Abstract
The nanocomposites of Chitosan (CS)-poly vinyl alcohol (PVA) reinforced with graphene oxide (GO) were fabricated by casting in a mold. The nanocomposite films were characterized by various techniques. The viscoelastic properties of the wound dressings were evaluated by dynamic mechanical thermal analysis (DMTA) since the healing quality of a wound dressing depends on such properties. DMTA results also were modeled by the Cole-Cole diagram. Finally, the effects of GO on antimicrobial and biological properties of films were studied. It has been shown that nanocomposite films support the initial attachment, proliferation, and growth of mouse fibroblast cells.
Keywords
Introduction
Graphene is a single layer of carbon atoms arranged hexagonally together by a backbone of overlapping sp2 hybridized bonds. 1 It has been the focus of extensive research studies due to its outstanding mechanical properties (e.g., stiffness, strength, and elasticity) high thermal stability and conductivity, impermeability to gases, high mobility of charge carriers, tunable band gap, and optical transparency. 2 Besides graphene, its functionalized counterpart, graphene oxide, also demonstrates excellent mechanical and thermal properties, together with biocompatibility, featuring it to have applications as a potent biomaterial. 3 The chemical groups (e.g., hydroxyl, epoxide, and carboxyl) on the surface of graphene oxide are well-known to be a practical and useful means to modify the graphene dispersion in water-based solutions. 4 Interestingly, graphene oxide is reported to be effective as antibacterial agent. 5 Therefore, by combining significant mechanical, thermal, and antibacterial properties, together with its water solubility, biocompatibility, and low processing costs, 2D graphene oxide nanosheets are considered promising candidates as nanoscale fillers for the future nanocomposites applied in the field of biomaterials. 6
Chitosan (CS) is a partially de-acetylated derivative of chitin containing the reactive amino groups. 7 Among naturally derived polymers, it is one of the most studied due to its biodegradability, biocompatibility, bio adhesivity, non-antigenicity, antibacterial activity, and responsivity. 8 However, its tensile strength and elasticity are not suitable for some biomedical applications, such as wound dressing and skin tissue replacement. CS mixed to other polymers, has become a window of research for modifying its properties. 9
Polyvinyl alcohol (PVA) is a non-toxic, water-soluble, extremely crystalline, biodegradable, and biocompatible polymer. 10 It has interesting physical and chemical properties, as well as good film-forming ability, due to plenty of hydroxyl groups, thus forming intermolecular hydrogen bonding. 11 Mixing CS with synthetic polymers, such as PVA, is an adequate method to prepare synthetic biodegradable polymers with fluent properties such as good water absorbance and appended mechanical properties while maintaining biodegradability. 12
It is possible to create a nanocomposite material merging desired properties by taking advantage of CS biocompatibility, excellent film-forming characteristics of PVA, and special physical properties of graphene oxide.13,14 To achieve this goal, several studies have been recently conducted on the fabrication of CS-PVA/GO nanocomposites, which are mainly based on the CS-PVA nanofibers. 15 Pandele et al. 16 prepared CS-PVA/GO nanocomposite films through casting in a mold and investigated the nanocomposite biocompatibility. Meanwhile, they showed the elastic modulus to be increased by over 200% by adding 6 wt% GO.
Despite the recent efforts, the viscoelastic behavior of CS-PVA/GO nanocomposite films is still unclear and should be unraveled for the future bioapplications, including wound dressing healing and skin tissue engineering. A successful wound dressing is required to exhibit acceptable structural, mechanical, and biological properties. Dressings are normally applied under tension to be conformed to wounded body surfaces. They may conform to an applied tension, which is less than that required to produce the limiting interface pressure (40 mmHg). However, since 40 mmHg pressure is the maximum value for tissue viability, no further increase in pressure is tolerable to smooth out any residual wrinkling. 17
If the wound dressing is a viscoelastic material, it can tolerate the pressure of more than 40 mm Hg from the bandage tension, and thus pressure will be relaxed with time. As a result, long-term conformability can be significantly increased above the immediate values based on elastic behavior; its advantage over plastic conformation is that subsequent joint motion is less likely to induce wrinkling as time-dependent viscoelastic deformations are recoverable. It is, therefore, necessary to investigate the viscoelastic properties of wound dressing. 18
Mazaheri et al. 19 have shown that the GO/CS films can be used as a template to enhance the proliferation of human mesenchymal stem cells (hMSCs) in vitro. Further, Pandele et al. 16 have demonstrated that MC3T3-E1 mouse osteoblastic cells can adhere and be developed on the CS-PVA/GO composite films.
