Abstract
Poly(methyl methacrylate)/silica nanocomposites membranes were prepared by simple solution mixing method using three different solvents (acetone, acetonitrile and chloroform). The structure and thermal properties of these nanocomposites were investigated by fourier transform infrared spectroscopy (FTIR), differential scanning calorimetry (DSC), thermogravimetric analysis (TGA) and environmental scanning electron microscope (ESEM). The infrared spectra confirm that the strongest H-bond interaction between the PMMA and silica was found when using chloroform solvent. In the case of thermal properties, the DSC and TGA analyses show significant improvement in thermal stability and glass transition temperature (Tg). Furthermore, the ESEM micrographs prove the structure stability of these synthesized nanocomposites. The results demonstrate that the solvent that lead to form the strongest H-bond interaction between the PMMA carbonyl groups and silica silanol groups (as chloroform solvent) will be more efficient in the synthesis of PMMA/SiO2 nanocomposites.
Keywords
Introduction
Polymer nanocomposites represent an attractive family of hybrid materials, in which inorganic nanoscale particles are added to the organic matrices. In the literature, various preparation techniques have been used to synthesize these nanocomposite materials. The most commonly methods used are in situ polymerization, 1 emulsion polymerization, 2 microemulsion polymerization, 3 solution mixing, 4 sol–gel method 5 and melt blending. 6 Many researchers have reported that the incorporation of inorganic fillers into polymer matrix can significantly improve a number of desirable properties, such as mechanical strength, 7 thermal stability, 8 barrier properties, 9 and wear resistance. 10 Among the pool of nanocomposite materials available, poly(methyl methacrylate)/silica nanocomposites have recently received considerable attention because of its numerous applications and advantages in many domains like scratch resistance, 11 bioactivity, 12 antimicrobial, 13 dielectric, 14 and optical applications. 15
The nature and the strength of the interactions between filler and polymer play a very important role in the final properties of nanocomposite materials. This interaction will condition the filler dispersion and distribution quality into the polymer matrix. In fact, the weak interactions lead to increase the amount of filler agglomerates, while the strong interactions form either mixed aggregates or individually disperse the primary particles. 16 In case of PMMA/SiO2, previous studies “based on FT-IR analysis” have indicated the presence of H-bond between the carbonyl groups of PMMA and the silanol groups of the SiO2. Their results revealed that the shift of O=C (PMMA) peak toward lower wave numbers (lower energies) is related to the strength of the H-bond interaction with OH (SiO2) groups.17-19
Moreover, the thermal stability and the decomposition mechanism of PMMA/SiO2 nanocomposites has been also studied. In fact, the improvement of the thermal stability is strongly depends on the dispersion state of SiO2 nanoparticles in PMMA matrix. The temperature of degradation of PMMA/SiO2 nanocomposites was increased compared to the pure PMMA, which depends on the amount of silica added20,21 (The majority of experimental works we reviewed indicated that the good reinforcement of PMMA by silica is between 1 wt% and 5 wt%). This increase in the temperature of degradation has been attributed to the trapping effect of polymeric radicals by the silica particles.22,23 Furthermore, the improvement of the thermal stability can be also attributed to the interaction strength of a hydrogen-bond interaction between the carbonyl groups of PMMA and the OH groups located on the surface of the silica. 24 It is worth to note that the interaction between nanoparticles and polymer chains affect highly the segmental mobility and dynamics of polymer nanocomposite. Consequently, the addition of SiO2 nanoparticles in PMMA polymer leads to shift the glass transition temperature (Tg) to the higher temperatures. This higher Tg is due to the immobilization and restriction of polymer segments by silica nanoparticles.25,26
The solvent-based methods like solution mixing are commonly employed because of their ease to use and to get the better nanoparticles dispersion in polymer nanocomposites. However, the most difficult problem is the solvent retention in polymer matrices which can significantly lower the glass transition temperature of nanocomposite materials. The interaction strength solvent/polymer and the evaporation procedure of solvent are the two main factors that influence the amount of the residual solvent. Indeed, the slow evaporating solvent with high boiling point lead to have a large amount of retained solvent, thereby increasing the polymer chain mobility and lowering the glass transition temperature. The same thing can be expected to happen if a strong interaction exists between solvent and polymer.27-29
The object of this work consist to propose, first, a simple approach for dispersing nanosilica in PMMA matrix through solution mixing method. This approach is based on studying the nature and strength of the molecular interaction created between the silica nanofillers and the PMMA matrix. Second, the validity of this approach will be tested and confirmed by analyzing the thermal properties of our synthesized nanocomposites
Experimental procedure
Materials
Commercially available PMMA was used as a matrix for preparing samples (grade: Acryrex® CM205, Mw ≈ 90,400 g/mol, manufactured by the Chi Mei Corporation). The fillers were fumed silica (Grade: Cab-O Sil® M-5P; manufactured by the Cabot Corporation). The fumed silica nanoparticles are nonporous, have a spherical shape of 7–15 nm in diameter with a specific surface area of 200 ± 25 m2/g. Solvent as acetone, acetonitrile and chloroform were used to prepare PMMA/SiO2 films.
