Abstract
This study is aimed to investigate the effect of the simultaneous incorporation of cellulose nanocrystals (CNC) and silver nanoparticles (SN) on the mechanical, biodegradability, and water vapor permeability of polylactic acid (PLA)-based films. PLA films and their nanocomposites containing different levels of CNC (0.333, 1 and 1.667 phr) and SN (0.333 phr) were prepared by solution casting method. CNC was reacted with acetic anhydride to improve its compatibility and miscibility with PLA. Fourier transform infrared (FTIR) spectroscopy, thermogravimetric analysis (TGA), tensile test, and water vapor permeability and antibacterial tests were employed to characterize the samples. The biodegradability was assessed by measuring the weight loss upon burial in the soil. FTIR spectroscopy confirmed the modification of cellulose nanocrystals. TGA test showed that partial acetylation slightly improved the thermal stability of CNC. The presence of cellulose nanocrystals increased the tensile strength and modulus of elasticity of the nanocomposite relative to pure polylactic acid. The biodegradability and water vapor permeability of the samples decreased upon CNC incorporation. The antibacterial properties of the films showed the higher resistance of the gram-positive bacteria as their cell walls include a peptidoglycan layer.
Introduction
Material science research is focused on producing efficient, renewable, and environmentally friendly materials to cope with petroleum scarcity while considering environmental concerns. Recently, the “green” bio-based nanocomposites are emerging in the market as they are biodegradable, low cost, abundant, biocompatible, nontoxic, and renewable. 1 Recently, several research groups started the preparation and characterization of various types of biodegradable polymer nanocomposites suitable for a wide range of applications. Most of the studied biodegradable polymers are starch and its derivatives, polylactic acid (PLA), poly (butylene succinate), polyhydroxybutyrate, and aliphatic polyesters such as Polycaprolactone. 2
Poly(lactic acid) (PLA) is a biodegradable and compostable aliphatic polyester, produced from renewable resources such as cassava, corn, starch, and sugarcane. 3 Its constituent monomers are lactic acid and 2-hydroxy propionic acid. It can be produced by ring-opening and condensation polymerization. 4 However, low elongation at break, slow degradation rate, and poor thermal stability have restricted its applications.5,6 To overcome these problems, methods such as mixing with other polymers, crystallization optimization, use of softeners, lubricants, and fillers have been reported. One of the approaches to improve the properties of this biopolymer involves the use of nano-scale fillers and the production of nanocomposite.7,8,9
Nanocomposites are a new class of composite materials combining nanotechnology and composite technology. The application of nanotechnology to these polymers may open new possibilities for improving not only the properties but also the cost-price efficiency. Owing to the nano-scale size of dispersed particles, these nanocomposites can exhibit markedly improved mechanical, thermal, barrier, and physicochemical properties.10,11,12 Nanoparticles such as nanoclay, nanocellulose, silver, titanium oxide, and carbon nanotubes can be used to reinforce the nanocomposites.
The size of silver nanoparticles ranges between 1 nm and 100 nm. Numerous shapes of nanoparticles can be constructed depending on their intended application. Commonly used silver nanoparticles are spherical, but diamond, octagonal, and thin sheets can be also applied. Their extremely large surface area permits the coordination of a vast number of ligands. Silver nanoparticles applicable to human treatments are under investigation in laboratory and animal studies to assess their potential efficacy, biosafety, and biodistribution. 13
In general, nanocellulose can be prepared in two main forms: nanofibers (CNFs) and nanocrystals (CNCs). Their main difference lies in their extraction method and appearance. Cellulose nanocrystals (CNCs) are the crystalline regions of CNFs and are known as cellulose monocrystalline regions. Due to the high crystallinity of CNCs, their elastic modulus is equivalent to that of crystalline cellulose (up to 140 GPa) which can be attributed to their hard nature and ability to form hydrogen bonds. Depending on their source of production, the size of CNCs varies from 10 to 1000 nm in length and 4–25 nm in diameter. 14
Cellulose nanocrystals can be obtained by acidic hydrolysis to remove the amorphous regions from cellulose chains. These structures have shown unique features such as availability, low cost, renewability, low density, and high surface-to-volume ratio, modulus of elasticity, tensile strength, stiffness, flexibility as well as good thermal, optical, and electrical properties. Moreover, they are considered biodegradable nanocomposites. 15 The most important limitation of cellulose nanoparticles in the production of nanocomposites is their incompatibility with non-polar polymers 16 which can be resolved by surface modification and to decline its polar nature.17,18,19
