Abstract
Nanoparticles need to be well dispersed in a polymer matrix to employ their advantages. Their agglomeration may cause defects and stress concentrations, deteriorating the final properties of polymer/graphene nanocomposite. Here graphene oxide (GO) was functionalized in several ways to improve its interaction with polyethylene and dispersion. Polyethylene-grafted maleic anhydride (PE-g-MA) with strong polar groups was used as the matrix to consider a high potential of enhanced interaction with the GO nanoparticles and therefore obtain a high-performance polyolefin composite. First, GO was produced by modified Hammer’s method from graphite. The GO was aminated by phenylene diamine and named FGO. Also GO was reduced chemically to obtain CRGO and thermally to obtain TRGO and then the recent ones were amino functionalized, named FCRGO and FTRGO, respectively. In another route GO was first amino functionalized and then was reduced chemically and named FCRGO+. The synthesized nanoparticles were characterized by visual tests, FTIR, XRD, SEM, and TGA analysis. Subsequently, the study investigates the fabrication of nanocomposites using these nanoparticles at 2 wt% in the PE-g-MA matrix by solution-casting method. The nanocomposites were characterized by optical microscopy, tensile and rheometric tests, FTIR, DSC, and SEM analysis. Based on the results, FCRGO+ was identified as the optimum nanoparticle having better dispersion, improved interfacial interaction, and stronger mechanical properties. The GO functionalized nanocomposites showed up to 36% in tensile modulus, 56% in tensile strength and 70% in elongation at break as compared to those of GO nanocomposites without functionalization of GO.
Introduction
Graphene, a nanoparticle with outstanding properties like strong mechanical properties and high thermal and electrical conductivity, is an excellent alternative for mixing with polymers to obtain high-tech nanocomposites. Polyolefin thermoplastics e.g., polyethylene (PE) and polypropylene own the major share of the polymer market and are growing in demand due to developments of properties and applications. Suitable physical-mechanical properties, toughness, chemical resistance, availability, cost, and processing are some of their advantages. Polyethylene (PE) has low modulus and strength so that improvement of these properties is of great interest to industry. Adding Nanoparticles to PE is one way to enhance its modulus and strength. PE-based nanocomposites suffer from poor interfacial interaction between PE and nanoparticles. 1 For example, a 58% decline in elongation at break was observed by Bhattacharyya et al. 2 when they blended ultra-high molecular weight polyethylene (UHMWPE) with 1 wt% thermally reduced graphene oxide (TRGO) even by a novel solution mixing. Recently, Kumar Sing et al. also incorporated reduced graphene oxide into UHMWPE for biomedical applications. They observed that the addition of 1 wt% nanoparticles resulted in optimal mechanical and thermal properties. 3 Castaelain et al. 4 studied the properties of high-density polyethylene (HDPE) nanocomposite containing 1 wt% graphene. Although they achieved a good dispersion of nano-platelets in HDPE matrix, they observed a 59% decline in elongation at break and a 15% decrease in Young’s modulus. Afterward, they chemically modified the graphene and observed a 31% enhancement in elongation at break and an insignificant change in Young’s modulus. 4 Another study on low-density polyethylene (LDPE) and graphene nanoplatelets (GNP) demonstrated that the addition of 3 wt% GNP increases Young’s modulus by 15%, reduces elongation at break by 94%, and compromises strain hardening behavior. 5 This effect is attributed to weak interactions between graphene and PE. On the other hand, modified graphene shows good compatibility with some polar polymers. For instance, 1.8 vol% chemically reduced graphene oxide (CRGO) improved poly(vinyl alcohol) (PVA) in tensile strength and Young’s modulus by 150% and 10 times, respectively. 6 Nanocomposites based on polar polymer matrix show improved mechanical properties due to good interactions between polar groups of the modified graphene and the matrix. Vinyl ester/TRGO thermoset composite showed improved mechanical properties. 7 Singh Bedi et al. 8 demonstrated that graphene nanoplatelets exhibit strong interaction with polyethylene terephthalate glycol, originating from π-π interaction, leading to mechanical improvement. However, they did not specify the properties of the graphene nanoplatelets such as aspect ratio and thickness. 8 Therefore, it is necessary to chemically modify graphene or PE to improve interfacial interaction between them. PE grafted maleic anhydride (PE-g-MA) is a good candidate as the chemically modified PE.1,9
The functionalization of graphene nano-platelets by organic molecules, which can interact with PE or the maleic anhydride groups on PE, improves the interfacial interaction of these nano-platelets and PE matrix. In 2014, Ren et al. 10 utilized dodecyl amine to functionalize graphene nano-sheets. They showed that dodecyl amine acts as a compatibilizer and improves the dynamical-mechanical and thermal properties, and the crystallinity of PE/functionalized graphene (1 wt%). 10 In another work, octadecyl amine was used to functionalize graphene oxide for compatibilization with PE. 11 They observed uniform dispersion and enhanced electrical conductivity due to the better interaction between nanoparticles and matrix. 11 Castaelain et al. 4 used three methods to functionalize graphene oxide and covalently graft with functionalized PE (polyethylene mono-alcohol). Their results showed a significant improvement in the mechanical properties compared to un-functionalized graphene oxide/PE nanocomposite. 4 In the previous work, our group synthesized and functionalized graphene oxide by ethylenediamine to graft with PE-g-MA. Ethylenediamine acted as a bridge between graphene sheets and PE chains and promoted the mechanical properties of the nanocomposite. 1 H. Madhad and D. Vasava summarized various graphene functionalization methods for fabricating polyamide/graphene nanocomposite. 12 Molecular dynamics simulation demonstrated an optimum value in grafting length and concentration to attain the best mechanical strength. 13
Many investigations have tried to functionalize graphene and blend it with polymers to achieve a nanocomposite with enhanced mechanical properties. However, for polyethylene, the attempts have face the issue of losing ductility and strain hardening behavior of PE with the addition of graphene. In this paper, seven graphene derivatives were synthesized and blended with PE-g-MA, as matrix, to compare dispersion and interaction of each nanoparticle with the matrix and finally reach a nanocomposite in which strain hardening behavior and high elongation at break of PE is preserved for the maximum value possible. Graphene oxide (GO) was synthesized and reduced using both chemical (CRGO) and thermal methods (TRGO). All oxidized graphene nanoparticles were aminated by phenylene diamine and named FGO, FCRGO, and FTRGO. FGO was also reduced chemically to achieve FCRGO+. They all were characterized and compared together. This paper attempted to provide comprehensive data about all of them and their synthesis method. Then, the synthetized nanoparticles were used to fabricate nanocomposite samples. Achieving uniform and high dispersion state along with establishing strong interaction within the matrix are necessary to attain enhanced mechanical properties for PE with preserved strain hardening behavior. Therefore, maleic anhydride groups on PE chains were grafted to the amino functionalized nanoparticles. The morphology, mechanical and rheological properties, and crystallinity of the nanocomposites were investigated. This paper aims to enhance interactions between graphene nanoparticles which are polar and PE chains which are non-polar to achieve an improved dispersion state of graphene nanoparticles in PE matrix. The mechanical and rheological properties of the nanocomposites are investigated. These nanocomposites hold potential for producing long-life PE materials suitable for various applications such as pipes and geomembranes due to their expected enhanced slow crack growth (SCG) resistance and creep resistance. Another work is being done on the use of these PE/graphene nanocomposite with an adequate dispersion state to enhance the SCG resistance and creep resistance of these nanocomposites. To best of our knowledge this is for the first time to develop a PE/graphene nanocomposite with enhanced modulus, yield strength and strength at break without losing high elongation at break of PE.
Experimental details
Materials
Graphite flakes purchased from Superior Graphite, PRC, with an average size of 500 microns. Sulfuric acid (97%–98%), hydrogen peroxide (30 wt%), 1,3-phenylene diamine (>98%), and isopropanol (99.7%) were supplied by Merck, Germany. Sodium nitrate (99%), potassium permanganate (99%), and hydrazine hydrate solution (50 wt%) were obtained from Sigma Aldrich, Germany. Methanol (98%) and xylene (97%) were obtained from Mojallali Co., Iran. Polyethylene-grafted-maleic anhydride, PE-g-MA, (ME0420) was purchased from Borouge, Saudi Arabia.
Synthesis of graphene oxide
A Modified Hummer’s method was used to prepare graphene oxide. The process progressed by utilizing Elhamnia’s report. 1 First, a 500 cc beaker was placed in a 0°C bath. 100 mL sulfuric acid, 2 g graphite flake, and 2 g sodium nitrate were added to the beaker and mixed strongly for 15 min. Then, 12 g potassium permanganate was gradually added to the solution. After 30 min of mixing and cooling, the ice bath was removed. The suspension was consistently stirred for 48 h at room temperature. Subsequently, 184 mL deionized water was added within 15 min. Afterward, the mixture was treated with 560 mL warm water (60°C) and 40 mL H2O2 solution (30%). The final suspension was washed with methanol for five times and then with deionized water. Eventually, graphene oxide was obtained after drying in a vacuumed oven at 60°C for 24 h.
Synthesis of reduced graphene nanoparticles
Thermally reduced graphene oxide (TRGO) was prepared by heating GO at 800°C for 10 min in
Functionalization of graphene nanoparticles
Full name of synthesized nanoparticles and their abbreviations.

Fabrication of polyethylene/graphene nanocomposites
All graphene nanoparticles were dispersed in the PE-g-MA matrix in boiling xylene while mixed with a magnet stirrer. The GO nanoparticles were dispersed in xylene using a solvent exchange process. First, 0.05 g of each nanoparticle was dispersed in water and sonicated for 2 h. Then, the particles were centrifuged for 15 min at 8000 rpm to separate water. The precipitated nanoparticles were dispersed in isopropanol, sonicated for 2 h, centrifuged for 15 min at 8000 rpm, to isopropanol be separated. Then, the nanoparticles were dispersed in 75 mL xylene and sonicated for 5 h. Subsequently, 2.5 g PE-g-MA was dissolved in this suspension while xylene was boiling in a reflux system for 1 h. Eventually, it was dried at 60°C for 24 h.
