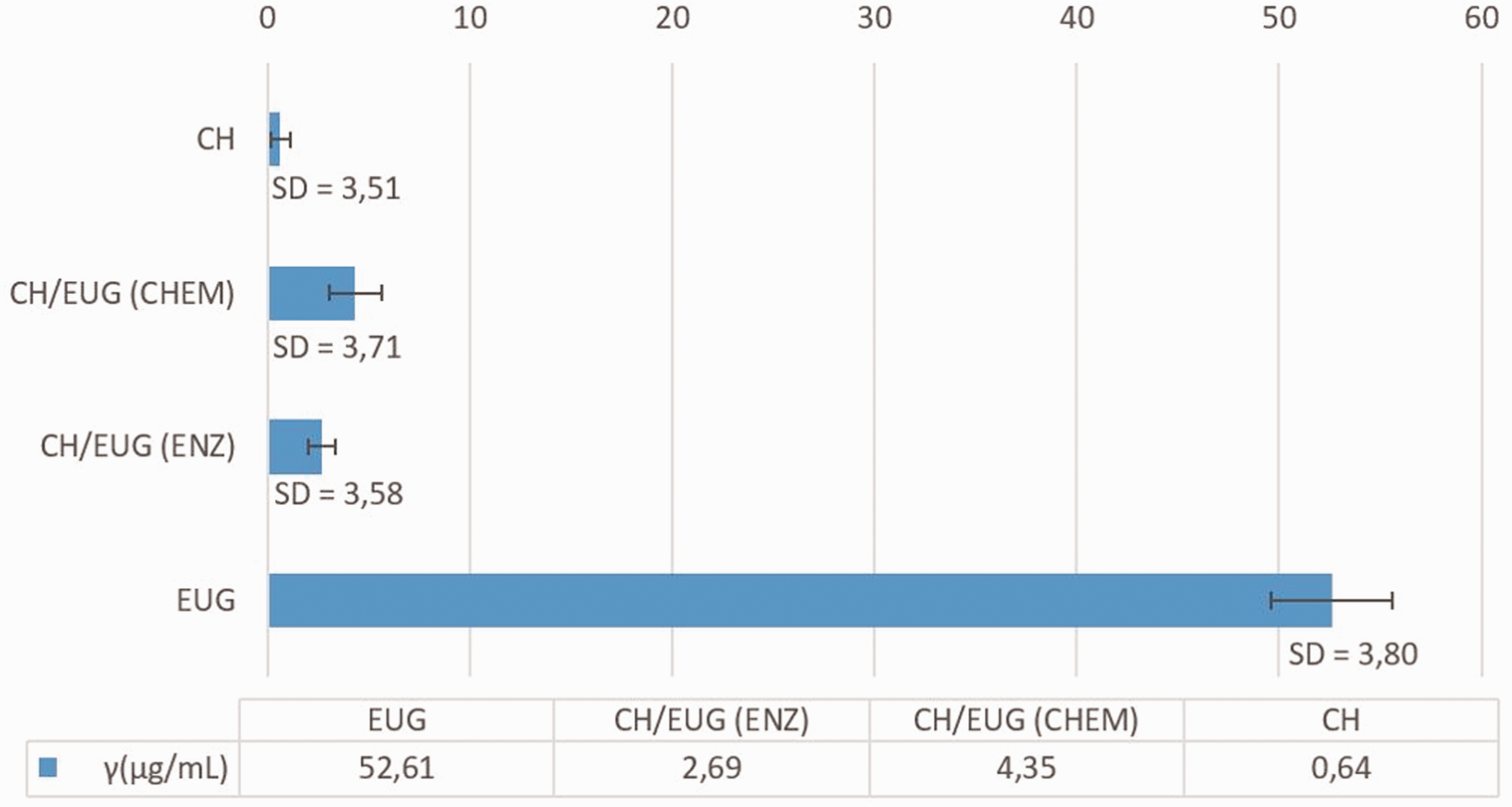
Content of total phenolic compounds expressed in µg/mL of gallic acid. The values are average values of three measurements and the standard deviation is in the range of 3.5–3.8 µg/mL.
DPPH• analysis:
The percentage of free DPPH• for EUG was 2.77% and the calculated inhibition of free DPPH• was 97.23% (Figure 10). As expected, the content of free DPPH• was highest in the chitosan solution (98.62%); the calculated free radical inhibition was 1.38%, since chitosan does not belong to the group of antioxidants.4–6 In the DPPH• analysis, the hypothesis was made that the integration of natural substances (i.e., eugenol in the chitosan backbone) contributed to the antioxidant activity of chitosan. It was found that the eugenol-modified chitosan obtained by chemical and enzymatic modification had a certain antioxidant activity. In CH/EUG (CHEM) an antioxidant activity of 30.69% was observed, while in CH/EUG (ENZ) a radical inhibition of 30.73% was found. Nevertheless, the antioxidative activity was lost compared to pure EUG, while the modified chitosan products showed an improved activity compared to chitosan itself. This was somehow to be expected, while, in the modification process, some reactive eugenol (phenolic) species were lost by the reaction with chitosan. It should be noted that these values were given at a time of 30 minutes. This effect can be improved by extending the time, which is quite often the case for natural substrates. On this basis, it can be concluded that the incorporation of natural substances such as EUG into the CH backbone has an inhibitory effect on the antioxidant properties compared to pure eugenol solutions, but enhances the antioxidant activity of eugenol-modified chitosan. Unfortunately, the determination of the antioxidative (reduction) potential of antioxidants with the DPPH• reagent alone was not completely sufficient. Not all antioxidants act as reducing agents but differ in their mechanism of action. As direct reducing agents they can remove radicals and other reactive species by chelating metal ions and prevent their formation. Nevertheless, the results of eugenol, CH/EUG (CHEM) and CH were consistent with the FC method. Some discrepancies were seen in CH/EUG (ENZ), where the FC method showed about half the TPCs of CH/EUG (CHEM). It can be concluded that, besides phenolic groups, some other parameters, such as structure, conformation, surface tension, etc., may have an influence on radical inhibition. For further evaluation of the antioxidative activity, the method for reduction of iron ions was, therefore, also part of the research.
