Abstract
Dense and translucent CA/PEG 1000/ZIF membranes were synthesized in acetone, utilizing solution casting. Membrane characterization was carried out using FT-IR, SEM and tensile testing. SEM proved presence of dense membranes and increase in the filler amount may have formed voids, raising the permeability of both gases. Single and mixed gas (CO2/CH4) permeation testing showed an increased permeability, on addition of more filler, which is probably due to formation of nano-gaps. A maximum selectivity of 39.49 and 34.86 for single and mixed gases respectively, and maximum permeabilities of 49.7685 and 1.41 barrers were observed. Tensile testing showed that strength peaked then decreased on increased loading, due to agglomeration on adding more ZIF, which introduced defects in structure. To conclude, selectivity of higher loaded membranes is favourable whereas tensile strength of lower loaded membrane is superior, but we have a trade-off between selectivity and tensile strength, so a higher-loaded membrane is most suitable.

Introduction
Metal organic frameworks (MOFs) are a class of crystalline solids, which compose of metal clusters joined by organic linkers. 1 MOFs are used in a multitude of applications, including clean energy, as a storage media for gases such as hydrogen and methane, as a high capacity adsorbent, and in membranes, thin films devices, catalysis and biomedical imaging. 2 In the field of gas separation, metal organic frameworks have been used for several decades. The process employed for gas separation when using MOFs only, is through selective adsorption of one gas with respect to another. 3 In this regard, Chen and Jiang synthesized a biological metal organic framework (Bio-MOF-11) and tested its adsorption capacity for CO2, H2 and N2. The adsorption selectivity for CO2/H2 in the gas mixture is found to be between 230 and 375, whereas the adsorption selectivity for CO2/N2 in the respective gas mixture is found to be in the range of 30–77. 4 In another work, Li and Yang synthesized and tested the selective gas adsorption of a zinc benzene-tri-benzoate metal organic framework (MOF), namely MOF-177. The gas adsorption characteristics showed that O2 is adsorbed more than N2 with a selectivity of 1.8 whereas the maximum H2 uptake is at a value of 1.5 wt%. 5
It is known that, when basic groups like –NH2 and –OH are present in any material, it would favourably adsorb carbon dioxide. This technology is very vital in the removal of carbon dioxide from natural gas, which would otherwise contribute to climate change. 6 Therefore, amine-functionalized MOFs are very suitable for carbon dioxide adsorption. A particular work in this field was carried out by McDonald et al. In this work, they synthesized a special kind of MOF with general formula M2(dobpdc) using 4,4-dioxido-3,3-biphenyl di-carboxylate (dobpdc), which was then functionalized using N, N-dimethylethylenediamine (mmen) to form the amine-functionalized MOF. Mmen-Mg2(dobpdc). The resulting MOF gives a CO2/N2 adsorption selectivity of 49000 from air, and a CO2/N2 adsorption selectivity of 200 from flue gases. 7 A lot more work was carried out in this aspect, after the discovery of the superior carbon capture ability of MOFs, after amine-functionalization. In one such work carried out by Wang et al., a MOF named MIL-101 was modified using tetra-ethylene-pentamine (TEPA) to form TEPA-MIL-101. Both un-modified and modified MOFs were used for CO2/CO separation. The subsequent product showed an increase in CO2 adsorption capacity whereas the adsorption capacity of CO drastically dropped, and the adsorption selectivity of CO2/CO was enhanced from 1.77 to 70.2 due to amine-functionalization. 8
With the passage of time, more and more materials were synthesized and tested for their gas adsorption and separation performance. A breakthrough was made when a special class of porous materials was found to have exceptional characteristics. These materials are called Zeolitic imidazolate frameworks (ZIFs). Zeolitic imidazolate frameworks (ZIFs) are a class of metal organic frameworks, which consist of a crystal structure based on alumina-silicate zeolites with the tetrahedral alumina-silicate and bridging O atoms replaced by transition metal ions and imidazolate linkers respectively. 9 An example of this can be shown in a work carried out by Prakash et al., in which computational analysis based on experimental data, of H2 uptake capacity of ZIF-95 and ZIF-100 was done, and it was found that the adsorption capacity of the ZIFs was 3.5 and 10.5 wt% respectively. 10 ZIFs are known to adsorb gases reversibly and the adsorption is weak. This was shown by a research work carried out by Saracco et al., where oxygen from air is selectively adsorbed by co-ordination with the outer Co (II) ions in a Co-ZIF-67 to form a superoxo species, which can be desorbed using light. This shows that not only will the oxygen be adsorbed from gas mixture, it can be easily separated from ZIF as well. 11