Characterization
The chemical structure of the synthetized nanoparticles was characterized using Fourier-Transformed Infrared (FTIR), EQUINOX 55 spectrophotometer, Bruker, Karlsruhe, Germany. The degree of exfoliation of nanoparticles was also measured by X-Ray Diffraction (XRD) PW 3040/60, Philips X’Pert, Netherlands (

Results and discussion
The results will be discussed in three different sections: the characterization of (I) oxidized graphene nanoparticles (GO, CRGO, and TRGO), (II) amino functionalized graphene nanoparticles (FGO, FCRGO, FTRGO, and FCRGO+), and (III) their nanocomposites with PE-g-MA.
Oxidized graphene nanoparticles characterization
Dispersion in different solvents (visual test)
As shown in Figure 1, CRGO and TRGO did not disperse in water, representing the separation of many oxygenated functional groups from graphene sheets and their hydrophobic behavior. They have also completely dispersed in xylene, which shows their non-polar characteristics. GO has been dispersed in polar solvents but aggregated in non-polar ones, which means hydrophilic properties due to oxygen-containing groups on the graphene surface. Dispersion of oxidized graphene nanoparticles in different solvents. (a) water, (b) iso-propanol, (c) xylene.
Fourier transform infrared spectroscopy analysis
Fourier Transform Infrared Spectroscopy (FTIR) is widely used for characterizing functional groups of nanoparticles and polymer nanocomposites. Figure 2 shows the spectra of GO, CRGO, and TRGO. The peak at 3406 cm FTIR spectra of GO, CRGO, and TRGO nanoparticles.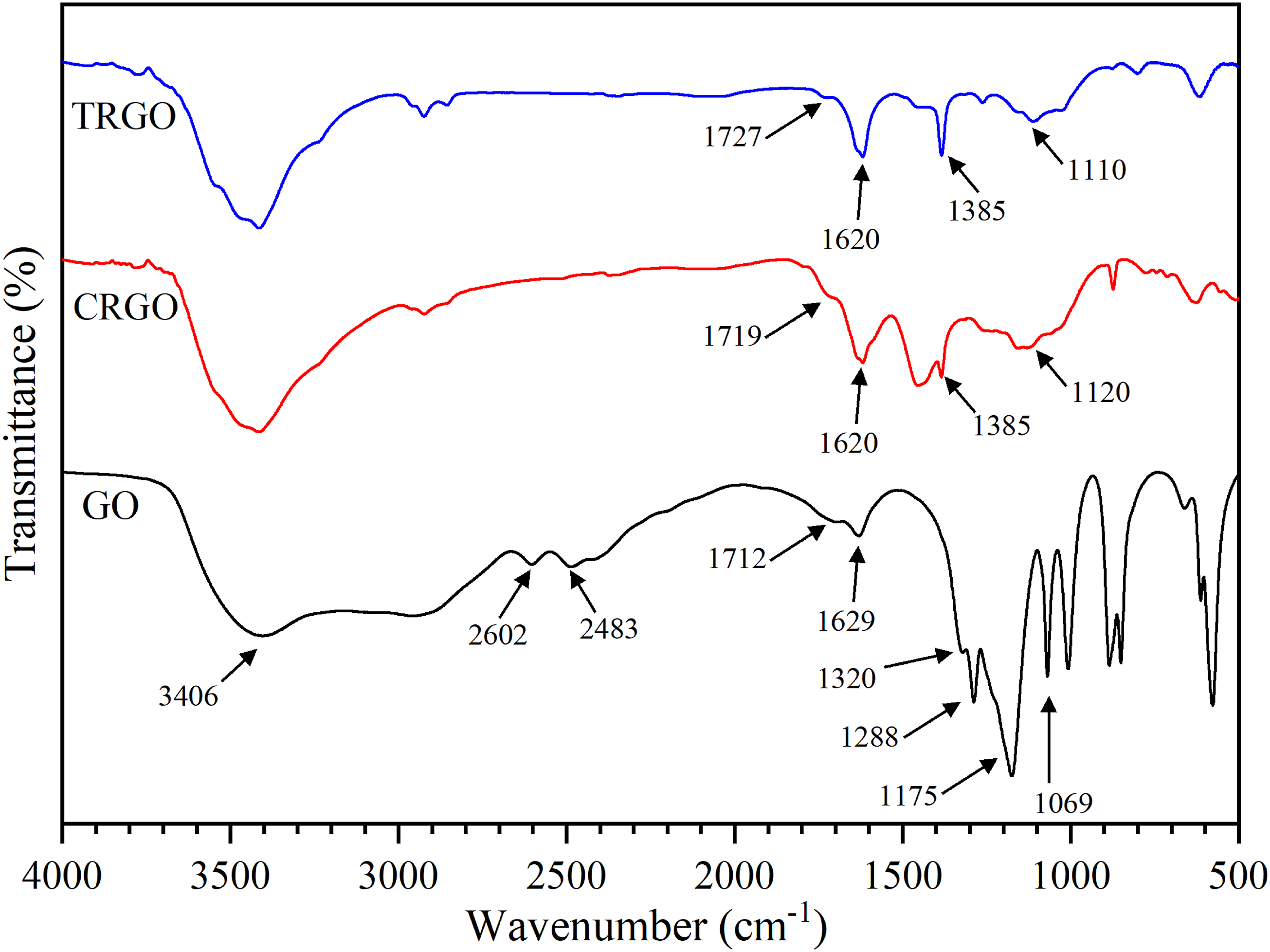
The percentage of the removed oxygen containing groups using FTIR spectra.

X-ray diffraction analysis
X-Ray Diffraction (XRD) analysis is a common and essential method to study the degree of exfoliation of graphene nanoparticles.
20
It is also used for measuring the layers’ thickness quantitatively. The d-spacing of layers will be studied using the peak degree ( XRD spectra of Graphite, GO, CRGO, and TRGO nanoparticles. XRD parameters of oxidized graphene nanoparticles. afull width at half maximum
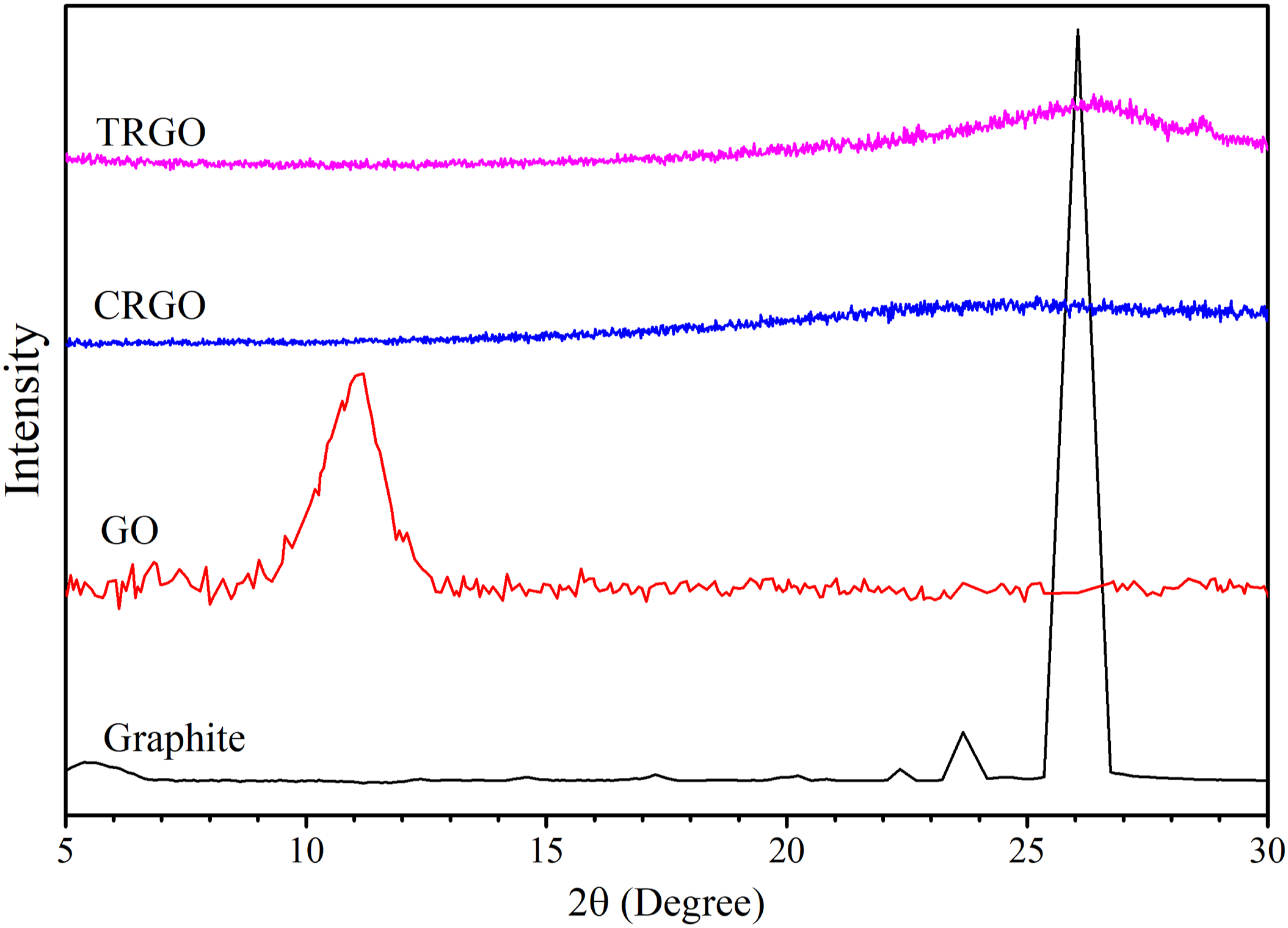
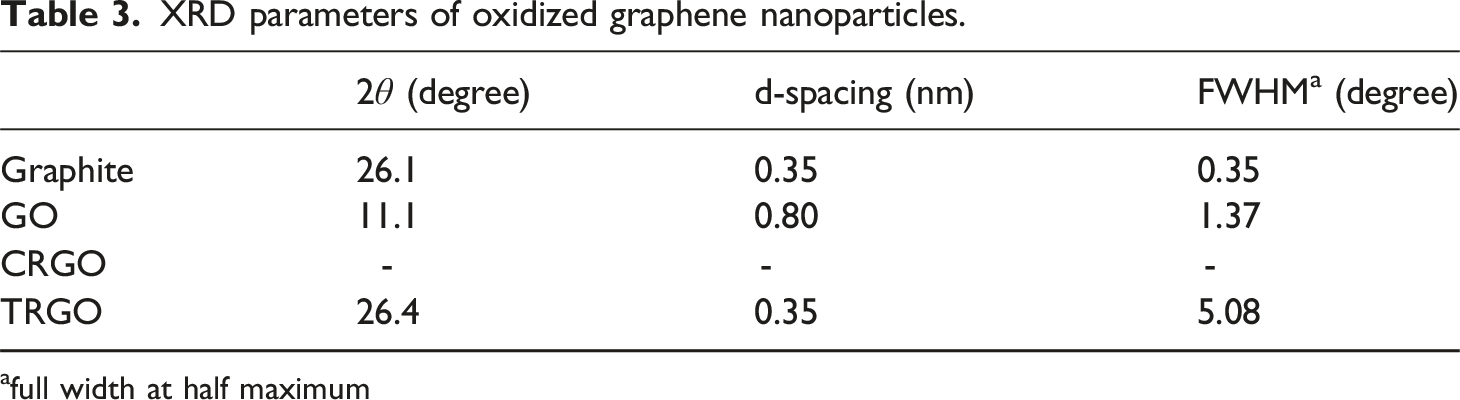
The sharp basal peak of graphite appears at 2θ = 26.1°, representing a d-spacing of 0.35 nm. This peak shifts to lower angles of
Field emission scanning electron microscopy analysis
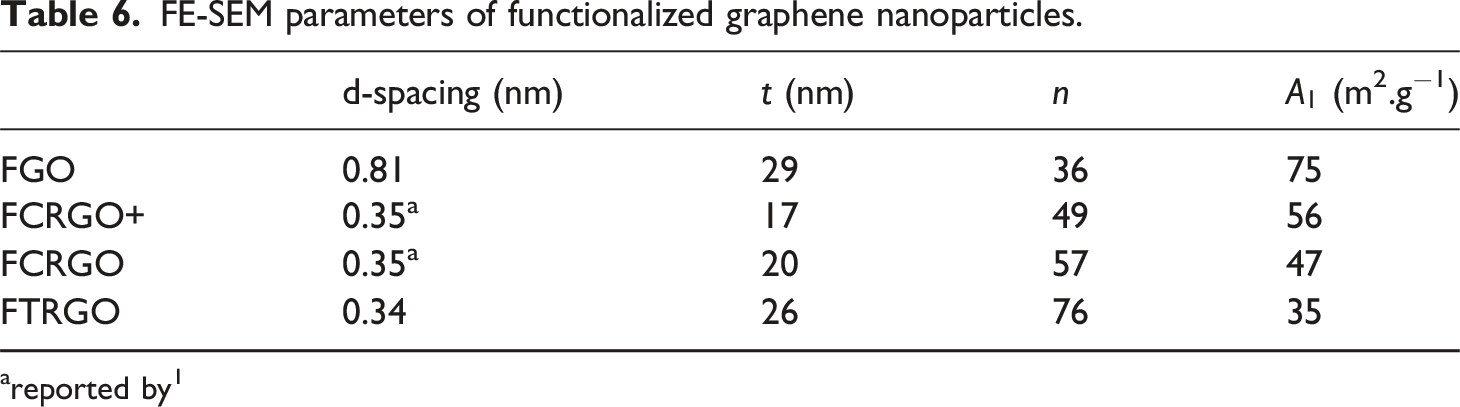
Figure 4 shows the FE-SEM images of all oxidized graphene nanoparticles. It is shown that all graphene derivatives have a 2D structure. More exfoliation is obvious due to the GO reduction. The nano-platelets’ thickness and length are measured and reported in Table 4. FE-SEM images of oxidized graphene nanoparticles. (a,b)GO, (c,d)CRGO, (e,f)TRGO. Scale bars in the left images are 500 nm, and the right ones are 2 μm. The calculated parameters of oxidized graphene nanoparticles extracted from FE-SEM images. areported by