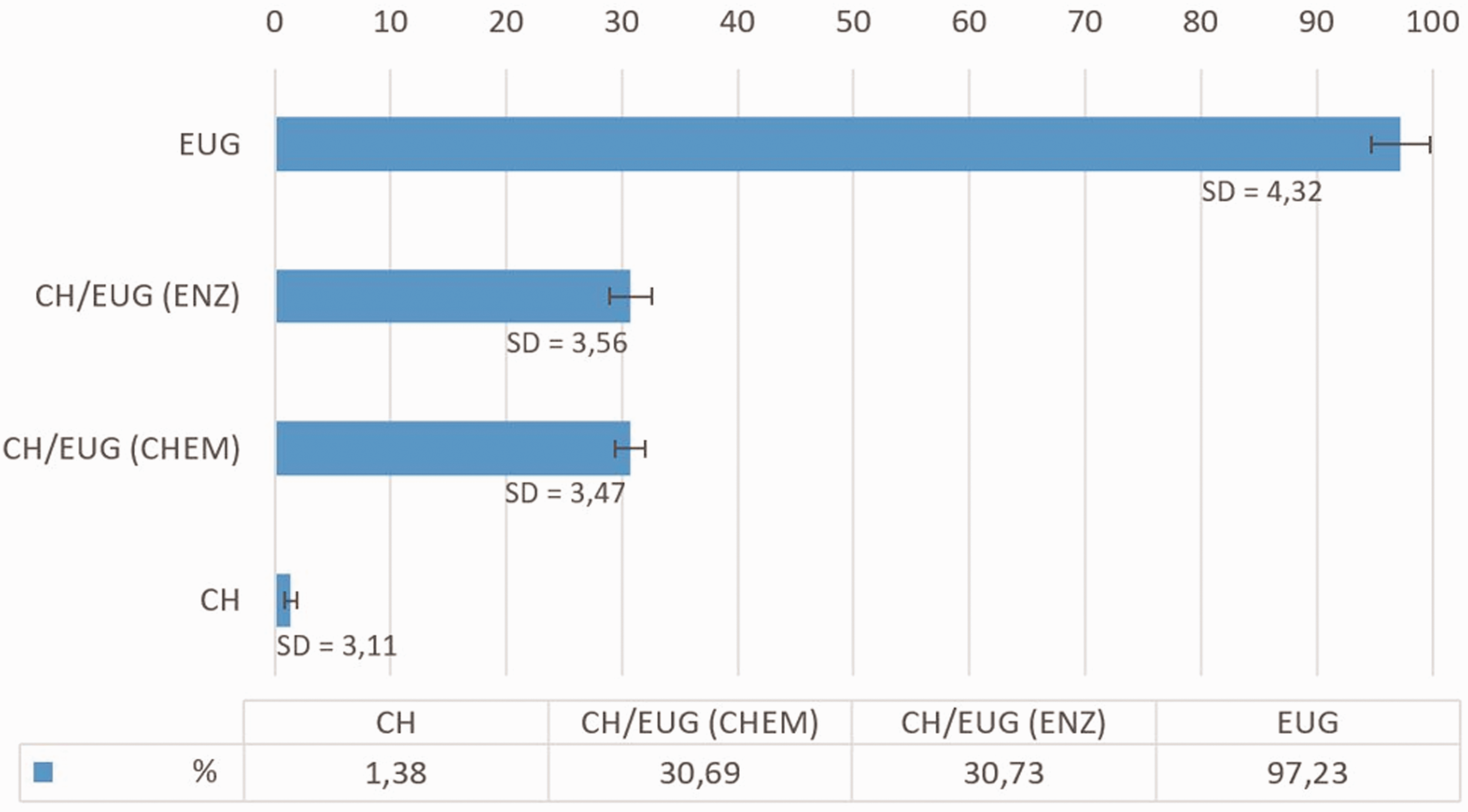
Proportion of a captured DPPH• at 30 minutes. The values are average values of three measurements and the standard deviation is in the range of 3.1–4.3%.
Fe3+ iron reduction capacity
The analysis of Fe3+ iron reduction capacity is one of the most important indicators of potential antioxidant efficacy. In this analysis, Fe3+ iron ions are reduced to Fe2+ in the presence of a reducing agent (antioxidant), which corresponds to a color change of the tested solution, measured at a wavelength of 700 nm. The results are shown in Figure 11.