Just like ordinary MOFs, the performance of ZIFs can also be improved and enhanced by amine-modification. Many research works have been published in this regard. An example of this was the research work carried out by Binti Yahya et al., in which they synthesized amino-functionalized ZIF-8, and the amine-modification was carried out using the wetness impregnation method, with two kinds of amine-containing compounds namely tetra-ethylene-pentamine (TEPA) and penta-ethylene-hexamine (PEHA). In this work, the adsorption capacity of amine-modified ZIF-8 was measured, and was compared with the un-modified ZIF-8. The CO2 adsorption capacity of the amine-modified ZIF-8 was measured to be 1.9983 mmol/g, and after amine-modification, the CO2 adsorption capacity was increased up to 199.6%. 12 Another work carried out by Zhang et al. detailed the advantages of amine-modification on carbon capture. Here, ZIF-8 is subjected to a process called ‘post-synthetic modification’. In this process, amine groups are incorporated into ZIF-8 using ethylene di-amine, and its adsorption capacity is greatly enhanced, whereas the CO2/N2 selectivity goes up to 23 at 0.1 bar and 13.9 at 0.5 bar. 13
There is always a limit to which, polymeric membranes can be used for gas separation. However, when mixed matrix membranes are formed, they would combine the advantages of inorganic fillers and organic polymeric membranes. 14 Similarly, in the case of MOFs, lot of research is being done with MOFs incorporated in polymeric membranes. In one such work, Basu et al. prepared a Matrimid based mixed matrix membrane, with different loadings of a MOF called MIL-53(Al) ranging from 0 to 30%. Results showed that the increase in loading of MIL-53 gives an increase in CO2 permeance up to 218%, whereas the CO2/CH4 selectivity also increased and was in the range of 28–35. 15 In one other research work, Perez et al. synthesized Matrimid based membrane with MOF-5 as filler in its structure. The incorporation of MOF-5 in the membrane increased the permeabilities of the gases increased by 120%. The CO2/N2 selectivity was found to be in the range of 36–44, and CO2/CH4 selectivity was found to be 40–51. 16
Similarly, as stated earlier, when MOFs are modified with amine groups, they can capture carbon dioxide more readily. When these amine-modified MOFs are used in membranes, they would naturally improve the permeation and selectivity of CO2 over other gases. This property of amine-modified MOF based mixed matrix membranes (MMMs) was exploited for CO2 separation from air and flue gases in a number of research works. In one particular research work carried out by Zornoza et al., the research team synthesized MIL-53 (Al) and modified it with amine groups to form NH2-MIL-53 (Al), which was in turn integrated into a poly-sulphone membrane to form the resulting matrix. This membrane gives much better separation of CO2 over CH4 and gives a maximum CO2/CH4 selectivity of 45. 17 In another similar work, MW Anjum et al. synthesized mixed matrix membranes (MMMs) composed of zirconium terephthalate UiO-66 as the filler with Matrimid as the base polymer, the MOF was functionalized by adding amine groups from 2-aminoterephthalic acid and 4-aminobenzoic acid. This resulting membrane had better CO2 separation as compared to unfilled (with no MOF) Matrimid membrane, and gives a maximum CO2/CH4 selectivity of 47.7. 18
Just like ordinary MOFs, ZIFs have also been used in membranes for facilitating gas separation. Lots of research is being carried out in this regard, to form a suitable blend for most effective gas separation. One research work carried out by Li et al., ZIF-7 was combined with poly (amide-b-ethylene oxide) (Pebax® 1657, Arkema) to form a mixed matrix membrane. The membrane thus formed has a maximum CO2 permeability (PCO2) up to 145 barrers, CO2/N2 selectivity up to 97, and CO2/CH4 selectivity up to 30. 19 In one research conducted by Bux et al., an ethane/ethane mixture was separated by adding ZIF-8 as filler on top of a titania-alumina support layer, which acted as the main membrane phase. This membrane gave an ethene/ethane selectivity of 2.8 and 2.4 at a pressure of 1 and 6 bar respectively. The fairly low selectivity of ethene over ethane is primarily due to two competing selectivities, superior ethane absorption selectivity and superior ethane diffusion selectivity. 20