1

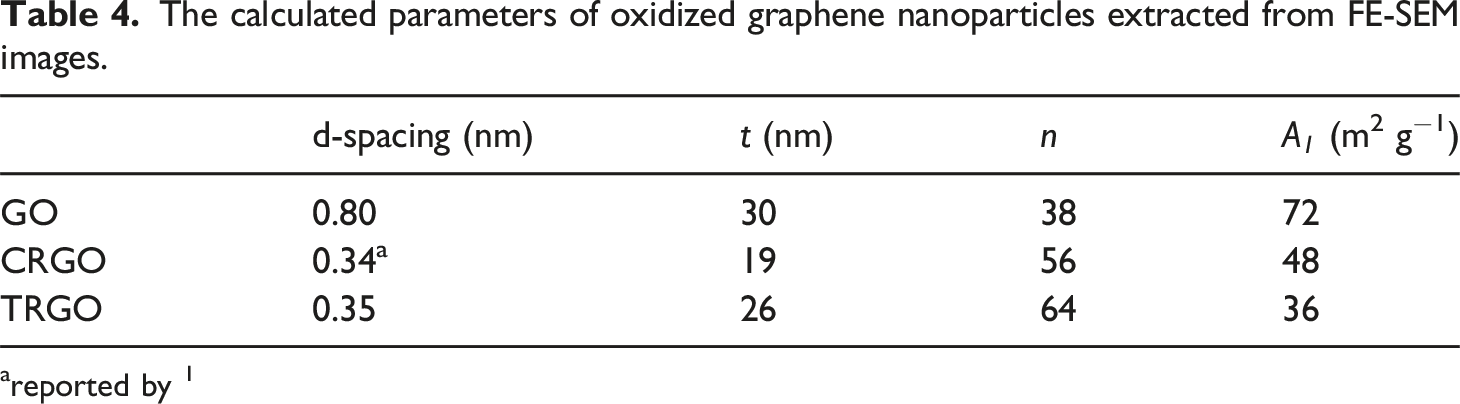
GO showed a specific surface area of 72 m2.g−1. It is shown that by reduction of GO in both chemical and thermal methods, it decreases to 48 and 36 m2.g−1, which corresponds to the destruction of the graphene sheet structure. As a result, it can be concluded that any thermal and chemical processes will break down the sheets and create defects in the structure of nanoparticles. Others have also reported this conclusion. 24
Thermogravimetric analysis
The thermal stability and comparison of functional groups quantity on the graphene surface studied by thermogravimetric analysis (TGA). They can be measured by following the weight loss of nanoparticles while temperature increases. Figure 5 shows TGA and DTG analysis of three different graphene nanoparticles. It is shown that their humidity vaporized at the first stage. GO has lost almost 59% of its weight until 800°C, representing the removal of moisture and oxygen-containing groups. CRGO and TRGO showed almost 30% weight loss, which shows fewer oxygen-containing groups than GO. It can be concluded that nearly 50% of oxygen groups were reduced after both types of reductions. However, As seen in the figure, the weight of CRGO and TRGO were decreased in diverse ways. CRGO lost major part of its weight between 100 and 250°C, but TRGO’s weight was lost at about 550°C. This observation revealed that the different oxygen-containing groups exist on the surface of TRGO and CRGO. According to Acik et al.
25
work, the amount and type of remaining oxygen-containing groups during the thermal reduction of GO depends on the initial oxygenated groups, temperature, and heating rate. Also, hydrogen atoms can be separated from hydroxyl groups and form radicals, which can react with the adjacent carbon and form ether groups. Thus, it can be concluded that although CRGO and TRGO have the same number of oxygen-containing groups, the types of these groups are different. TGA and DTG analysis of GO, CRGO, and TRGO nanoparticles.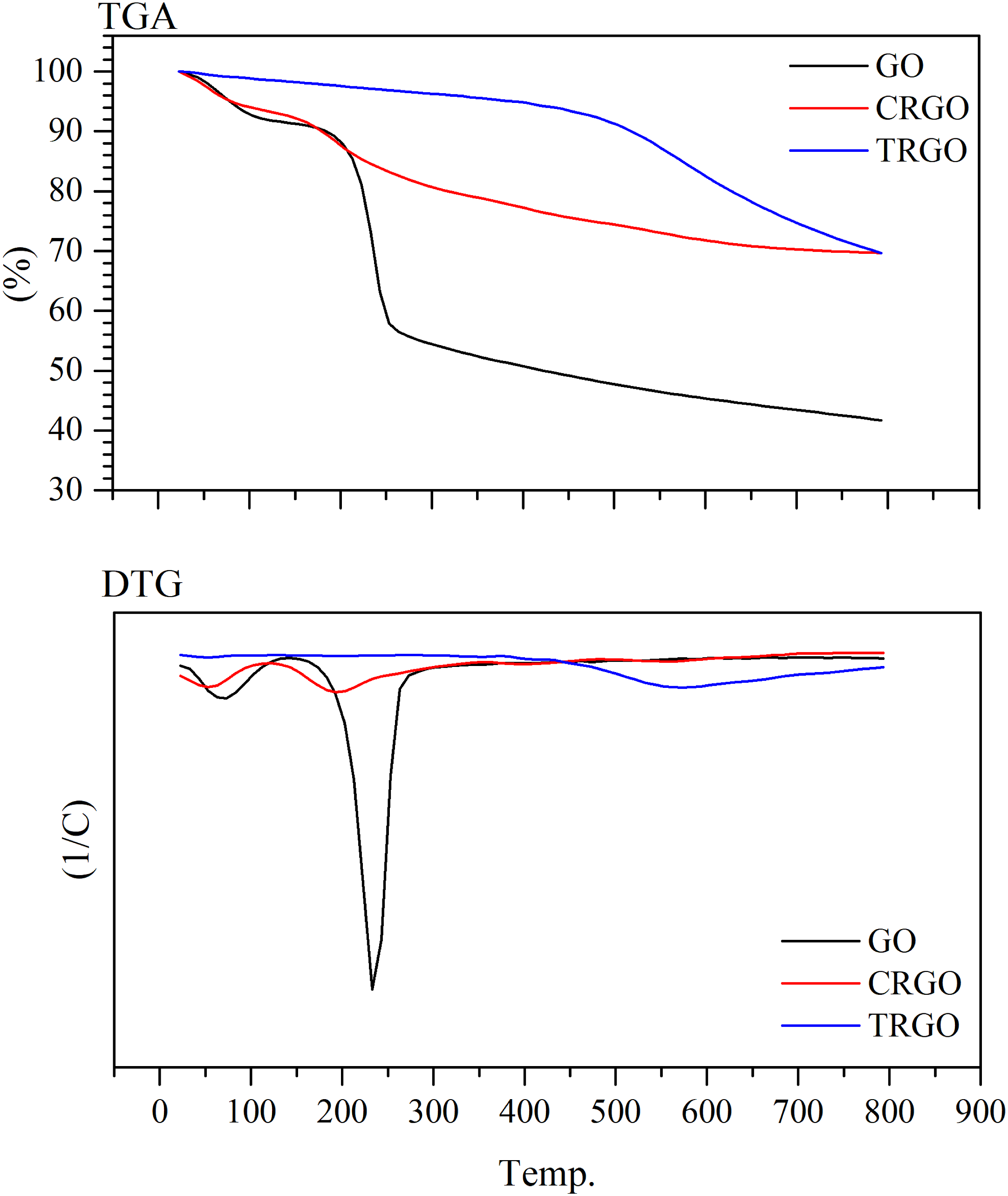
Functionalized graphene nanoparticles characterization
Dispersion in different solvents (visual test)
As Figure 6 shows, FGO dispersed in all three solvents. During functionalization, some oxygen-containing groups were replaced by phenylene diamine groups. Phenylene diamine has benzene groups with Dispersion of amino functionalized graphene nanoparticles in different solvents (a) water, (b) iso-propanol, (c) xylene.