Based on Reza Arjmandi et al., the biodegradability of polylactic acid composites increased by partial replacement of MMT (montmorillonite) with MCC (microcrystalline cellulose) filler as compared to neat PLA. 20 Juan I. Moran et al. prepared poly(lactic acid)/Cellulose-nanowhisker nanocomposites for packaging applications and showed that the mechanical properties of the nanocomposites were higher than those of the neat matrix. 21 According to the thermogravimetric analysis (TGA) studies by Achal Bhiogade and colleagues, PLA is thermally more stable than PLA-based composites. 22 C. Prapruddivongs et al. reported higher permeability and antibacterial properties in the samples containing cloisite® 30B. 23
In the present study, the compatibility of CNC with polylactic acid was improved by chemical surface modification of the nanoparticles with acetic anhydride. The water vapor permeability of CNC/nanosilver-reinforced polylactic acid nanocomposite was also investigated for the first time. Their water absorption and weight loss in deionized water, as well as the mechanical performance, were also explored. Finally, the antibacterial properties of the films were assessed against Gram-positive
Materials and methods
Materials
Polylactic acid (2003D; Natureworks; US) was used as the polymeric base material. Cellulose nanocrystals were obtained from chemical treatment of pure cellulose fibers (2.5% dryness) as produced by Nano Novin Polymer (Iranian company) in the form of a white gel. Silver nanoparticle powder with a density of 10.5 g/cm3, purity of 99.99%, and size of 20 nm was purchased (Research and Market, US). Chloroform (Merck, Germany) was used as a solvent. Acetic acid and pyridine (Merck, Germany) were employed for the chemical modification of the CNC, Acetic anhydride was also supplied from the Sigma Aldrich Company.
Preparation of the films
Components of the samples.

CNC: cellulose nanocrystals; PLA: polylactic acid.
Chemical modification of cellulose nanocrystalline surface
Chemical modification of nanocellulose surface can increase its compatibility. Acetylation was performed by constant mixing under a nitrogen atmosphere in a balloon equipped with a condenser. To remove water, 40 g CNC suspension with 2.5% dryness was mixed with 80 mL acetic acid before acetylation. After 10 min of gentle mixing, the nanocrystals were separated from acetic acid through centrifugation. The remaining nanocrystals were transferred to a balloon to which 30 mL acetic acid and 40 mL acetic anhydride were added in one step followed by the addition of a pyridine catalyst at 5wt% relative to nanocrystals.
Figure 1 schematically shows the reaction. The resulting suspension was reacted at 80°C for 5 h under a magnetic stirrer and nitrogen atmosphere. Then, the acrylic nanocrystals were washed in an acidic medium using a centrifuge; followed by washing with the acetone-methanol mixture (1:2) four times. The acetylated CNCs were dried in an oven at 60°C for 8 h.
24
Chemical modification of nanocellulose with acetic anhydride.
25

Fourier transform infrared spectroscopy
Infrared spectroscopy was used to examine the chemical structure of cellulosic nanocrystals before and after chemical modification. The fourier transform infrared (FTIR) spectra of the films were recorded in the range of 2500-500 cm−1 using an FTIR spectrometer (RXI FTIR).
Tensile properties
Tensile test was carried out under ASTM D882 standard using a stretching device model STM 250 (Santam, made in Iran). The dumbbell-shaped films (0.5 × 10 cm2) were placed between the two jaws of the device at an initial distance of 50 mm and a loading speed of 2 mm/min. Five replications were considered for each sample.
Thermal gravimetric analysis
Thermal stability and thermal degradation of the samples were evaluated by TGA TMA1000 (Sanaf, Iran) using 30 mg film samples at the temperature range of 50–500°C and heating rate of 10°C/min under nitrogen atmosphere.
Water vapor permeability
Water vapor permeability was measured according to ASTM E96-95 standard. Accordingly, special vials were employed with a diameter of 2 cm and a height of 4.5 cm. The lid of these vials had a hole with a diameter of 8 mm, in which a piece of film was placed. After placing the cut pieces of the desired films and the initial weighing, the vials were transferred to a desiccator containing water placed in an incubator at 25 ± 1°C. The weight of the vials was measured every few hours for 4 days. The amount of water vapor transferred from the films is determined by measuring the weight of the vials. The water vapor transfer rate (WVTR) can be obtained by plotting the weight gain of the vials versus time. Using equation (1), water vapor permeability (WVP) was calculated
In the above equation:
WVTR: water vapor transfer rate (g/s.m2)
WVP: water vapor permeability (g/Pa.s.m)
X: film thickness (m)
P: pure water vapor pressure at 25°C (Pa)
R1: relative humidity in the desiccator (100%)
R2: relative humidity within the vial (0%).
This test was performed in triplicates.
