Abstract
In the present study, it was aimed to investigate the thermoelectric (TE) properties of polyaniline/hexagonal boron nitride (PANI/h-BN) composites. First, h-BN was synthesized from boric acid and urea. Then, PANI-HCl was synthesized by oxidative chemical polymerization. Finally, the composites were prepared using different weight ratios of h-BN. The composites were characterized using attenuated total reflection accessory attached Fourier-transform infrared spectroscopy, UV-vis spectroscopy, X-ray diffraction, and scanning electron microscopy/energy dispersive X-ray analyzer. TE investigation of the composites showed that the addition of h-BN significantly contributes to the TE properties of PANI-HCl. The addition of h-BN increased the power factor of PANI-HCl from 0.07 μWm−1K−2 to 143.05 μWm−1K−2. Furthermore, all the composites showed negative Seebeck coefficients which are the characteristics of n-type semiconductors.
Introduction
In recent years, as a result of the depletion of fossil fuels, the most common energy source, and increasing environmental concerns, researchers have started to search for alternatives to environmentally friendly energy sources. Among the renewable energy sources such as hydroelectric energy, wind energy, and solar energy, thermoelectric (TE) energy generators become a promising alternative to overcome the waste heat and greenhouse gas emissions caused by the use of fossil fuels. 1 TE devices convert the applied temperature gradient directly to electrical energy as a result of the Seebeck effect. 2
The efficiency of TE materials is determined by the dimensionless figure of merit ZT = σS2T/κ, where σ, S, T, and κ are successively related to electrical conductivity, Seebeck coefficient, absolute temperature, and thermal conductivity.3–5 Highly efficient TE generators are obtained by combining both p-type and n-type semiconductors. The high TE performance is observed at low and medium temperatures with the use of such devices. 6 However, the instability under ambient conditions and electron trapping problems are the main drawbacks of n-type semiconductors.7,8
Due to the high cost, hardness, and difficult processing of traditional inorganic TE materials, the researchers focused on conductive polymers as a promising alternative. There are numerous studies in the literature on TE properties of polyaniline (PANI), polypyrrole, polythiophene, and derivatives. 9 The power factor (PF) is the main concept used in determining the TE performance of conductive polymers due to their low and similar thermal conductivity. 10 PANI is one of the most widely studied conductive polymers in TE applications due to its easy synthesis method, environmental stability, lightweight, tunable electrical conductivity method, and low cost.11–13 The solubility and electrical properties of PANI can be enhanced by doping organic acids such as camphor sulphonic acid (CSA), dodecylbenzene sulfonic acid, p-toluene sulfonic acid, or mineral acids namely hydrochloric acid (HCl), sulfuric acid, fluoroboric acid, and orthophosphoric acid. 14 The electrical conductivity and the Seebeck coefficient of CSA-doped PANI reach up to 30 Scm−1, 8–12 µVK−1, respectively. 15 In addition to the doping process, the TE properties of PANI can be increased by the contribution of organic/inorganic additives such as graphene,16,17 carbon nanotube,18–20 bismuth telluride, 21 and graphitic carbon nitride. 22 In these studies, the PF of PANI was enhanced up to 321 µWm−1K−2. 20
Two-dimensional materials become an attractive alternative as a TE material due to their electrical and thermal transportation, stretchability, unique chemical, mechanical, and physical properties.23,24 With the developing technology, several two-dimensional materials such as graphene, black phosphorus, transition metal dichalcogenides (MoS2, WS2, etc.), and nitrides (boron nitride) have been successfully produced. 25 In the literature, there are various studies on the TE performance of PANI composites filled with graphene, which is the most popular 2-dimensional material.16,17,26,27 In these studies, it was reported that the PF of PANI increased to 214 μWm−1K−2. 27
Among the two-dimensional materials, boron nitride (BN) has become an attractive material due to its stiffness, good mechanical and thermal properties, resistance to oxidation and corrosion, and chemical inertness. 28 Boron nitride can be obtained in different polymorphs, namely, nanoparticles, microparticles, nanotubes, nanorods, nanowires, nanosheets, and nanoribbons. Hexagonal boron nitride (h-BN) is the most popular polymorph compared to its counterparts due to its graphite-like isoelectronic structure and high melting point (3000°C). 29 There are several studies in the literature on the TE properties of h-BN. Chen et al. 30 reported that with the addition of h-BN, the Seebeck coefficient of graphene single layer reached up to −99.3 μVK−1. Furthermore, Wang et al. 31 mentioned that the PF of boron nitride-filled PEDOT:PSS composites enhanced to 100.1 μWm−1K−2.
