Abstract
Recently, antibacterial coatings based on graphene oxide (GO) nanocomposites have attracted many studies around the world. The use of polymers as the matrices of GO nanofillers in the nanocomposites has been explored to produce efficient coatings against bacteria. One of the most prospective applications is the incorporation of GO into biocompatible polymers, which can produce antibacterial coatings. Here, recent progresses on the antibacterial coatings of nanocomposites based on biocompatible polymers and GO are reviewed. The effect of GO filler concentrations, biocide materials, and biocompatibility are discussed to find the most efficient antibacterial activity and biocompatibility of nanocomposites. Among biocompatible polymers, chitosan (Cs), poly vinyl alcohol (PVA), and poly lactic acid (PLA) are the most popular matrices used for the nanocomposites. This review also elaborates challenges in the use of other biocompatible polymers. Future works on biocompatible antibacterial coatings should be conducted by considering the concentration of GO nanofillers or adding other materials such as essential oils to suppress the toxicity toward functional cells.
Introduction
The colonization of bacteria on surfaces can give an adverse impact on the function of instruments such as petroleum pipelines, aquatic systems, textiles, food packages, contact lenses, and medical devices.1–3 These negative impacts include the worsening of surface functions and properties 4 ; surface corrosion 5 ; bacterial contaminations6,7; biofouling 8 ; chronic infection 7 ; and even deaths. 9 Food packaging without antibacterial coatings may cause quality deterioration and spoilage of foods. The microorganisms attaching to these surfaces, e.g.: Escherichia coli (E. coli) 10 ; Staphylococcus aureus (S. aureus), 7 commonly form biofilm that may resist several antibiotics. Moreover, the type of antibacterial coating depends upon the mechanism in which the coating interacts with the bacteria, e.g.: silver nanoparticle (AgNP) coating is effective in treating E. coli, but not so toward S. aureus. This leads to a challenge in biomedical innovation focusing on the development of antibacterial surfaces by the application of surface coatings or modification of the surface architecture.11,12 With recent progress in nanotechnology, the effort to design and fabricate new antibacterial coatings based on nanomaterials has been conducted.13–15 Antibacterial coatings have several advantages including high efficiency of bactericidal activity, low toxicity, low cost, environmentally friendly, and long lifetime.16,17 Antibacterial coatings based on nanomaterials are widely used in clinics, industries, and households to prevent bacterial colonization and the spread of infections.16,17
The nanomaterials based on graphene, such as graphene oxide (GO) and reduced GO (rGO) have been widely used in coating technologies18–21 including antibacterial coatings.22–26 Although rGO has been used as an antibacterial agent, 27 GO is more potential for antibacterial coatings due to its functionalized structure, 28 good dispersion, remarkable thermal stability,29,30 and synergistic effect with other biocide agents.28,31–33 GO is accepted as the most effective antibacterial nanomaterial due to its edge cutting effect,34,35 oxidative stress effect created by reactive oxygen species (ROS) or charge transfer, 35 and cell entrapment ability. 36 Oxidative stress may damage bacteria cells by the release of ROS, whereas the cell entrapment is envisioned by confining the bacteria on the layers of GO limiting its cell division ability. 35
