Abstract
The high-density polyethylene (HDPE) and fish scale particulates are in the ratio of 10:3 (matrix 100 g:filler 30 g) which is filled and blended with thermoplastic composites and are then fabricated by the injection molding machine. In this work, the thermal energy absorption is obtained as 103.9 J g−1 that melts onset temperature at 121.75°C and melts peak temperature at 129.98°C of the specimen using differential scanning calorimetry instrument. A gradual mass degradation and decomposition of the prepared samples were analyzed from the thermogravimetric analysis. Evaluation of microstructure, surface morphology, and elemental analysis was carried out using field emission scanning electron microscope. The presence of functional groups in the polymeric materials was identified using Fourier transform infrared spectroscopy. The cytotoxicity testing of composites has been carried out using MG 63 cell line. In these studies, five different volumes of liquid extract of the prepared specimen having different concentrations (10, 20, 30, 40, and 50 μL) were allowed to interact with fresh cell culture medium for 24 h. The cell viability, cell morphology, and the levels of cytotoxicity of the composite specimen were studied as per ISO 10993:12 and ISO 10993:5 test standards. It was found that the natural fiber filled composite showed none to slight cytotoxic reactivity to MG-63 cells after 24 h contact. The cytotoxicity level of fish scale particulate filled HDPE composite material was compared with standard reactivity level and it was confirmed to have low toxic level (none to slight).
Keywords
Introduction
Many materials have been used for bone defect repair, but there are no ideal and fully approved implants. Among the materials, high-density polyethylene (HDPE)-based composites can be used as an implant, because of their high mechanical properties (E-modulus, strength, and hardness) and good biological properties (no toxicity and biocompatibility). 1 Recently hydroxyapatite (nHA) [Ca10(PO4)6(OH)2] has been introduced as a bone graft material in medical applications due to its similar chemical composition as that of bone. 2 The calcium deficient nHA mineral phase which is found in the fish scale (Pagrus) has higher tensile strength of 90 MPa and the demineralized scales have lower tensile strength of 36 MPa, which depends on the interactions between the apatite and collagen fibers. 3 Fish scales (Lobelia rohita) are reinforced with epoxy composites that exhibit a tensile strength of 66 MPa (5 wt%), 64 MPa (10 wt%), and 61 MPa of which (15 wt%) were estimated with neat epoxy that is about 70 MPa. 2,4 The scale (Megalops atlanticus) consists of collagen reinforced apatite. The tensile strength of scales was found at the head, middle, and tail regions in the possible three primary orientations 0° (24 MPa), 45° (20 MPa), and 90° (15 MPa), respectively. 5 The nHA from pork bones was extracted via hydrolysis, pre-calcination at 600°C, and calcination at (750–950°C). 5 It can also be extracted from scales (Tilapia) by calcination and acid–base treatment. 6 It was extracted from fish scale by enzymatic hydrolysis and it has an increased MG 63 growth nHA and can be considered as a potential biomaterial for nonnatural bone fabrication. 7
Scales found on teleost fish accommodate both flexibility and protection. These are the tissues which form a natural flexible armor, which protects the underlying soft tissue and vital organs. 8 The Fourier transform infrared spectroscopy (FTIR) spectrum of fish scale and extracted collagen is in the range of 450–4000 cm−1 N–H of amide І (1640 cm−1), amide ІI (1534 cm−1), and amide ІІІ (1226 cm−1). The visible bands in fish scales at 1073, 600, and 545 cm−1 indicate the asymmetric stretching, symmetric, and asymmetric bending vibrations of phosphate group (PO4 3−), respectively. 9 Sha et al. 9 investigated cell viability, cell morphology, and cytotoxicity of titanium dioxide (TiO2) nanoparticles (NPs) in vitro using different liver cell lines: (i) carcinoma (SMMC-7721), (ii) human liver (HL-7702), (iii) rat liver (BRL-3A), and (iv) rat hepatocarcinoma (CBRH-7919). TiO2 particles were exposed to varying concentrations (0.1–100 µg mL−1) and period of time (12–48 h). The results revealed that carcinomatous liver cells and human liver cells exhibited more tolerance to TiO2 NPs exposure for 24 h, comparing to normal liver cells and rat liver cells. 10