Preparation of nanocomposites
As mentioned above, the nanocomposites films of PMMA/SiO2 were synthesized by solution mixing method (Figure 1). The PMMA (0.2 g) was dissolved in the solvent (20 ml). Silica nanoparticles

Experimental setup of samples preparation.
Characterization
FTIR
Fourier Transform Infrared (FTIR) transmission spectra were recorded on a JASCO V-530 spectrometer. The results were collected in wave numbers between 4000 and 450, 1 cm−1 resolution, 128 scans, and 40 kHz scanning velocity using an MCT detector. Silica samples were made in the transmission mode from dried casts deposited on KBr pellet. PMMA/SiO2 nanocomposites were characterized directly in the form of thin films.
TGA
Thermogravimetric analysis was performed on TGA (LabSysevo de Setaram) to study the thermal stability as well as the rate of degradation of PMMA and synthesized nanocomposites. The various samples were subjected to a ramp of ambient temperature at 500°C carried out with 10°C min−1 under dry nitrogen. The mass of samples introduced is of the order of about (10 mg).
DSC
Conventional differential scanning calorimetric measurements were recorded using a DSC SETARAM 92 with an intracooler for low temperature. DSC was used for the investigation of glass transition temperature variation of PMMA and synthesized nanocomposites. The appropriate amount of samples (10 mg) was sealed in aluminum sample pans. DSC analyses of these hybrid materials were then conducted under dry nitrogen at a heating rate of 5°C min−1 from 25 to 500°C.
ESEM
The surfaces of the samples were observed with the Thermo-Scientific™ Quattro S environmental scanning electron microscope. The film samples were mounted on a stub with double sided adhesive tape. The images were obtained using the experimental conditions of 5 kV and 50 Pa in order to assess the consequences of the gas pressure in the ESEM. 30
Results and discussion
PMMA/silica interaction
Infrared spectra of synthesized hybrid membranes and pure PMMA were shown in Figure 2(a) to (d). All samples showed distinct infrared absorptions regions. The absorbance and shift frequencies of the C=O bond stretching vibrations in the investigated fragments are presented in Table 1. For apolar solvents such as chloroform, the absence of polar (side) groups implies a weak interaction with polar polymer repeat unit (PMMA) compared to the polar solvents (as acetone and acetonitrile). These mechanisms favor the interaction between the PMMA molecules and the SiO2 particles for samples prepared by chloroform solvent. The weak interactions of the chloroform with PMMA and with the silica allowed the presence of strong H-bond interactions. These results were confirmed by the significant shift of the peak C=O in the case of PMMA/chloroform/silica system (22 cm−1).

FTIR spectra: Nc-Ac (a); Nc-Acn (b); Nc-Chl (c); pure PMMA (d).
The shift frequencies of the carbonyl group (C=O) for synthesized nanocomposites in acetone, acetonitrile and chloroform.
