Abstract
The focus of this research is to synthesis different nanocomposites by in situ chemical polymerization. The nanocomposites were prepared by intercalating monomers such as aniline, iodoaniline or combination of aniline and 2-iodoaniline into modified montmorillonite clay. The syntheses were carried out using ammonium persulfate as oxidant with different molar ratios of oxidant to monomer. The resulting nanocomposites were investigated and confirmed by X-ray fluorescence spectroscopy, XRD, UV-visible, FTIR spectroscopy, cyclic voltammetry and transmission electron microscopy techniques. XRD patterns show that, the intensity of diffraction peaks for polymers/(M-Na) composites are lower than that for (M-Cu). This is attributed to the interaction of montmorillonite with polyaniline (PANI), polyiodoaniline (PIANI) and poly (ANI-co-2-IANI) molecular chains. The FTIR and UV-vis spectroscopic results also confirmed the presence of iodine in the nanocomposite. When looking at the yield of the process, highest yield is obtained for the poly (ANI-co-IANI) (80:20 molar ratio) and the lowest yield is for PANI/M-Na composite.
Introduction
Nowadays polymers are extensively used materials in many industrial applications such as electronics, drug and food packaging. It is expected to be exposed to various environmental conditions such as solar radiation, humidity, pollutants and microbial flora that affect their thermo-mechanical properties.1-5 Polyaniline (PANI) is one of the most widely studied conducting polymer with a wide variety of applications.6-9 Due to its high thermal stability and low cost, it is widely used in batteries, sensors, 10 transistors, 11 printed boards, 12 electromagnetic interference (EMI) shielding and flat panels. 13
Polymer composites incorporated with nanofiller is of great interest due to its enhanced properties.14-16 Polymer nanocomposites can be prepared by mixing technique,9,17,18 injection molding,19,20 compression molding,21,22 stir casting,23,24 freeze drying 25 and continuous lamination process. 26 Nano-PANI and its derivative can be synthesized by emulsion polymerization techniques 27 and phase transfer polymerization. 28 PANI can also synthesize in an inorganic host with the help of an oxidant.29,30 Different types of clay are also employed in the synthesis of polymer nanocomposites. Among them, expandable-layered clay silicates are most commonly used clay. Being maghnite (Mag), Algerian montmorillonite (MMT) sheet silicate clay possess very high cation exchange property. 31
Composites of several conducting polymers with montmorillonite were synthesized under different conditions.22-35 Lee et al. 36 synthesized intercalated PANI/MMT (sodium treated) nanocomposites with various PANI fractions. Their study shows clearly that, the physical interaction between the clay and PANI ameliorated the electrical conductivity and thermal stability of the composites. Moreover, they are consistently related to the structure of the nanocomposites as found from X-ray diffraction analysis. Çelik and Önal 37 also found that, the intercalated PANI/MMT nanocomposites have high thermal stability because of the intercalation of PANI between clay layers, which was confirmed by the increase in the interlayer spacing by about 0.53 nm.
Bereket et al. 38 have synthesized polyaniline (PANI), poly (2-iodoaniline) (PIANI), and poly(aniline-co-2-iodoaniline) Poly (ANI-co-IANI) by electrodeposition of the monomer aniline and iodoaniline. The polymer formation was confirmed through FTIR and UV-vis spectroscopy analysis. Moreover, a blue shift in the spectrum was observed due to the presence of iodine. Salavagione et al. 39 studied the effect of intercalated cation on the properties of poly (O-methylaniline)/Maghnite clay nanocomposites. TEM, FTIR and GC-MS analysis were performed, and changes were observed in the properties of nanocomposite depending on the interaction of cations between the clay layers. Also, the bands corresponding to montmorillonite vibrations were appeared around 1040 and 925 cm−1. Similar studies were performed by Toumi et al. 40 on 2-aminobenzene sulfonic acid with aniline and montmorillonite (sodium treated). They analysed the effect of intercalated cation exchange using XRD, FTIR and UV-vis spectroscopy analysis. It is observed that, there is an increase of d-spacing was demonstrated to be shifted due to the intercalation of the polymer between the M-Cu layers. Do Nascimento et al. 34 have conducted a spectroscopic study on aniline polymerization into montmorillonite clay using UV, XRD and Raman spectroscopy techniques. The polymerization was confirmed using the UV bands obtained around 620 and 670 nm and Raman spectrum at 632.8 nm.