The biodegradable nHA (fluoridated) is coated on Mg–Zn alloy via electrodeposition method and was observed to enhance the interface area on bioactivity in bone healing. None toxicity was evaluated and even more cells viability was exposed on the 7th day against the normal culture reference (control). 11 The phosphate (alkaline) and calcium release were noticed in bone alternates which were prepared with Terminalia arjuna (TA) extract when compared with control. The cell line MG 63 was revealed to exhibit the potential influence in cell differentiation. 12 Huang et al. investigated the cytotoxicity behavior of virgin HDPE and incorporated it with theNPs (graphite) of different weight percentage. 13
The virgin HDPE and all HDPE/graphite NPs (GNPs) polymer composite specimens that interacted with wish cell line exhibited no cytotoxicity. Bioglass HDPE composite is bioactive and biocompatible and has a stimulatory effect on human osteoblast cells. In vitro and polyether urethane film containing 0.1% zinc diethyldithiocarbamate biomaterials is carried out in vitro studies in BABL/3T3 cells. The indirect cytotoxicity test was carried out in hydrogel membrane (composed of PVP, PEG and agar) and it revealed the absence of toxicity. 4,14,15 Sarkar et al. investigated the growth of skin in the fibroblast cells over polyurethane outer surfaces and the investigation revealed a good proliferation and no cytotoxic effect, and it has been proved that the synthesized polyurethane to be a suitable material for tissue engineering. 16 Diaz and Puerto have investigated in vitro poly lactide-co-E-caprolactone/nHA composites biomaterials to be used as a scaffold in bone engineering. The characterization was done in the in vitro degradation mechanism of this device in an aqueous media, phosphate buffered saline (PBS). 17
Ramani Deepachitra et al. carried out an investigation in graphene oxide, this research helped to enhance the composite strength, cell adhesion, and nHA-induced more osteomineralization sites on cells (MG-63). The functionalized graphene oxide possess superior osteoinductive potential and it would be a capable composite for orthopedic implants. The collagen pulled out from fish scale is an ionic liquid pretreatment that is a natural-made biopolymer and also has wide applications for medical uses. 18 The rapid development of biocomposites has led to the fabrication of more and more natural fibers filled composites to perform a particular function, such as artificial bone for human implants. nHA has been used in the medical field as a bone repair material because of its significant bioactive tendency and biocompatibility. 8 In vitro studies, which were carried on virgin HDPE/GNPs filled composites with cell line (WISH), revealed the absence of cytotoxicity and it was considered as a suitable biomaterial for hip joint replacement. 19
The objective of the study is to develop composite materials using naturally available nHA from fish scale particulates, which is similar to the human bone chemical composition. It is used as filler in HDPE, as PE thermoplastic has excellent biocompatibility and mechanical properties. 12 Since both are having biocompatibility properties as per the literature review, a new composite has been developed on the injection molding and cytotoxicity studies have been carried out.
To carry out the in vitro cytotoxicity test, five different concentrations (10, 20, 30, 40, and 50 μL) of liquid extracts were allowed to interact with MG 63 cell line for 24 h. In this analysis evaluation, the morphology of cell viability of natural particulates filled HDPE polymer composite specimens was studied, and the results were compared with (control) reference fresh cell culture medium. The preliminary study results provided the level of non-cytotoxicity (low level), which are essential to carry out the in vivo studies of the specimen to be used as a biomaterial. The natural particulates filled HDPE polymer composites specimen exhibits no cytotoxicity level to the MG 63 cell line. Fish scale filled HDPE composite has been considered for this present analysis as core mixture, as there is no relevant literature related to this composite mixture.
Experimentation
Materials
The waste Catla (Labeo catla) grey colored fish scale particulates were used as the filler to produce HDPE composite. The fish scale has a density of 0.9 g cm−3. The commercially available HDPE, procured from Varsha Poly Products (India), was used as a matrix. It has a density of 0.91 g cm−3, melting point of 125° C, tensile strength of 16 MPa, and melt flow index of 6 g/10 min at 190°C and 2.16 kg.
Cell line
The cell line MG-63 (organism: human, origin tissue: bone, and morphology: osteosarcoma) was procured from National Centre for Cell Science (NCCS) (Pune, India) and cell culture was developed at biotechnology laboratory to carry out in vitro studies.