Fourier transform infrared spectroscopy analysis
Figure 7(a) shows the FTIR of GO and FGO. The peak at 3322 and 3150 cm FTIR spectra of nanoparticles, (a) GO and FGO, (b) CRGO, FCRGO+ and FCRGO, (c) TRGO and FTRGO.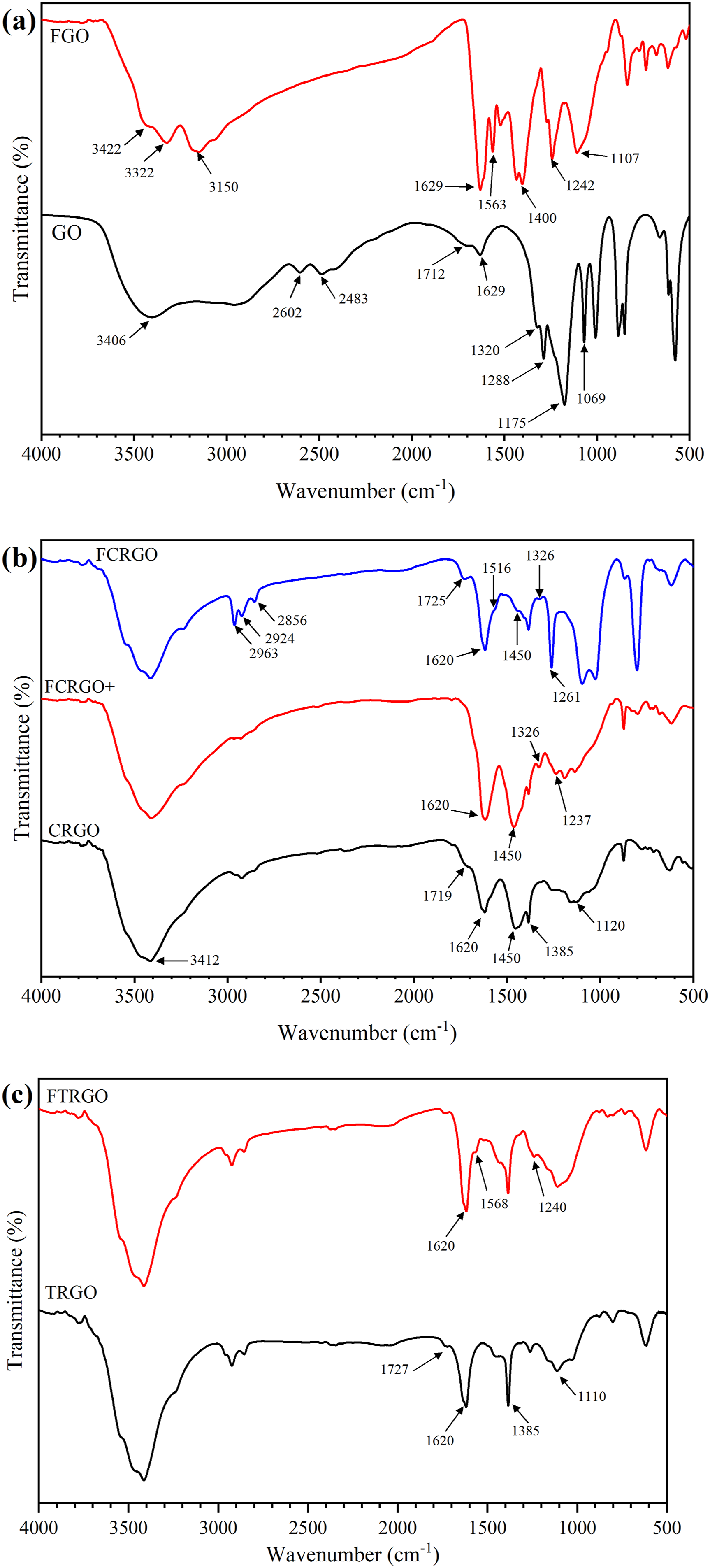
Figure 7(b) shows the FTIR spectra of CRGO, FCRGO+ and FCRGO. The characteristic peak at 1237 cm
X-ray diffraction analysis
Figure 8 shows the XRD spectra of FGO, FCRGO+, FCRGO, and FTRGO. A basal peak at XRD spectra of amino-functionalized nanoparticles. XRD parameters of amino functionalized nanoparticles.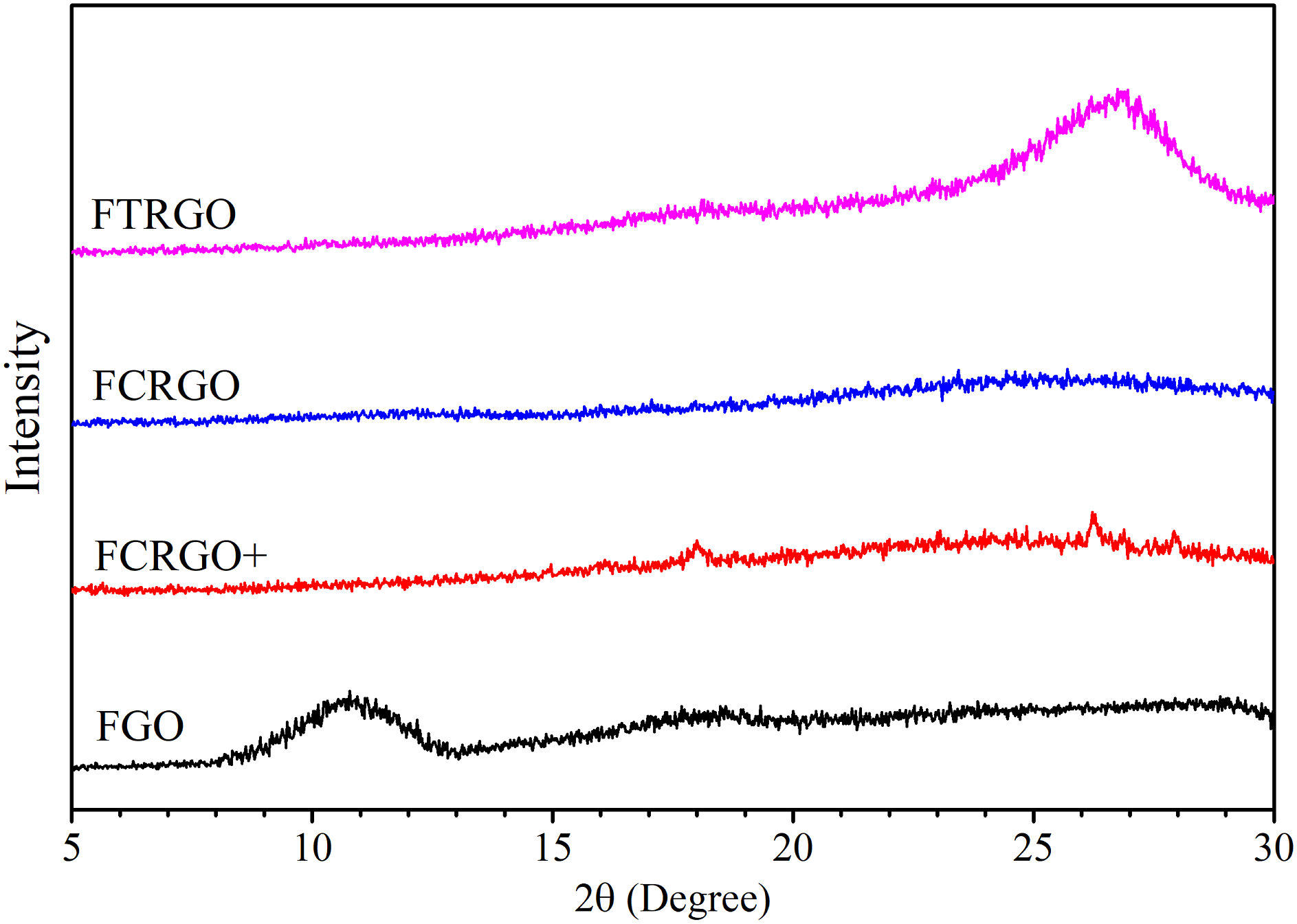
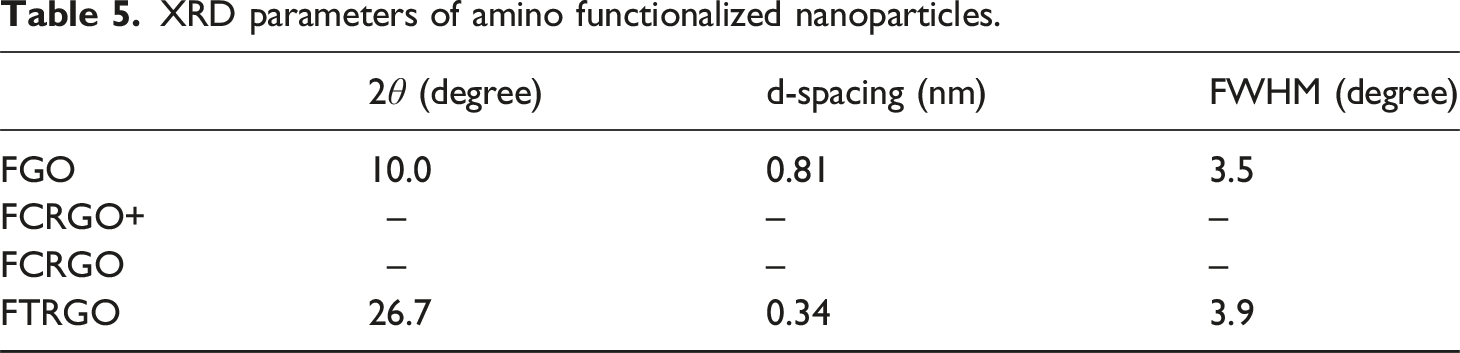
All four materials showed wide and weak basal peaks, corresponding to high exfoliation. FCRGO and FCRGO + did not show a clear peak, indicating excellent exfoliation, similar to CRGO. Surprisingly, FTRGO showed lower exfoliation than FCRGO, FCRGO+, and even TRGO. Our results showed that chemical reduction makes more exfoliation than thermal reduction. However, decreasing the degree of exfoliation by amino functionalization of TRGO can be attributed to the tendency of amine groups to each other.