GO exhibits biocompatibility that makes it a promising nanofiller for some biocompatible polymers to form antibacterial coatings.37–39 GO activates antibacterial effect and enhances the mechanical strength of the polymers.40,41 In general, there are three strategies to fabricate biocompatible an antibacterial coatings based on biocompatible polymers and GO nanocomposites as shown in Figure 1. The first strategy is to directly disperse GO nanofillers into a biocompatible polymer, e.g.: melt mixing GO or rGO with poly lactic acid (PLA). 42 For some biocompatible polymers, GO filler can readily activate the antibacterial effect on the nanocomposites. The second strategy is to add biocide materials such as AgNPs, metal oxides, and antibiotics together with GO as nanofillers of biocompatible polymers. 43 The concentration of these biocide agents and the synergistic effect with GO strongly affect the antibacterial activity of the coatings. However, the excessive use of the biocide agents might decrease the biocompatibility of the coatings. The third strategy is covalently modifying GO before being dispersed into the polymers. 44 The biocompatibility and antibacterial activity can be adjusted by tuning the concentration of the functionalized GO. 45
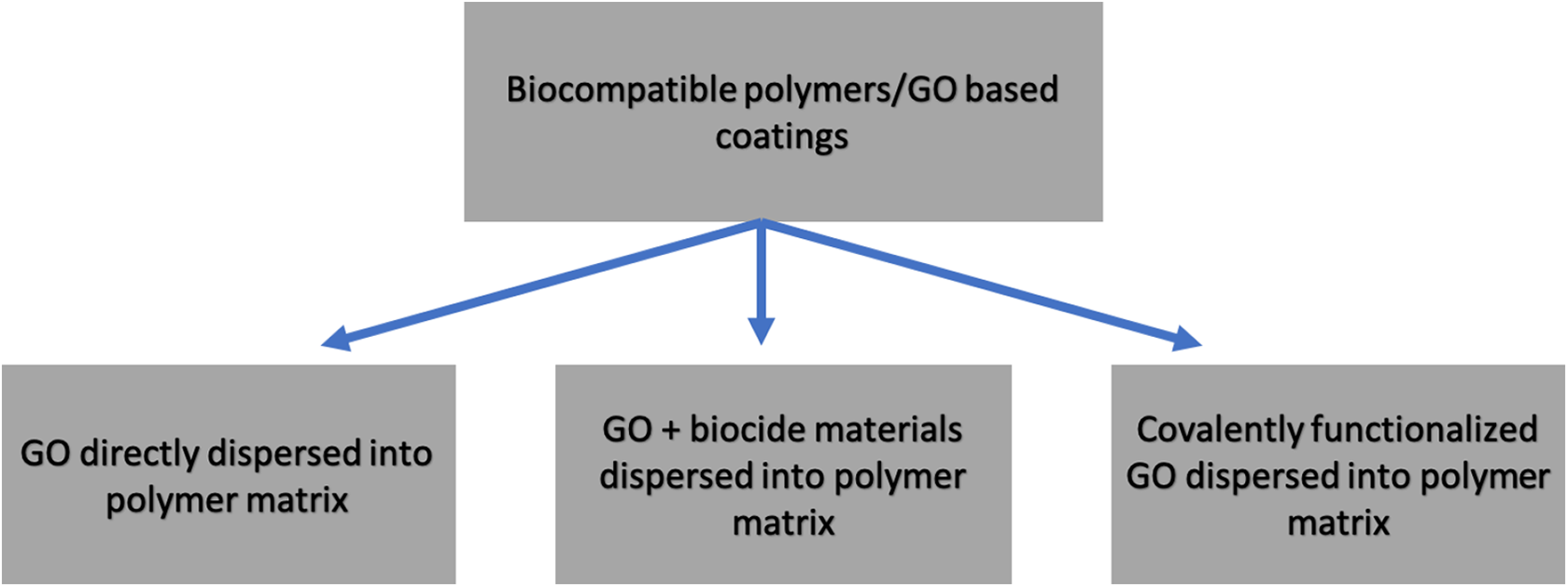
Three different strategies to obtain antibacterial coatings based on biocompatible polymers and GO.
The concentration of GO nanofillers cannot be arbitrary in order to hold or improve biocompatibility of the polymers. Optimizing GO and biocides in the biocompatible polymer matrix is crucial and challenging for fabricating biocompatible antibacterial coatings. This is because the nanocomposites should kill bacteria, but not kill normal and functional cells, e.g.: erythrocyte and human mesenchymal stem cells (hMSCs). 44 Recent studies show the possibility to produce nanocomposites with this multipurpose property.44,45 The coating process of the nanocomposite films can also influence the biocompatibility and antibacterial activity. The solution-casted film of chitosan/GO nanocomposite, for example, shows cytotoxicity when the concentration of GO is 3 wt.% 44 while the electrophoretic-deposited film of the same composite exhibits biocompatibility even when the concentration of GO is 30 wt.%. 45 The coating technique does affect the biocompatibility of these two films yet both of them still exhibit a comparable bactericidal effect.44,45
Searching nanocomposites with excellent antibacterial properties and less toxicity is still a challenge. The availability of review papers on this topic is necessary to provide the path and outlooks toward future research on biocompatible antibacterial coatings. Han et al. wrote an overview of antibacterial coatings based on graphene nanomaterials with an emphasis on antibacterial activities. 46 Henriques et al. reviewed the relation between the production process of antibacterial coatings based on graphene nanomaterials and their antibacterial performances. 47 However, this paper overviews the antibacterial performance of antibacterial coatings that have biocompatibility property. Recent progresses on the antibacterial coatings of nanocomposites based on biocompatible polymers and GO are reviewed. The effect of GO filler concentrations, biocide materials, and biocompatibility are discussed to find the most efficient antibacterial as well as biocompatible nanocomposites.