In this study, the nanocomposite films are first characterized by FT-IR, XRD, TGA, and SEM. Thermal gravimetry analysis (TGA) and viscoelastic behaviors are then used to elucidate the dispersion of GO in the 50CS-50PVA polymer matrix. Modeling based on the Cole-Cole diagram is developed to study the viscoelastic behavior of polymers.
In the case of a good fit with experimental data, the parameters can be identified and eventually interpreted in terms of the width of the relaxation spectrum and effect of cross-linking.20,21 This modeling method can provide the predictive information about elastic and viscous properties independently. 22 The present study aims to evaluate the viscoelastic properties of films by the Perez model since it gives a good fit of Cole-Cole plots. 21
The antibacterial properties of the nanocomposites are evaluated against both gram-positive and gram-negative bacteria. Finally, in vitro viability assay by mouse fibroblast cells (L929) is performed.
Experimental
Materials
CS powder of medium molecular weight (MW = 161,000 g/mol), a degree of deacetylation (DD) equal to 75.6%, and viscosity of 1406 m.Pas (1% in 1% acetic) was purchased from Aldrich Chemical, being used without any further purification. PVA with 98–99% degree of hydrolysis and molar weight of MW = 31,000–50,000 g/mol was supplied from Aldrich Chemical (Cat. # 363138). GO nanosheets were produced via the well-known Hammers method with concentration and diameter of 4 g/lit and 2.7–6.8 μm, respectively.
Preparation of CS-PVA hybrid polymer and CS/PVA/GO nanocomposites
The polymer blend and the nanocomposites were fabricated based on the method suggested by Pandele et al. 16 with some modifications. Briefly, CS powder (1 g) was first dissolved in acetic acid solution (1%; 100 ml). Simultaneously, 100 ml of PVA aqueous solution was obtained by dissolving PVA powder in distilled water at 80°C in an autoclave. Blends were prepared by mixing 1:1 stoichiometric ratio (50PVA-50CS) of the two polymer solutions, after which the as-prepared solution was cast onto a transparent Petri glass dish. After drying at room temperature for 72 h, the films were peeled off from the mold and thermally treated in a vacuum oven.
An ultrasonic device (Bandline Company, Germany) with a maximum power of 75 W and a frequency of 20 kHz, HD 3200 series was used to distribute GO nanosheets in the base polymer mixture. The composite mixtures with different GO contents (0.5, 2, 3 wt%) were prepared by gradually adding the suitable amount of GO solution to that of 50CS-50PVA followed by sonication for 1 h at a power of 60 W. The films were prepared accordingly using a similar method as in the case of 50CS-50PVA blend.