However, in the studies described above, only sodium treated montmorillonite was employed for the synthesis of polymer nanocomposites. Therefore, the use of modified montmorillonite with different cations is expected to affect positively the properties of the nanocomposites and merits research efforts. Moreover, the prepared nanocomposite can be utilized in different mechatronic applications.
The main objective of this work is to synthesize nanocomposites of aniline and iodoaniline with a natural montmorillonite (Maghnite) clay modified with two inorganic cations (i.e., Na+ and Cu2+). The physical properties of the developed nanocomposites and the polymers extracted from them are also studied. The effect of the intercalated cation-exchanged on the nanocomposites structure of the modified montmorillonite as a dispersant is examined. The in-situ polymerization method has been performed with aniline and it has been extended to 2-iodoaniline (IANI), which is an N-substituted derivative of aniline. Several techniques have been used to analyse the nanocomposites, such as X-ray fluorescence spectroscopy (XRF), X-ray diffraction (XRD), UV-visible, FTIR spectroscopy, cyclic voltammetry and transmission electron microscopy techniques.
Experimental
Materials
Aniline (ANI, 99%) and 2-iodoaniline (IANI, 98%) were supplied by Aldrich Chemical Company. Figure 1 shows the chemical structure of aniline and 2-iodoaniline. Ammonium persulfate (NH4)2S2O8 solution was supplied by Merck. CuSO4 aqueous solution was applied to Maghnite-Cu (M-Cu) to prepare the montmorillonite modified by copper (M-Cu). NaCl aqueous solution was applied to Maghnite-Na (M-Na) to prepare the montmorillonite modified by sodium–montmorillonite clay (named as Maghnite) and obtained from Tlemcen (Algeria). The water employed for the preparation of the solutions was obtained from an Elga Labwater Purelab Ultra system.
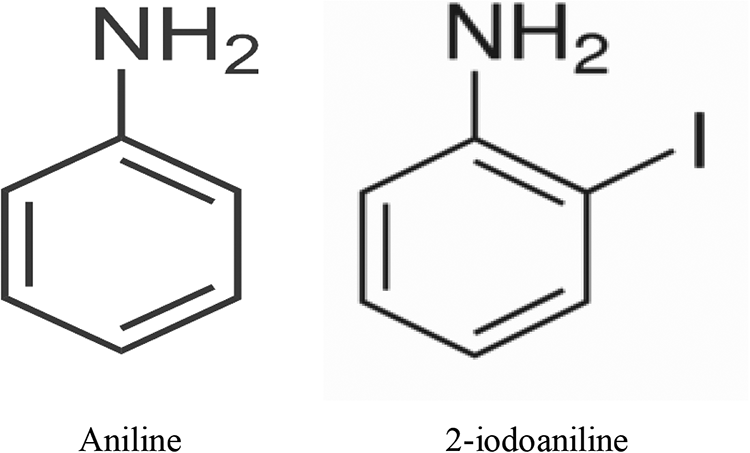
Chemical structure of aniline and 2-iodoaniline.
Methods
Preparation of Maghnite (M-Na) and (M-Cu)
In its natural state, clay contains a number of impurities such as silica, in different forms (e.g., quartz, cristobalite), oxides and hydroxides of iron, organic matter, carbonates etc. These impurities are easily identified from their chemical composition by chemical analysis or by their type of crystallization by using X-ray fluorescence (XRF). To improve operating reliability of the results and to avoid secondary interactions an important purification procedure was implemented.
In order to purify the clay, two different treatments were conducted. These are (1) dispersion followed by sedimentation in water, and (2) removal of supernatant by chemical treatment.