Chitosan/GO based antibacterial coatings
Chitosan (poly (1,4-b-d-glucopyranosamine)) has excellent antibacterial, 48 non-toxicity, high biocompatibility, and bioadsorbility properties.49,50 However, its mechanical strength is lacking. GO nanofillers can improve chitosan’s mechanical strength without hampering its antibacterial properties. 51 This combination may result in excellent film-forming ability with antibacterial and biocompatibility so that it is suitable for active food packaging and medical device coatings. 44 Table 1 summarizes nanocomposites materials based on chitosan/GO for biocompatible and antibacterial coatings.
Nanocomposites materials based on chitosan/GO for antibacterial coatings.

The biocompatibility and antibacterial activities of chitosan/GO nanocomposites depend on the GO concentration and the coating process. Wronska et al. reported that the addition of GO filler of 3 wt.% was excellent to combat S. aureus and E. coli. The coating also displayed improved mechanical strength compared to pure chitosan, but slightly cytotoxic. The biocompatibility of chitosan/GO can be achieved by adjusting the GO nanofiller concentration. 44 Mazaheri et al. demonstrated that by adding only 1.5 wt.% of GO into chitosan matrix polymer, the film becomes biocompatible for high proliferation of hMSCs without affecting its antibacterial activity. 51 By changing the coating method, Ordikhani et al. reported that chitosan/GO film caused no toxicity on human osteosarcoma cell line (MG-63 osteoblast-like cells) even with 30 wt.% of GO nanofiller. Moreover, the film still exhibited antibacterial activity on S. aureus with bactericidal rate of 62% in 2 hours. 45 It is noteworthy that the viability of osteoblast-like cells on the coating highly depends on the concentration of GO nanofiller.45,61
Many attempts have been done to incorporate small amount of GO nanofiller into chitosan to suppress the cytotoxicity of chitosan/GO nanocomposite yet still holding enough mechanical properties. However, this decreases the antibacterial activity. Therefore, adding biocide materials could be an efficient way to reinforce the antibacterial effect. AgNPs can be used as a biocide into chitosan/GO nanocomposites. The main reason of using AgNPs is due to their good synergistic antibacterial effect when combined with other antibacterial agents.52,62–64 Marta et al. demonstrated a stable chitosan/GO/AgNPs film that can effectively combat bacteria due to the synergistic effect of all constituents, which depends on the ratio of chitosan to AgNPs–GO. 52 Khawaja et al. fabricated chitosan/GO/AgNPs by directly dispersing GO and AgNPs into the chitosan solution. The resulted film has synergetic effects and inhibits several bacteria, e.g.: S. aureus, S. mutans, E. coli, K. pneumonia, P. aeruginosa, and S. typhi. 65 Jena et al. coated Cu-Ni alloy with chitosan/GO/AgNPs by means of electrophoretic deposition such that GO can be suppressed to only 0.025 wt. %. The coating exhibits an antibacterial effect on Pseudomonas sp. 53 The chitosan/GO/AgNPs exhibits higher antibacterial activity than most of the antibacterial agents based on AgNPs or AgNPs–GO reported so far.
