Abstract
Hydroxyapatite (HAp) is the major inorganic component of natural bone which exhibits better biocompatibility with various kinds of cells and tissues, making it an ideal candidate for dental and orthopedic applications. The naturally extracted HAp (Ca10(PO4)6(OH)2) from fish scale and seashell is exactly matched with the chemical composition of bone minerals. Nowadays, soft chemistry is used for the synthesis of bioceramics such as HAp. This is a chemical route that yields more homogeneous solid-state materials. In this study, the extracted powder from fish scale and seashell was heated in the furnace and maintained at 700°C for 3 hours and the powder was naturally cooled. The derived CaO was used for preparing HAp by the microwave irradiation techniques. The HAp was filled with High-Density Polyethylene (HDPE) in the ratio of 10:3 (Matrix 100 g: Filler 30 g) and composite was fabricated by the injection molding. The functional groups present in the HAp-HDPE specimen was identified using Fourier Transform Infrared (FTIR) spectroscopy analysis. The thermal stability of 30 wt. % HAp-HDPE composite was analyzed using Differential Scanning Calorimetry
Introduction
The hydroxyapatite (HAp) was extracted from the seashell and utilized for biomedical application. It’s a low cost simple and eco-friendly microwave irradiation technique to prepare HAp from seashell (Anadara granosa). 1 Also, wet chemical reaction route was used to synthesize the nano-hydroxyapatite from the powdered sea shells. 2 Over the past several years biologically derived natural materials such as fish bone, 3 shell,4,5 bovine bone, 6 egg shells, 7 and coral8,9 have been converted into useful biomaterials like hydroxyapatite nanoparticles. The hydroxyapatite nanocrystals have exhibit important properties such as biocompatible, bioactive, and thermodynamically stable in the body fluid in biomedical applications.10,11
The HDPE based composites can be used as an implant, because of their high mechanical properties (E-modulus, strength, and hardness), and good biological properties (no toxicity and biocompatibility). 12 HAp could be synthesized via various techniques such as follows: i) dry methods ii) wet methods iii) hydrothermal, iv) high temperature processes 13 Ozawa et al. (2002) developed the hydroxyapatite from fish bone (Japanese sea bream) through heat treatment ≤1300°C. This derived ceramics would be useful as inexpensive biocompatible material. 14
Balaji Ayyanar and Marimuthu synthesized Hydroxyapatite (HAp) from fish scale and filled with HDPE. The surface morphology of composite specimens was investigated through Field Emission Scanning Electron Microscope (FESEM). The cell morphology, cell viability, and levels of cytotoxic of the HAp-HDPE specimen were studied as per ISO 10993:12, and ISO 10993:5 standards. It was found that 30 wt. % HAp-HDPE showed none to slight cytotoxicity reactivity to MG-63 cells after 24 hours contact. The cytotoxicity level of HAp-HDPE material was compared with standard reactivity level and it was confirmed to have low cytotoxic level. 15
The objective of this study was to develop a new composite using naturally extracted hydroxyapatite from fish scale Catla (Labeo catla) and sea shell (Laevistrombus canarium) through dry heat treatment. It was utilized as filler in HDPE, because polyethylene thermoplastic has excellent mechanical and biocompatible properties. 16 Since both the extracted HAp are having minerals which are similar to the chemical composition of bones, a new combination HAp-HDPE composite has been developed by injection molding and cytotoxicity studies have been carried out. The previous studies was carried out using HAp extracted from fish scale, but in this present study HAp was extracted from both fish scale and seashell equally filled with HDPE. Also carried out the morphology studies of composite using Filed Emission Scanning Electron Microscope (FESEM).
In order to carry out the in vitro MTT assay, five different concentrations (10, 20, 30, 40 and 50 μl) of liquid extracts of the 30 wt. % HAp-HDPE composite were allowed to interact with MG 63 cell line for 24 hours. In this present study, the morphology and cell viability of the composite specimens were studied and results were compared with (control) reference cell culture medium.
Experimentation
Materials
The fish scale, and seashell have a density of 900 kg/m3, and 1200 kg/m3, respectively. The commercially available HDPE procured from Varsha Poly Products (India) was used as a matrix. It has a density of 910 kg/m3, the melting point of 125°C, and tensile strength of 16 MPa, and Melt Flow Index (MFI) of 6 g/10 min at 190°C, and 2.16 kg. The cell line MG-63 (Organism: human, origin tissue: bone and Morphology: Osteosarcoma) was procured from National Centre for Cell Science (Pune, India) and cell culture was developed a biotechnology laboratory to carry out in vitro studies.
Methodology
The methodology carried out to develop the HAp-HDPE specimen is shown in Figure 1. In order to produce the HAp-HDPE specimen, the required quantity of mixtures in the ratio of 10:3 wt. % (Matrix 100 g: Filler 30 g) of HDPE and HAp was taken to heating chamber and preheated at 60°C. The temperature 90 to 100°C was preferred in injection molding machine to control the melting of matrix and thermal degradation of fillers in the composite. The operating pressure was around 2 to 3 bar during the injection molding.