Thermal stability of PMMA/silica nanocomposites
The results of thermogravimetric analysis (TGA) of pure PMMA and synthesized nanocomposites are shown in Figure 3. The thermal decomposition of PMMA exhibit only a single degradation stage occurred from 250°C to 500°C. In contrast, the thermal decomposition of the synthesized nanocomposites gave similar thermograms with tow degradation stages. The first stage was in the range of 50–200°C, which resulted from the elimination of the residual solvent that have not been released during the vacuum drying process. The second stage was in the range of 250–500°C, which can be ascribed to a degradation of the nanocomposite membranes. The values of complete degradation temperatures (TC) for the pure PMMA and its nanocomposites are listed in Table 2. All nanocomposites showed an increase in TC compared to that of pure PMMA. Accordingly, the thermal degradation properties of the pure PMMA were improved by the addition of silica fillers due to the high thermal stability of SiO2 nanoparticles. Nevertheless, the obvious difference in TC between different nanocomposites provides clear evidence that the solvent type has an effect on the interaction quality between the silica particles and the polymer matrix. Otherwise, the type of solvent determines the energy strength of the hydrogen bonding between the carbonyl groups of PMMA and the hydroxyl groups of the silica. As a result, the highest final degradation temperature is corresponding to the films synthesized in chloroform solvent. The chloroform has very weak interactions with PMMA and even with silica, which favors the presence of strong hydrogen bonds between them (Results were confirmed by FTIR spectroscopic analysis). Furthermore, for PMMA/chloroform/silica system, the high thermal stability confirms the good dispersion of the silica nanoparticles in the PMMA matrix compared to PMMA/acetonitrile/silica and PMMA/acetone/silica systems.
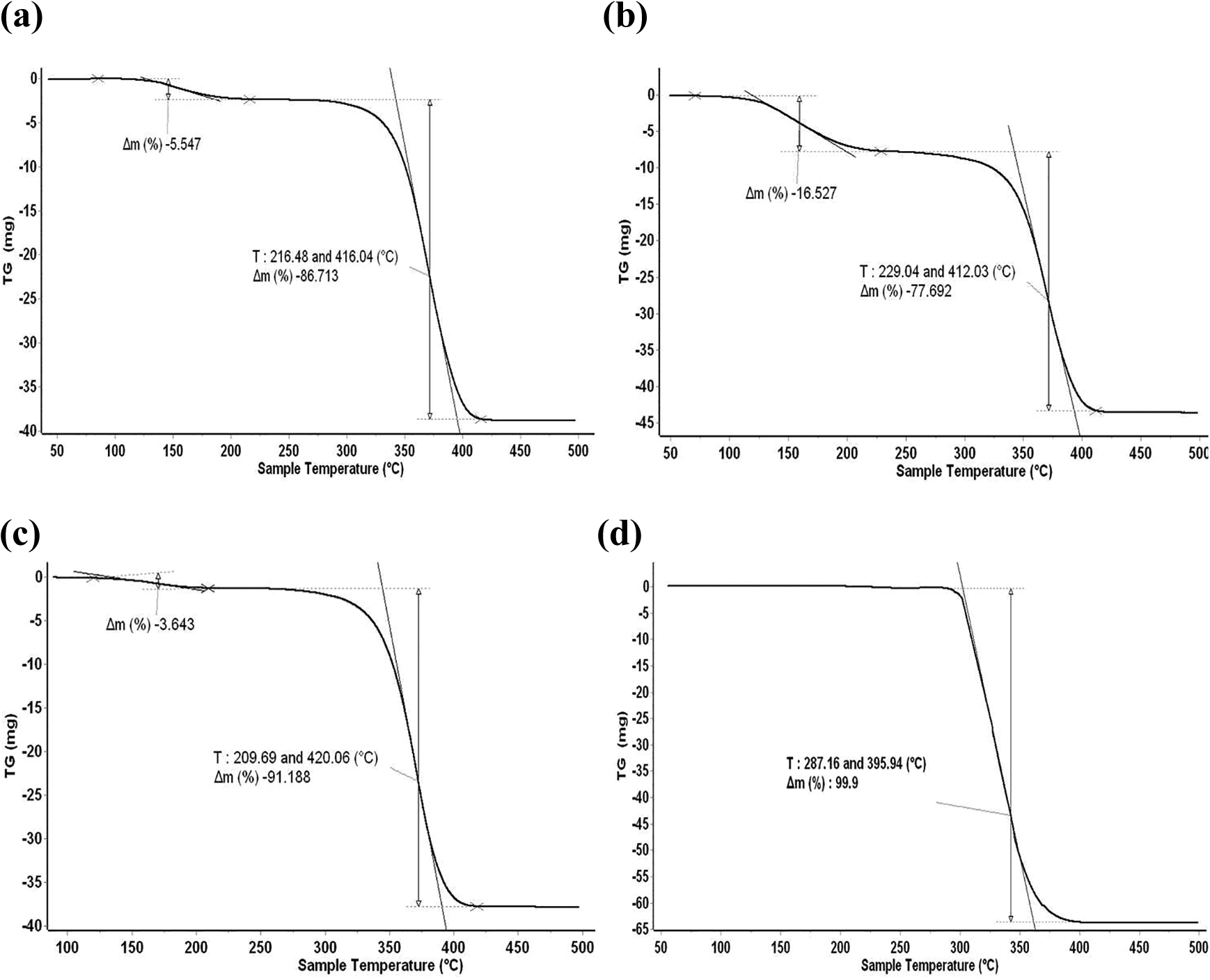
TGA curves: Nc-Ac (a); Nc-Acn (b); Nc-Chl (c); pure PMMA (d).