First, the clay samples were washed with distilled water to remove impurities. The raw-montmorillonite (10 g) was crushed for 20 min using a Prolabo ceramic ball grinder. The samples were then dried at 149.85°C for 24 h and stored in tightly stoppered glass bottles for later use (M-Na). To obtain M-Na with copper intercalated (M-Cu), the M-Na was dispersed into a 1 M CuSO4 solution and stirred for 24 h. Then the solid was recovered by centrifugation and washed with abundant water. The absence of chloride was confirmed using silver nitrate41,42 and the absence of sulphates is confirmed using barium nitrate.43,44 The products were dried at 110°C, overnight and their composition was measured by X-ray fluorescence, obtaining the data is given Table 1.
Elementary compositions of copper exchanged sample sodium–montmorillonite.

Synthesis of nanocomposites
Three different nanocomposites based on aniline, 2-iodoaniline or aniline and 2-iodoaniline were prepared. To obtain the nanocomposites by monomer, 0.022 M of aniline or 2-iodoaniline or aniline and 2-iodoaniline (monomers) were added to 0.5 g of the M-Cu (to get Maghnite-Cu/polymer nanocomposite) or M-Na (to get Maghnite-Na/Polymer nanocomposite). The mixture of Maghnite with monomers was stirred at room temperature for 1 hour. The chemical polymerization was triggered by slowly adding 100 ml of 0.5 M ammonium persulfate (NH4)2S2O8 in solution to the mixture. The oxidant amount was calculated in order to obtain a molar ratio equal to 1:1.
The resulting precipitate was collected on a filter which was washed several times with distilled water. The nanocomposites PIANI/M-Cu powder was dried under vacuum at 50°C. Finally, the dried powder of nanocomposites was ground to fine powder and preserved. The reaction was carried out under magnetic stirring for 24 hours at room temperature and then the solid product was filtered under vacuum and washed with distilled water to remove all traces of oxidant. All the materials were dried at 60°C to obtain the polymers which remained in the catalyst layers. The same procedure was used for the copolymer synthesis, keeping always the mass ratio M-Cu/monomers (aniline and/or 2-iodoaniline were added with molar ratios: 20/80, 50/50 and 80/20, 60/40, 40/60 to a total concentration of (0.22 M.) at 0.5 g (see Figure 2). The obtained yield was calculated for all the prepared composites. UV-Visible spectroscopy, XRD and FTIR analysis were carried out to verify the formation of intercalated MMT clay-polymer composites. However, the results were found identical for all molar ratio of the same monomer. Hence, representative results are presented in each section of results and discussion

Synthesis of nanocomposites PIANI/M-Cu via in-situ chemical polymerization.
Results and discussion
X-ray fluorescence
X-ray fluorescence spectrometry (XRF FX or X-ray fluorescence) is an elemental analysis technique that allows to characterize the chemical elements present in a sample and to quantify these elements. The X-ray fluorescence (XRF) analysis is based on the excitation spectral line X. The composition of M-Na and M-Cu are found to be almost similar except the presence of sodium in M-Na is replaced by Copper in M-Cu.
XRD spectroscopy
X-ray diffraction was performed using Bruker CCD-Apex equipment with an X-ray generator (Cu Ka and Ni filter) operated at 40 kV and 40 mA. The scanning speed and the step size were 0.08°/min and 0.05°, respectively to analyse the d-spacing between the montmorillonite sheets. Figure 3 shows the XRD spectrum the clay and the polymer /montmorillonite nanocomposites. Table 2 presents the calculated d-spacing values (d001) between the montmorillonite sheets using Bragg equation d = 2π/q, where q is the magnitude of scattering vector defined as q = (4π/λ) sin (θ); λ is the X-ray wavelength, and 2θ is the scattering angle. 45

XRD spectra of (a) M-Na nanocomposites and (b) M-Cu nanocomposites.
Maximum peaks and d-spacing of the nanocomposites intercalated into sodium montmorillonite and copper montmorillonite.