Destruction of films in deionized water
The degradation of films in deionized water was evaluated as described by Hossain et al.
26
through a weight difference method. Typically, 25 mm × 25 mm films were cut and their initial weight was recorded. The samples were then immersed in glass bottles containing 25 mL of deionized water at 25 and 40°C for 10 days. The samples were then dried in an oven at 50°C until reaching a constant weight. The weight loss (%) was calculated by equation (2).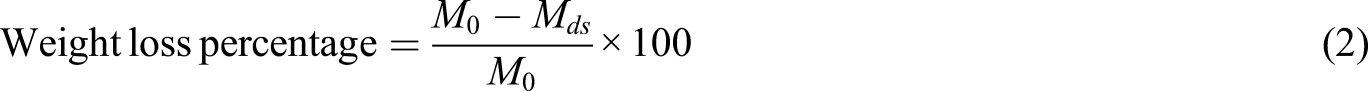
In the above equation:
M0: sample weight before immersion
Mds: weight of the dried sample after immersion
Biodegradability
The biodegradability of the samples was assessed by measuring their weight at 50°C in the beginning and after drying in the oven. The samples were then buried in the soil at a depth of 5 cm (inside plastic boxes and in the laboratory). During the test, the soil inside the boxes was kept moist by adding distilled water. To measure the extent of degradation, the samples were removed from the soil at 10-day intervals and their surface soil was washed with distilled water followed by oven-drying for 12 h. Finally, the weight of the samples was recorded. Weight loss percentage was calculated using equation (3).
In the above equation:
W0: Initial dry weight
Wd: dry weight after burial in soil
Antibacterial properties
The antibacterial properties of the films were evaluated by the disk diffusion method according to Clinical & Laboratory Standards Institute 1 standards. For this purpose, plates containing Mueller-Hinton agar medium were prepared. Wells with a diameter of 6 mm were made on the medium. Then, 100 μL bacterial suspensions with turbidity equivalent to 0.5 McFarland were uniformly cultured on the medium. The dried extract was dissolved in dimethyl sulfoxide to a concentration of 30 mg/mL extract 10 μL/well (300 μg) was poured into the wells and the plates were incubated at 37°C for 24 h. The antimicrobial activity was determined for each microorganism by measuring the growth inhibition zone.
Results and discussion
Fourier transform infrared spectroscopy of cellulose nanocrystal
Infrared spectroscopy was used to investigate the chemical structure of cellulose nanocrystals after chemical modification. Figure 2 shows the FTIR spectrum of cellulose nanocrystals before and after chemical modification. The FTIR analysis of modified cellulose nanocrystals showed an absorption peak at 1706 cm−1 due to the stretching vibration of the C=O bond. But this peak is transferred toward lower wavenumbers (1780-1750) due to the effect of the methyl group attached to alpha carbon (reaction with acetic acid). This effect reduced the dual bond property of C=O and thus declined the absorption number. The stretching vibration of the C-O bond of the acetyl group at 1050 cm−1 and the stretching vibration of the methyl (C=O)-CH3 groups confirmed the formation of acetylated groups in the modified cellulose nanocrystals.
27
Fourier transform infrared spectroscopy of pristine and modified cellulose nanocrystals.
Thermogravimetric analysis of cellulose nanocrystals
The effect of acetylation on the thermal stability of cellulose nanocrystals was evaluated by TGA, as shown in Figure 3. During thermal decomposition, nanocellulose showed a gradual and multi-stage weight loss which can be divided into three temperature zones. The first zone started at 50°C and continued up to 150°C, which can be attributed to the evaporation of water and other solvents. The second temperature range lied at 150–350°C while the third range was at 350–500°C. In the second temperature zone, approximately 50% weight loss occurred in the CNC due to the removal of the side groups formed in the preparation of cellulose nanocrystals such as sulfate. In the third stage, the rest of the compounds were decomposed.
28
According to Figure 3, the onset of thermal degradation for pristine and modified cellulose nanocrystals was at 210 and 245°C, respectively, close to the report of Fortunati et al. (220 and 260°C for pristine and modified cellulose nanocrystals, respectively).
29
The thermal stability of CNCs depends on their degree of crystallinity, that is, it decreases with declining the degree of crystallinity. As can be seen, the partial acetylation slightly improved the thermal stability of the cellulose nanocrystals. The reason could be assigned to the effect of acetylation on the crystalline structure of nanocrystalline cellulose (removal of some amorphous areas, hence, increasing the crystallinity degree). Tingaut et al. used acetylated cellulose microfibrils to improve the properties of PLA films. Their results indicated a decrement in the thermal stability of nanocellulose with increasing acetylation, due to severe degradation of crystalline areas of cellulose.