Final degradation temperatures: Pure PMMA and synthesized nanocomposites in acetone, acetonitrile and chloroform.

Glass transition temperature of PMMA/silica nanocomposites
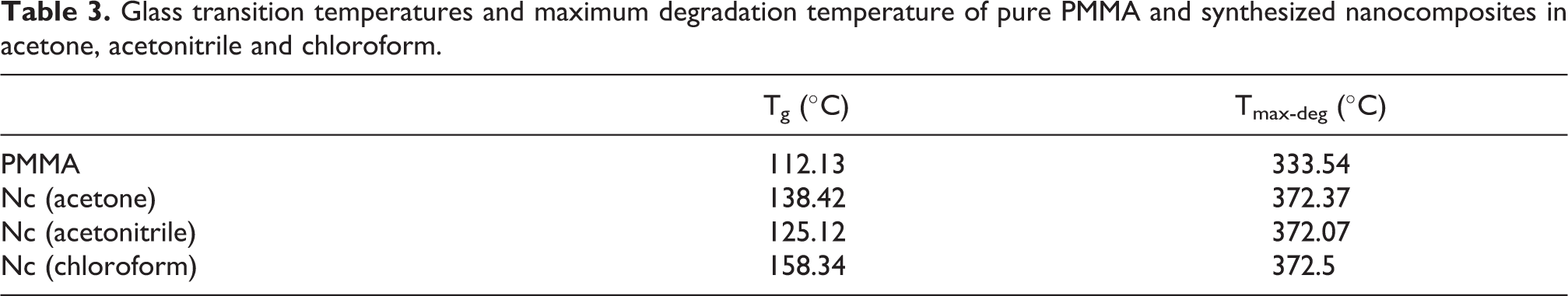
The results of differential scanning calorimetry (DSC) are shown in Figure 4(a) to (d). The values of the glass transition temperature (Tg) and the maximum degradation (Tmax-deg) for the pure PMMA and the PMMA/SiO2 nanocomposites are listed in Table 3. The Tg of our pure PMMA was remarkably increased by the incorporation of the silica filler. In fact, the strong interfacial bonding created between the silica nanoparticles and PMMA molecules leads to increase the Tg by impeding the polymer chain flexibility. 29 This increase observed is depending on the nature of the solvent used to prepare the nanocomposites. The presence of a slight endothermic peak occurring at 112°C is related to the Tg of the pure PMMA; while, the endothermic peaks at 125°C, 138°C 158°C are related to the glass transition temperatures of PMMA/SiO2 nanocomposites prepared with acetonitrile, acetone and chloroform, respectively. The solvent-based methods (mixing solution) are commonly used to get better dispersion of nanoparticles in polymer hybrid materials. In our experiments, the peaks of glass transition temperature in the DSC curve were markedly different, despite that the nanocomposite films shared the same drying process (at 45°C temperature for 4 h). This difference suggests firstly the effect of the residual solvent retained in the nanocomposite films. Some solvent have an inefficient evaporation process due to the strong interaction with polymer chains. Consequently, the residual solvent in polymer matrix lead to increase the polymer chain mobility, then the Tg will be lower. The amount of residual solvent in the films was measured by TGA analysis, as shown in Table 4. The results showed that all the samples still contain solvent after the evaporation process. acetonitrile has a high boiling point (81°C) and relatively strong interaction with the PMMA molecules, which slows the evaporation process at the drying temperature. This can explain the large amount of the acetonitrile solvent retained (16 wt%) in the film, causing the lower glass transmission temperature observed (125°C). In the same manner, acetone has the lowest boiling point (56°C) and strong interaction with the PMMA molecules. In this case, the evaporation process allows obtaining a slight residual quantity of acetone solvent (5 wt%) with a lower glass transition temperature (138°C). The lowest residual quantity was found in the PMMA/chloroform/silica system (3 wt%) accompanied by the highest glass transmission temperature (158°C). This is due because of the low boiling point (61°C) of chloroform solvent and its weakest interaction with PMMA molecules.