Field emission scanning electron microscopy Analysis
As discussed, FE-SEM images were captured to study the nano-structure of the nanoparticles. Figure 9 shows the FE-SEM images of FGO, FCRGO+, FCRGO and FTRGO. The planar structure for all nanoparticles can be seen in the images. Also, the exfoliation of nanoparticles was observed. Table 6 demonstrates the specific area, number of layers, and thickness of stacks. As concluded before, any chemical or thermal treatment breaks the structure of graphene nanoparticles. Thus, the functionalization of nanoparticles reduces the particle size. FE-SEM images of (a,b)FGO, (c,d)FCRGO+, (e,f)FCRGO, and (g,h)FTRGO nanoparticles. Scale bars in left images are 500 nm and right ones 2 μm.
Thermogravimetric analysis
The presence of functional groups can be verified by pursuing the weight loss of the graphene nanoparticles at different temperatures. Figure 10(a) shows the TGA and DTG analysis of GO and FGO. They both showed a slight weight loss until 130°C, which refers to the vaporization of humidity. FGO showed higher humidity than GO. The other weight loss for GO from 180 to 270°C shows the removal of oxygen-containing groups. FGO lost its weight from 180 to 400°C with two peaks at 220 and 325°C. The first peak represents the removal of oxygen-containing groups, and the second one can be attributed to the removal of aminated and residual oxygenated functional groups, which indicates higher thermal stability of aminated groups than oxygenated groups. GO has lost almost 59% of its weight up to 800°C, but 76% of FGO lost. TGA and DTG analysis of nanoparticles, (a) GO and FGO, (b) CRGO, FCRGO, and FCRGO+, (c) TRGO and FTRGO.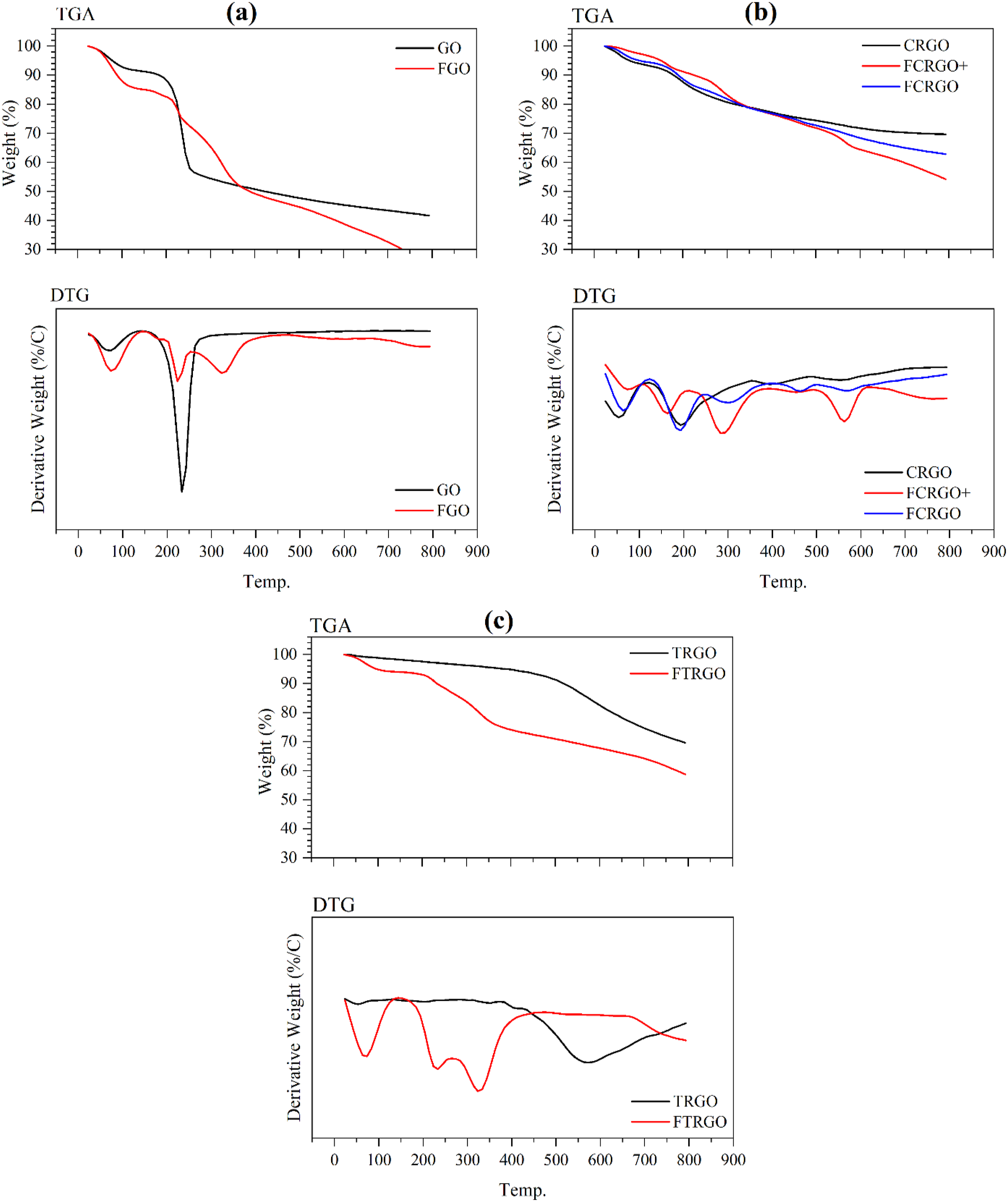
Figure 10(b) shows the thermal stability of CRGO, FCRGO+ and FCRGO. All three nanoparticles lost their humidity until 110°C. CRGO and FCRGO showed a peak at 190°C, indicating the removal of their oxygen-containing groups. FCRGO also illustrated two other peaks at 300 and 460°C, attributing to aminated functional groups. Nevertheless, FCRGO + showed different thermal behavior. After water vaporization, three weight loss peaks were seen for FCRGO+. The peak at 160°C represents the removal of oxygenated functional groups, and at 280°C represents the removal of aminated functional groups. Furthermore, the peak at 560°C can be attributed to the removal of residual functional groups and the thermal decomposition of carbons. 32
Conversely, in Figure 10(c), TRGO showed insignificant weight loss up to 400°C, indicating a low amount of oxygenated functional groups. Therefore, the peak at 560°C for TRGO shows the removal of those oxygen-containing groups that are thermal stable and were not removed in thermal reduction. At this temperature, the decomposition of carbons is possible. 32 Nevertheless, FTRGO lost weight at a wide temperature range from 170 to 400°C due to removing oxygenated and aminated functional groups. However, these functional groups were not as many as FCRGO and FCRGO+.
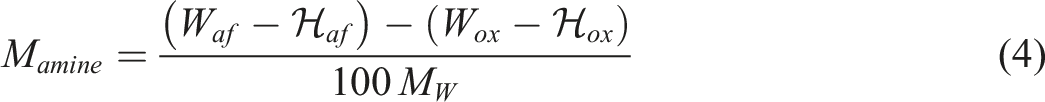
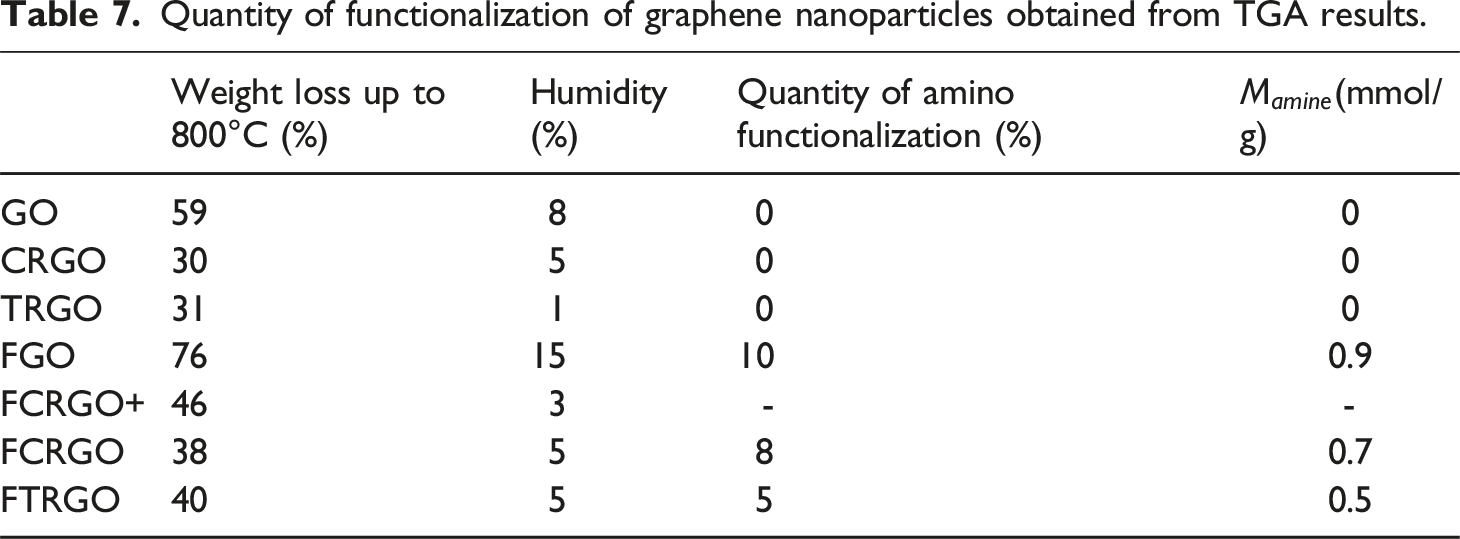
Quantity of functionalization of graphene nanoparticles obtained from TGA results.