Methodology
To develop the required composite materials, a systemic approach was carried out. The methodology and process carried out to make natural particulate-filled HDPE composite specimen is shown in Figure 1. To produce the composite specimen mixtures, required quantity has been taken for analysis. The HDPE and fish scale filler in the ratio of 10:3 was used to form the composite. To produce the composite specimen mixtures of the required quantity, the ratio is 10:3 wt% (matrix 100 g:filler 30 g) of HDPE and fillers have been taken.

Methodology.
The sample was preheated at 60°C for about 60 min and introduced in the heating chamber for further heating. The temperature 90–100°C was preferred in injection molding to control the melting of matrix and thermal degradation of fillers in the composite. The manual operating pressure was 2–3 bar during the injection.
Preparation of fish scale particulates
The fish scales were collected from a fish market in local area Omalur, Salem, Tamil Nadu (India). The scales were cleaned over and over again with warm water to remove the adhering dust and soluble impurities from their surface. The washed scales were kept in the sunshine for 1 day and dried. It was then grinded into powder having particulates of 15–30-µm size.
Injection molding
To melt the sample mixtures, the required quantity of preheated fish scale particulates and HDPE has been taken in the heating chamber for few minutes. The heated and blended semisolid pressurized mixture was placed into the cavities in the die to fabricate the molded composite specimen. The obtained composite specimens were then removed from the die and unwanted portion was trimmed out. The temperature was slowly controlled to keep the fillers in the matrix without any remarkable change. The temperature (80–100°C) was preferred in the injection molding process to control the melting and thermal degradation of fish scale particulates present in the composite.
The die is made of steel which has upper and lower die replica of specimen shape in the form of a cavity with a suitable size. The dies are provided with a cavity at the junction to mix together the melted semi-solid mixture of fish scales particulates with HDPE. Fish scale particulates filled HDPE and mixture were fed into a heated barrel pushed into a cavity where it is cooled and hardened to the configuration of the mold cavity. The filled mixture of required quantity was pushed to specimen cavities through the small circular passage to get the cured specimens. The specimen cavity has both ends extended portion to accommodate the excess mold material. Figure 2 depicts the molded composite specimen. The molded specimens are used for further studies.

Natural fiber fish scale particulates filled with HDPE specimen.
Characterization
Fourier transform infrared spectroscopy
FTIR analysis of fish scale particulate’s powders was carried out in the range from 500 cm−1 to 5000 cm−1. The data were used to identify and confirm the functional groups present in the molded sample. The test was carried out at PSGR Krishnammal College for Women, Coimbatore.
Microstructure and EDX analysis
The surface morphology, microstructure, and energy dispersive X-ray (EDX) analysis of fish scale particulates filled HDPE specimens were examined using a ZEISS Sigma 300 scanning electron microscope (Bangkok, Thailand) at Coimbatore Institute of Technology, Coimbatore. The molded samples were gold sputtered before examining the specimen.
Differential scanning calorimeter
The DSC analysis is under flowing nitrogen (nitrogen 50.0 mL min−1). The sample with the mass is in the order of 9.5 mg which was heated gradually from 10°C to 300°C at a rate of 20°C min−1 at Amrita Vishwa Vidyapeetham, Coimbatore (India). The specimens were analyzed in the ratio of 10:3 wt% compositions, and the crystallization, peak melting temperatures, and thermal energy required for phase changes were obtained from the result.
Thermogravimetric analysis
TGA was conducted using a Q600 SDT (TA Instruments, Lindon, USA) at Amrita Vishwa Vidyapeetham, Coimbatore (India). The sample with a mass of 10 mg was heated gradually from the temperature of 50–600°C at a constant heating rate of 20°C min−1 under flowing nitrogen (20 mL min−1).
Ethylene oxide sterilization
Ethylene oxide (EtO) sterilization and in vitro direct cytotoxicity test of natural particulate filled HDPE composite specimens were carried out as per standard ISO 10993:12 and ISO 10993:5 at the Centre of Excellence for Medical Textiles, South India Textile Research Association (SITRA), Civil Aerodrome Post (Coimbatore, India).
Biocompatibility studies
EtO sterilization
EtO sterilization process is carried out to kill the microorganism present in the specimens for further investigating the in vitro cytotoxicity studies. The specimens were pre-conditioned and humidification was carried out for 20–30 min to attain 60 lbs pressure and temperature of 50°C. The sample was exposed to 100% level of EtO for the duration of 7 h and aeration was done at overnight for 12 h. Finally, the sample was sterilized by EtO sterilizer. A color change observed which indicates the completion of the sterilization process is shown in Figure 3.