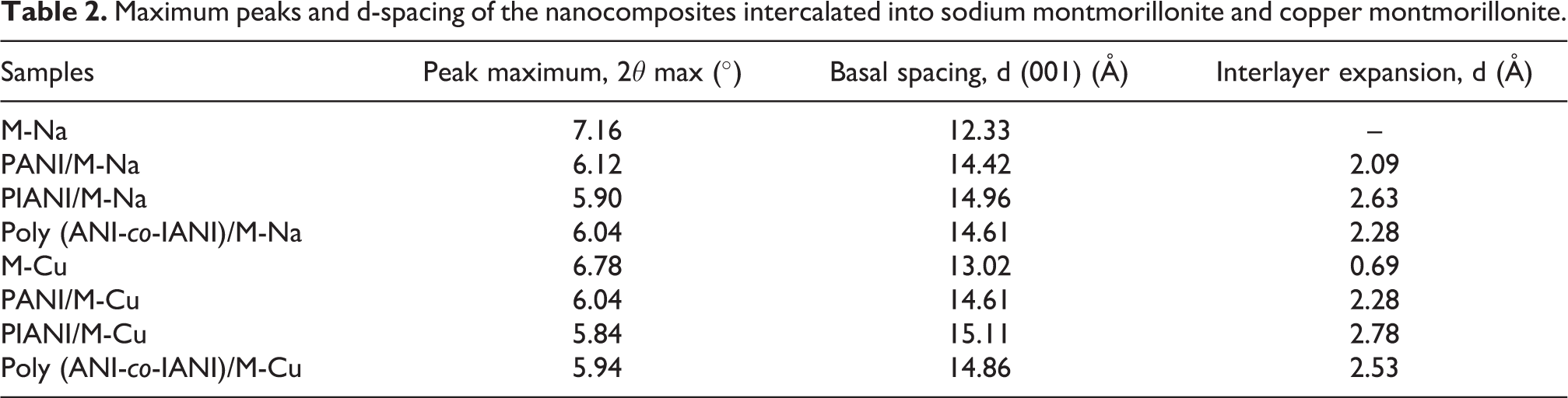
The change in the interlayer spacing of maghnite after ion exchanging Na+ with Cu++ was found to increase from 12.33 Å in the M-Na to 13.02 Å in the M-Cu. As compared with the M-Na and M-Cu, it can be seen that the diffraction peak of PANI/M-Na and PANI/M-Cu is shifted to the lower degrees due to the intercalation of the polymer between the clay layers. The intercalation of PANI/M-Na and PANI/M-Cu leads to an increase in the basal distances increase from 14.42 A to 14.61 A respectively. We can remark also from the diffraction peaks related to Poly (ANI-co-IANI)/M-Na and Poly (ANI-co-IANI)/M-Cu, which have decreased at a lower angle of 2θ = 6.04, corresponds to the d-spacing of 14.61 Å and 2θ = 5.94, corresponds to the d-spacing of 14.86 Å respectively. The same effect has been seen by many researchers in different polymer/clay composites. 46 For PIANI/M-Na and PIANI/M-Cu interlayer spacing is limited due to the presence of iodine in chain polymers (Table 2).
UV-vis absorption
The absorbance measurements of the homo- and co-polymer were dissolved in (DMSO) and UV-vis spectra were recorded on a SPECORD 200 plus spectrophotometer in the range of 200–800 nm. Figure 4 shows the UV-vis absorbance for PANI, PIANI and Poly (ANI-co-IANI) solutions. It can be noticed that, there are two broad absorption bands located between 300–450 nm (band 1) and 525–625 nm (band 2). Band 1 is assigned to π–π* transitions which denotes the bandgap of the polymer whereas band 2 is due to the n–π* transition of benzenoid to quinoid attributed to middle oxidation state.47-49 However, a blue shift has been observed from the actual band positions of PANI. This is due to the iodine incorporation into the polymer composite. Many researchers have reported this effect.49-51 These spectra also suggest that the synthesized materials are PIANI and Poly (ANI-co-IANI). Moreover, the bandgap of the composites was extracted using Tauc plot. For PANI/MCu, the bandgap is found to be 2.85 eV where as for PIANI/MCu and Poly (ANI-co-IANI)/MCu it was reduced to 2.8 eV due to the presence of iodine.

(a, b) UV-Vis spectra of MMT, PANI/M-Na, PIANI/M-Na, Poly (ANI-co-IANI)/M-Na, PANI/M-Cu, PIANI/M-Cu and Poly (ANI-co-IANI)/M-Cu nanocomposites.