If the structures of ZIFs are augmented by adding amine groups, and then these amine-impregnated ZIFs are included in the membrane structure, they can also introduce several desirable CO2 separation properties within the structure. In one such study, Nordin et al. synthesized the zeolitic imidazolate framework ZIF-8, which was then modified using ammonium hydroxide. The resulting amine-modified ZIF-8 was then dispersed in the base polymer membrane made of poly-sulphone. The resulting membrane is tested for its permeability and selectivity with CO2/CH4 mixture, the CO2/CH4 selectivity was found to be in the range of 25–43. 21 Another research on the CO2 separation from gas mixtures using amino-modified ZIF based membranes, was carried out by Huang et al., who synthesized and modified ZIF-90 using 3-aminopropyltriethoxysilane (APTES). The amino-modified ZIF-90 membranes were synthesized by coating the α-alumina support with APTES and then immersing the coated support material in the synthesized ZIF-90 solution. This forms the amino-modified ZIF-90 membranes. The resulting membrane is then tested for gas separation and for this purpose; an equi-molar CO2/CH4 mixture was used. The CO2/CH4 selectivity was up to 4.7 in this work. 22 In a fairly recent work carried out by MZ Ahmad et al., 23 CO2/CH4 separation performance for mixed matrix membranes consisting of ZIF-8 and a novel polymer, 6FDA-bisP was tested. It was found that maximum CO2 permeability of 35.3 barrers was achieved with CO2/CH4 selectivity of 25.6. In another work carried out by HBT Jeazet et al., 24 ZIF-8 was used as a filler for PSf membranes. It was found that a maximum CO2/CH4 selectivity of about 35, with corresponding CO2 permeability of 10 barrers, for a ZIF-8 loading of 8 wt%. Furthermore, work was also done by Jomekian et al., 25 ZIF-8 was used as a filler for Pebax® 1657 based mixed matrix membranes which were further used to synthesize composite membranes, with PES as the porous support layer. It was found that a maximum CO2/CH4 selectivity of 16.1 with CO2 permeability of 758 barrers was obtained. All the reported literature show the usefulness of ZIF-8 as a filler for CO2/CH4 separation, and therefore these results encouraged us to use the said filler for CO2/CH4 separation performance enhancement in pristine CA membranes as well.
In our work, we are supposed to capture and separate CO2 from different gas mixtures. So, after doing this extensive research, we had decided on using amino-modified ZIF-8. This was done primarily, because amine groups are naturally CO2 selective, 6 and ZIF-8 was used because it has exceptionally high thermal stability (stable up to 550°C) and chemical stability (it is resistant to alkaline water as well as organic solvents). 9 To fabricate ZIF-8 based membranes, we used cellulose acetate (CA) as our base polymer, because it was easily available, and polyethylene glycol (PEG) MW 1000 as a plasticizer to incorporate flexibility into the membrane and improve its mechanical properties. 26 We prepared a membrane from the solution containing 10% w/v cellulose acetate with 15% w/w (plasticizer-to-polymer) PEG MW 1000, and incorporated varying amounts of as-synthesized amino-modified ZIF-8 filler ranging from 5% to 20% (filler-to-polymer) w/w. The membranes exhibited excellent mechanical and gas separation properties for all the reported amounts of ZIF used, with an increased separation performance of CO2 over CH4 on increasing amount of the filler. Hence, the maximum mechanical properties and gas separation performance was found for 20% amine-modified ZIF-8 in CA/PEG membranes.
Experimental section
Materials
Cellulose acetate (CA), Mn = 50,000 g/mol by Gel Permeation Chromatography (GPC) was obtained from Sigma-Aldrich, St. Louis, MO, USA. PEG, Mw = 1000 g/mol, was procured from Siheung, Daejung, Korea. Ultra-pure acetone (Mw = 58.08, 99% purity GC) was purchased from Sigma-Aldrich, and used without further purification. As-prepared amino-modified ZIF-8 was obtained from another research student within the research group. For gas permeation purposes, CO2 (99.999 vol%), commercial grade N2 (99.995 vol%) and CO2/CH4 (10:90 by volume) gas mixture was used.
Membrane synthesis
Amino-modified ZIF-8 incorporated membrane was synthesized by dissolving separately, 10% w/v CA and 15% w/w (filler-to-polymer) PEG in acetone, and different weight fractions of the above mentioned ZIF in acetone, ranging from 5% to 20% w/w (filler-to-polymer). First, 10% w/w CA and 15% w/v PEG was dissolved in acetone, by stirring overnight. Then, the required amount of ZIF was subjected to 2 hours of sonication, separately in acetone. After 2 hours of sonication, the two solutions (i.e. CA/PEG in acetone and amino-modified ZIF-8 in acetone) were combined and the resulting solution was stirred overnight again. The concentration of the final solution was 10% w/w CA in acetone after optimization of concentration.
The resulting solution was cast onto solvent-washed petri dishes, and placed in the air for 30–45 minutes under atmospheric conditions. After that, the petri dishes were transferred to the vacuum oven and dried at 40°C for 3–4 hours. The fabricated membranes were tested for permeation of pure CO2, CH
Testing and characterization
Fourier Transform Infra-Red spectroscopy
Fourier Transform Infra-Red (FT-IR) Spectroscopy was conducted using Perkin-Elmer Spectrum 100 Spectrometer, to determine the different functional groups present in the fabricated membranes. The analyses were carried out in the wave number range of 4000–400 cm−1 at a resolution of 4 cm−1. Thin strands of the different membrane samples were cut to be used for the analysis.