Characterization
The structure of the nanocomposites was investigated by FT-IR measurements performed on the BRUKER Germany Tensor 27 Infrared Spectrometer in the waveguide range of 400–4000 cm−1. The phase and crystallinity of the structures were characterized by an X-ray diffractometer (Bruker XRD machine D8 Advance) and Cu-Kα radiation over a 2θ range of 5–80°, a step size of 0.02, as well as a scan speed of 4°/min at 30 kV and 40 mA. Steady shear viscosity curves were obtained at ambient temperature (23–25°C) by a controlled stress Couette rheometer (Anton-Paar, MCR300). The water absorption ability of the scaffold was studied using the following procedure. Dry scaffolds were first weighed (Wdry) and then immersed in distilled water for 24 h to reach the swollen equilibrium. Later, the scaffolds were gently removed from the beaker and placed on a wire mesh rack. Excessive water was drained, and scaffolds were weighed (Wwet) again to determine the absorbed water. The swelling ratio was calculated using the following equation:11,23

TGA curves were obtained using the Shimadzu TGA 209 F1thermogravimetric instrument. The temperature range applied was 20 to 700°C, with a ramp rate of 10°C min−1. The viscoelastic behavior was evaluated by performing dynamic mechanical thermal analysis using a DMA Netzsch 242 machine. For these measurements, rectangular specimens of 4 mm width, 25 mm length, and 0.1 mm thickness were prepared. The tests were carried out in three-point bending mode at a frequency of 1 Hz in the temperature range of 25 to 140°C, with a heating rate of 3°C/min (ASTM: D5023-01). The surface structures of the films were studied via an FE-SEM (TESCAN-Mira III).
The antibacterial activity of 50CS-50PVA and 50CS-50PVA/GO nanocomposite films was investigated via the agar diffusion method against the gram-negative (Escherichia coli) and the gram-positive (Staphylococcus aureus) bacteria. An amount of 50 μcc of the bacterial suspension was spread out on a Mueller-Hinton agar surface. The nanocomposite samples were then cut into small circular pieces (diameter = 10 mm) and included on the inoculated agar plates. Finally, the plates were incubated at 37°C for 12 h, after which the radius of the inhibition zone was measured.
In vitro biocompatibility was studied using mouse fibroblast cells (L929) provided by the national cell bank (The Iran Pasteur Institute, Tehran, Iran) and cultured in Dulbecco’s modified Eagle’s medium (DMEM, Sigma–Aldrich, USA) with 10% Fetal Bovine Serum (FBS, Sigma–Aldrich, USA) at 37°C and 95% moisture with 5% CO2. Before cell seeding, the specimens were sterilized using UV treatment for 40 min. For each cellular experiment, three replicates were used per group. Tissue culture polystyrene (TPS) was used as a negative control group for all tests.
Cell viability was measured using the standard (ISO 10993-5) 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) assay protocol. Briefly, cell culture with a density of 2.5 × 104 cm2 was seeded on the specimens within a three-well plate and incubated at 37°C and 95% moisture in 5% CO2 for 24 and 48 h. After each interval, MTT at a concentration of 5 mg/ml was added to each well, and the cells were incubated for 2 to 4 h. Upon removal of the MTT solution, the formed formazan crystals were solubilized in isopropanol for 15 min. Absorbance at 570 nm was measured by an ELISA reader (BioTekmicroplate reader, USA) and reported separately for each well.
For cell morphology, mouse fibroblast cells (l929) (with a density of 2.5 × 104 cm2) were cultured on the electrodeposited coatings and incubated in DMEM supplemented with 10% FBS at 37°C and 95% moisture in 5% CO2 for 24 and 48 h. The specimens were gently washed three times with PBS; then, they were fixed with Karnovsky (2.5 vol% glutaraldehyde and 4 vol% paraformaldehyde) solution for 90 min followed by dehydration through a graded series of ethanol. Finally, the specimens were vacuum-dried and gold-coated for SEM study.
Result and discussion
Structural analysis
FT-IR spectra of the PVA, CS, 50CS-50PVA, and 50CS-50PVA/3 wt% GO nanocomposite films are shown in Figure 1(a) and (b). The main spectral bands and their assignments are given. The infrared spectrum of PVA shows an absorption peak at 3271 cm−1, referring to the intermolecular hydrogen bonding and –OH-stretching vibration. The vibrational band observed at 2930 cm−1 is associated with the C–H stretching from alkyl groups. 24 The absorption corresponding to the –C–O stretching occurs at 1096 cm−1. The spectrum of the CS film demonstrates broad band at 3300 cm−1 due to the –OH stretching. The band at 1540 cm−1 is devoted to the –NH bending (amide II) (NH2). 12

(a) FT-IR spectra of CS, PVA and 50CS-50PVA and (b) FT-IR spectra of GO, 50CS-50PVA and 50CS-50PVA/3 wt% GO.