FTIR spectra
The Fourier Transform Infrared (FTIR) spectra were recorded on a crystal diamond reflection spectrometer, in the range 500–4500 cm−1. The IR spectra of the chemically prepared samples are shown in Figure 5. The bands and associated bonds are listed in the Table. 3.

(a, b) FTIR absorption spectra of the Mag-Na, Mag-Cu, PANI/M-Na, PIANI/M-Na, Poly (ANI-co-IANI)/M-Na, PANI/M-Cu, PIANI/M-Cu and Poly (ANI-co-IANI)/M-Cu nanocomposites.
It can be noticed that there is no change for the peak at 1460 cm−1 which represents the presence of benzenoid units. Moreover, two peaks at 1300–1200 cm−1 and 1025 cm−1 represented the presence of Iodine the composite structure. This confirms the incorporation of Iodine into the PIANI/M-Na, Poly (ANI-co-IANI) M-Na, PIANI/M-Cu and Poly (ANI-co-IANI) M-Cu composites.
Electrochemical response
Electrochemical behaviour of nanocomposites synthesized by chemical method has been tested using cyclic voltammetry studies in 1 M HClO4 solution at a scan rate of 50 mV s−1 (Figure 6). Cyclic voltammograms of PANI/M-Cu show two peaks anodic at 0.27 V and 0.49 V. The cathodic response electrochemical show two peaks at 0.32 V and 0.62 V. The redox process is due to the oxidation of the benzenoid form of polyaniline and the second one to the oxidation of the quinoid form of polyaniline. The voltammetry profiles for PIANI/M-Cu show one anodic peak at 0.65 V. And the cathodic branch display one peak centred at 0.42 V. However, in the case of poly (ANI-co-IANI)/M-Cu two anodic peaks are observed at 0.28 V and 0.63 V. And the cathodic response display two intensive peaks centred at 0.25 V, 0.61 V. The voltammetric profiles for PANI and poly (ANI-co-IANI) from M-Na, show two redox pairs were clearly observed at (0.44/0.25, 0.73/0.35 V) and (0.48/0.32, 0.68/0.64 V), respectively. In the case of PIANI/M-Na one redox process centred is observed at 0.49/0.32 V.
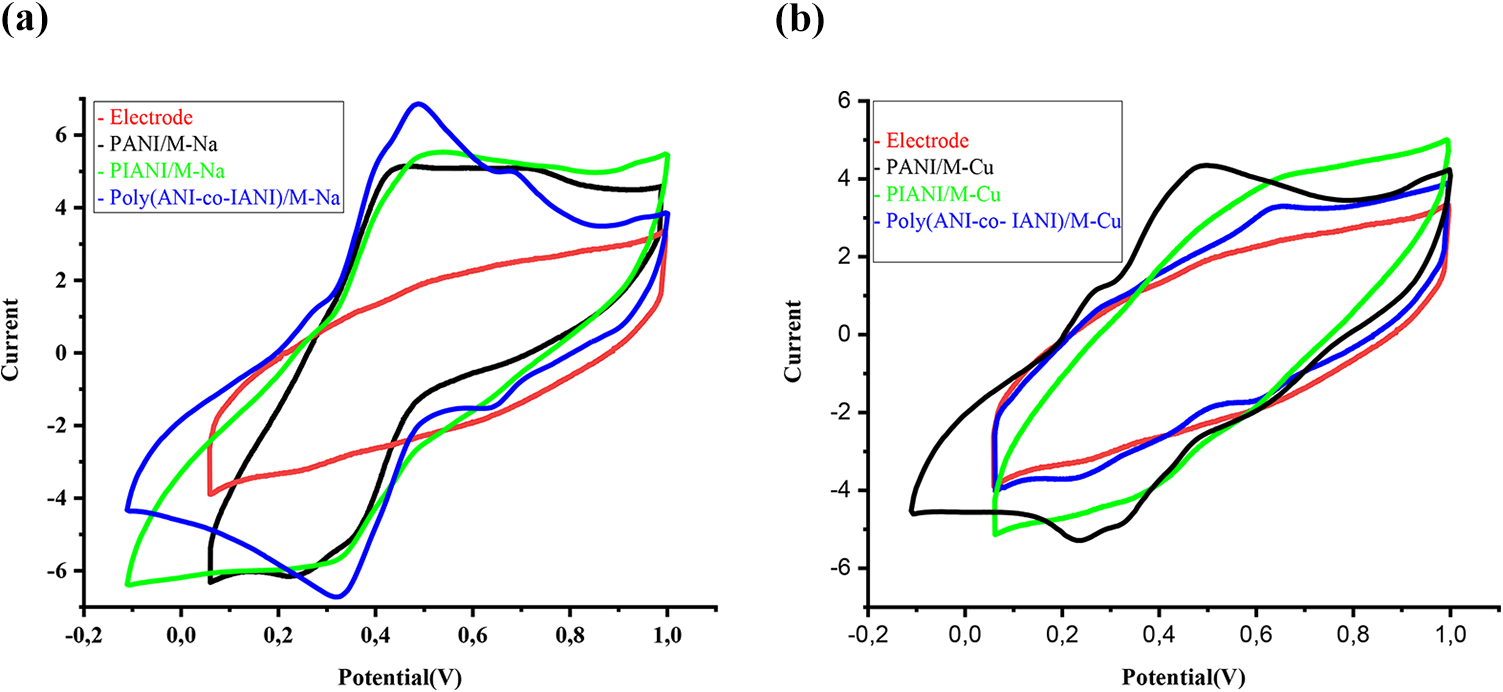
(a, b) Cyclic voltammograms recorded for PANI/M-Na, PIANI/M-Na, Poly (ANI-co-IANI)/M-Na, PANI/M-Cu, PIANI/M-Cu and Poly (ANI-co-IANI)/M-Cu nanocomposites.
It is interesting to note that each polymer structure within different layered host’s present completely different cyclic voltammograms; these results confirmed that the chain in the backbone polymers prepared with montmorillonites is different. In general, all polymer molecules within the intra-lamella of (M-Cu and M-Na) show electroactivity clearly.