There are several studies in the literature examining various properties of polyaniline/hexagonal boron nitride (PANI/h-BN) composites such as photocatalytic and adsorptive activity,28,32,33 dielectric properties, 29 thermal stability, and fire safety performance, 34 and biosensing properties. 35 However, to the best of the authors’ knowledge, there is no study on the TE properties of PANI/h-BN composites.
In the present study, the TE properties of PANI-HCl/h-BN composites containing different weight ratios of h-BN were investigated.
Materials and methods
Materials
Aniline (≥99.5% pure), ammonium persulfate (98% pure), urea (≥99.5% pure), n-methyl-2-pyrrolidone (anhydrous, 99.5% pure), acetone (≥99.5% pure) were purchased from Merck, Germany. Boric acid was purchased from ETİ Maden, Turkey.
Synthesis of PANI-HCl
First, 0.01 mol of aniline dispersed in 80 mL of 1 M HCl solution. Immediately after, 20 mL of 0.02 mol ammonium persulfate solution in 1 M HCl was added dropwise into the medium. The polymerization reaction was carried out under ambient conditions for 16 h. After polymerization, the obtained dark-green powder was washed with distilled water and acetone to remove impurities and by-products.
Preparation of h-BN
The preparation of h-BN was carried out as described in literature. 36 First, boric acid and urea were dissolved in water, respectively, and an aqueous solution with a B:N molar ratio of 1:6 was prepared. Then, the solution was dried at 80°C in an oven under a vacuum for 16 h. The dried sample was powdered in a mortar. The powdered sample was heated to 600°C at a temperature rate of 10 ˚C/min in a furnace for 2 h. Subsequently, the heating process was continued at 800°C (10 ˚C/min) temperature for 3 h.
Preparation of PANI/h-BN composites
The composites were prepared by dispersing the components with an ultrasonic homogenizer. First, PANI was dispersed in n-methyl pyrrolidone to form a 1% solution by weight. Then, different weight ratios of h-BN were added to the dispersion (10%, 20%, 30%, 40%, and 50% with respect to the polymer weight). The samples were sonicated at a frequency of 20 kHz for 30 min.
Finally, the composites were dried at 100°C for 24 h. Composite samples were prepared by compressing with a hydraulic press under a pressure of 100 MPa to form a circular pellet with a diameter of 10 mm and a thickness of 1 mm.
Characterization
The chemical properties of the samples were examined using Fourier-transform infrared spectroscopy (FTIR-ATR) (THERMO Nicolet Is 10), and UV-vis (SHIMADZU UV2600) spectroscopies. The crystalline nature of the samples was investigated by X-ray diffraction (XRD) operated at 30 mA and 40 KVP MAX (T&T TT-90). The surface properties and the dispersion behavior of h-BN in the matrix were explored using SEM and EDX mapping (Zeiss EVO® LS 10). The conductivity and Seebeck coefficient measurements were carried out using Four Point Probe (Entek Electronic, Turkey) and Seebeck coefficient measurement system (Entek Electronic, Turkey).
Results and discussion
X-ray diffraction patterns of h-BN, PANI-HCl, and the composite with 50% h-BN are shown in Figure 1. The characteristic peaks of PANI-HCl were observed at 15.4°, 21.3°, and 25.2°. These broad peaks indicate the semi-crystalline nature of PANI-HCl.37,38 For the spectrum of h-BN, the broad peak at 26.2° and the weaker peak at around 42.3° can be attributed to the graphite-like and turbostratic structures, respectively. The turbostratic crystals show a similar to the hexagonal layered structure; however, they are composed of randomly oriented layers.