30
Thermal analysis of modified and pristine cellulose nanocrystals.
Mechanical properties of polylactic acid-based nanocomposites
The results of the mechanical test on polylactic acid films containing different ratios of CNC and Ag NPs are presented in Figure 4. As seen, the incorporation of CNC into the PLA matrix enhanced the tensile strength and modulus. In general, the mechanical performance of composites, especially nanocomposites, depends on several factors such as compatibility of the polymer matrix with the filler, stress transfer to the filler, volume fraction of the filler, filler aspect ratio, filler orientation, and matrix crystallization.
31
Here, the improvement of mechanical properties of nanocomposites by adding CNC to the PLA matrix can be attributed to the high crystallinity and good mechanical properties of these nanoparticles.
32
As can be seen in Figure 4, the increasing trend of strength and tensile modulus by adding 1 phr of CNC was higher than other samples. The reason could be due to the formation of a rigid three-dimensional network of CNCs within the substrate and the reduced mobility of polymer chains. At higher nanoparticle contents, this increase was not observed in properties, probably due to improper dispersion and particle aggregation.
33
Mechanical properties of polylactic acid and its bionanocomposites.
Water vapor permeability of polylactic acid-based films
The use of nano-scale structures results in a more uniform distribution of materials and increases the crystallinity of the film, thus, hindering the passage of water vapor. In the presence of the filler, the permeability was affected by the zigzag path which is itself changed by factors such as the aqueous activity, the filler shape, surface ratio, distribution, amount, orientation, adhesion to the substrate, crystallization, purity, porosity, and pore size.34,35 The results of water vapor permeability of PLA-based films are presented in Figure 5. Water vapor permeability of polylactic acid and its bionanocomposites.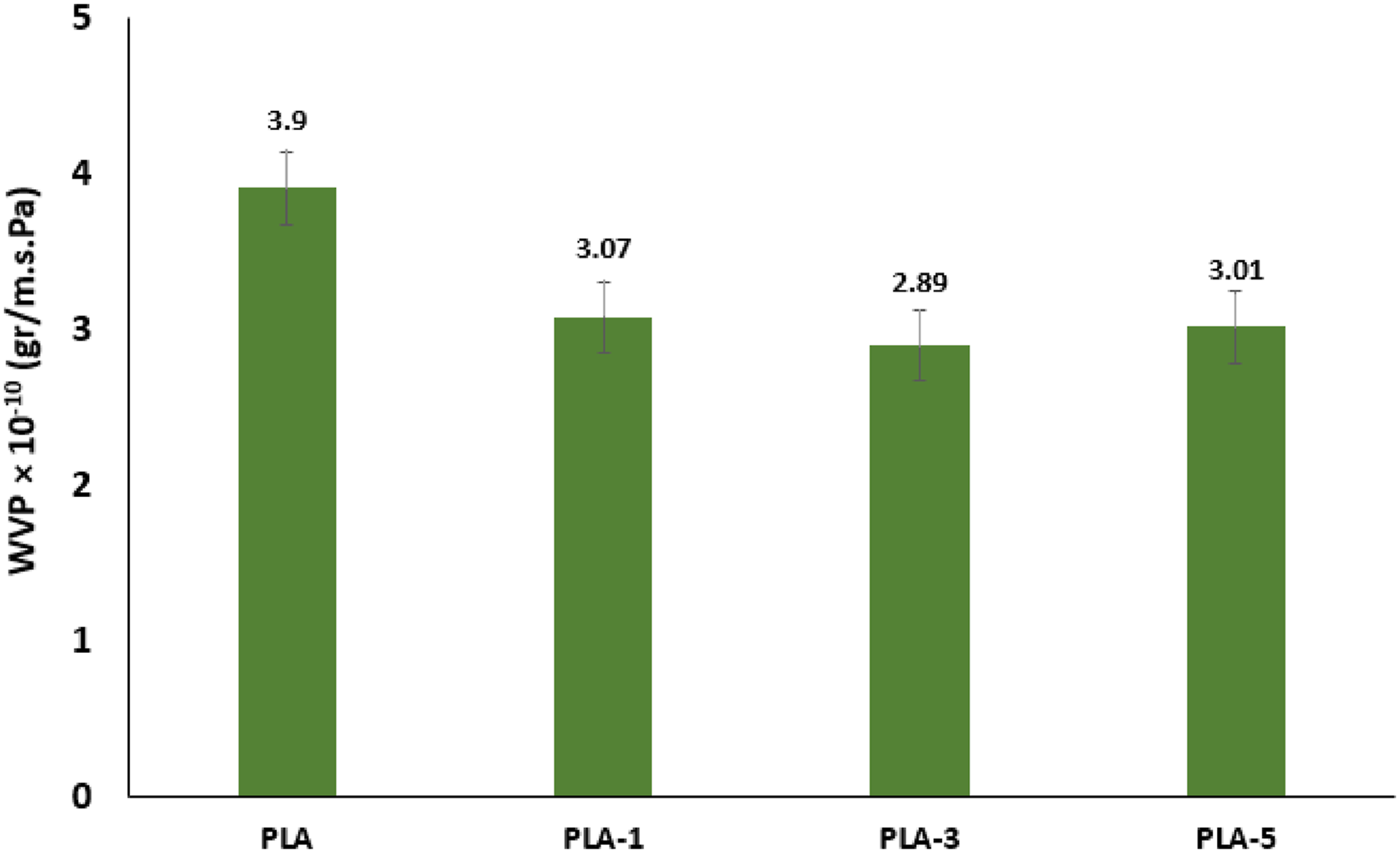
As can be seen, by increasing the CNC level, water vapor permeability decreased due to the high crystallinity of cellulose nanocrystals, which acted as a nucleating agent in the polymer matrix. The presence of cellulose nanoparticles in the polymer matrix prolonged the path of water molecules. Increased crystallinity of the polymer due to the presence of cellulose nanoparticles enhanced the cohesion and density between the polymer chains and reduced the inter-chain free spaces, which is another reason for the declined permeability of nanocomposite films. It was observed that the sample containing 1.667 phr CNC did not exhibit reduced permeability compared to the sample containing 1 phr, which is probably due to clumping and improper distribution at this CNC percentage. 36
Degradation of polylactic acid films in deionized water
Weight loss of films at 25 and 40°C
Weight loss percentage of nanocomposites at 25° and 40°C.

PLA: polylactic acid.
Water absorption of the films at 25 and 40°C