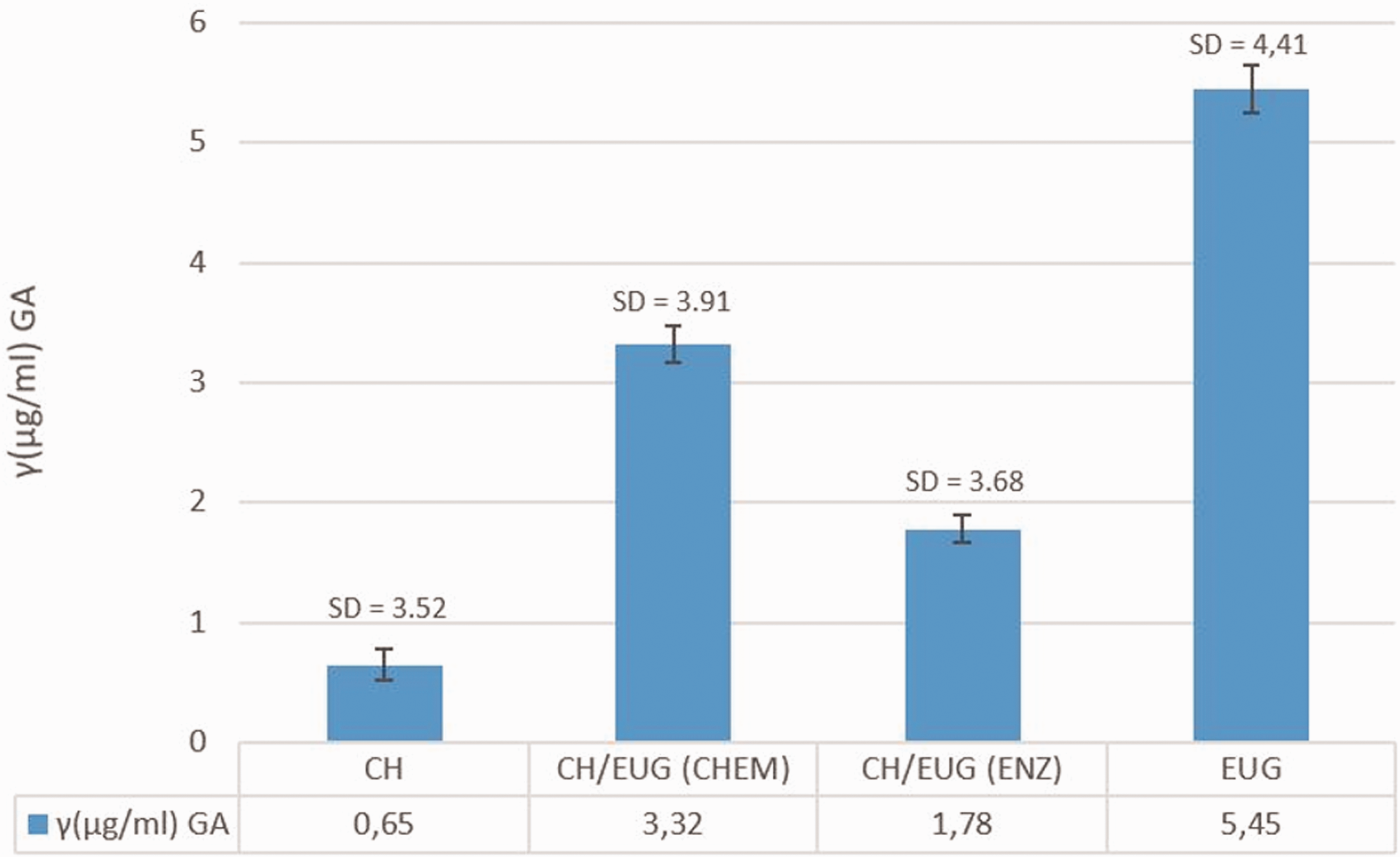
Fe3+ iron reduction capacity. The values are average values of three measurements and the standard deviation is in the range of 3.5–4.4 µg/mL. GA: gallic acid.
Among the newly formed eugenol-modified chitosans, CH/EUG (CHEM) showed the best reduction capacity, with an average reduction capacity of 3.32 µg/mL of GA, followed by CH/EUG (ENZ) with an average reduction capacity of 1.78 µg/mL of GA. The trend between these two samples was like the FC method. CH showed the worst reduction capacity of Fe3+ ions, and again indicated a poor antioxidant effect of chitosan. The obtained results of the reduction capacity supported the FC method.