39
Furthermore, the sharp peak observed at 28.2° indicates that h-BN has partially crystalline and amorphous states due to the low-temperature synthesis. Furthermore, it can be clearly seen that the addition of h-BN contributes to the crystallinity of the composite. X-ray diffraction patterns of polyaniline-hydrochloric acid (a) h-BN, (b) and the composite with 50% h-BN (c).
SEM images of the pristine PANI-HCl and PANI/h-BN composite containing 50% h-BN are illustrated in Figure 2. From the images, it was observed that PANI particles were totally covered with h-BN. It can be seen that h-BN particles were dispersed homogeneously and were buried inside PANI. SEM images of the pristine polyaniline (a) and composite with 50% h-BN with different magnifications (b), (c), (d).
The mapping images of the composite with 50% h-BN are given in Figure 3. The mapping images indicate the homogenous distribution of B atoms in the h-BN structure. Since aggregate formation negatively affects the properties of composites, the homogeneous distribution of additives is very important to obtain uniform properties in composites. The homogeneous distribution observed even with the highest contribution rate of h-BN can be attributed to the compatibility between the additive and the matrix. The mapping images of the composite with 50% h-BN.
Fourier-transform infrared spectroscopy spectra of h-BN, PANI-HCl, and the composite with 50% h-BN are shown in Figure 4. In the spectrum of h-BN, the peak at 3198 cm−1 is related to B-OH stretching while the peak at 1030 cm−1 corresponds to B-OH plane bending. The peaks at 1320 cm−1 and 752 cm−1 are attributed to B–N bonding. Furthermore, the peaks of BN–O bonding are at 1185 cm−1, 920 cm−1 (Figure 1(a)).40,41 For the pristine PANI-HCl, the peaks at 1550 cm−1 and 1440 cm−1 belong to C–C stretching of quionoid and benzenoid rings. The peaks at 1272 cm−1 and 1214 cm−1 correspond to C-N stretching of benzenoid ring. Moreover, the peak of aromatic C-H vibrations at 1167 cm−1.
42
Fourier-transform infrared spectroscopy spectra of h-BN (a), Polyaniline-hydrochloric acid, and the composite with 50% h-BN (b).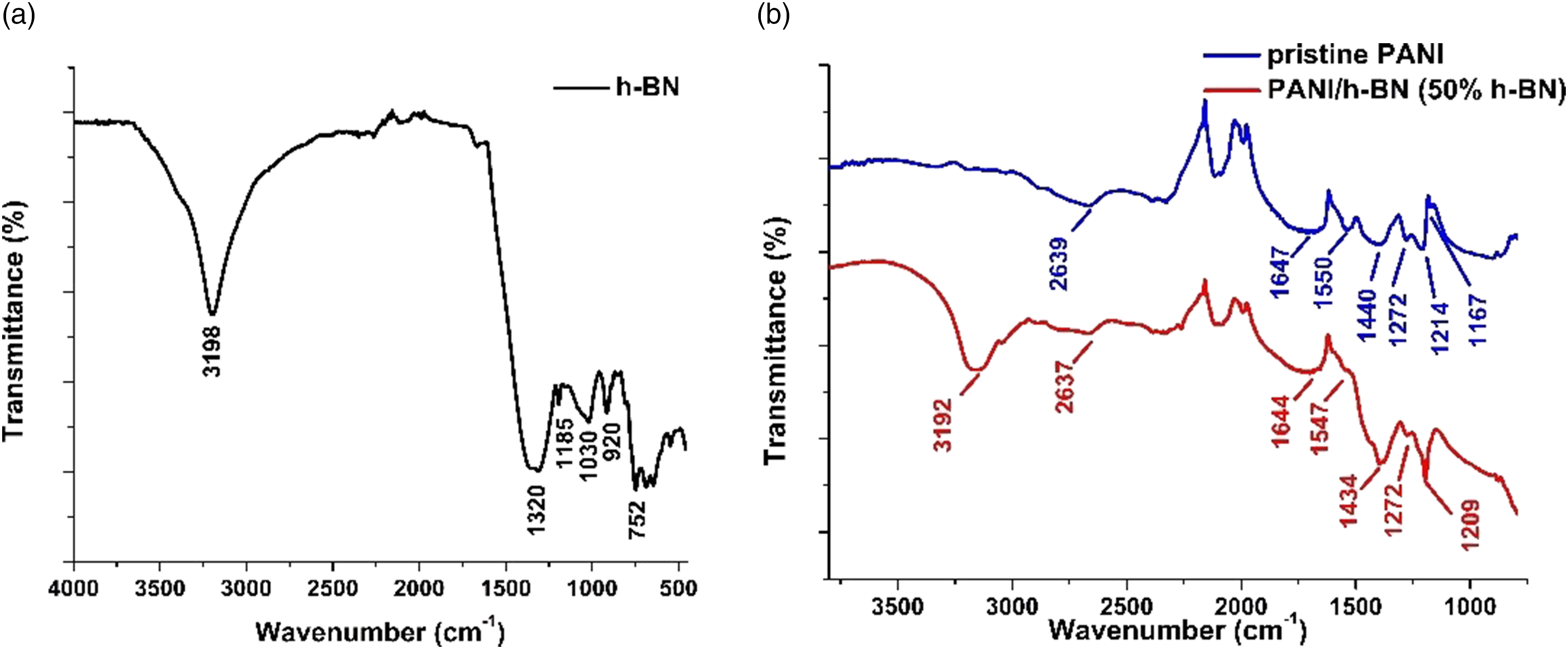
In the spectrum of the composite, it was observed that the peaks shifted to lower wavenumbers. These shifts which are also called bathochromic shifts are attributed to the extended electron conjugation and also reduction of the bandgap between the highest occupied molecular orbital and lowest unoccupied molecular orbital. 43 This indicates that the addition of h-BN particles contributes to the electrical conductivity of pristine PANI-HCl.