Metal oxide nanoparticles are biocide materials which can be incorporated into chitosan/GO nanocomposite. Metal oxides such as iron oxide,54,55 ZnO, 56 and TiO2 57 are recognized as safe materials and have inherent antibacterial activities.57,66,67 Konwar et al. incorporated 0.1 wt.% of GO/iron oxide into chitosan to produce chitosan/GO/iron oxide nanocomposites with antibacterial activity against Methicillin-resistant S. aureus, S. aureus, and E. coli. Furthermore, reducing the concentration of GO/iron oxide down to 0.05 wt.% leads to very low hemolytic activity toward human erythrocytes. 54 Another advantage of incorporating iron oxide or metal oxide is the ability to adsorb liquid waste. Jiang et al. reported the use of chitosan/GO/iron oxide to combat bacteria and adsorb methyl orange. 55 Sanmugam et al. demonstrated the chitosan/GO/ZnO film exhibited strong antibacterial activity against E. coli and S. aureus and good dye adsorption behavior for methylene blue and chromium complex. 56 In addition, Xu et al. found that the nanocomposite based on chitosan/GO/TiO2 exhibited excellent antibacterial activity and biocompatibility. 57
Another way to improve the antibacterial activity of chitosan/GO nanocomposite is to reduce GO,58,63 and functionalize GO into phosphorylated GO, trimethylsilylated GO, 45 guanidine functionalized GO, 59 or chitosan-grafted graphene. 60 However, the use of more than 1.5 wt.% of the first three modified GO cause cytotoxicity, erythrocyte hemolysis, adsorbing the released hemoglobin, oxidizing hemoglobin, and changing catalase activity. 44 It is an open question for searching the minimum content of modified GO to be nanofillers of chitosan polymer that has good mechanical properties, antibacterial activity, and biocompatibility. The use of antibiotics may be another solution. Ordikhani et al. reported that adding 0.5 g/l of vancomycin into chitosan/GO completely kills bacteria in only 2 hours. 45
PVA is a synthetic polymer which is highly biocompatible, water-soluble, and non-toxic. It has excellent film-forming, emulsifying, and adhesive properties yet has poor mechanical properties and brittle.68–71 Incorporating small amount of GO nanofiller, i.e.0.1 wt.% to the PVA matrix improves the thermal, mechanical, and electrical properties of the film. 69 However, the PVA/GO nanocomposite does not exhibit obvious antibacterial performance. 70 Therefore, PVA/GO nanocomposite can be modified by means of adding biocide materials or functionalizing GO as shown in Table 2. Cao et al. reported that adding methylated melamine grafted polyvinyl benzyl chloride (mm-g-PvBCl), into PVA/GO nanocomposite activated the bactericidal effect which increases with increasing concentration of the biocide up to 5 wt.%. 70 The bactericidal effect is due to the positive charge density of active groups in the biocide, which attracts the negatively charged bacteria cells and damages them. This PVA/GO/mm-g-PvBCl nanocomposite could be a promising material for antibacterial coatings in food packaging and hygiene products industry.