Assessment color of sterilization indicator. (a) Before sterilization and (b) after sterilization.
Direct cytotoxicity
The EtO sterilized specimens were investigated for direct in vitro cytotoxicity studies using MG 63 cell line. The liquid extract from the sterilized sample was allowed to interact with MG 63 cell line using five different (10, 20, 30, 40, and 50 μL) concentration levels by direct cytotoxicity method. The percentage of cytotoxicity and cell viability of the specimen were analyzed.
Cell culture
MG-63 is an established and well-characterized cell line that has demonstrated reproducible results. Cell culture medium was developed by minimum essential medium supplemented with fetal bovine serum. Since this is a suitable serum which has the least level of antibodies, it contains more growth factors that permit versatility in many different cell culture applications.
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay
The cell viability was assessed using an 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay after natural particulate polymer composite specimen exposure, as only the viable cells have functional mitochondrial dehydrogenase enzymes which can reduce MTT to formazan. After fish scale particulates and cell interactions, cells were washed three times with PBS (pH 7.3) and culture media; MTT (0.5 mg mL−1) was added to each cell and incubated at 37°C for 4 h.
Result and observation
FTIR analysis
Figure 4 shows the FTIR spectrum of fish scale particulates in the wave number range from 500 cm−1 to 5000 cm−1. The characteristic band around 3743 cm−1 is observed and it shows the presence of OH group.

FTIR spectra of fish scale particulate.
The bands appear around 3273 cm−1 show the presence of amide which is most prominent in fish scale. Apart from that, the band at 1642 cm−1 confirms the presence of bending vibration of N–H in fish scale. The amide ІІ has vibration band around 1532 cm−1 which is also present in the scale. The band at 1030 cm−1 is assigned to amide ІІІ. Moreover, the bands at 1030 and 606 cm−1 indicate the vibrations of a phosphate group (PO4 3 −) in fish scale. 5
Figure 5 shows the FTIR spectrum of HDPE. The characteristic band around 3743 cm−1 is observed and it shows the presence of OH group. The bands around 2913 cm−1 show the presence of C–H which is most prominent in HDPE. Apart from that, the band at 2847 cm−1 confirms the presence of stretching vibration of alkenes C–H. The C=0 ester stretching vibration band was found around 1742 cm−1. The C–H bend alkene has vibration band around 1465 cm−1 which is also present in the scale. The band at 1465 cm−1 is assigned to amide ІІІ, which is also revealed in HDPE. Moreover, the bands at 721 cm−1 indicate the presence of the vibrations of groups (C–H) in HDPE.