Scanning electron microscopy (SEM) analysis
SEM analysis was done using JEOL JSM-6490LA analytical low vacuum SEM to determine the morphology and pore distribution of the different membrane samples containing 5%, 10%, 15% and 20% amino-modified ZIF-8 in a CA/PEG membrane matrix. An accelerating voltage of 15 kV was used for all the samples, with subsequent magnification of 1000×, 5000×, 10,000× and 20,000×.
Ultimate tensile strength measurement
The ultimate tensile strength measurement was carried out using SHIMADZU AGS-X series precision ultimate tensile tester with a maximum load of 50 kN. All of the samples were tested using the ASTM standard D882-02 at a strain rate of 0.5 mm/min. The stress-strain behaviour was studied for the samples containing 5%, 10%, 15% and 20% ZIF in CA/PEG mixed matrix membrane.
Gas permeation measurement
Gas permeation properties were measured using a stainless steel permeation rig. The properties were measured using pure CO2, pure CH4 and a mixture of CO2/CH4 gases. The active area of the rig is found to about 8 cm2. Measurements were obtained against pressures of 1, 2, 3 and 4 bar respectively, keeping temperature constant at atmospheric conditions. The measurements were taken manually using a bubble flow metre. The single gas permeabilities and selectivities are then obtained at the different values of pressure using this arrangement, whereas for mixed gases i.e. CO2/CH4 mixture, the permeabilities and selectivities are measured using Perkin-Elmer Clarus 580 Gas Chromatograph.
Results and discussion
Characterization by FT-IR spectroscopy
As shown in Figure 1, the presence of different functional groups is confirmed by analysing the various membrane samples using FT-IR spectroscopy. It is seen here that, there is an inverted bell-shaped curved peak at a wave number of 3484.71 cm−1 for the membrane containing 5% ZIF-8. It is because of stretching vibrations of the O-H bond. Then, there is another peak, due to the stretching vibration of Csp3-H bond at wave number of 2879.05 cm−1. Another major peak of the spectra is at a value of 1749.89 cm−1 which confirms the presence of a C=O double bond. Finally we see that there is a broadening of the peak at about 3400–3500 cm−1, which is broadened from its original bell-shape. This can be explained by the coupling of the hydrogen bonding interaction and FT-IR absorption of both O-H and N-H bonds which are found in the amine-modified ZIF. 27 Similarly, for 10%, 15% and 20% ZIF loading, we see comparable peaks ranging from wavenumber values of 3300–3400 cm−1, 2800–3000 cm−1 and 1600–1800 cm−1, which confirm the presence of N-H, O-H, Csp3-H and C=O bonds in all the membrane samples. Therefore, FT-IR spectroscopy has given us the desired results and proved to us, that our desired components are indeed present in the fabricated membranes. These same peaks are also seen in the FT-IR spectra of CA/PEG and pure CA, which proves that the pure CA and CA/PEG membranes have actually been fabricated properly and successfully, along with the CA/PEG/ZIF-8 membranes (Figure 1). Characterization by tensile testing equipment

FT-IR spectrum of (a) CA/PEG/ZIF-8, (b) CA/PEG, (c) pure CA membrane samples.
The ultimate tensile strength of all the membrane samples is measured at an elongation rate of 0.5 mm/min. This value of elongation is picked after several runs with both high and low elongation rates ranging from 0.1 to 1 mm/min. When the elongation rate is too low, the tensile strength is much higher than it should be; whereas if the elongation rate is too high, the tensile strength is much lower than expected. Only, at a moderate value of elongation rate such as 0.5 mm/min, will the membrane strength be more accurately represented.
Once the elongation rate is chosen and the experiment is carried out, it can be seen from Figure 2, that the tensile strength increases to a very high value on adding small amounts of ZIF, such as 5% and 10% ZIF. However, when the amount of ZIF is increased beyond 10%, the value of tensile strength of the membranes drops even lower than that of the membrane with no ZIF incorporated in its matrix. This shows that the mechanical properties of the membranes are best at lower ZIF loadings up to 10%.

Tensile strength of 5%, 10%, 15% and 20% ZIF based membranes.