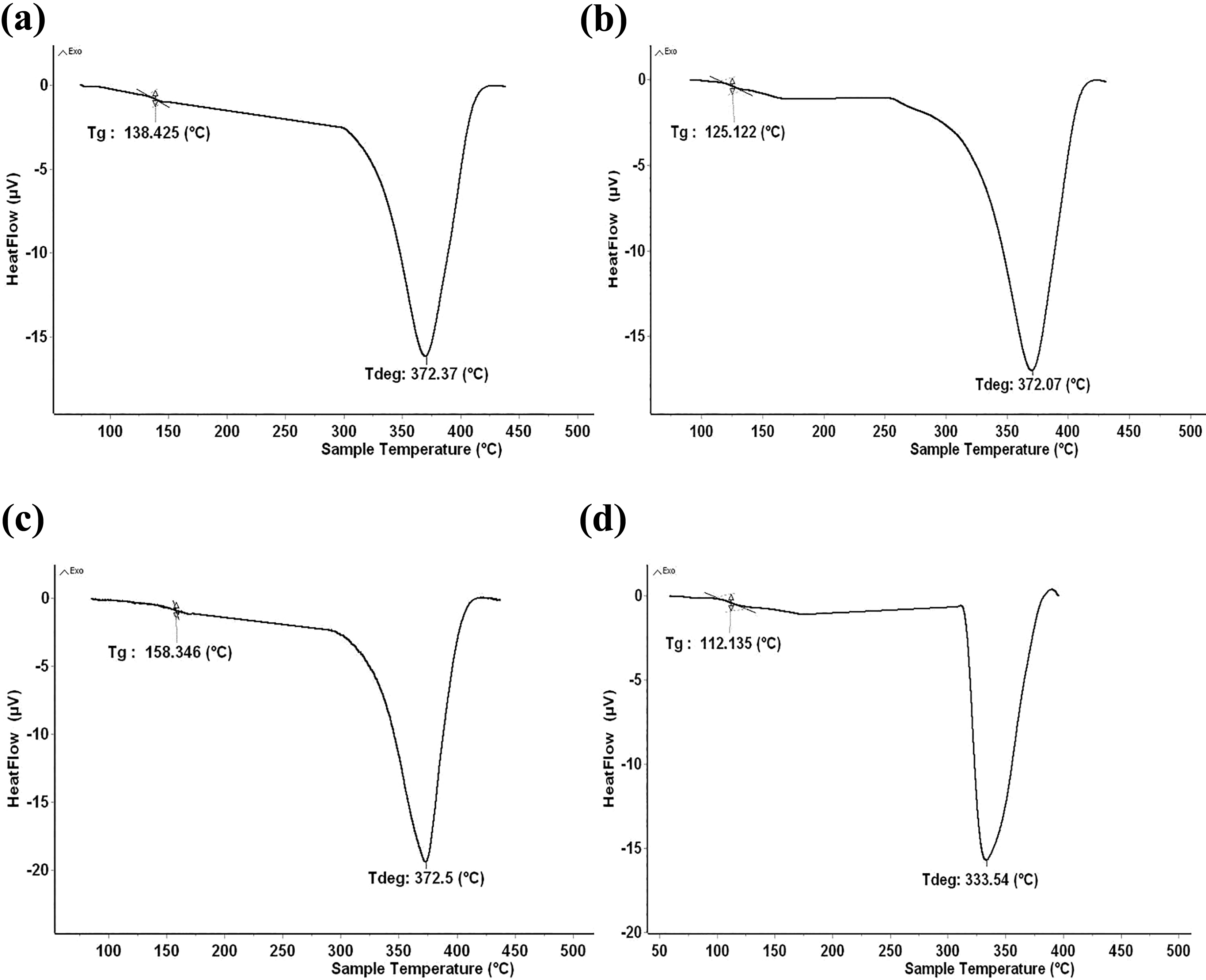
DSC curves: PMMA (a); Nc-Ac (b); Nc-Acn (c) and Nc-Chl (d).