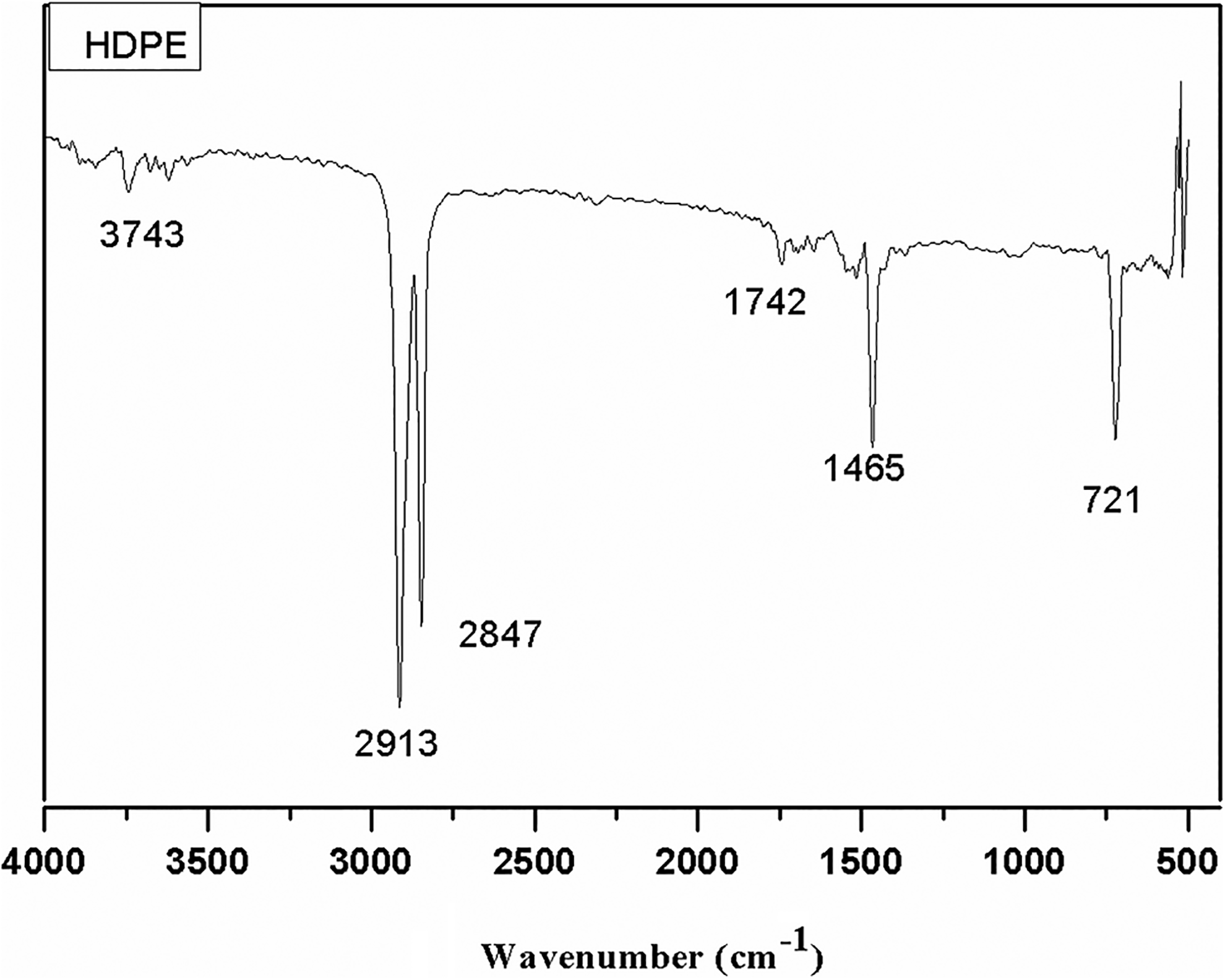
FTIR spectra of HDPE.
EDX analysis
The EDX spectra of the fish scale particulates filled HDPE are shown in Figure 6. The EDX analysis of fish scale particulates filled HDPE composite is shown in Table 1.

EDX analysis of fish scale particulates filled HDPE.
EDX analysis of fish scale particulates filled HDPE.

AN: atomic number; EDX: energy dispersive X-ray analysis; HDPE: high-density polyethylene; Unn. C: unnormalized concentration in weight percentage of the element; Norm. C: normalized concentration in weight percentage of the element.
The different elements present in the specimens were identified as follows. The wt% of O, C, Ca, P, and K is 51.44, 27.40, 12.65, and 8.51, respectively. The Ca/P ratio is found to be 1.48.
Surface morphology
Field emission scanning electron microscope (FESEM) image of fish scale particulates which is dispersed in the HDPE matrix specimens was investigated. The inner portion samples were mounted on stubs with gold sputtering to enhance the conductivity of the samples. The inner surface morphology of the specimens was examined directly using a SEM. The morphology of a fish scale particulates filled HDPE in the in-plane level is presented in Figure 7. Fish scale particles filled HDPE are sometimes clustered in the specimens due to improper blending. This formation can be avoided by further reducing the size of the particulate and by feeding them into the cavity to disperse homogeneously.

FESEM micrograph of exposed fish scale particulates filled HDPE.
The results show that particulates filled HDPE was sometimes clustered in the fasteners which are revealed in microstructure image. This formation can be avoided by reducing the size of the particulate and feeding into the cavity to disperse homogeneously. By preparing the composites with nano size particulates, the agglomeration trouble can be condensed to some extent. Particulates were agglomerated and poor intimate contact with HDPE is revealed. The crater formation can be reduced by adjusting the melting temperature and proper blending, mixing of scale particulates, and HDPE pellets. Figure 8 shows the micrograph of the homogeneously filled fish scale particulates with HDPE. The morphology of fish scale particulates filled with HDPE patterns shows the equally disbursed fish scale particles.