Methodology.
Microwave technique
In microwave technique, 0.25 M calcium nitrate tetra hydrate was added drop by drop to 0.15 M potassium dihydrogen phosphate until colloidal suspension occured. During the process, Ca/P ratio was maintained at 1.67. The mixture was stirred for about 4 hours after which it was placed inside the microwave oven at 720 W. The obtained white precipitate was washed and dried in a muffle furnace at 700°C for 3 hours.
Evaluation of cytotoxicity
MG-63cell line is an established and well-characterized cell line that has demonstrated reproducible results. The cell viability was assessed using an MTT (3-(4, 5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay after HAp-HDPE specimen exposure, as only the viable cells have functional mitochondrial dehydrogenase enzymes which can reduce MTT to formazan. After 30 wt. % of HAp-HDPE liquid extracts of the specimen were allowed to interact with MG-63 for 24 hours, they were washed three times with Phosphate Buffer Saline (PBS) (pH 7.3) and MTT reagent (0.5 mg/ml) was added to each cell and incubated at 37°C for 4 hours.
The Ethylene Oxide (EtO) sterilized 30 wt. % of HAp-HDPE specimen was investigated for indirect in vitro cytotoxicity studies using MG 63 cell line. The liquid extracts from the sterilized sample was then allowed to interact with MG 63 cell line using five different (10, 20, 30, 40, and 50 μl) concentration levels by indirect cytotoxicity method. The percentage of cytotoxicity and cell viability of the specimen was evaluated.
The 30 wt. % of HAp-HDPE specimen after EtO sterilization was incubated in 1X PBS at 37°C for 24 hours. The culture medium from the MG-63 cells was replaced with a fresh medium. The liquid extracts of the 30 wt. % of HAp-HDPE specimen at five different volumes in triplicate was added to the cells. After