Nanocomposite materials based on PVA/GO for antibacterial coatings.

PVA/GO/AgNPs nanocomposites show antibacterial activity depending on GO and AgNPs precursor concentrations. Surudzic et al. reported that by adding 0.01 wt.% of GO and 3.9 nM of AgNPs precursor into PVA, the resulted nanocomposite film had better mechanical and thermal properties and had antibacterial activity against S. aureus and E. coli. Its bactericidal effect was higher than the Ag/PVA film. 71 Abudabbus et al. reduced the concentration of AgNPs precursor down to only 0.25 mM yet the PVA/GO/AgNPs film still had antibacterial activity toward the same bacteria. A synergistic action of both silver and graphene causes the physical damage on bacterial membranes and accelerates the bactericidal rate. Furthermore, the PVA/GO/AgNPs nanocomposite has no cytotoxic toward peripheral blood mononuclear cells (PBMC). 72 Cobos et al. studied the antibacterial dependency of PVA/GO/AgNPs nanocomposite on GO and AgNPs concentration. They compared two PVA/GO/AgNPs nanocomposites, i.e.: the film with 1 wt.% of GO and 0.5 wt.% of silver precursor; and the film with 2 wt.% of GO and 1 wt.% of silver precursor. They concluded that the more concentration of GO and AgNPs in the film the longer the inhibition of bacterial growth. 73 Furthermore, Cobos et al. reported that the incorporation of 5 wt.% of GO/AgNPs has antibacterial activity. However, the biocompatibility of the nanocomposite toward functional cells was not studied. 74 Usman et al. combined PVA, GO, AgNPs, and starch through one pot synthesis into a nanocomposite. The starch was used to reduce GO and Ag salt in the PVA matrix. The film showed inhibition zone against S. aureus and E. coli due to synergistic effect of GO and AgNPs. 75
Functionalizing GO is also used to obtain biocompatible antibacterial nanocomposites. Pan et al. modified GO with polymeric N-halamine then mixed the functionalized GO with PVA solution to obtain a novel hybrid nanocomposite. 76 The N-halamine compound is known as an efficient antimicrobial, non-toxic, and environmentally friendly material, which can enhance the antibacterial efficacy of GO. They used the as-prepared polymeric N-halamine or poly [5,5-dimethyl-3-(30-triethoxysilylpropyl) hydantoin] (PSPH). The PSPH was covalently grafted onto GO, chlorinated, and mixed with PVA. It was concluded that GO and polymeric N-halamine enhanced the antibacterial activities with bactericidal rate nearly 100% in 30 minutes against S. aureus and E. coli. Liu et al. reported functionalized GO with zwitterionic citicoline (CDPC) to improve the dispersibility of water-based polymers and put La3+ ions to enhance antibacterial properties. The resulted PVA/GO-CDPC-La film inhibited the growth of S. aureus and E. coli. Furthermore, in vivo wound healing experiments demonstrated that the film healed wound faster post-operatively and promoted the formation of mature epidermal architecture. This finding could lead the use of the film as a wound-dressing material. 77