FESEM micrograph of fish scale particulates filled HDPE.
The background grey dark color depicts matrix phase (HDPE) and white silver metallic shining color indicates the fish scale (fillers) particulates. The particulate size is found to be measured in the range of 19.96–29.44 µm. Further, the particulates are of reduced size with fish scale filled HDPE composite without any agglomeration and crater formation. This exhibited an arrangement of different size of the fish scale particulates filled in the matrix. This clearly shows the matrix and fiber interface.
Differential scanning calorimetry
About 9.5 mg of fish scale particulate filled HDPE powder sample was heated gradually from 10°C to 300°C at a rate of 20°C min−1. The results revealed that the melt onset temperature was 121.75°C and the melt peak temperature was found to be 129.98°C, as shown in Figure 9. The energy absorbed by sample 103.9 J g−1 is due to an endothermic reaction as the sample has absorbed the heat.

DSC curves of a fish scale particulate filled HDPE composite material.
This lower energy state that filled composite absorbs heat, which is seen as a negative peak on the graph above. At this transition, the filled polymer composite is changed from a relatively hard and glassy solid to a softer, more stretchy or rubbery material. As the temperature was increased above 129.98°C, the polymer structure gains a sufficient amount of flexibility to reorganize amorphous into a lower energy crystalline form of the composite. In the crystallization stage, the atoms or molecules are highly organized into a bonded structure. Finally, as the heat is increased continuously, the HDPE blend fish scale powder is melted so that the peak point is reached. The temperature can be rapidly increased further that it can produce a fully amorphous polymer.
Thermogravimetric analysis
Figure 10 shows that the thermal stability analysis of the fish scale filled HDPE sample (10 mg) was carried out using TGA analysis in the temperature range of 10–700°C. The TGA measures the amount of abrupt weight degradation of fish scale particulate filled HDPE composite material.

TGA curves of a fish scale particulate filled HDPE composite material.
Since, it is a function of progressively increasing temperature (°C), or isothermally as a role of time duration (seconds), in an atmosphere of nitrogen 300 (mL min−1) environment. The graph depicted the decreasing weight of a composite material with a function of increasing temperature. In the first stage, when the temperature is increased gradually from 28.23°C to 325.85°C, the weight of the sample is nearly stable decreasing from 9.34 mg to 9.01 mg with 3.5% weight loss. In the second stage, the temperature has increased further from 467.76°C to 497.75°C, and the weight got decreased from 8.01 mg to 2.03 mg with 78.26% weight loss. In the third stage, further, it increases the temperature gradually from 504.22°C to 690.27°C, the entire remaining weight of the sample (100%) is exhausted and oxidized. This corresponds to the removal of water and organic matter. The chemical reaction describes that the sample is Ca10(PO4)6(OH)2 + CH2 = CaHPO4 + H2O + Ca (dicalcium phosphate, water, and calcium).
Evaluation of cytotoxicity
The samples after ETO sterilization were incubated in 1× PBS at 37°C for 24 h. The culture medium from the MG-63 cells was replaced with fresh medium. The liquid extract of the test specimen at five different volumes in triplicates was added on the cells. After incubation at 37 ± 1°C for 18 h, MTT assay (1 mg mL) was added in all the wells and incubated for 4 h. After incubation, a small quantity of dimethyl sulfoxide (DMSO) and organic sulfur was added in the wells and cytotoxicity results were revealed at 570 nm using photometer. The cell viability and cytotoxicity were calculated using the following equations
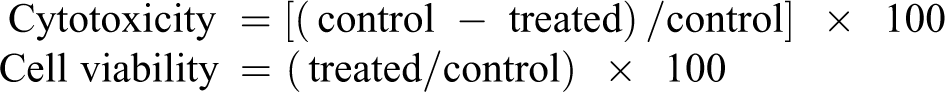
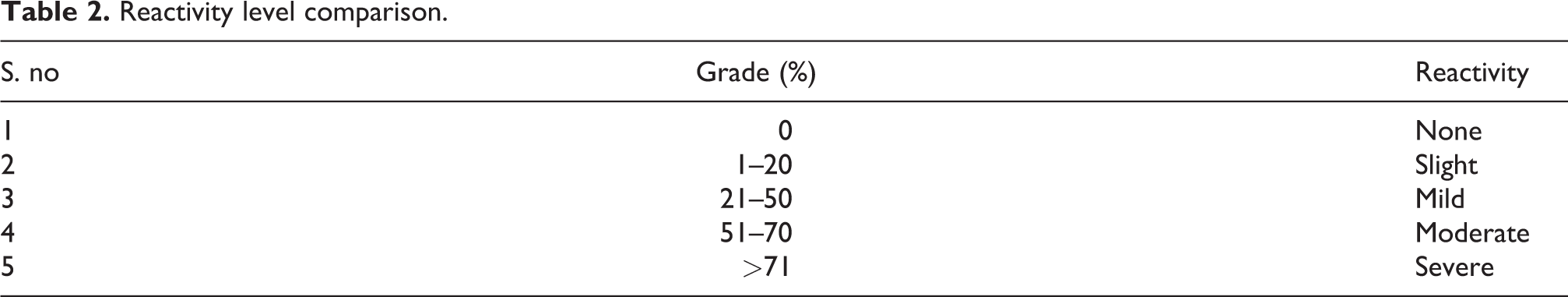
The grade (%) of cytotoxicity was calculated from the direct cytotoxicity method and the results compared with standard reference data are presented in Table. 2.