incubation at 37 ± 1°C for 18 hours, MTT assay (0.5 mg/ml) was added in all the wells and incubated for 4 hours. After incubation, the small quantity of dimethyl sulfoxide organic sulfur was added in the wells and cytotoxicity results were revealed at 570 nm using a photometer. The cell viability and cytotoxicity were calculated by using the following Equations.
The percentage grade of cytotoxicity was calculated from the indirect cytotoxicity method and the results were compared with standard reference data as shown in Table 1.
Comparisons of reactivity level. 13
Cytotoxicity = [(Control − Treated)/ Control] × 100 - (1)
Cell viability = (Treated/Control) × 100 - (2)
Results and discussion
FTIR spectrum of the 30 wt. % HAp-HDPE composite
Figure 2 shows the FTIR spectrum 30 wt. % HAp-HDPE composite. The characteristic band around 3838, 3745, 3676, 3647, and 3564 cm−1 is observed and it shows the presence of OH group. The bands around 2913 and 2847 cm−1, show the presence of C–C which is the most prominent in HAp-HDPE composite. Apart from that, the band at 2369 and 2315 cm−1 confirms the presence of stretching vibration of C=O. Moreover, the band at 1648 cm−1 indicates the presence of vibrations of NH groups and 1540 cm−1 indicates the presence of the vibrations of 1426 and 1020 cm−1 indicate the presence of the vibrations of groups CH and CO groups in HAp-HDPE composite. The vibration band of 719 cm−1 indicates (C–H rock) alkanes, and band around 671 cm−1 indicates (C–Br stretch), alkyl halide groups.
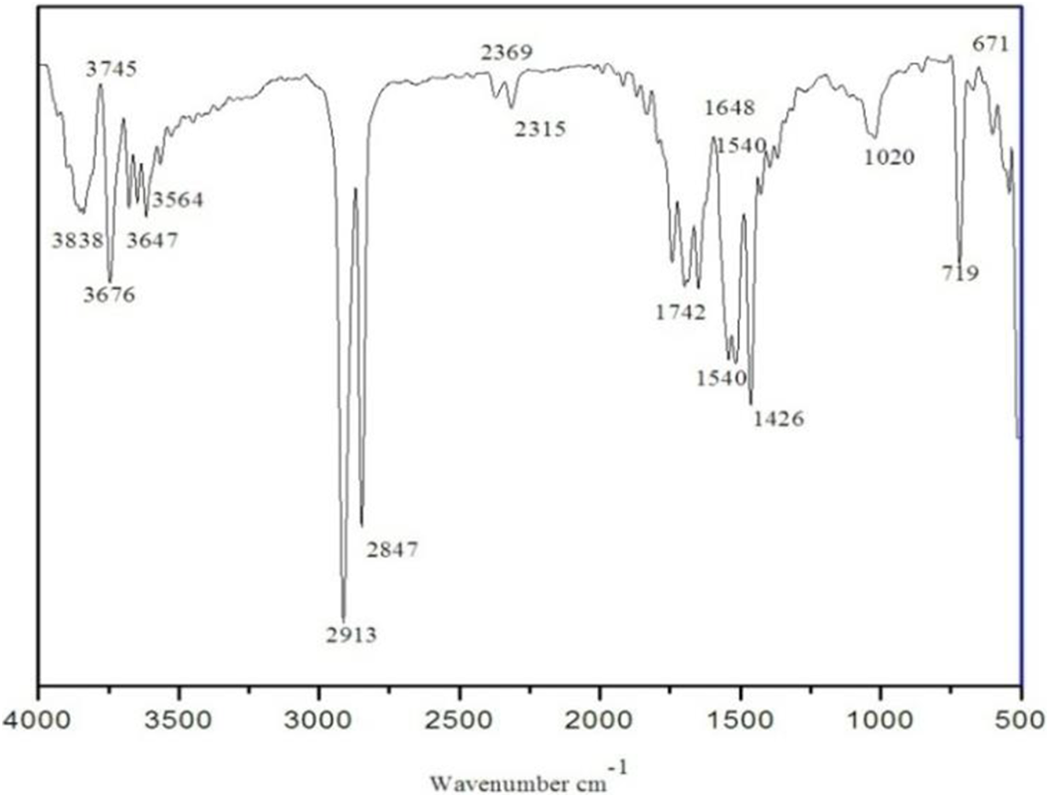
FTIR spectrum of the HAp-HDPE composite.
X-Ray diffraction of the 30 wt. % HAp-HDPE composite
The XRD pattern of 30 wt. % HAp-HDPE composite is shown in Figure 3. The diffraction peaks are in good agreement with Joint Committee on Powder Diffraction Standard (JCPDS) data.