According to Li et al., the explanation for the decrease in the tensile strength of the mixed matrix membranes (MMMs) on increasing the ZIF loading is due to the deteriorating stiffness of the polymer/filler interface. The reason is that when the area around the filler particles is stiffer than the main polymer phase, then there is less chance of crack formation and propagation. Therefore, as the amount of ZIF is increased, the area softens around the filler particle and thus the crack starts to propagate more. This reduces the tensile strength of the membrane on increasing ZIF loading, because of increased crack propagation. 28 Furthermore, it can be said that the decrease in the tensile strength of the membrane on increasing the filler loading can be attributed to chain scission of the polymeric matrix during fabrication process, while the filler loading is being increased, as was explained by H Wu et al. 29
Characterization by scanning electron microscopy analysis
As shown in Figure 3, the different samples were tested using Scanning Electron Microscopy at magnifications at 5000×, 10000×, 15000×, 20000×, 25000× and 30000×. All these samples showed perfectly dense structures, with white spots dispersed throughout the surface which showed the presence of ZIF filler particles. These analyses showed that increasing the amount of ZIF does not greatly affect the pores structure and morphology of the membranes. Moreover, the spread of the white spots shows proper dispersion of the ZIF in the membrane, which gives the membranes their superior gas separation performance. Also, tracks can be seen in the polymer structure, which is due to effect of permeation testing on the membranes, as SEM analyses are carried out after membranes are tested for gas permeation properties. However, increase in amount of ZIFs causes a formation of nano-gaps, which can be justified by increase in permeation. The formation of nano-gaps in the polymer chain on adding larger amounts of ZIFs, this can