As observed in Figure 1(a), the band intensity at 840 cm−1, assigned to the C–H rocking region of PVA, disappears from the spectra of the CS film and decreases in the 50CS-50PVA blend film. Also, the reduction in the intensity of the band at about 3271 cm−1 occurs in the 50CS-50PVA blend film. This may be due to the –OH stretching vibration of PVA with secondary –NH groups of CS. Further, an increase in the intensity of the absorption band corresponding to the C–H stretching vibration was observed at approximately 2925 cm−1 in the 50CS-50PVA blend film. This indicates that when two or more polymers are blended, the occurrence of physical blends and chemical interactions causes changes in the characteristic peaks of the spectra. These observations reveal good miscibility between PVA and CS. The most likely reason is the formation of intermolecular hydrogen bonds between the –OH and –NH groups in CS and the –OH groups in PVA. 24
The FT-IR spectrum of GO in Figure 1(b) depicts a strong –OH peak at 3300 cm−1, and other C–O functionalities, including C=O (1717 cm−1), C–O (1378 cm−1), and C–O–C (1015 cm−1), are clearly visible. The spectrum also shows a C=C peak at 1624 cm−1 corresponding to the remaining sp2 character. 25 The 50CS-50PVA/3 wt% GO nanocomposite shows a new peak at 1706 cm−1 assigned to carboxyl groups from the GO surface, indicating the presence of GO within the 50CS-50PVA composite. In the curve of 50CS-50PVA/3 wt% GO, the peaks of the characteristic absorption are approximately similar to those in the 50CS-50PVA curve. The FT-IR spectrum of the 50CS-50PVA/3 wt% GO shows a combination of characteristics similar to that of the pristine 50CS-50PVA and GO.
XRD patterns
Figure 2 displays the XRD patterns of CS, PVA, 50 CS-50PVA, and 50CS-50PVA/3 wt% GO nanocomposite films. In the case of CS, prominent diffraction peak was observed at 2θ = 20.3°, presenting the CS crystalline structure. 26 PVA showed a distinct diffraction peak at 2θ = 19.31° and a shoulder at 2θ = 22°, typical of the crystalline PVA. 27 The 50CS-50PVA hybrid polymer included two characteristic peaks attributed to the crystalline phase at 2θ = 11.8° and the amorphous state at 2θ = 19.4°. The CS peak at 2θ = 20.3° vanished in 50CS-50PVA polymer, confirming the good compatibility between CS and PVA. Finally, two generic PVA peaks at 2θ = 19.31° and 2θ = 22° became broad in the 50CS-50PVA polymer blend, denoting the intermolecular interactions between the CS and PVA molecules. The addition of CS clearly resulted in the decrement of the original crystallinity of PVA. The GO characteristic peak in the composite is difficult to detect. The disappearance or weakening of GO peak reveals most of the GO nanosheets to be diffused in the polymeric matrix and overlapped with the CS peak. As a result, GO nanosheets are uniformly distributed into 50CS-50PVA hybrid polymer beneficial for achieving nanocomposite films with improved properties.

XRD patterns of CS, PVA, 50CS-50PVA and 50CS-50PVA/3 wt% GO nanocomposite film.
Morphology studies
The microstructures of 50CS-50PVA and 50CS-50PVA/2 wt% GO films were probed by FE-SEM; the corresponding images are presented in Figure 3. The 50CS-50PVA nanocomposite shows a smooth surface (Figure 3(a)), while 50CS-50PVA/2 wt% GO one exhibits a relatively rough surface (Figure 3(b)). This indicates the proper distribution of GO nanosheets in the 50CS-50PVA matrix and the placement of GO within the polymer matrix.

(a) FE-SEM images of the 50CS-50PVA film and (b) 50CS-50PVA/0.5 wt% GO nanocomposite film.