The XRD pattern of 30 wt. % HAp-HDPE composite.
Sharp and narrow peaks show good crystallinity. 2θ value for the 30 wt. % HAp-HDPE specimen is found as 25.90, 31.91, 36.90, 41.40,46. 18,50.13, and 53.20 corresponding to crystal plane 002, 211, 202, 310, 222, 213, 321, and 004, respectively.
FESEM surface morphology of 30 wt. % HAp-HDPE composite
The surface morphology of 30 wt. % HAp-HDPE composite specimens was investigated through FESEM. The FESEM morphology in the-plane level is presented in Figure 4 depicts the exposed HAp. The morphology exhibits the homogeneously dispersed HAp in HDPE. The background white with gray color depicts the matrix phase HDPE and white silver metallic shining color of particulates indicates the HAp fillers. It can be concluded that FESEM morphology exhibits an arrangement of different sizes of the HAp extracted from the fish scale and sea shell.

FESEM surface morphology of 30 wt. % HAp-HDPE composite.
DSC analysis of the HAp-HDPE composite
The DSC curve illustrates the behavior of HAp-HDPE composite10:3 ratio (Matrix 100 g: Filler 30 g) with a function of increasing temperature as shown in Figure 5. The results reveal that the melt onset temperature is 129.7°C, and the melt peak temperature is found at 140. 6°C.
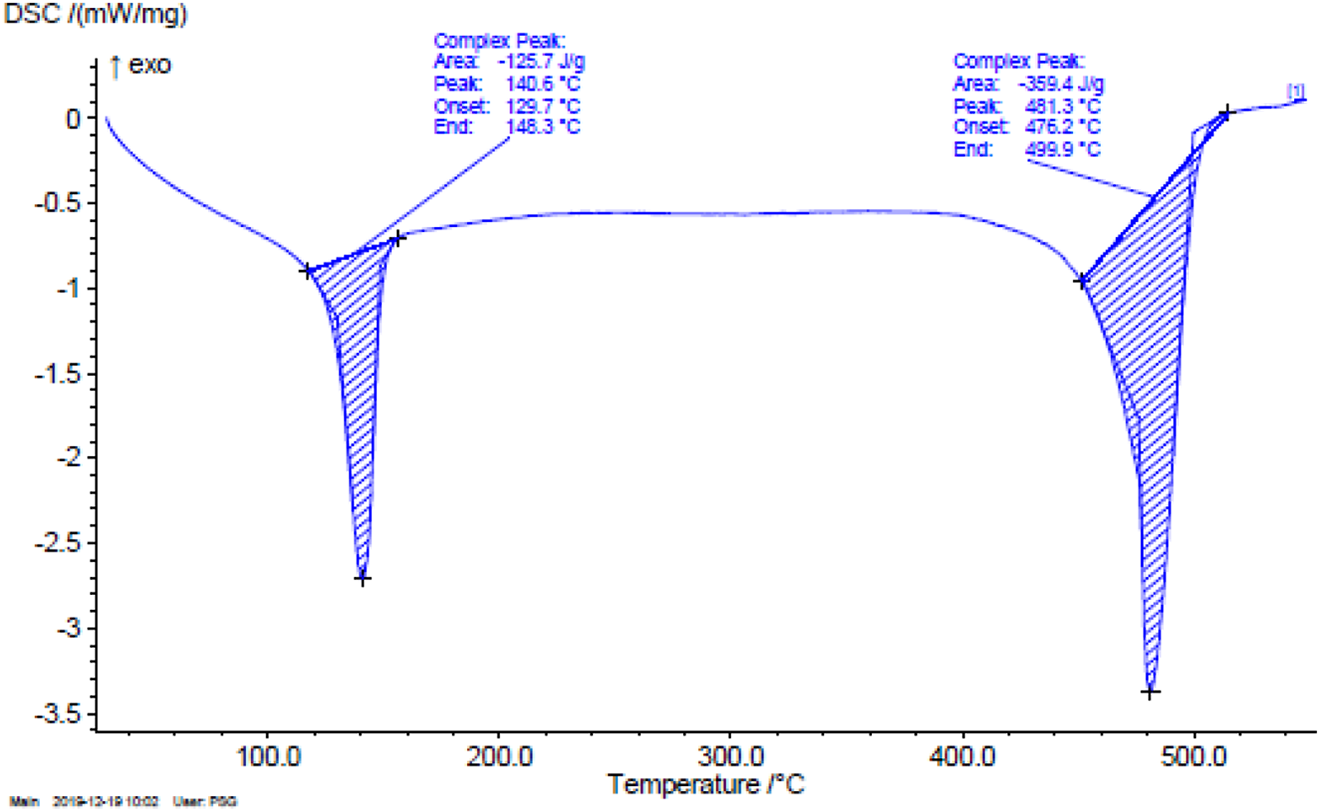
DSC Analysis of the HAp-HDPE Composite.
The energy absorbed by sample was found as 125.7 J/g due to an endothermic reaction while the sample absorbed the heat. This lower energy state that HAp-HDPE composite absorbs heat, which is seen as a negative peak on the graph. At this transition, the HAp-HDPE composite is changed from a relatively hard and glassy solid, to a softer, more stretchy or rubbery material when the temperature is increased above 148.3°C.
TGA analysis of the HAp-HDPE composite
The TGA curve depicts the decreasing weight of the HAp-HDPE composite 10:3 (Matrix 100 g: Filler 30 g) with a function of increasing temperature as shown in Figure 6 and the percentage of mass reduction (%) is given in Table 2.

TGA curves of a 30 wt. % HAp-HDPE composite.
TGA properties of matrix and filler (10:3) wt. %.