Evaluation of viscose functionalized by eugenol–modified chitosan
ATR FT-IR spectroscopy
The results of ATR FT-IR spectroscopy for reference viscose (VIS) and viscose functionalized by CH, CH/EUG (CHEM) and CH/EUG (ENZ) are shown in Figure 12. VIS characteristic signals can be seen at 3334, 2914–2848, 1471 and 1020 cm−1. The signal at 3334 cm−1 was attributed to the O–H tensile vibrations of the hydroxyl groups (–OH). The signals at 2914–2848 cm−1 corresponded to the C–H tensile vibrations and the signals in the spectral range between 1500–800 cm−1 belonged to different C–H, O–H, C–O and C–O–C vibrations of the glucoside ring. 48 Based on the ATR FT-IR spectra of functionalized viscose with CH/EUG (CHEM) and CH/EUG (ENZ) (spectrum VIS CH/EUG (CHEM) and spectrum VIS CH/EUG (ENZ)), it was possible to confirm the functionalization of viscose with eugenol-modified chitosan, since it was possible to see increased signals in the range around 3334 cm−1 and new signals at 1615 and at 1150 cm−1, which are also shown in the ATR FT-IR spectra of the two samples CH/EUG (CHEM) and CH/EUG (ENZ). Unfortunately, there was no evidence of chemical (covalent) bounds or ionic interactions among adsorbate/CH/EUG (CHEM), CH/EUG (ENZ) and adsorbent, VIS, nor of significant changes in the infrared (IR) spectrum due to hydrogen bonds. However, our earlier study showed that chitosan is bound to cellulose mainly by physical interactions, which was also assumed in this study. 49
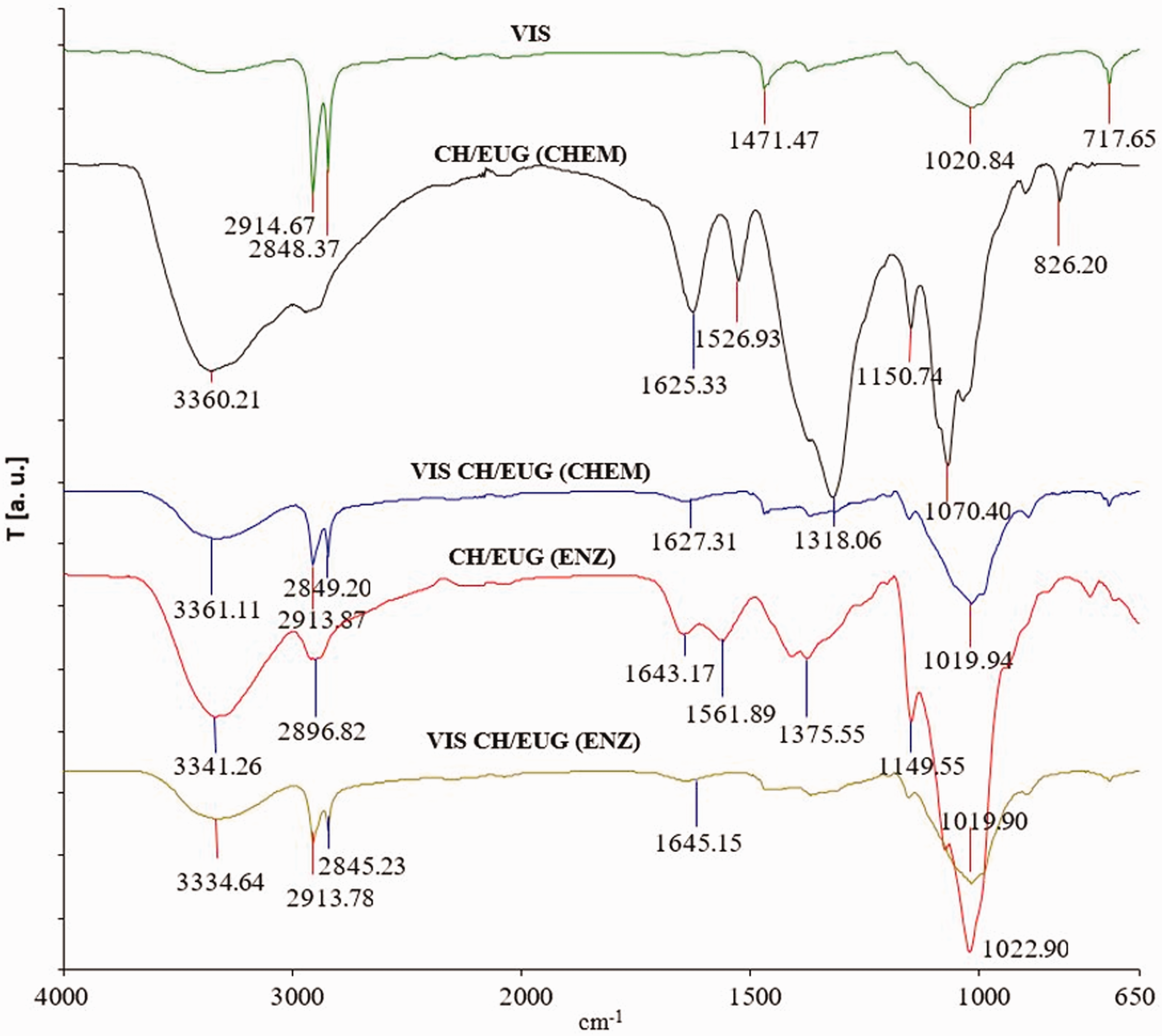
Attenuated total reflection Fourier transform infrared spectra of VIS, CH/EUG (CHEM), VIS CH/EUG (CHEM), CH/EUG (ENZ) and VIS CH/EUG (ENZ).
Scanning electron microscopy
Figure 13(a) shows an image of a reference viscose showing the typical viscose fiber morphology, with its characteristic longitudinal channels running parallel to the fiber axis. This morphology is the result of the wet formation of viscose fibers made of cellulose xanthate. 50

Scanning electron microscopy image of (a) viscose (VIS), (b) viscose functionalized with chitosan (VIS CH), (c) viscose functionalized with CH/EUG (CHEM) and (d) CH/EUG (ENZ).