PLA/GO based antibacterial coatings
PLA is commonly known as a biodegradable and biocompatible polymer. Unfortunately, PLA does not exhibit antibacterial capacity and suffers from brittleness. The addition of different ratio of GO nanofillers into PLA can produce multifunctional nanocomposites with mechanical strength, antibacterial behavior, and improved biocompatibility. 39 Table 3 summarizes different route for producing antibacterial coatings of PLA/GO based nanocomposites. Liu et al. reported that incorporating 1 wt.% of GO into PLA via direct dispersion method does not give antibacterial effect. 78 However, increasing the concentration of GO up to 5 wt.% caused completely bactericidal effects toward S. aureus and E. coli within 24 hours. 42 Furthermore, the nanocomposite was biocompatible toward osteosarcoma cells. 42 The use of 10 wt.% of rGO in PLA/rGO film showed lower antibacterial than the use of GO and more toxic toward osteosarcoma cells. However, by giving an electrical stimulus over the PLA/rGO film the efficiency of bactericidal increases to 100%. 42 Kang et al. produced Poly (L-lactide) PLLA/GO film via grafting method, which enables GO to be functionalized by PLLA. 79 The nanocomposite has good polymer matrix/GO compatibility and homogeneous distribution of GO with only 0.5 wt.% of GO via electrospinning method. 79 However, physical mixing and covalent functionalization can either cause phase segregation or compromise the intrinsic properties of GO. 80 Yang et al. reported different technique to fabricate PLLA/GO film via π-π stacking process to form PLLA-conjugated GO (PLLA-c-GO), which then dispersed into PLLA solution. By using 4 wt.% of GO, the film has advantages including antibacterial activity, uniform distribution of GO in the PLLA matrix, and very low toxicity toward mammalian cells, e.g.: L929 and macrophage cells. 80
Nanocomposite materials based on PLA/GO for antibacterial coatings.
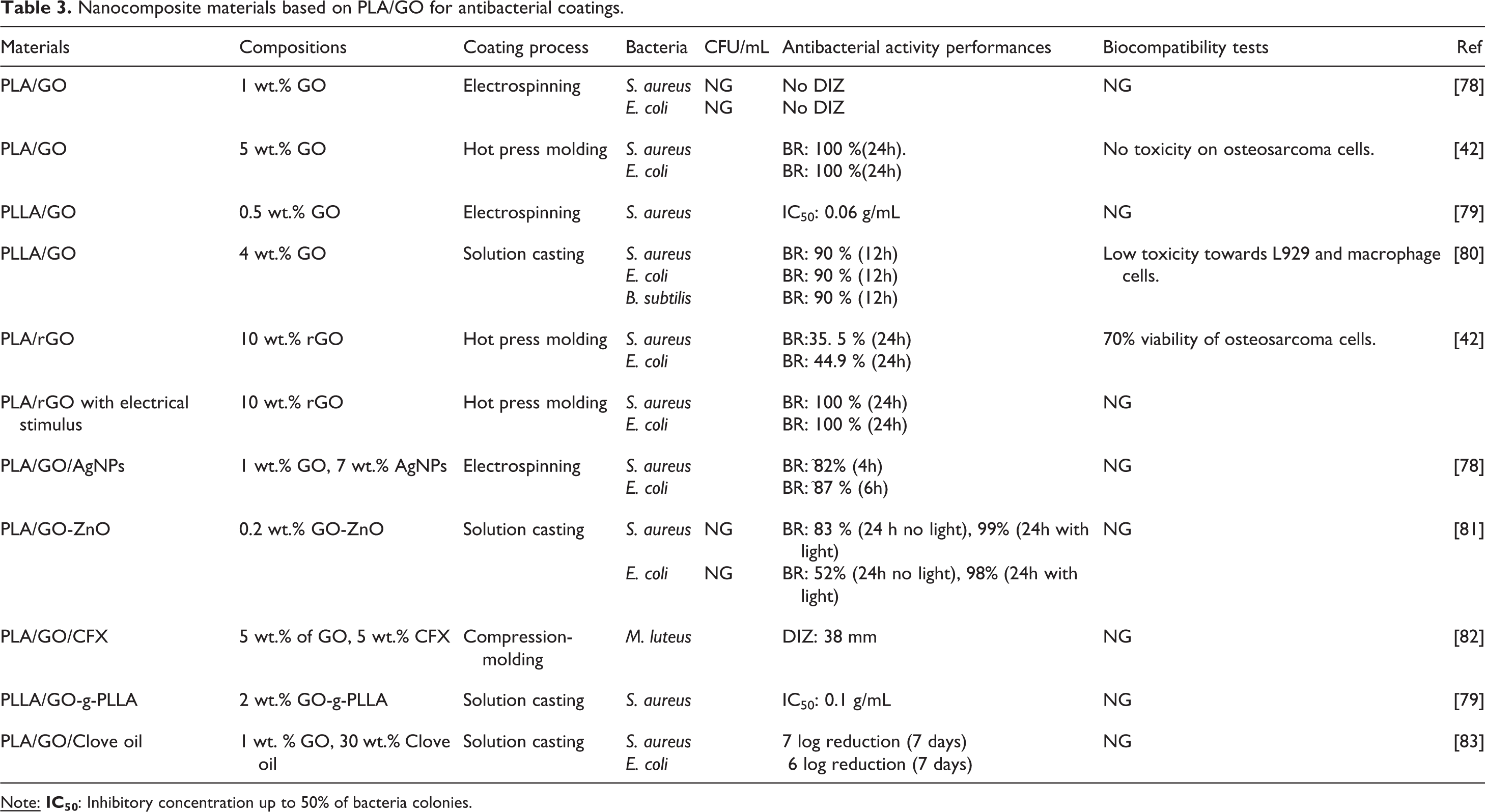
The incorporation of GO-inorganic nanocomposites into PLA matrix also offers an opportunity to develop enhanced mechanical, antibacterial, and biocompatible properties. Liu et al. developed PLA-based coatings with GO/AgNPs nanofillers through electrospinning method. They used 1 wt.% GO and 7 wt.% AgNPs incorporated into the PLA matrix to improve the tensile and bactericidal performances. 78 The film exhibits synergistic antibacterial properties that exceed the performances of individual GO and AgNPs components. 78 GO/metal oxide nanocomposites attract attention from researchers because GO could bring enhanced properties and improved performance of metal compounds. 84 Huang et al. reported the use of GO/ZnO nanofillers to improve the properties of PLA polymer. The resulted film not only keeps the preferable properties of GO and ZnO, but also exhibits the synergetic effect between them. 81 Interestingly, this film can lead to photo-induced antibacterial nanocomposite. The PLA/GO/ZnO film with 0.2 wt.% of GO-ZnO can kill 83% and 52% of S. aureus and E. coli, respectively, without light exposure. Under light exposure, the killing efficiency increases up to 99% and 98% toward S. aureus and E. coli, respectively. 81