be explained by the fact that polymer chains could not properly pack with each other, which gives rise to the small voids which are the basis of nano-gaps. 30 In addition to formation of nano-gaps, the high degree of ZIF-8 loading may cause the inorganic filler particles to agglomerate within the polymer matrix, which can cause the permeability to increase as is shown by the gas permeation testing results. Moreover, this may also cause reduction in the tensile strength of the particles, as is shown by the tensile testing results to follow. 31

SEM analysis of membrane samples using 5%, 10%, 15% and 20% ZIF in CA/PEG membranes.
Gas permeation testing analysis
The mixed matrix membranes containing 5%, 10%, 15% and 20% ZIF-8 in CA/PEG membrane, are tested for the permeation of single gases using the stainless steel gas permeation rig, at subsequent pressures of 1,2,3 and 4 bars respectively. The permeation results can be summarized in Table 1.
Single gas permeation testing of CO2 and CH4 gases through MMMs.
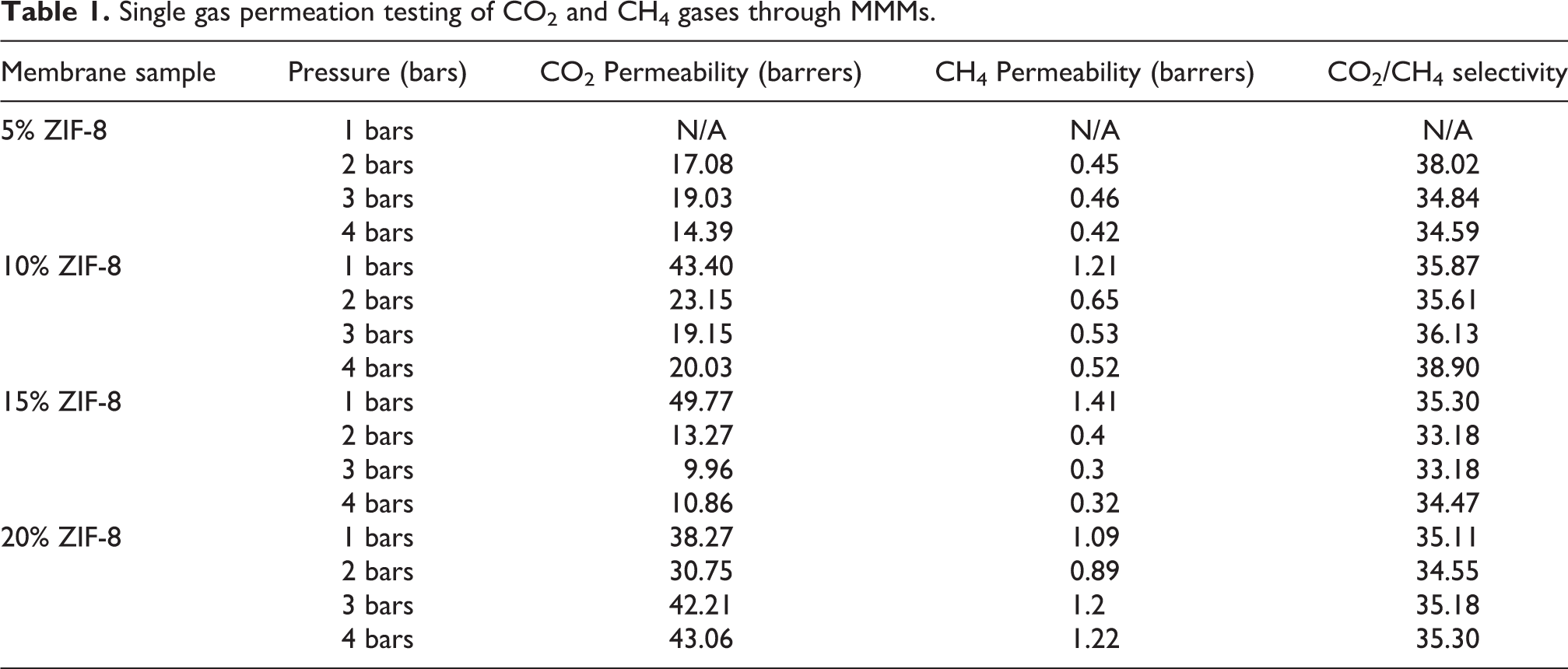
As we can see from the graph in Figure 4 and 5, the permeability mainly decreases with increasing pressure. This can be explained by the phenomenon of membrane compression on increasing gas pressure. 32 Whereas, the permeability decreased somewhat, on incorporation of ZIF up to 15%, but increased drastically on increasing ZIF loading beyond 15%, as explained by the formation of nano-gaps as described by SEM analysis. The initial decrease in permeability can be explained by the fact that there may be a more tortuous path or the gas molecules to travel to get to the permeate side, as the filler is increased. Moreover, as the amount of filler increases, the molecular chains of the polymer become less mobile and more rigid as more filler is added, which would hinder the permeability of gas molecules. 33
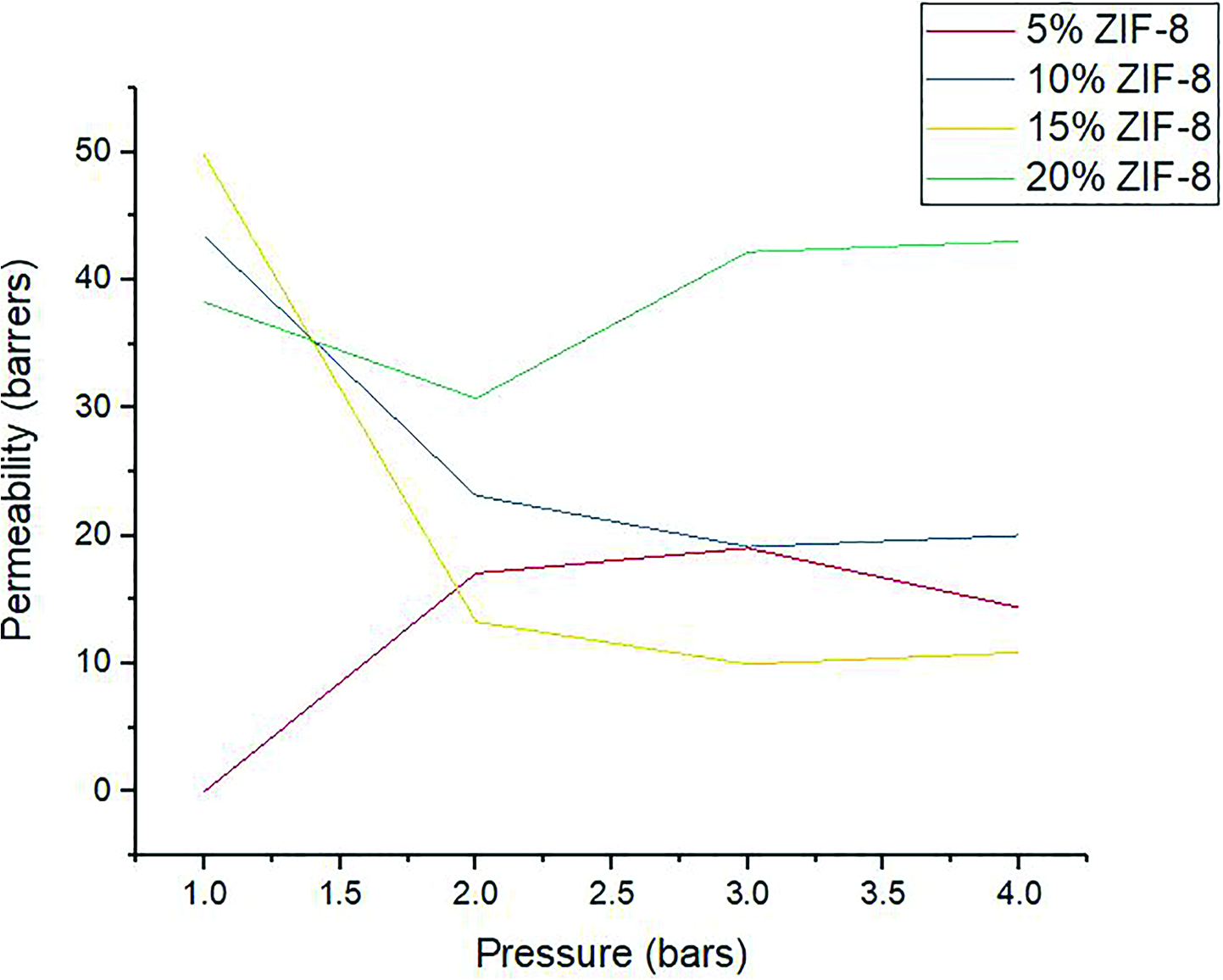
CO2 permeability of ZIF/CA membranes at different ZIF loadings over a range of pressures.