The image (Figure 13(b)) shows that the surface of viscose becomes somewhat rougher when chitosan is applied and covers the surface of the fibers with a thin, evenly distributed layer. The roughness is due to the deposited chitosan particles; their length is a few microns. The layers formed on viscose by the adsorption of solutions from CH/EUG (CHEM) show a completely different surface morphology (Figure 13(c)). When applying the prepared eugenol-modified chitosan solution as a coating, two morphological phenomena can be highlighted: (a) formation of a layer of smaller precipitated particles and (b) formation of discontinuous polymer films, which are preferably adsorbed in the longitudinal channels of the fibers. In most cases, the spaces in the fiber channels represent an area where different treatment solutions accumulate to a large extent; in the present case, the spaces between the channels serve as anchorage for the treatment solution of eugenol-modified chitosan adsorbed on the viscose.
Changes in the surface morphology of viscose after surface modification with a solution of enzymatically eugenol-modified chitosan CH/EUG (ENZ) are shown in Figure 13(d). SEM images of the functionalized viscose surface show clearly the changes caused by the deposition of undissolved or suspended particles present in the aqueous suspension of the enzymatically eugenol-modified chitosan. Compared to the adsorption of the chemically eugenol-modified chitosan, the adsorbed layer shows a higher degree of particle agglomeration, which is also less evenly distributed over the fiber surface, than the chemically eugenol-modified chitosan product. The sample CH/EUG (ENZ), in turn, indicates the occurrence of preferential adsorption of particles in the interstices of the longitudinal channels. The results of the SEM analysis of the functionalized viscose compared to the reference sample clearly showed the presence of deposits of the treatment bath components on the viscose. The surface of the viscose layers of the deposited eugenol-modified chitosan product is rougher compared to the viscose functionalized with chitosan solution only. There were also noticeable differences between the surface layers of chemically and enzymatically eugenol-modified chitosan products. The chemical and enzymatic modification also affected the morphological changes of the functionalized viscose, which were otherwise clearly visible in the SEM images.
Acid Orange VII method
The number of the remaining accessible amino groups of chitosan on non-woven viscose determined by the Acid Orange VII method is shown in Figure 14.
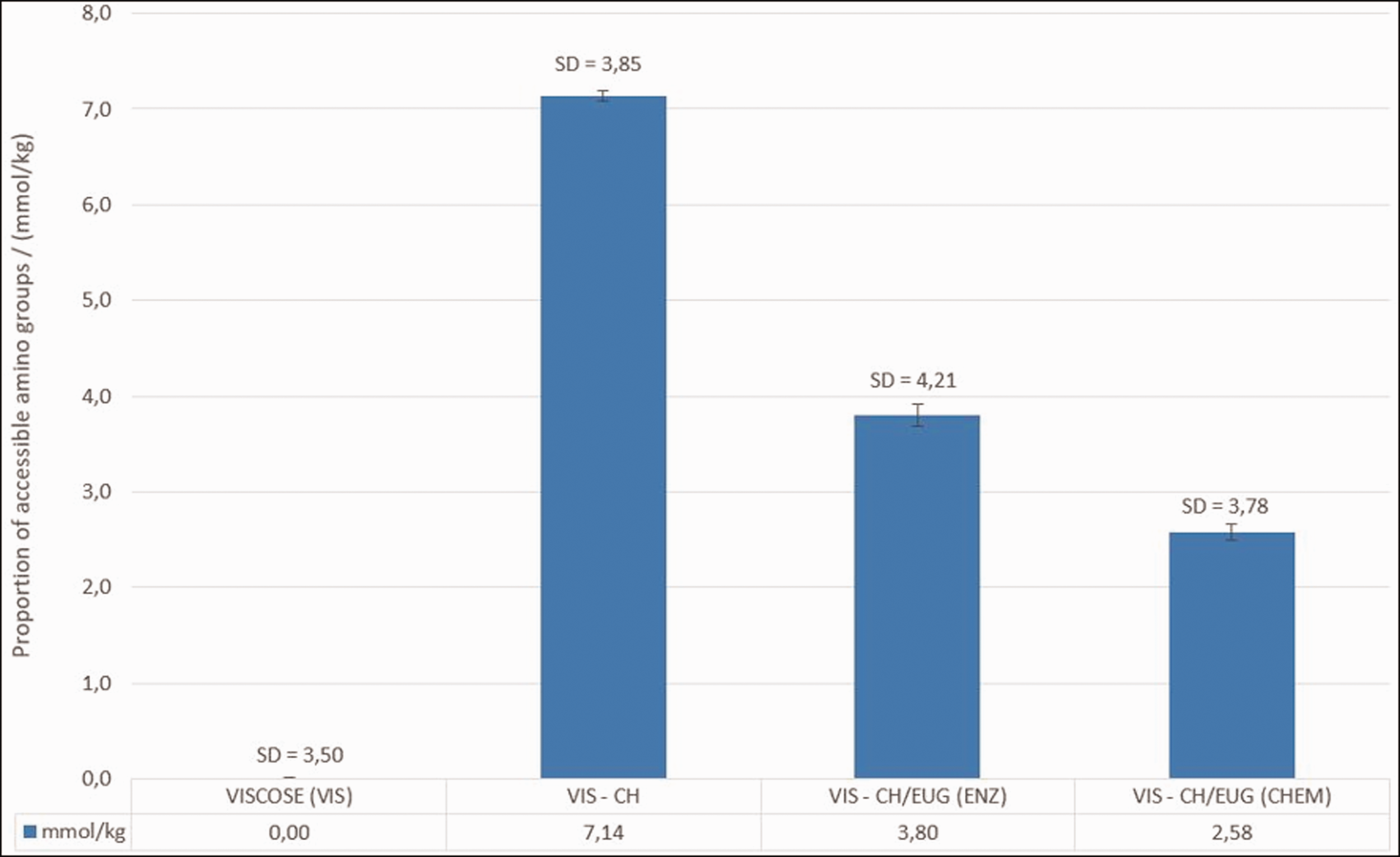
The number of remaining accessible amino groups of reference viscose (VIS) and viscose functionalized with separate functionalization baths (CH/EUG (ENZ) and CH/EUG (CHEM)), determined by using the Acid Orange VII method. The results presented are average values of three measurements. The standard deviation is in the range of 3.5–4.2 mmol/kg.