Rheological behavior
Figure 4 compares the plots of viscosity versus shear rate for the 50CS-50PVA and the nanocomposite films at different GO weight percentages. GO clearly affects the solution viscosity of the nanocomposites, and as the concentration of GO increases in the blend, the absolute viscosity also increases. Yan and Han 23 have reported similar behavior in the case of CS/GO composite. After adding the 2 and 3 wt% GO, the sharp shear thinning behavior is observed, which can be enhanced by dispersion and exfoliation of GO nanosheets in the CS-PVA blend.

The viscosity curves of 50CS-50PVA and nanocomposite films.
Water absorption
The ability to maintain water in films is essential in skin tissue engineering. According to Figure 5, by increasing the amount of GO in the films, the swelling ratio decreases, although it still shows good water absorption. This apparently originates from the fact that GO is well dispersed inside the polymer blend; therefore, the surface becomes more hydrophilic due to the presence of OH functional groups. On the other hand, the reduction in the surface pore size and pore modification by GO insertion is responsible for the decreased water absorption. A similar observation has been reported by El Achaby et al. 28 for CS-polyvinyl pyrrolidone (PVP)/GO composites, where the water absorption dramatically decreases for the composite as compared to the CS-PVP blend. The nature of the matrix and filler generally determines the water absorption of the nanocomposite films, and the reduced swelling ratio is connected with the strong interactions between filler and the polymer blends. Therefore, as the content of GO increases, more potent interpenetrated network forms within the 50CS-50PVP blend matrix that, in turn, impedes the swelling of the films and diminishes the water absorption. 28

Water absorption of 50CS-50PVA and nanocomposite films.
Thermal behavior of polymer and nanocomposite films
TGA is considered as the most important method to study the thermal stability of polymers. 29 The corresponding weight loss curves for CS, PVA, 50CS-50PVA blend, and nanocomposite films are plotted in Figure 6. As can be seen, the CS degradation occurs in a single step in the range of 237 to 275°C. The major weight loss is about 30%, which is attributed to the degradation of the polymer backbone. 30 On the other hand, the thermal behavior of PVA film proceeds in two stages. The first weight-loss step (about 62%) at 180–376°C is due to the rapid decomposition of PVA segments. The second weight-loss step at 380–490°C occurs due to the degradation of the species generated by PVA during its thermal degradation process. 24 In the range of 200–300°C, CS shows higher thermal stability (lower weight loss) as compared to PVA. Consequently, the residual mass of CS and PVA are 40% and 4%, respectively. Moreover, the corresponding residual mass of 50CS-50PVA film lies in between CS and PVA polymers (about 19%). The results suggest that these two polymers are well blended together. 31

The TGA curves of CS, PVA, 50CS-50PVA and nanocomposite films.
As indicated in Figure 6, the degradation stages are shifted toward higher temperatures when GO is loaded into the 50CS-50PVA film since the major decomposition imitation temperature (Ti) for 50CS-50PVA and nanocomposite films is 240°C and 257°C, respectively. It is noted that the thermal stability of nanocomposites is enhanced due to hydrophilic and electrostatic interactions between CS and GO. 32
For the 50CS-50PVA/GO film, the major weight loss takes place at a temperature range between 256 and 465°C. Consequently, a residue of 18% is obtained for 50CS-50PVA against 21.5 and 25% at the same temperatures when the amount of 0.5 and 3 wt% GO is, respectively, added to the polymer matrix. The improvement of nanocomposite thermal stability is mainly attributed to the effect of the physical barrier of GO nanosheets, leading to a decomposition delay of the membrane with temperature.