CH4 permeability (Y-axis) for different ZIF/CA membrane samples.
The trend of selectivities of the various membrane samples with change in pressure can be represented as in Figure 6.
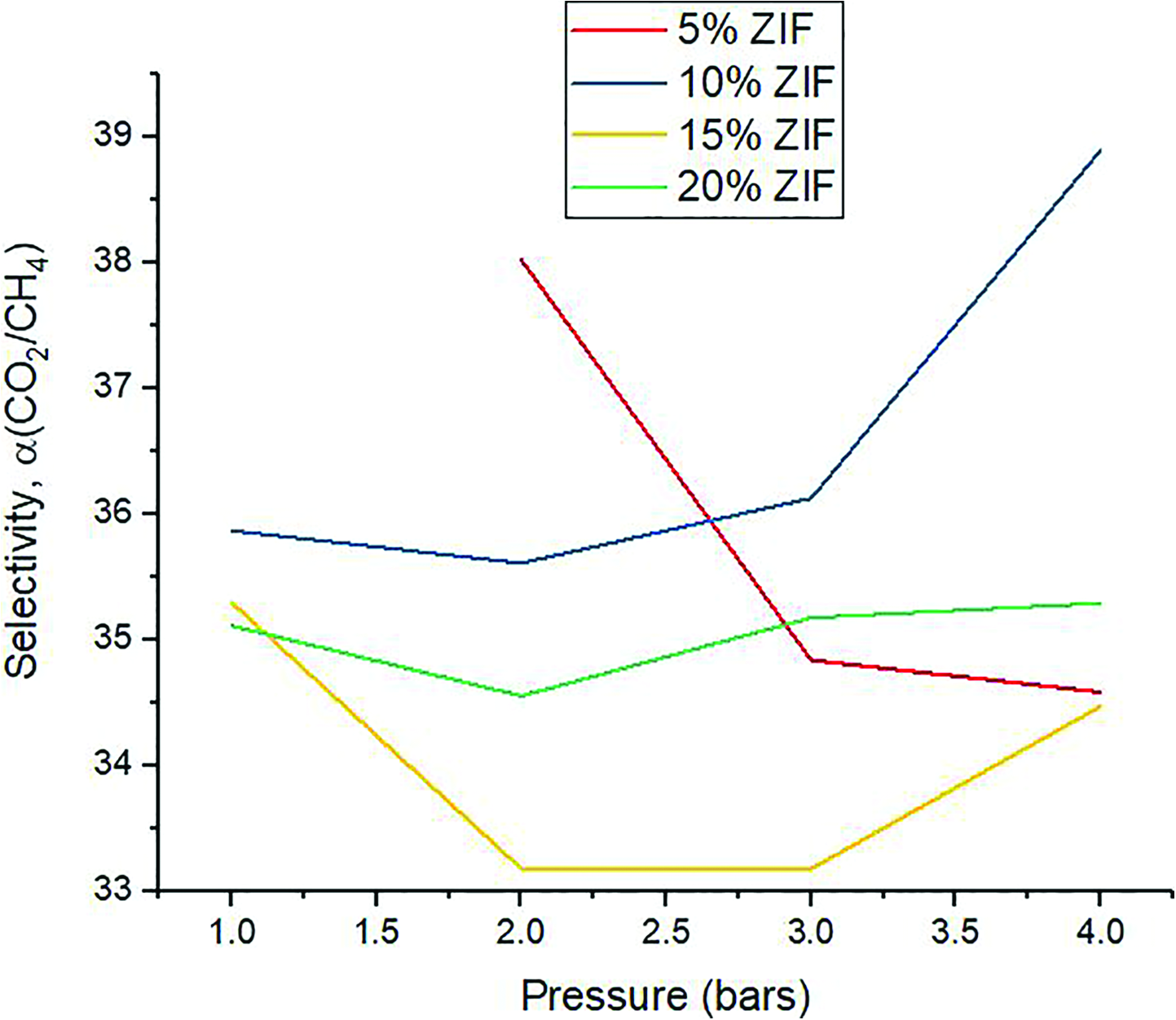
CO2/CH4 single gas selectivities for different membrane samples.