A combination of GO and essential oil could make better antibacterial films when incorporated into PLA matrix. 83 GO is used to control the release of essential oil to combat the bacteria. However, limited studies have been carried out on the PLA based films incorporated with GO and essential oils. Arafat et al. demonstrated the use of GO nanofillers and clove essential oil into PLA matrix enhances its flexibility and lowers the oxygen permeability. 83 The film has excellent antibacterial activities against S. aureus and E. coli. Antibiotic could be used as antibacterial agent slowly released from nanocomposite. Scaffaro et al. incorporated ciprofloxacin (CFX) into PLA/GO film, which inhibits M. luteus. Moreover, the incorporation of graphene nanofiller enhances the stiffness of the film and tunes the release of CFX to combat the bacteria. 82
Table 4 summarizes other biocompatible polymers with excellent antibacterial properties by incorporating GO nanofillers.85–93 Mahdavi et al. reported the use of 1 wt.% of GO and 1 wt.% of rGO/AgNPs as nanofillers for PAM matrix. The PAM/GO was synthesized by in situ polymerization while the PAM/rGO/AgNPs was obtained via in situ reduction of silver nitrate using sodium borohydride as the reducing agent. The PAM/GO has inhibition ability toward S. aureus and Pseudomonas. Moreover, the inhibitory zone increases via reducing GO and incorporating AgNPs into the solution. 85 It is well known that AgNPs has an ability to pass through cell membranes then accumulates inside the bacteria to lead cell malfunction. 64 Hu et al. studied the different antibacterial effects of PDMS/GO and PDMS/rGO from PDMS/aerogel GO and PDMS/aerogel rGO. The antibacterial activity of PDMS/GO and PDMS/rGO is inferior only by 40% bactericidal rate toward S. aureus and E. coli. Moreover, the modification of rGO into aerogel rGO does not improve the bactericidal ability of PDMS/rGO. However, PDMS/aerogel GO exhibits the bactericidal rate rises up to 88% (3 hours) and 82% (3 hours) toward S. aureus and E. coli. 86 The study of biocompatible antibacterial coatings is still highly open for these two polymers.
Nanocomposite materials based on other polymers/GO for antibacterial coatings.
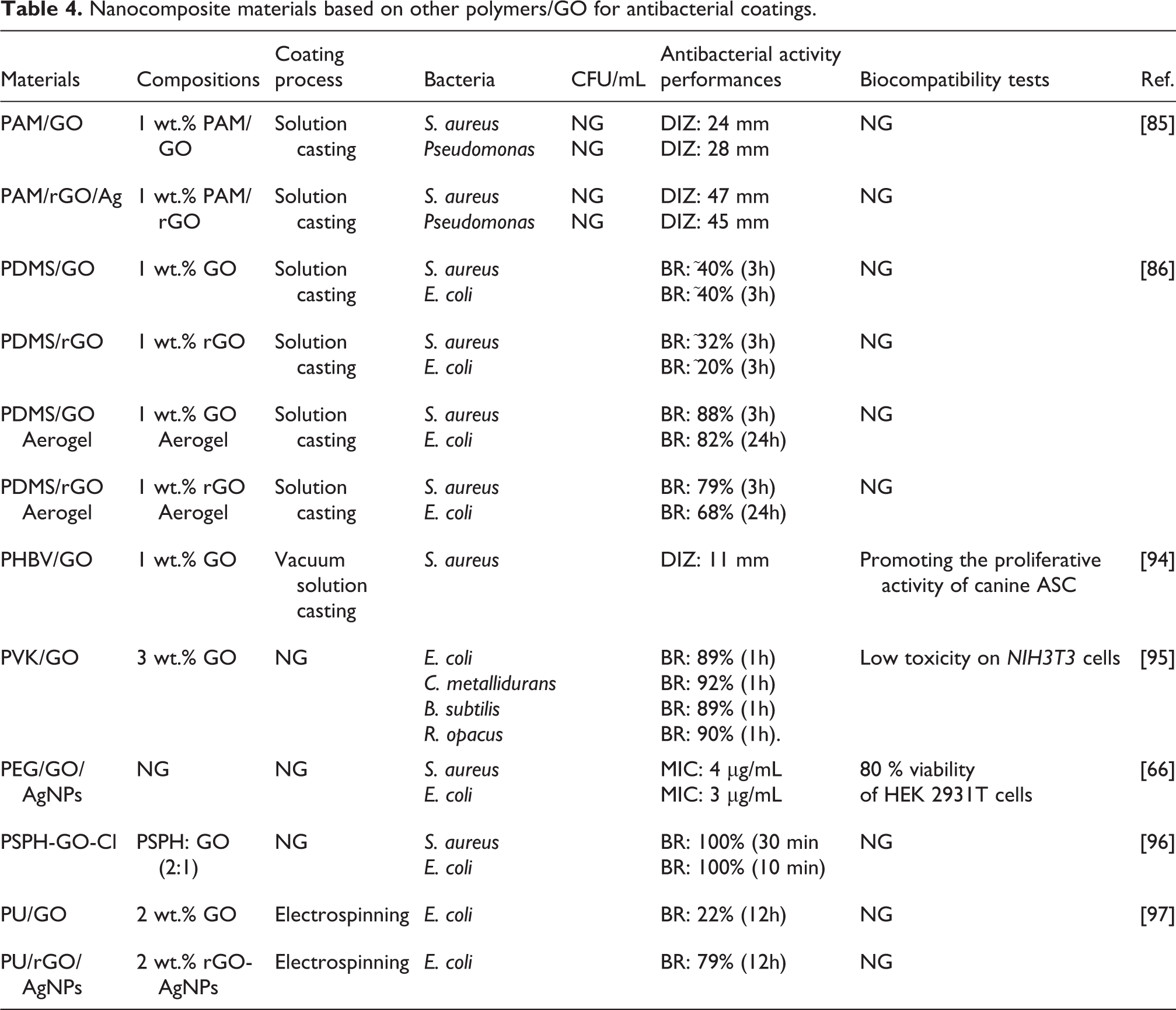
Rivera-Briso et al. prepared Poly(3-hydroxybutyrate-co-3-hydroxyvalerate) (PHBV) filled with 1 wt.% of GO nanosheets. The PHBV/GO (1 wt.%) can inhibit the growth of S. aureus and promotes the proliferative activity of canine adipose derived mesenchymal cells (ASC). The film had improved mechanical strength due to the GO. 94 Carpio et al. reported the use of 3 wt.% of GO as nanofillers of polyvinyl-N-carbazole (PVK). The resulted film presented excellent antibacterial properties without significant cytotoxicity to mammalian cells. In addition, PVK/GO film completely inhibited the growth of bacterial film in 48 hours and still presented a significant toxic effect with a bactericidal rate of 89%, 92%, 89%, and 90% toward E. coli, C. metallidurans, B. subtilis, R. opacus, respectively. 95 Pan et al. have modified PSPH/GO by changing GO into chlorinated GO to kill S. aureus and E. coli with efficiency of 100% in 30 minutes and 10 minutes of incubation time, respectively. 96