Glass transition temperatures and maximum degradation temperature of pure PMMA and synthesized nanocomposites in acetone, acetonitrile and chloroform.
Solvent boiling points and the amount of solvent retained in nanocomposites.

Second, the chloroform is a nonpolar solvent, which interacts weakly with the polar surface of SiO2 and PMMA molecules. This weak interaction facilitates the existence of a highly interconnected hydrogen bond between PMMA and silica, although the PMMA/chloroform/silica system allows the good distribution and dispersion of silica nanoparticles in PMMA polymer compared to PMMA/acetone/silica and PMMA/acetonitrile/silica systems.
Furthermore, the hybrid membranes present higher thermal stability than pure PMMA. The addition of the SiO2 nanoparticles increases also the maximum degradation temperature (Tmax-deg) of the nanocomposites compared with the pure PMMA polymer. In contrast, the maximum degradation temperatures of different PMMA/SiO2 nanocomposites are almost identical. The solvent nature has no effect on (Tmax-deg) because the PMMA molecules are the great majority.
Surface morphology of PMMA/silica nanocomposites
The surface morphology of our synthesized nanocomposites was observed using environmental scanning electron microscope. ESEM images (Figure 5) show that all samples have a co-continuous structure with homogenous and smooth surface. Indeed, the surface morphology of nanocomposite samples is closely related to the nature and the strength of the interactions between silica and PMMA. These interactions are resulted from a competition between the crosslinking kinetics (SiO2/SiO2 and SiO2/PMMA) and the phase separation kinetics (immiscibility of SiO2 clusters in the PMMA polymer). Clearly, the samples prepared in chloroform solvent exhibit more homogeneous and smooth surface compared with samples prepared in acetone and acetonitrile. The explanation is due because of strong the hydrogen bonding built between the SiO2 and PMMA. These strong physical links can inhibiting phase separation and lead to good dispersion and distribution of the silica nanoparticles in PMMA polymer.

Surface ESEM images of Nc-Ac (a); Nc-Acn (b); Nc-Chl (c).
Conclusion
PMMA/SiO2 nanocomposites have been successively synthesized by means of solution mixing method. The effect of solvents (acetone, acetonitrile and chloroform) on the molecular interaction strength between the filler (SiO2) and the matrix (PMMA) were characterized by structural and thermal studies. From the FTIR analysis, the strong molecular interaction at the interface SiO2/PMMA was found in nanocomposite prepared with chloroform solvent. Based on the DSC analysis, notable increases in the glass transition temperature in the order: Pure PMMA (112.13°C) < Nc/Acetonitrile (125.12°C) < Nc/acetone (138.42°C) < Nc/chloroform (158.34°C). Furthermore, from TGA analysis, the synthesized nanocomposites exhibit enhanced thermal stability of 16 (Nc/acetone) to 24°C (Nc/ chloroform) with respect to pure PMMA. The improved structure stability of these nanocomposites was shown by the ESEM micrographs (especially the Nc/chloroform sample). In general, the results show that the solvent that facilitate the presence of strongest H-bond interaction between the PMMA carbonyl groups and silica silanol groups will be more efficient in the synthesis of PMMA/SiO2 nanocomposites
Footnotes
Acknowledgement
The authors gratefully acknowledge the constructive suggestions and the critical revision given by Professor Djamal Benbertal on the preliminary version of the present article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