As can be seen from the graphic representation in Figure 13, VIS has no amino groups, which is due to the chemical structure of the viscose, that is, cellulose that only have hydroxyl and aldehyde (as end) groups. The highest number of amino groups is found in viscose functionalized with CH solution only (7.14 mmol/kg), followed by viscose functionalized with enzymatically eugenol-modified chitosan, CH/EUG (ENZ) (3.80 mmol/kg). The lowest number of the remaining accessible amino groups is found in viscose functionalized with chemically eugenol-modified chitosan (2.58 mmol/kg for CH/EUG (CHEM)). From the results of the Acid Orange VII method, which alone corresponded to the results of potentiometric titration in modified chitosan products, it can be seen that the number of remaining accessible amino groups in the viscose was influenced to a certain extent by the type of modification process. In addition, the adsorption of the eugenol-modified chitosan solution was carried out as a coating at pH 6, whereby only a few protonated amino groups of the chitosan were available in eugenol-modified chitosans and, thus, negligible ionic interactions with anionic carboxyl groups of the viscose are possible. On this basis, physical interactions between viscose and eugenol-modified chitosan were predicted. Moreover, more available deprotonated amino groups in eugenol-modified chitosan products (as solutions) at pH 6 led to a more tortuous structure of the chitosan, which is, preferably, adsorbed more extensively in solid matrices than in long-chain chitosan.51,52 Since CH/EUG (ENZ) has somewhat higher amino groups in the mass (solution), the above explanation may be the reason for a somewhat higher accumulation or availability of copolymer amino-chitosan groups.
Microbiological testing
The main problem of antimicrobial substances used for textiles is their harmlessness to the human body. In this context, chitosan has gained much attention as an environmentally friendly antimicrobial agent for textiles. 53 The antimicrobial activity of the functionalized viscose was tested according to ASTM E2149-13 (Standard test method for determining the antimicrobial activity of immobilized antimicrobial agents under dynamic contact conditions) against pathogenic micro-organisms, that is, Staphylococcus aureus (Gram-positive bacteria), Escherichia coli (Gram-negative bacteria) and Candida albicans (fungi). The results are collected in Table 1. The results are the average value of three measurements, and the standard deviation is within the interval of 1–5%.
Inhibition of micro-organisms

The results show that all functionalized viscose samples, except for the viscose functionalized with chitosan (VIS/CH) (65%), showed a high reduction of the Gram-positive bacteria Staphylococcus aureus up to 75% (Table 1), which is the limit for successful inhibition.1,2 The best inhibition of Staphylococcus aureus was shown with VIS functionalized with CH/EUG (CHEM) (95%); a satisfactory reduction was also shown with VIS functionalized with a solution of CH/EUG (ENZ) (79%) (Table 1). The Gram-negative bacteria Escherichia coli was inhibited sufficiently by VIS functionalized with the solution of CH/EUG (CHEM). In this case, the reduction was 90%. For all other samples, the reduction was below 75%, and, thus, not effective for Escherichia coli. All functionalized viscose samples showed a high degree of inhibition toward the fungus Candida albicans. The best result was obtained in the case of the viscose functionalized with chitosan solution (VIS/CH = 96%), followed by viscose functionalized with a solution of the two eugenol-modified chitosans (VIS CH/EUG (CHEM) and VIS CH/EUG (ENZ); 92% for both samples). From the results (Table 1), it can be concluded that the chemically modified eugenol–chitosan has proved to be multifunctional in biological activities and exhibited a broad spectrum of micro-organism inhibition. It can also be concluded that not only chitosan amino groups are responsible for antimicrobial activity in general. If amino groups have a leading inhibitory effect, VIS/CH should show the best inhibition (see Figure 13). From this, it can be concluded that other factors, such as morphology, hydrophilicity/hydrophobicity, etc., are also responsible for the antimicrobial activity of the material. Furthermore, the clear added value of eugenol for microbial inhibition is evident, while its phenolic groups are also responsible for the antimicrobial character. The latter was expressed more by CH/EUG (CHEM) in comparison to CH/EUG (ENZ), which is correlated to the accessible phenolic groups amounts, as shown by the bulk methods, FC and Fe3+ iron reduction capacity.