Transmission electron micrographs (TEM)
The TEM images of pure M-Cu, P (ANI-co-IANI)/M-Cu and M-Na, P (ANI-co-IANI)/M-Na nanocomposites are shown in Figure 7. Figures of M-Na and M-Cu show sheet-like plates of the montmorillonite. In figure of M-Cu, P(ANI-co-IANI)/M-Cu and M-Na, P(ANI-co-IANI)/M-Na, it can be seen that the clay layers are dispersed uniformly and homogeneously in the polymer matrix and the interlayer spacing of M-Na and M-Cu are expanded, which indicates to the intercalated morphology. This morphological pattern was also observed by other researchers.33,60 These TEM results were in good agreement with the results of the XRD patterns.

TEM images of Mag-Na, Mag-Cu, poly (ANI-co-IANI)/Mag-Na and poly (ANI-co-IANI)/Mag-Cu nanocomposites.
Yield (%) of nanocomposites synthesized by M/Cu and M/Na
The reactions yields were evaluated for all composites and presented in Table 4. The yields have mass values between (48.97–75.39%) where the highest and lowest yield are obtained for poly (ANI-co-IANI) at 80:20 molar ratio and PIANI/M-Na respectively. The mass values do not depend on the polymer composition.
The yield (%) of nanocomposites synthesized by M/Cu-M/Na.

PANI: polyaniline; PIANI: poly (2-iodoaniline).
Conclusion
In this paper, nanocomposites using in-situ chemical polymerization were synthesized successfully. The composition of M-Na and M-Cu clays were verified using XRF analysis. The nanocomposites were synthesized by intercalating modified montmorillonite (MMT) clay into PANI, PIANI and Poly (ANI-co-IANI). XRD and TEM observations suggest that polymers are intercalated into the layers of the clay for Cu-M and Na-M samples. The FTIR and UV results confirmed that, there is a strong interaction between polymer chains and the montmorillonite. Good electrochemical response was observed for the polymer grown into the montmorillonites in which the cyclic voltammetry indicate the polymerization into M-Cu and M-Na is electroactive .When comparing the yield of reaction, poly (ANI-co-IANI) with 80:20 molar ratio has obtained a maximum of 75.39% and the lowest yield of 48.97was reported for PANI/M-Na composite.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