Viscoelastic behavior of the films
DMTA tests are powerful tools to investigate viscoelastic properties. The main DMTA properties for 50CS-50PVA and nanocomposite films are summarized in Table 1. The storage modulus (E′) and loss modulus (E″) determined at 1 Hz frequency is plotted in Figure 7 as a function of temperature for both 50CS-50PVA and the nanocomposite films. The glassy storage modulus at 30°C increases by introducing GO in the blend (Figure 7(a)). The maximum modulus values for the 50CS-50PVA film, as well as the composite with 0.5 and 3 wt% GO, were 4350, 7040, and 7343 MPa, respectively, showing a 61.8 and 68.8% enhancement for the CS-PVA film at 30°C. A good correlation between the dispersion of GO nanosheets into the polymer matrix and the improvement of viscoelastic properties is observed, as illustrated in Figure 3. As a comparison, Han et al. 23 also have investigated the addition of different GO contents to CS; they report the storages modulus to be about ∼1.8 and ∼2.5 GPa for CS and CS/0.5 wt% GO, respectively, at 25°C. The improved viscoelastic properties are correlated with adding PVA and making 50CS-50PVA/GO composites. As given in Table 1, the storage modulus of the composite increases at higher temperatures, in the rubbery state between 80°C and 140°C. This behavior can be explained in terms of interactions between the GO and the polymer film due to the high surface area and good exfoliation of GO nanosheets in the 50CS-50PVA matrix. The rubbery modulus of the composite containing 0.5 and 3 wt% GO was 132 and 153% higher than that of 50CS-50PVA at 120°C. Indeed, in the rubbery state, the molecular motion and the amplitude of this motion are very high, and the macromolecule is practically in contact with nanosheets. The interaction between GO and 50CS-50PVA, therefore, prevents the reduction of rubbery modulus at high temperatures. On the other hand, good exfoliation of GO nanosheets leads to a better dispersion in 50CS-50PVA film. The glass transition temperature (Tg) values of 50CS-50PVA and nanocomposites collected from Figure 7(a) are listed in Table 1. The addition of 0.5 and 3 wt% GO to the 50CS-50PVA blend increases Tg to about 13.2 and 20.7%, respectively. The mobility of the 50CS-50PVA around GO is reduced due to the interaction between GO and neat polymer.33-35 Hence, the affinity of GO to 50CS-50PVA is expected to reduce the mobility of the polymer matrix around the GO that, in turn, gives rise to higher thermal stability (Figure 6). This effect basically appears around and above Tg due to the limited potential movement of the polymeric matrix. 36
Summary of key DMTA properties.


(a) Storage modulus for 50CS-50PVA and nanocomposite films at 1 Hz frequency and (b) loss modulus for 50CS-50PVA and nanocomposite films at 1 Hz frequency.
The loss modulus (E″) determined at 1 Hz frequency is plotted as a function of temperature for 50CS-50PVA hybrid polymer and the nanocomposite films (Figure 7(b)). This modulus, at the maximum peak, is improved after adding GO nanosheets. It demonstrates the energy that has been changed into heat and can thus be applied as an evaluation of viscous component or unrecoverable oscillation energy dissipated per cycle. Improvement of loss modulus implies the resistance of polymer chains against to viscoelastic deformation and sliding of polymer chains. It is concluded that a satisfactory dispersion of GO would assist in dissipating energy under the viscoelastic deformation of the surrounding hybrid polymer matrix. Also, broadening of nanocomposite films peak could be the result of the interaction of 50CS-50PVA polymer with GO nanosheets.35,36
Cole-Cole diagram
Different approaches have been used to study the viscoelastic properties in the temperature range between glassy and rubbery domains; thus, various models have been proposed to predict these properties. These models generally represent the curve of E″ versus E′; the curve is called the Cole-Cole diagram. The theoretical model must be validated by the experimental data obtained by DMTA tests. After DMTA tests, an asymmetric Cole-Cole diagram is plotted (Figure 8). According to the Perez model, the behavior of polymers can be explained by the bi-parabolic model presented as the following equation:
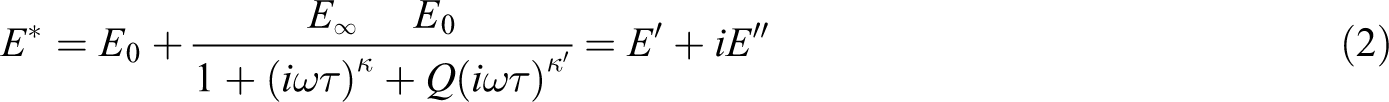

Cole-Cole plots for (a) 50CS-50PVA, (b) 50CS-50PVA/0.5 wt% GO and (c) 50CS-50PVA/3 wt% GO.