Antioxidant activity
Figure 15 shows the results of the antioxidant activity of the coated viscose fibers. As can be seen from the results, the lowest antioxidative activity was shown for the reference (VIS = 19%) and VIS functionalized with chitosan (CH) as a macromolecular solution (28.13%). The antioxidative activity of the viscose functionalized with both eugenol-modified chitosan products was strongly increased, that is, both coatings introduced a good antioxidative activity into the viscose. The viscose coated with a solution of eugenol-modified chitosan achieved almost the same results in a time of 30 minutes. This can be explained by the fact that there was a synergy effect between the viscose fibers and eugenol-modified chitosan, which, together, contributed to the valuable antioxidant activity, which, in equilibrium, accounted for around 60%.

Antioxidant activity of VIS, VIS EUG, VIS CH, VIS CH/EUG (CHEM) and VIS CH/EUG (ENZ) by the DPPH• method. The results presented are average values of three measurements. The standard deviation is in the range of 3.0–3.8%.
It is also interesting to note that, with the time extension (to 120 minutes), better results were achieved by the functionalized viscose compared to the time of 30 minutes for the pure eugenol-modified chitosans (Figure 15). It is obvious, as already speculated, that more time is needed until natural substances are active as oxygen scavengers. At the time of 120 minutes a similar antioxidant activity (≈60%) was found for both functionalized viscose samples (i.e., VIS CH/EUG (CHEM) and VIS CH/EUG (ENZ)). This was somewhat expected while a higher amount of CH/EUG (ENZ) was bound to the fibers, although these copolymers themselves had a lower number of the available phenolic groups.
The good antioxidant properties were due to the phenol groups in eugenol that can release hydrogen to free radicals and, thus, inhibit oxidation processes. According to the results of the study by Gülçin, 54 eugenol had the strongest antioxidant activity and radical scavenging effect. In addition, the antioxidant activity of natural antioxidants, including eugenol, has been shown to play a critical role in tissue repair under normal and pathological conditions with antibacterial and anti-inflammatory effects, by scavenging the reactive oxygen species (ROS) resulting from oxidative damage, thereby improving wound healing and other tissue infections.50,55 In addition, some recently published data suggest a positive cost–benefit balance for the treatment of venous ulcers with eugenol-based antioxidant dressings.53,56 It must be pointed out that the functionalization process is technologically and economically suitable to be transferred into real-life conditions, while some process design and cost aspects must be studied for real applications.54,57
Desorption
Release of eugenol-modified chitosan from viscose
The desorption study gives an answer as to whether the release of active substances from the material surface into the desorption bath (represented by the deionized water) is to be expected. The kinetics of active substance release was indirectly followed by the DPPH• method, which was also used to determine antioxidant activity. This was done for desorption baths as described in the experimental section. The indirect method used is based on a rough estimate of desorption. If desorption of eugenol has occurred, the antioxidant activity for that desorption bath is predicted based on the presence of eugenol. The results are shown in Figure 16.
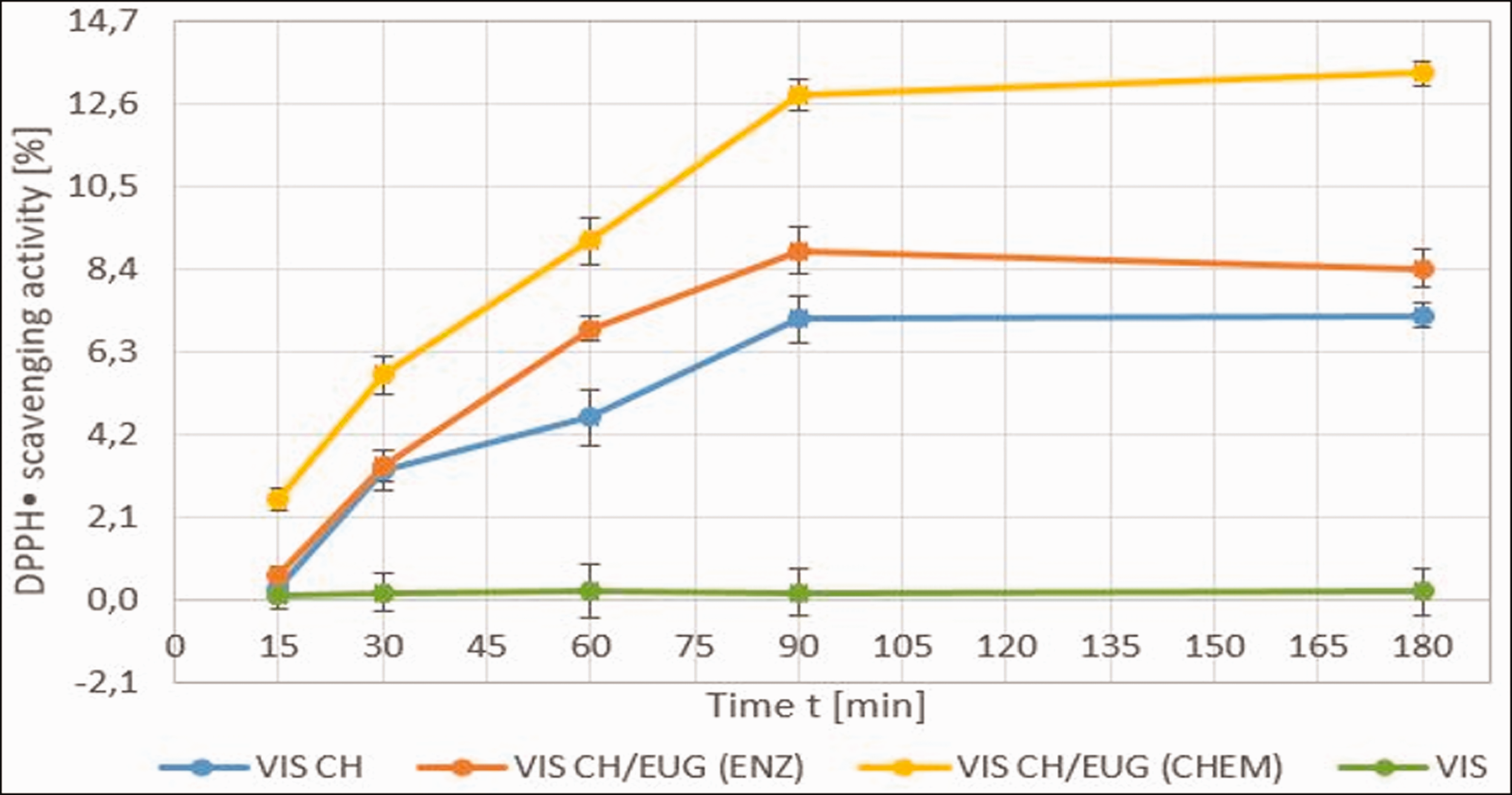
The eugenol-modified chitosan release–desorption kinetics. The results presented are average values of three measurements. The standard deviation is in the range of 3.0–3.2%.