For high moisture-sensitive packaging materials, exposure to high-humidity environments not only alters the functional properties of the packaging material but also reduces the durability of the packaging product due to moisture absorption.
Water absorption of nanocomposites at 25° and 40°C.

PLA: polylactic acid.
Biodegradability of nanocomposites
The weight loss of PLA films and composites is shown in Figure 6. As can be seen, the weight loss of the samples decreased with increasing the amount of cellulose nanocrystals. The weight loss rate of the samples over time followed a linear trend and increased by prolonging the burial time in the soil. The growth of microorganisms requires adequate physical and chemical conditions such as moisture, temperature, and oxygen. Thus, the process of composite degradation depended on providing the necessary conditions for the growth and attack of microorganisms.39,40 The decrease in the degradability of the samples due to the presence of cellulose nanocrystals can be attributed to the strong tendency of free hydroxyl groups to form complexes with polar mineral compounds of the soil, which made them impermeable to microorganisms. The occurrence of strong reactions with minerals at the soil surface indeed limited the accessibility of functional groups and reduced the biodegradability of samples.29,41,42 Weight loss of polylactic acid film and nanocomposites after burial in soil.
Cellulose nanocrystals formed long paths in the polymer substrate, prolonging the diffusion of water and oxygen molecules, thereby delaying the growth of microorganisms. Therefore, the degradability of composites decreased by elevating the CNC content.
Antibacterial properties of nanocomposites
Inhibitory zone diameter for

PLA: polylactic acid.
Conclusion
Polylactic acid was used in the current research to prepare biodegradable films. To improve the functional properties of the films, CNC and Ag NPs were employed. Various tests were performed to measure the film properties. The results can be summarized as 1. Acrylication of CNC was confirmed by FTIR analysis. Dispersion and TGA results also showed that the use of acetic anhydride was effective in acetylation treatment. 2. The incorporation of cellulose and silver nanoparticles into the polylactic acid film improved its mechanical properties compared to the pure PLA film. By adding more than 1 phr nano cellulose, the mechanical properties decreased. 3. Cellulose and silver nanoparticles increased the moisture absorption inhibition of films and also reduced the water vapor permeability of films. So that the film containing 1 phr CNC and 0.333 phr Ag NPs exhibited the highest reduction compared to other films. 4. According to the biodegradability test results, the presence of nanoparticles in the PLA polymer substrate leads to the formation of complexes between hydroxyl groups and polar mineral compounds in the soil, thus reducing the biodegradability of the nanocomposites. 5. The antibacterial properties of the films showed an antibacterial resistance in the samples containing Ag NPs in comparison with the Ag-free samples which were more resistant to gram-negative bacteria than the gram-positive ones.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