Other examples of biocompatible polymers incorporated to GO are PEG and polyurethane (PU). These are biocompatible polymers that have no antibacterial effect even after being combined with GO.66,97 Incorporating AgNPs could improve the bactericidal effect of PEG/GO and PU/GO. Chen et al. developed PEG/GO film incorporated with AgNPs, which exhibited antibacterial activities toward S. aureus and E. coli and low toxicity toward mammalian cells. 66 Pant et al. reported the production of PU coating containing rGO-AgNPs via the electrospinning technique. Even though the distribution of GO nanosheets in the nanocomposite was uniform, its bactericidal rate was only 22% (12 hours) toward E. coli. It indicates that PU/GO nanocomposite does not have clear antibacterial behavior. However, the bactericidal rate of PU/GO was improved up to 79% (12 hours) by reducing its GO with addition of AgNPs. 97
Perspective and conclusions
This mini review highlighted the recent development on biocompatible polymers/GO nanocomposites for antibacterial coatings. GO nanofillers are very useful to enhance mechanical strength of the polymers and to combat bacteria. However, there is a limit for GO addition on the polymer to cause a cytotoxicity effect, which is undesired for functional biocompatible coatings. The biocompatible polymers emphasized in this mini review are chitosan, PVA, and PLA. The cytotoxicity is one of the main hurdles for the optimum coatings. Even though the bactericidal rates of some nanocomposites are high, the cytotoxicity toward mammalian and functional cells increases with a large concentration of GO fillers. The toxicity may even be higher in the presence of biocide materials especially metal oxides and AgNPs. Therefore, the optimization of biocompatible antibacterial coatings depends on the concentration of GO fillers, addition of biocide materials, and coating processes. There are three main challenges for future studies, viz.: controlling the GO filler, modifying the polymer/GO composites with bactericidal agents, and testing the nanocomposite’s biocompatibility. Studies on adding essential oils such as clove oil are very prospective for developing antibacterial coating with biocompatibility. Further works related to theoretical studies and developing available antibacterial coatings are strongly needed to study exact mechanism of the antibacterial effect. Future studies may also include the potential applications of the coating toward viruses and fungus.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to thank UNY for providing financial support through the Research Group (RG) grant with grant number: B/189/UN34.13/PM.01.01/2020.