Nanocomposites characterization
The uniform dispersion of nanoparticles and the strong interfacial interaction between the fillers and polymers are two main factors in improving different properties of polymer nanocomposites, including mechanical properties. 34 Herein, the synthesized nanoparticles were blended with maleated PE to reach the best nanocomposite with favorable properties.
Optical light microscopy test
Optical light microscopy was used to study the macro-scale dispersion of nanoparticles in the PE-g-MA matrix. As shown in Figure 11, FGO was dispersed more uniformly than GO. FCRGO + showed the best dispersion among the whole of nanoparticles. Other nanoparticles, including CRGO, FCRGO, TRGO, and FTRGO, were poorly dispersed in the PE-g-MA matrix due to the weak interfacial interaction, which demonstrates the low number of functional groups on their surface, as discussed in TGA results. Dispersion of nanoparticles in PE-g-MA observed by optical microscopy.
Field emission scanning electron microscopy analysis
The fracture surface of the nanocomposites was observed by FE-SEM to study the dispersion of nanoparticles in micro-scale. In Figure 12, FE-SEM images of pure PE-g-MA showed a smooth surface. The nanocomposites containing GO, CRGO, and FCRGO have a high number of agglomerations on their surface. The nanocomposites with FGO and FCRGO + showed lower agglomeration than their unfunctionalized ones, which means a stronger interfacial interaction between the nanoparticles and the matrix. PE-g-MA/TRGO and PE-g-MA/FTRGO did not show a sensible difference because of low functional groups and almost same degree of exfoliation. FE-SEM images of PE-g-MA and its nanocomposites. The scale bar is 10 μm, and the magnification is 12 KX.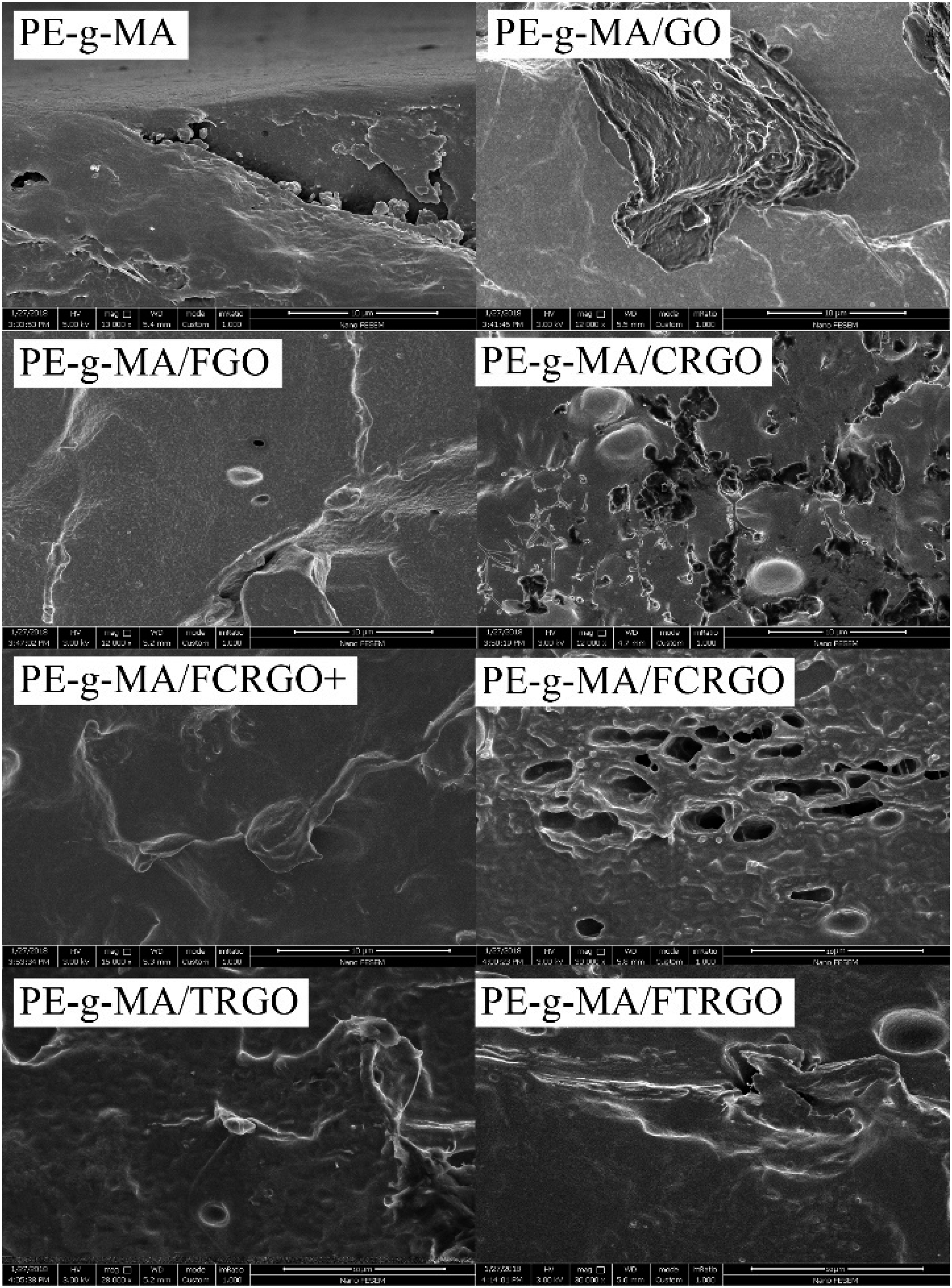
Fourier transform infrared spectroscopy analysis
Attenuated Total Reflectance – Fourier Transform Infrared (ATR-FTIR) was used to characterize the bonding between maleic anhydride in PE chains and amine groups on the surface of nanoparticles. Figure 13 compare all nanocomposites with each other. FTIR spectra of PE-g-MA and its nanocomposites with (a) GO and FGO, (b) CRGO, FCRGO+, and FCRGO, (c) TRGO and FTRGO.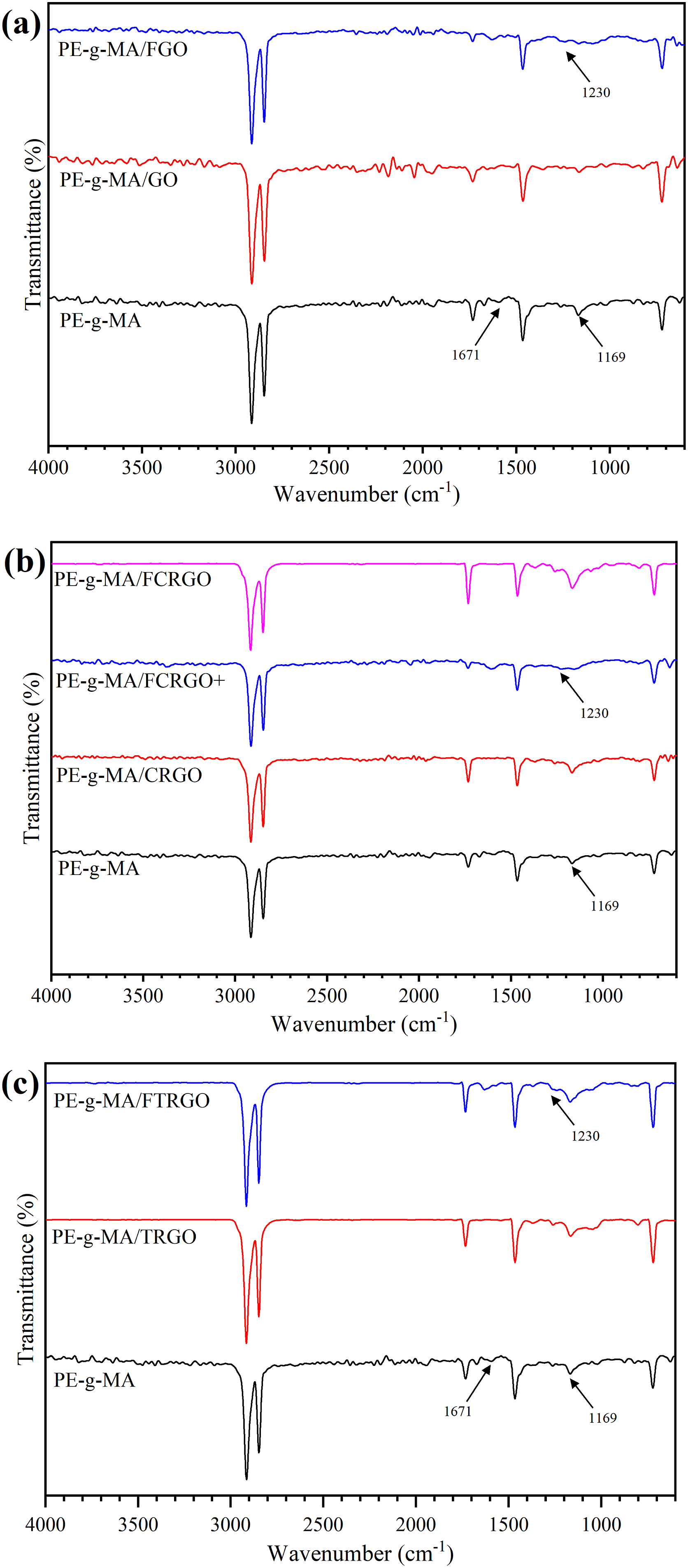
The characteristic bond at 1169 cm−1, which represents the C-O-C bond in maleic groups of PE-g-MA, has become wide after the fabrication of nanocomposites. 35 The C-N bond appears at 1230 cm−1 for grafted composites, corresponding to the grafting between PE-g-MA and functionalized graphene. 36 The peak at 1671 cm−1, corresponding to the C = C bond, shifted to higher wavenumbers for grafted nanocomposites. However, low grafting density between nanoparticles and matrix makes it difficult to characterize this reaction by the ATR-FTIR analysis, and other evidence is needed. Thus, rheological and tensile test were done which presented in the next sections.