with


and

where k, k′, and Q are the constants of this model. ω = 2πf is the angular frequency(f = frequency), and E* is the complex shear modulus; E∞ and E0 are the value of shear modulus at the glassy and rubbery states, respectively. k and k′ values depend on the slope of the tangents at the beginning and the end of the Cole-Cole diagram, Q is a constant related to the maximum value of E″, and τ is the average relaxation time.
A numerical method allows plotting the theoretical Cole-Cole diagram to fit it on the experimental curve. The theoretical curve fits perfectly with the experimental results, signifying the bi-parabolic model’s ability to accurately predict the viscoelastic behavior of 50CS-50PVA and nanocomposite films.
The calculated parameter values of the Cole-Cole diagram (Figure 8) for neat polymer and nanocomposite films are provided in Table 2. From the presented results this table, the following conclusions can be taken:
The calculated parameters of Cole-Cole diagram.

E∞ and E0 values for nanocomposite films are higher than that of 50CS-50PVA film. Therefore, the addition of 0.5 and 3 wt% GO into neat polymer causes the improvement of mechanical properties.
The parameter k shows molecular motion and the intensity of the effects of correlation involved during the expansion of the Somigliana dislocation (smd). 37 The values of k for CS-PVA and also 0.5 and 3 wt% GO nanocomposites are 0.37, 0.29, and 0.28, respectively. Therefore, the addition of GO into the 50CS-50PVA decreases the values of k; such a decrease indicates weaker molecular motion. 37
The parameter k′ accounts for the difficulty with which local shearing occurs; it is the most significant parameter in the Perez model. When k′ decreases, the movement of polymer chains becomes slower. Once a local shearing takes place, the molecular orientation resulting from it makes molecular mobility more difficult. Generally, in a cross-linked system, the value of k′ is low. The addition of GO to neat 50CS-50PVA decreases the values of k′. In other words, in α region, local shearing occurs hardly, and Tg increases. In fact, in α region, where the molecular motion and local shearing are very high, the interaction between functional groups of GO and 50CS-50PVA prevents the movement of polymer chains and the reduction of Tg. The result of the calculated parameters of the Cole-Cole diagrams verifies the nanocomposites’ thermal behavior (Figure 6). The parameters of the Perez model indicate a good correlation between viscoelastic experimental results and the model used. Montazeri has investigated the viscoelastic behavior of epoxy/carbon nanotubes by plotting the Cole-Cole diagram using the results of the DMTA test. According to this research, there is a good agreement between the Perez model and the viscoelastic behavior of the compositespecimen. 36
Antibacterial activity assay
The antibacterial activities of 50 CS-50PVA and 50CS-50PVA/3 wt% GO nanocomposite films were investigated against gram-negative Staphylococcus aureus and gram-positive E. coli using zone of inhibition tests. The results are illustrated in Figure 9. The antibacterial activity of nanocomposite films increased with increasing GO nanosheets against both bacteria. Meanwhile, the antibacterial activity of 50CS-50PVA/3 wt% GO nanocomposite film was higher than that of 50CS-50PVA film. The radius of the inhibition zone attained 13 mm and 18 mm for 50CS-50PVA and 50CS-50PVA/3 wt% GO film, respectively.

Photographs of the inhibition zones of 50CS-50PVA and nanocomposite films against (a) E. coli and (b) S. aureus. The numbers 0, 1, 2 and 3 denote 50CS-50PVA, 1 wt% GO, 2 wt% GO and 3 wt% GO respectively. (c) The histogram of the antibacterial activity of 50CS-50PVA and nanocomposite films against E. coli and S. aureus.