In the first stage, temperature is steadily increasing from 30.21°C to 465.21°C and the weight is approximately stable and starts decreasing from 4.87 to 4.05 mg.
In the second stage, the temperature increases further from 465.21°C to 475.25°C the quantity of weight decreases from 4.05 to 3.37 mg. In the third stage, the temperature increases further from 475.21°C to 485.214°C and the huge quantity of weight were decreased from 3.37 to 2.025 mg. In the fourth stage, the temperature was increasing further from 485. 214 to 495.214°C and a huge quantity of weight decreases from 2.025 to 1.17 mg.
In the fifth stage, the temperature increases further from 495.21°C to 545.21°C and a quantity of weight decreases from 1.1723 to 0.703 mg. Finally, further increasing the temperature, the remaining entire weight was exhausted. From the figure it can be concluded that peak temperature of weight degradation was found at 465.21°C. The total amount of weight loss is around 94.05%. The major loss of weight is attained between the temperatures of 440 to 510°C. If further increasing the temperature of the materials, it becomes oxidized and finally gets decomposed.
Evaluation of cytotoxicity HAp-HDPE composite
The grade (%) of cytotoxicity and cell viability of the 30 wt. % HAp-HDPE specimen were calculated for different volume of the extracts (10, 20, 30, 40, and 50 μl) by indirect cytotoxicity test and results are given in Table 3. The obtained results were compared with standard reference data of cytotoxicity reactivity in Table 1 and the results were tabulated.
Level cytotoxicity of 30 wt. % HAp-HDPE composite.

Figure 7 presents the level of cytotoxicity (%) of 30 wt. % HAp-HDPE specimens with five different volumes of the extracts (10, 20, 30, 40, and 50 μl) allowed to interacted with fresh culture medium through indirect cytotoxicity test.

Cytotoxicity 30 wt. % of HAp-HDPE composite.
The level of cytotoxicity (%) obtained was maximum of 50 for 50 µl liquid extracts of the specimen interacted with fresh medium. The levels of cytotoxicity 50% is compared in Table. 3.
The level of cytotoxicity of 50% is between (21-50%). This grade of 3 is reveals that reactivity is under mild level. Figure 8 depicts the level of cell viability (%) of the composite specimen having five different volumes of the extract (10, 20, 30, 40, and 50 μl).

Cell viability 30 wt. % HAp-HDPE composite.
Cell morphology
The cell morphology of fresh medium (control) is shown in Figure 9(a) and cell morphology of different level of liquid extracts of 30 wt. % HAp-HDPE specimen interacted with fresh culture medium (10, 20, 30, 40, and 50 μl) is shown in Figure 9(b) to (e). The cell viability for different volume of the extracts was calculated and compared with control (fresh medium). The cell viability was found based on image and intensity of dark purple color, cell morphology, size of cells, orientation, and number of cells counts for five different level of liquid extracts which was added in the fresh medium (10, 20, 30, 40, and 50 μl) by using 570 nm photometer and the results compared with the fresh culture medium. The liquid extracts of 10, 20, 30, 40, and 50 µl were separately added in the fresh cell culture medium and the cell viability was revealed. The round bubbles and hairlines indicated the nonliving cells and living cells in the culture medium, respectively.

(a) Control, (b to e) Cell morphology of MG63 cells after interacting with liquid extracts of the HAp-HDPE Specimen (10, 20, 30, 40, 50 µl).
The culture medium interacting with 10 µl and 20 µl extracts exhibited 81 and 77% cell viability and 19 and 23% Cytotoxicity, respectively. The culture medium interacting with 30 µl exhibited 62% cell viability and 38% slight cytotoxicity. The culture medium interacting with 40 µl exhibited 57% and 43% slight cytotoxicity. The culture medium interacting with 50 µl exhibited 50% slight cytotoxicity.
Conclusion
The organic and inorganic functional groups in the HAp-HDPE specimen were found and confirmed using the FTIR spectrum. The effect of HAp content on the structural and indirect cytotoxic properties of HAp-HDPE biocomposites was investigated in this study. At HAp contents of 30% wt., the presence of HAp-HDPE contributed to the sight toxicity of the composite after interacting with the MG63 cell line. Fish scale and sea shell ceramics resulting from the heat treatment of fish waste at 700°C have a major hydroxyapatite phase. Also, the preliminary in vitro experiments revealed the cytotoxicity of the 30 wt. % HAp-HDPE specimen gave none to slight cytotoxicity reactivity to MG-63 cells and after 24 hours interaction. The 30 wt. % HAp-HDPE specimen exhibited 50% cell viability and 50% of toxicity. Such an extracted HAp would be useful as inexpensive ceramics are biologically and environmentally compatible materials. Fish scale and sea shell originated hydroxyapatite is a potential new ceramic resource in the coastal areas. The present results also suggest a possible materials-recycling technology for future waste management.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