Rheometry test
The rheometric test was used to study the behavior of molten nanocomposites, processing behavior, and the interaction between nanoparticles and matrix. Frequency sweep analysis was done to study the dynamic behavior of nanocomposites in linear viscoelastic region. The specimens were tested from 0.01 to 1000 rad s−1 in 190°C at 1% strain.
The storage modulus of the nanocomposites is represented in Figure 14(a). The nanocomposites containing GO, CRGO, and TRGO showed insignificant change in storage modulus than pure PE-g-MA. Nevertheless, by amino functionalization a remarkable increase in storage modulus was seen. This change in rheological behavior is due to the formation of a network of nanoparticles in the matrix.
37
The nanocomposite with FCRGO showed a lower storage modulus than the nanocomposite containing FCRGO+. This observation demonstrates that FCRGO + create a stronger interfacial interaction with the matrix than FCRGO. However, the FGO nanocomposite showed the highest storage modulus due to the highest amino functional groups on the FGO surface which is attributing to the grafting of maleic groups in the PE-g-MA chains and amine groups on the FGO surface. Our previous study showed the similar results.
1
(a) Storage modulus, (b) Complex viscosity, versus frequency for PE-g-MA and its nanocomposites with graphene derivatives.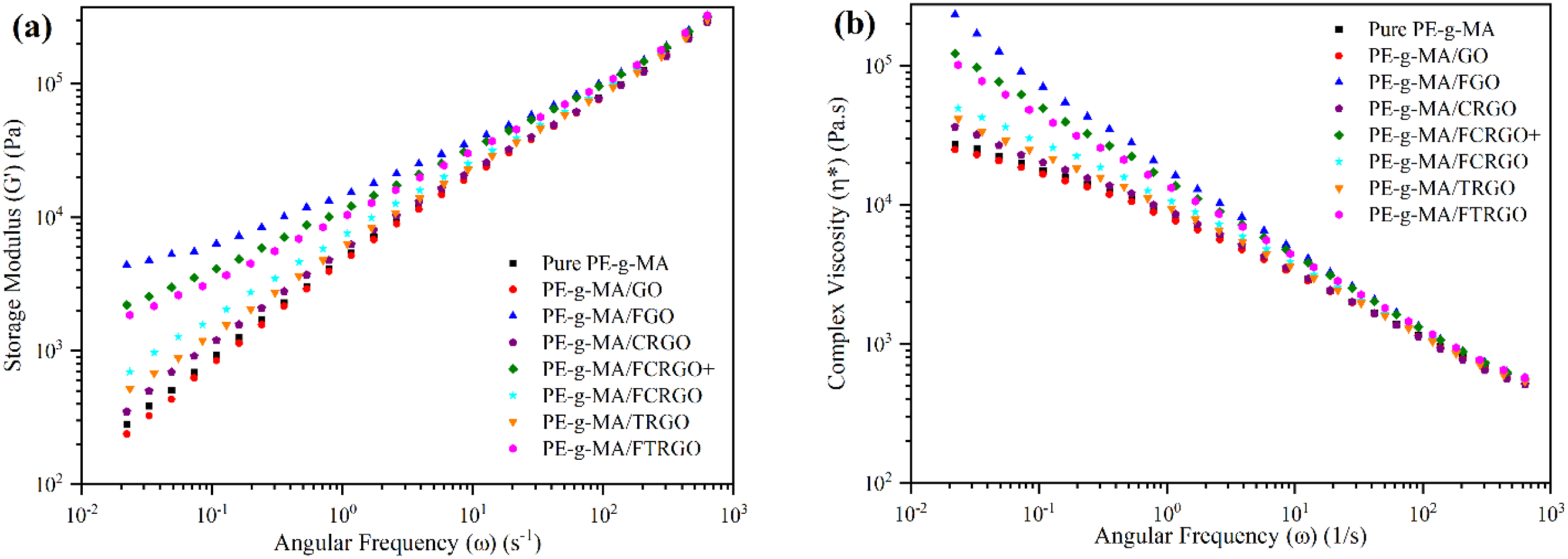
Complex viscosity is another parameter to investigate the interaction between nanoparticles and polymer matrix. Figure 14(b) shows the change in complex viscosity versus frequency. All specimens showed shear thinning behavior. The nanocomposites with functionalized nanoparticles showed a viscosity upturn in low frequencies due to strong interfacial interaction, originating from nanoparticle-matrix grafting.
Cole-Cole plot was utilized to find the structural difference between nanocomposites and pure PE-g-MA. In Cole-Cole, imaginary viscosity ( (a) Cole-Cole plot, (b) Han plot, of nanocomposites.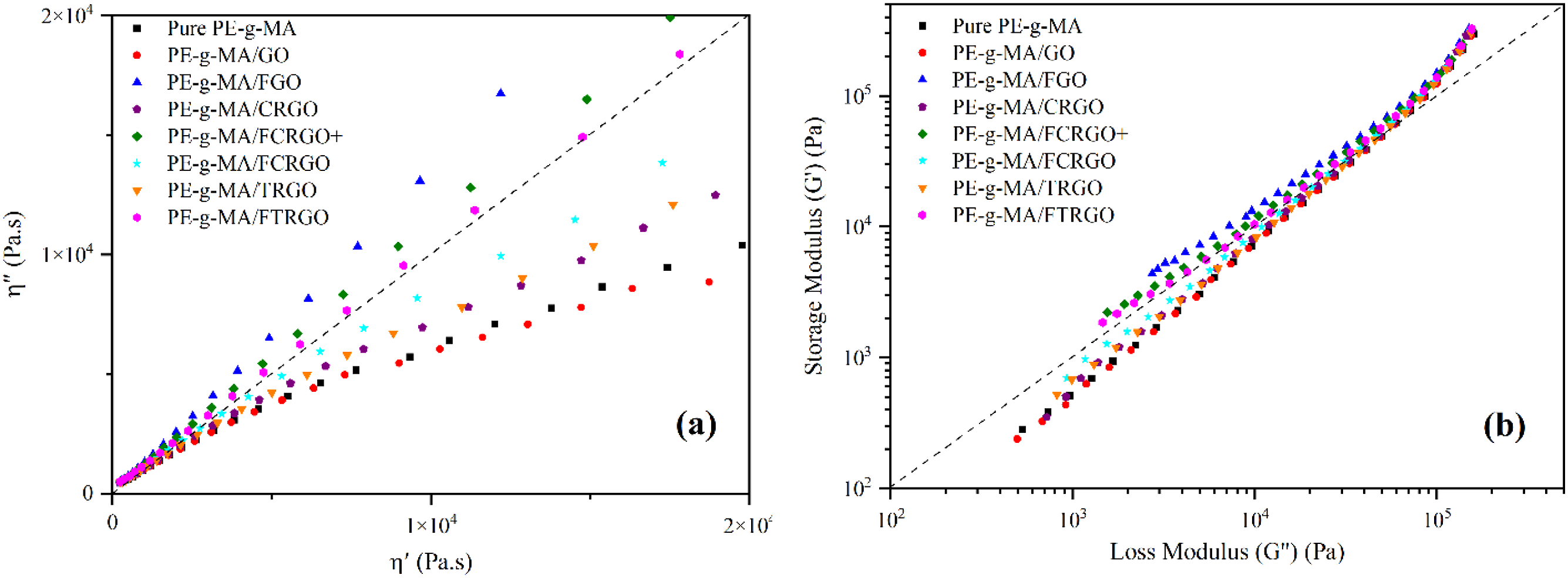
Finally, the Han plot is a criterion for studying nanocomposites’ viscose and elastic behavior. In this curve, loss modulus draws in terms of storage modulus on a logarithmic scale. Figure 15(b) confirms that grafted nanocomposites behave elastic, proving the strong interfacial interaction between nanoparticles and matrix. It is clear that FGO and FCRGO + nanocomposites show the more elasticity than others.
Tensile test
Tensile results for PE-g-MA and its nanocomposites. Tensile property of an extrusion grade of HDPE is represented for comparing.