Reactivity level comparison.
The grade (%) of cytotoxicity and cell viability of the specimen was calculated for different volume of the extract (10, 20, 30, 40, and 50 μL) by direct cytotoxicity test and the results are presented in Table 3. The obtained results are compared with standard reference data of cytotoxicity reactivity in Table 2 and the results were proven.
Level cytotoxicity of fish scale particulate filled HDPE composite material.

HDPE: high-density polyethylene.
Figure 11 depicts that the level of cytotoxicity (%) of composite specimens with five different volumes of the extract (10, 20, 30, 40, and 50 μL) allowed is interacted with fresh culture medium by direct cytotoxicity test. The level of cytotoxicity (%) obtained was a maximum of 5.8 for 50 μL liquid extract of the specimen interacted with fresh medium. The level of cytotoxicity 5.8% was compared in Table 2. The level of cytotoxicity of 5.8% fell between the grades of 2 (1–20%). The reactivity level was under slight level which is within an acceptable range.

Cytotoxicity (%) of fish scale particulate filled HDPE composite material.
The above graph Figure 12 depicts the level of cell viability (%) of the composite specimen having five different volumes of the extract (10, 20, 30, 40, and 50 μL) separately interacted with fresh culture medium by direct cytotoxicity test.

Cell viability (%) fish scale particulate filled HDPE composite material.
Cell morphology
The cell morphology of fresh medium (control) is shown in Figure 13, and the morphology of different levels of liquid extract is added to the fresh medium (10, 20, 30, 40, and 50 μL) and is shown in Figure 14. The cell viability for different volume of extract added in the control was counted and compared with control (fresh medium). The cell viability was found in the image based on their intensity of dark purple color, cell morphology size, orientation, and number of cell counts for five of the different level of liquid extract which are added in the fresh medium (10, 20, 30, 40, and 50 μL) using 570 nm photometer and the results compared with the fresh culture medium.
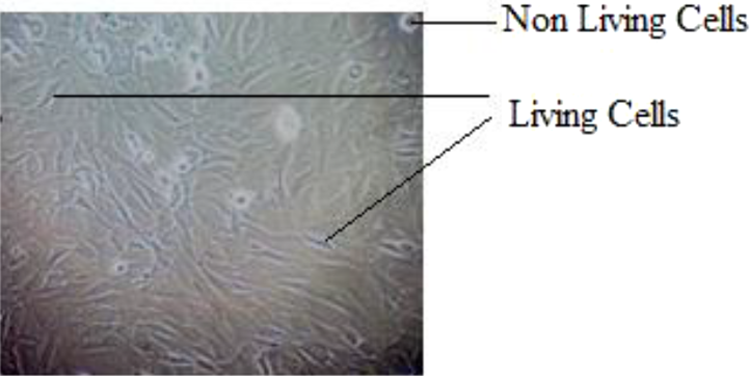
MG-63 cell morphology of control.

MG-63 cell morphology (10, 20, 30, 40, and 50 μL) of liquid extract added control.