The selectivity trend can be related to the individual permeabilities of the gases, and the resulting selectivities of the different membrane samples for the gas mixture come out to be as shown in Figure 6. The results obtained for membranes containing varying amounts of amino-modified ZIF-8 must be compared with samples without ZIF-8 to gain a better perspective. In this regard, M Mubashir et al. 34 carried out gas separation tests on such membranes, and it was found that CA membranes without filler had CO2 permeability of about 3.75 barrers and a CO2/CH4 single gas selectivity of about 9.5. These values showed a considerable increase on addition of filler, as can be seen from our results. The single gas permeation results show a maximum selectivity of 38.897. This is an increase of about 309% in the selectivity, on addition of 5% amino-modified ZIF-8.
For the verification of our permeation results, mixed gas permeation was also carried out for the mixed matrix membranes. For this purpose, CO2/CH4 mixture (50:50 by volume) was used. The permeabilities of CO2 and CH4 as well as CO2/CH4 selectivities through the mixed matrix membrane can be summarized in the Table 2.
Permeation testing of CO2/CH4 (50:50) gas mixtures through MMMs.

The graphs in Figures 7 through 9, consolidate the results obtained by single gas testing through the same membrane samples, by confirming that gas permeabilities indeed decrease on increasing pressure, which was due to the membrane compression on increasing gas pressure as detailed earlier. Whereas, the permeability decreases on loading up to 15% due to the formation of a tortuous path on addition of ZIF which is also explained earlier. However, the permeability increases once the ZIF loading is increased beyond 15% and this is also explained earlier to be due to the formation of nano-gaps. All these results have been shown to be verified by the mixed gas testing experiments. In both single and mixed gas testing, it can be seen that generally the permeability of both gases decreases with increasing pressure. This can be explained by first understanding what goes on inside the membrane samples when gas feed pressure is increased. When pressure is increased, it results in two competing procedures happening within the membrane. One is the increase in permeability due to increased sorption of the gas in the membrane. Second phenomenon is the decrement in permeability because of the increase in pressure, which in turn is due to the collapsing of free volume on increasing pressure. This has been quoted earlier as well, and is explained by the process of membrane compression due to the pressure increase. 35

CO2 permeability from CO2/CH4 gas mixture through different membrane samples.

CH4 permeability from through various membrane samples from CO2/CH4 gas mixture.

CO2/CH4 mixed gas selectivities through various membrane samples.
Conclusion
As a whole, our work focused on the study of the effect of incorporating an increasing amount of inorganic filler, such as amino-modified ZIF-8, on the CO2/CH4 selectivity as well as mechanical strength of the membranes. Moreover, the improvement of selectivity on incorporation of amine groups into the ZIF was also studied as compared to un-modified ZIF-8. It was found that the selectivity of CO2 over CH4 increased slowly as the amount of ZIF added was increased, from a maximum of 34.586 at 5% ZIF loading to a maximum of 39.49 for 20% ZIF loading, for single gas permeation testing. Whereas, for mixed gas testing, the selectivity increased from a maximum of 32 for 5% ZIF loading to a maximum of 34.86 for 20% ZIF loading. The increase in the selectivity of CO2 over CH4 on increased ZIF loading, can be attributed to the presence of amine groups in the ZIF, which being basic have an inherent affinity for the acidic CO2 gas, as described earlier. Furthermore, it was seen that on increasing the ZIF loading from 5% to 20%, the maximum CO2 permeability increased from 21.41 barrers to 36.891 barrers at 4 bars pressure, whereas the maximum CH4 permeability increased from 0.669 barrers to 1.185657 barrers. This was explained by H Cong et al., to be due to the formation of nano-gaps, as described earlier. Also, it was seen that increasing the pressure caused a decrease in permeability in most cases. This was explained by previously published literature, to be due to membrane compression on increasing pressure, which was also explained earlier. Finally, it was seen that the tensile strength of the membranes increased from 20.67 MPa for unfilled CA/PEG membrane to 43.77 MPa for CA/PEG membranes with 5% ZIF. However, this value then continued to decrease on adding more ZIF, until it reached its minimum value of 14.2988 MPa which was even lower than the strength obtained with no ZIF incorporated in the matrix. This was aptly explained by literature to be due to the increased crack propagation at high ZIF loading due to the stiffening up of the polymer chains around the dispersed ZIF particles. Therefore, it was concluded that CA/PEG membrane with 20% ZIF loading was the most suitable for gas separation purposes, as it gives much better selectivity, in spite of the fact that it has much lower tensile strength values.
Footnotes
Acknowledgements
We acknowledge the support of MEMAR Lab at School of Chemical and Materials Engineering, National University of Sciences and Technology.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