The UV-vis spectra of the pristine PANI-HCl and the composites consisting of different weight ratios of h-BN are given in Figure 5. The sample solutions used in the analysis were prepared in equal concentrations. In the spectrum of PANI-HCl, the absorption bands at 355 nm and 800 nm correspond successively to the characteristic π-π* transitions of benzenoid and quinoid rings.
44
From the spectra of the composite samples, it was observed that the peaks evidently shift to higher wavelengths. This indicates that the addition of h-BN particles increases the delocalization of polarons and led to extend the PANI chains.
17
The UV-vis spectra of the pristine polyaniline-hydrochloric acid and the composites containing different weight ratios of h-BN.
The TE properties of the samples are illustrated in Figure 6. The conductivities of the samples were measured at room temperature, whereas the Seebeck coefficient measurements were carried out at the temperatures of 283 K and 333 K at the cold and hot ends of the samples. The PF of the samples was calculated using the equation of PF = σS2 where σ and S refer to electrical conductivity and Seebeck coefficient, respectively. Thermoelectric properties of polyaniline-hydrochloric acid and the composites; conductivity (a), Seebeck coefficient (b), and power factor (c).
The comparison of the TE performances of the composites prepared in this study and some PANI/2D material composites in the literature.

TE: thermoelectric; PANI: polyaniline.
Conclusion
In this study, the effect of the addition of h-BN on the TE properties of PANI-HCl was investigated. For this purpose, first h-BN was synthesized using boric acid and urea, as precursors. Then, PANI-HCl was synthesized by oxidative chemical polymerization. Finally, the composites containing different weight ratios of h-BN were prepared. X-ray diffraction analyzes indicated that the addition of h-BN contributed to the crystallinity of the composites. SEM mapping images proved the homogenous dispersion of h-BN in the matrix. FTIR-ATR and UV-vis analyzes showed that the addition of h-BN enhanced the electronic conjugation. As a result of the conductivity measurements, the highest conductivity was obtained from the composite containing 30% h-BN with 254 Scm−1 which is almost 20 times higher compared to pristine PANI-HCl. The Seebeck coefficient measurements demonstrated that all the composites have negative values corresponding to n-type conductors. Besides, the composite with 50% h-BN showed the highest PF with 143.05 μWm−1K−2 which is 2000 times greater compared to pristine PANI-HCl. In this study, we concluded that h-BN is a promising material to be used as an additive in TE applications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