VIS samples functionalized with the solution of chemically eugenol-modified chitosan showed a rapid release after 15 minutes (Figure 16). The amount of captured DPPH• was 2.56% for VIS functionalized with the solution CH/EUG (CHEM). All other samples, CH/EUG (ENZ) and VIS CH, showed lower amounts of captured radicals over a period of 15 minutes. After 90 minutes, more active substances were released into the desorption bath. For VIS CH/EUG (CHEM) this release accounted for 12.8% of the DPPH• capture activity, while VIS CH/EUG (ENZ) showed 8.8%. A certain rinsing effect was also observed for VIS CH, that is, 7.1%. Comparing these results with the antioxidant activity of the viscose itself, it can be concluded that about 20% of the active substance release can be predicted. This is only a rough estimate, whereas this method is an indirect method for desorption assessment. In Figure 16 two areas were identified. As mentioned earlier, the first area showed an increase in release with time up to 90 minutes. After 90 minutes, a plateau of the release area was evident, that is, no more release occurred (equilibrium point, Figure 16). A clear linear correlation of the kinetics relays was not observed, suggesting that the release mechanism is more complex, and the order of kinetics may change with time. It could be concluded that the release is mainly due to a combination of two mechanisms, diffusion and possible swelling processes.
Conclusions
The first phase of the research involved chemical and enzymatic modification of chitosan in combination with the natural active compound polyphenol eugenol. The success of the newly formed synergistic formulations was investigated and confirmed using a combination of spectroscopic methods (ATR FT-IR, 1H NMR, UV/VIS), potentiometric titration and methods for evaluation and detection of antioxidative efficacy (FC, DPPH•, Fe3+ reduction).
Furthermore, these two chitosan–eugenol formulations were applied in the form of colloidal solutions as green coatings on viscose fibers. ATR FT-IR analysis of the functionalized viscose substrates and Acid Orange VII spectrophotometric method confirmed the adhesion of the eugenol-modified chitosan solutions to the viscose by detecting typical functional groups present in both copolymers. The analyses of SEM showed that in all cases of treatment, increased surface roughness was observed compared to the surface of the untreated viscose and that the colloidal chitosan–eugenol formulations were integrated on the fibers as particles or thin films. Also noteworthy was the appearance of different shapes of particles and film layers when colloidal solutions of chemically and enzymatically modified chitosan with eugenol are applied, suggesting that the different modification of chitosan affects not only the chemical but also the morphological changes of the coatings. The best inhibition of micro-organisms in terms of antimicrobial activity was shown for viscose functionalized with solutions of chemically eugenol-modified chitosan, while the enzymatic modification of chitosan with phenolic agents showed a little less antimicrobial activity. The first sample successfully inhibited Staphylococcus aureus, Escherichia coli and Candida albicans, whilst the second one only inhibited Staphylococcus aureus and Candida albicans. Both eugenol-modified chitosan solutions brought good antioxidant activity to the viscose (≈60%), which was not present in the reference viscose or in the viscose functionalized with chitosan alone. It was proved that eugenol and chitosan act synergistically from the bioactive point of view and present an efficient coating for viscose.
Desorption experiments, indirectly determined by the DPPH• method, showed fast initial release within 90 minutes and the release reached a plateau/equilibrium) from this time on.
The surface modification of viscose by the eugenol-modified chitosan solutions (CH/EUG (CHEM), CH/EUG (ENZ)) due to the synergistic antimicrobial and antioxidant activity showed the potential for use in medical and hygienic applications, especially in the field of wound healing (gauze, plasters, bandages), where antimicrobial and antioxidant properties are important simultaneously.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.