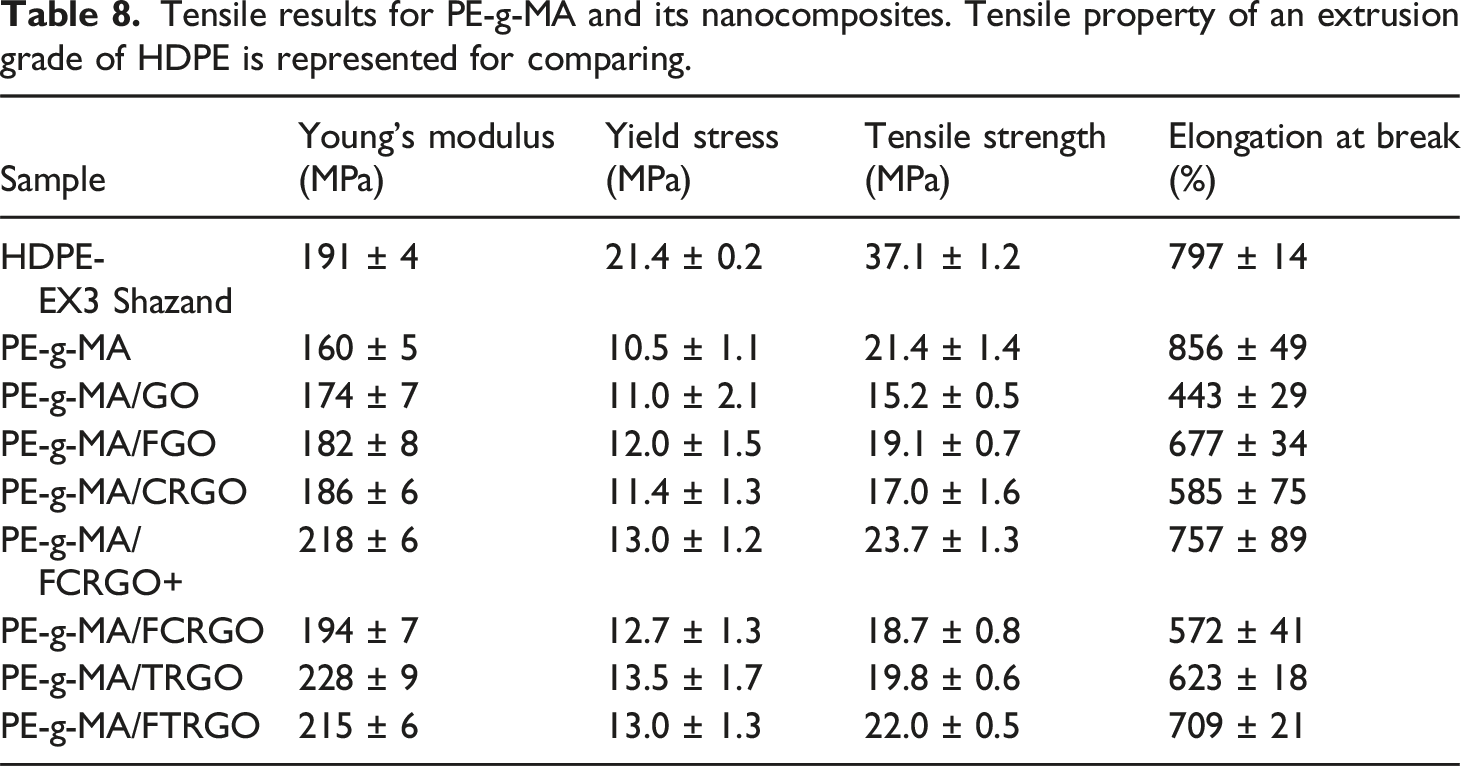
Adding unfunctionalized nanoparticles i.e., GO, CRGO, and TRGO to the polymer matrix decreased Young’s modulus, tensile strength, and elongation at break. Tensile strength declined 30%, 20%, and 7% rather than pure PE-g-MA, respectively. It is noteworthy that nanocomposites containing TRGO exhibited superior tensile properties compared to those containing CRGO and GO. This improvement can be attributed to the reduced hydrophilicity of nanoparticles resulting from the removal of oxygen-containing groups. 38 By replacing functionalized GO (FGO) instead of GO, 25% improvement in the tensile strength was seen. Using FCRGO+ and FCRGO instead of CRGO also showed 40% and 10% increase in the tensile strength. Comparing TRGO and FTRGO also indicates 11% increase in the tensile strength due to functionalization. In general, it was found that the tensile properties of nanocomposites increase by functionalization of the nanoparticles. Interestingly, PE-g-MA/FCRGO+ and PE-g-MA/FTRGO nanocomposites show the higher tensile strength than pure PE-g-MA. The results showed that the functionalization by phenylene diamine improves the interfacial interaction between nanoparticles and PE-g-MA, resulting in a better dispersion in nanoparticles, a decrease in particle agglomeration, and an improvement in the stress transfer from matrix to nanoparticles. It is worth noting that not only tensile strength but also elongation at break and yield stress were significantly increased by amino functionalization which confirms grafting between matrix and nanoparticles.
In general, relatively smaller deviations in the tensile properties of the composites containing amino functionalized GOs are observed suggesting good interactions between the particles and matrix.
Differential scanning calorimetry analysis
DSC parameters for all nanocomposites.
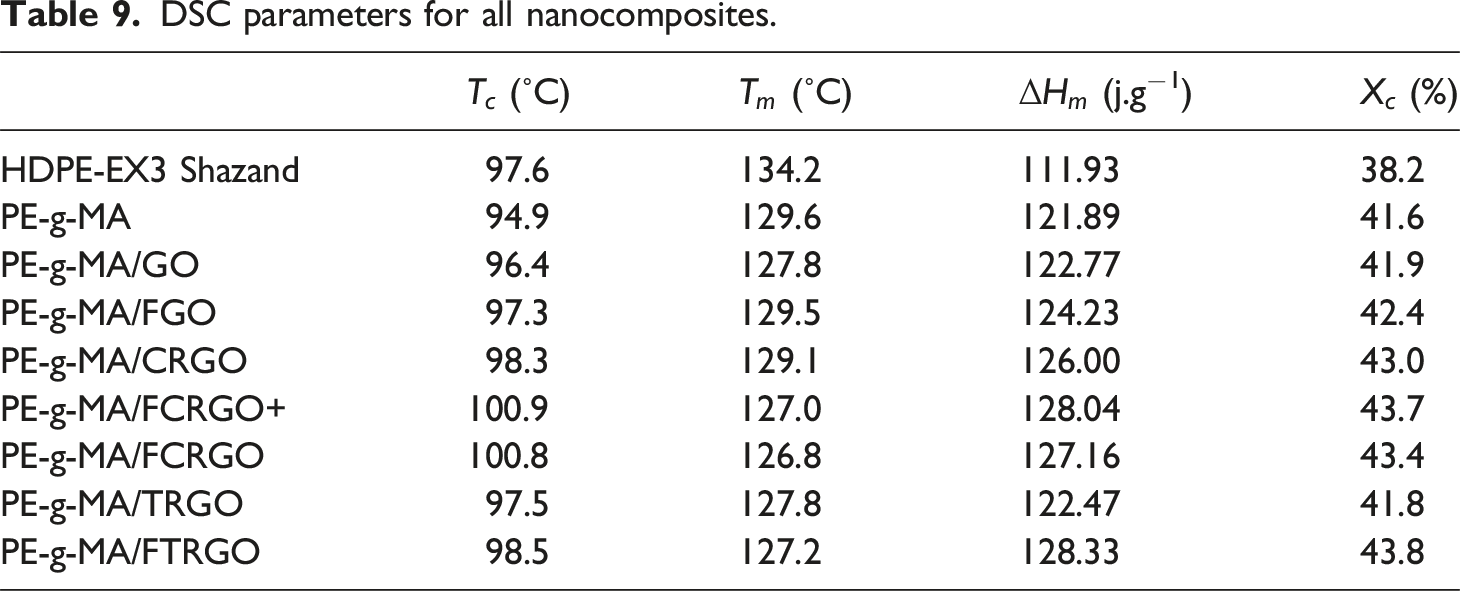
The next parameter that DSC can display is the crystallization percent (
Summary & conclusion
This article focused on several methods to functionalize graphene nanoparticles with the aim of imparting strongest interactions to polyethylene grafted maleic anhydride (PE-g-MA). Graphene oxide (GO), chemically reduced GO (CRGO), thermally reduced GO (TRGO) were synthesized. Then they were amino functionalized to obtain FGO, FCRGO, FTRGO, respectively. In a different way FGO was chemically reduced and called FCRGO+. The synthesized graphene nanoparticles were characterized by dispersion in different solvents, FTIR, XRD, FE-SEM and TGA. The study investigated different properties, such as the stability of nanoparticles in solvents, degree of exfoliation, and thermal stability. Then, these nanoparticles at 2 wt% were incorporated into PE-g-MA by a solution-casting method.
The results of FE-SEM, XRD and FTIR showed successful synthesis of GO and functionalized/reduced GOs. Although the functionalization/reduction of GO led to breakage of nanoplatelets, but higher aspect ratios and improved exfoliation were attained. Additionally, the FTIR spectra revealed the formation of amide bonds and nucleophilic substitution reactions. The increased d-spacing of functionalized graphene nanoparticles compared to unfunctionalized ones further confirmed the presence and reaction of phenylene diamine molecules on the surface of GO nanoparticles. The FTIR, rheological properties and tensile properties of the nanocomposites showed that the amino functionalized graphene nanoparticles exhibit improved dispersion and interfacial interaction in PE-g-MA matrix. Particularly, FCRGO + showed the best performance with increases of 160 to 218 MPa in tensile modulus, 10.5 MPa to 13.0 MPa in yield strength, 21.4 to 23.7 MPa in tensile strength, and 856% to 757% in elongation at break. The latest is a great achievement since the addition of nanoparticles to polyethylene can severely decrease its elongation at break or even eliminates its yield behavior. For comparison, the elongation at break of PE-g-MA, PE-g-MA/GO and PE-g-MA/FCRGO + are 856%, 443%, and 757% respectively where the latest is very close to the pristine PE-g-MA. GO, CRGO, and TRGO not only did not show improvement in mechanical and rheological properties, but also showed a decline in these properties.
This work showed that with proper functionalization of GO, polyethylene/functionalized-GO nanocomposites can be developed with improved mechanical properties. Improvements of 36% in modulus, 56% in tensile strength and 70% in elongation at break for PE-g-MA/FCRGO + as compared to those of PE-g-MA/GO are technically promising for future development of cost-effective PE nanocomposites with enhanced modulus and mechanical strength without losing ductility and necking. These nanocomposites are going to be considered for the pipe grade polyethylene with high SCG and environmental stress cracking resistance (ESCR) properties.
Supplemental Material
Supplemental Material - Multi-approach synthesis of functionalized graphene oxide as reinforcement for polyethylene: Make a strong interaction
Supplemental Material for Multi-approach synthesis of functionalized graphene oxide as reinforcement for polyethylene: Make a strong interaction by Mustafa Azizi, Mehdi Elhamnia and Ghodratollah Hashemi Motlagh in Journal of Thermoplastic Composite Materials.
Footnotes
Acknowledgements
We would like to acknowledge the support of Dana Plastic Lab in providing test apparatuses. Also, thanks to Amirkabir Nanotechnology Research Institute (ANTRI) of Amirkabir University of Technology members for their sincere advice.
Author contributions
All authors contributed to the study concept and design. Material preparation and data collection were performed by Mustafa Azizi. Analysis and Discussion were performed by Mustafa Azizi, Mehdi Elhamnia, and Ghodratollah Hashemi Motlagh. The first draft of the manuscript was written by Mustafa Azizi and Mehdi Elhamnia, and all authors commented on the previous version of the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.