Referring to some investigations, GO has an inhibitory effect on the growth of gram-positive and gram-negative bacteria.5,38 Damage to the cell membrane of bacteria arises once bacteria come into contact with GO; this can be caused by the sharp atomic edges of GO, penetrating the cell membrane and disturbing its totality. Besides, oxidative stress is considered as a necessary member of antimicrobial activity for bacterial cells subjected to GO. 5
In vitro biocompatibility evaluation
Results of MTT assay 50CS-50PVA polymer and 50CS-50PVA/3 wt% GO scaffolds are summarized in Figure 10. It can be seen that the viability of fibroblast cells on the nanocomposite surface is higher than the 50CS-50PVA hybrid polymer (Figure 10(a)). This finding is in line with previous reports; for example, Pandeleet al. 16 have shown that GO has a positive effect on MC3T3-E1 mouse osteoblastic adhesion and proliferation. Also, Ordikhani et al. have reported higher viability for the CS-30GO film than the pristine CS, while the CS-55GO coating exhibits more cytotoxicity. 39
Cell-matrix adhesion with the cooperation of other pathways regulates critical cellular processes, such as migration, proliferation, differentiation, and homeostasis. For this purpose, the optical microscope was used to observe the morphological changes of the cell mass exposed to the films for 48 h. Figure 10 (b) to (d) shows cell growth after 48 h. As seen in the figure, the cells adhered to the surface of the culture medium, and no suspended or dead cells were found. In Figure 10(c), the cells have grown; however, small particles have been observed in the cell culture medium, reducing the number of cells compared to the control sample. This decrease in the number of cells can be attributed to the relative smoothness of the polymeric composition film surface.16,40 There is no apparent difference between the cells grown and adhered to the culture medium of film containing GO and control sample cells (comparing Figure 10(b) and (d)). These findings suggest GO as an excellent supplement for scaffolds in tissue engineering applications.

(a) The quantification of cell proliferation rate on 50CS-50PVA and 50CS-50PVA/3 wt% GO, as revealed by MTT test after 48 h cells cultured on the surface. (b) Light microscopic images 48 hours after cells cultured control, (c) 50CS-50PVA and (d) 50CS-50PVA/3 wt% GO nanocomposite film.
Conclusion
In summary, nanocomposite films of 50CS-50PVA reinforced with GO nanosheets (50CS-50PVA/GO) were successfully prepared at different contents up to 3 wt%. FT-IR spectra and FE-SEM observations confirmed GO to be uniformly dispersed in the matrix and well bonded with the polymers. The viscosity test indicated a sharp shear thinning behavior after adding 2 and 3 wt% GO to the 50CS-50PVA film. The TGA curves showed that when GO was loaded into the 50CS-50PVA film, the degradation stages were shifted toward higher temperatures, thus improving the thermal stability. According to viscoelastic behavior, the storage and rubbery modulus values at 25°C for composite containing 0.5 wt% GO were enhanced by about 54 and 132%, respectively, as compared to the 50CS-50PVA film. The parameters of the Perez model showed a good correlation between viscoelastic experimental results and the theoretical predictions. The result of viscosity, thermal stability, and viscoelastic behavior proved a good dispersion of GO into the polymer matrix.
50CS-50PVA/3 wt% GO film showed acceptable antibacterial activity against both gram-negative E. coli and gram-positive S. aureus using relevant tests.
The value of 3 wt% GO within the 50CS-50PVA film led to a significant increase in the cell proliferation rate. The improved thermal stability, viscoelastic and antibacterial properties, together with good cell proliferation of 50CS-50PVA/3 wt% GO nanocomposite films, are promising for future bioapplications, such as wound dressing materials and skin tissue engineering.
Footnotes
Acknowledgement
The authors are grateful to the Department of Resin and Additives, Institute for Color Science and Technology, for conducting tests.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