The liquid extract of 10, 20, 30, 40, and 50 μL was separately added in the fresh cell culture medium and the cell viability was revealed. The round bubbles and hairlines indicate the nonliving cells and living cells in the culture medium, respectively. The culture medium with 10 and 20 μL extract exhibited 99% cell viability and 0% cytotoxicity. The culture medium with 30 μL exhibited 99.4% cell viability and 0.6% slight cytotoxicity. The culture medium with 40 μL exhibited 98.3% and 1.7% slight cytotoxicity. The culture medium with 50 μL exhibited 98.3% and 5.8% slight cytotoxicity.
Conclusion
In the present research work, fish scale filled HDPE composite microstructure was investigated and elemental analyzed by EDX analysis using FESEM. The atomic wt% of C, O, Ca, P, and K was found to be 83.19, 14.37, 1.89, 0.51, and 0.04, respectively. The size of particulates was measured in the range of 19.96 ± 0.1 to 29.44 ± 0.1 µm. Further by utilizing it as filler in a polymeric matrix to form a composite material with nano size particulates, the agglomeration trouble was condensed to some extent. The crater formation was reduced by adjusting melting temperature and proper blending mixture. Organic and inorganic polymeric materials were identified and confirmed using FTIR as 1642 cm−1 confirms the presence of bending vibration of NH of amide І in fish scale. The amide ІІ has vibration band of 1532 cm−1 and band at 1030 cm−1 assigned to amide ІІІ which is also confirmed in fish scale. The thermal energy absorbed during endothermic reactions was found at 103.9 J g−1; the melt onset temperature was 121.75° and melt peak temperature was at 129.98°C; and peak melting temperature was found using DSC instrument.
Thermal stability and mass degradation of the sample were evaluated by varying the temperature (10–700°C) using TGA instrument. The results showed in the first stage that when the temperature was increased gradually from 28.23°C to 325.85° C, the weight of the sample was nearly stable and started decreasing from 9.34°C to 9.01 mg of 3.5% loss. In the second stage, the temperature was increased further from 467.76°C to 497.75°C and the maximum quantity of weight was decreased from 8.01 mg to 2.03 mg of 78.26% loss. Finally, further increasing the temperature gradually from 504.22°C to 690.27°C, the entire remaining weight of the sample (100%) was exhausted and oxidized. The tensile strength of the fish scale particulate filled HDPE composite specimens was found as 25 ± 0.5 MPa.
Investigation of direct in vitro cytotoxicity work was done as per ISO Standard 10993:5. The preliminary in vitro experiments revealed that the cytotoxicity of the test samples of a natural fiber filled composite showed none to slight cytotoxic reactivity to MG-63 cells after 24 h contact. Through this study, percentages of cell viability and the level cytotoxicity were investigated. However, further in vivo investigations of a natural fiber filled HDPE composite specimens are to be studied. Since there was no toxic in the specimens.
This research work opens up a new opportunity for utilization of a fish scale particulates as filler filled with HDPE matrix. In this study, it is concluded that biodegradable waste, mainly fish scale, can be effectively used for engineering and medical applications by preparing injection molding with the HDPE. A new composite has been (10:3 wt%) developed which would reduce the usage of HDPE around 30–40%. Plenty of available biowaste fish scale can be converted into suitable material for various applications.
The next phase of the fish scale filled HDPE composite is to be investigated indirect cytotoxicity, cell adhesion, and in vivo biocompatibility studies to ensure the other properties of biocompatibility. Since it has no toxicity in the specimens, it opens a wide opportunity for the development of by-products from the fish scale wastes.
Footnotes
Acknowledgments
The authors are grateful to the Department of Mechanical Engineering, Coimbatore Institute of Technology, Coimbatore, for providing the required instrument for this work. We are also grateful to Mrs Vijaya Chinnappan, Ms Dhivyya Dharshinii Maniyam, Mrs B Gayathri, Dr V Arthy, Mr Sankar Rajan, Mr Edwin Jebaraj, Dr C Vivek, Dr G Sivakumar, Mr K Vellieyangiri, Mr P Aravindhan, Mr U Gurusundar, Mr V Keerthivasan, and Ms RM Kiruthika and University of Amrita Vishwa Vidyapeetham, PSGR Krishnammal College, and Centre of Excellence for Medical Textiles, South India Textile Research Association (SITRA), Civil Aerodrome Post (Coimbatore, India) for helping with the experiments.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
