Abstract
Superabsorbent polymers (SAPs) were synthesized by aqueous solution polymerization of carboxymethyl cellulose (CMC)/acrylic acid (AAc) and polyvinyl alcohol (PVA)/AAc blend using benzoyl peroxide as an initiator. SAPs were synthesized by a facile method under mild conditions. A comparative study was done between CMC/AAc as a biodegradable superabsorbent and PVA/AAc copolymer. The effects of cross-linking, reaction time, temperature, and initiator molar ratio on water absorbency were studied herein. The highest swelling capacity was obtained for CMC/AAc biodegradable superabsorbent composite. The water absorption capacity of SAPs increased with increases in benzoyl peroxide content, immersion time, and reaction temperature of polymerization. The gel fraction of SAPs decreased with increases in the amounts of N,N′-methylenebisacrylamide as a cross-linker. Fourier-transform infrared spectroscopy was used to examine the molecular interactions. The morphology and structure of superabsorbent composites were investigated by scanning electron microscopy.
Keywords
Introduction
Superabsorbent polymers (SAPs) can be described as having a three-dimensional, hydrophilic network systems and a moderate degree of cross-linking with the ability to absorb water considerably greater than their dry mass. 1 -3 The SAPs contain carboxylic acid, partially neutralized carboxyl groups, carboxylate salt, and carboxamide. They draw surrounding water into the network by diffusion while the SAPs backbone chains maintain their structure due to cross-linking, so the polymer network expands and swells. Also, the covalent cross-links can prevent the polymer network from being dissolved in water. They are able to absorb water without dissolving, exceeding 1000- to 1500-fold their dry weight. 4 -11
Due to such significant properties, the SAPs are produced for different applications. For example, they are used in disposable baby diapers, 12,13 agriculture as soil amendments, 14,15 coal dewatering, 16 controlled release of drugs as carriers, 17,18 cosmetics 19 and absorbent pads, and wastewater treatment. 20,21
The structure of polyacrylic acid contains an ionizable group on each repeat unit (–COOH). These chains are then cross-linked. SAPs are prepared from acrylic acid (AAc) and a cross-linker by suspension or solution polymerization. 22 -24 The type and amount of cross-linker control both the stretchability and swelling capacity.
SAPs are classified into three types, namely semi-artificial (cellulosic derivatives), synthetic polymers, and natural (polysaccharide derivatives). Demand has arisen for natural polymer-based superabsorbent materials that are derived from feedstock such as cellulose. 25 -28
Compared to synthetic petroleum-based polymers, these polymers have the advantages of sustainability, high hydrophilicity, nontoxic base components, and biodegradation properties. Natural polymers can be made from carboxymethyl cellulose (CMC), which has a naturally occurring polysaccharide cellulose base. 29
Feng Cheng et al. reported that the mechanical properties of polyvinyl alcohol (PVA) were improved by incorporating PVA-grafted graphene oxide. 30
Ali Akl et al. synthesized PVA core and poly(N-isopropylacrylamide/acrylic acid) shell. 31
In this study, biodegradable CMC was chosen instead of graphene oxide or other chemical component. The SAPs were synthesized by grafting polyacrylic acid onto CMC and PVA via solution copolymerization.
Synthesized superabsorbents were also compared in terms of the polymerization conditions along with investigating their effects on absorbency. Fourier-transform infrared (FTIR) spectra were used to characterize successful graft copolymerization. The morphology and structure of superabsorbent composites were investigated by scanning electron microscopy (SEM).
Experimental
Materials
AAc of 99.8% purity, benzoyl peroxide, CMC (AR grade), sodium hydroxide, and N,N′-methylenebisacrylamide (MBA) (AR grade) were purchased from Aldrich (Singapore), E-Merck, Merck KGaA Corporate (Frankfurter straβe, Germany), and Fluka (Buchs SG, Switzerland), respectively, the two latter of which were used in alkaline hydrolysis and as cross-linker, respectively. PVA and reagent grade ethanol were procured from Aldrich Chemical Co. and E-Merck, respectively.
Preparation of superabsorbents
As shown in Figure 1, AAc (7.2 g) was dissolved in 15 ml distilled water/ethanol solution and then neutralized with sodium hydroxide solution (2 M) in a three-neck round bottom flask reactor. Then, benzoyl peroxide, CMC (0.01 g), and MBA (0.15 g) as cross-linkers were added and stirring was continued under reflux condition to dissolve them. Polymerization reaction was carried out for 15 min while keeping the reaction temperature at 90°C. Finally, the resulting product was washed several times with distilled water, and dried at 60°C to a constant weight, followed by then screening.

Schematic representation of synthesis of CMC/AAc SAP.
The reaction was repeated for AAc (7.2 g), benzoyl peroxide, MBA (0.15 g), and PVA (0.01 g) at the same condition. Polymerization reaction was carried out for 25 min.
Water absorbency measurement
A weighed quantity of the SAPs was immersed in distilled water at room temperature (approximately 27°C) to reach the swelling equilibrium. Swollen samples were then separated by screening and weighed after being drained on a sieve for 15 min. The experiment was repeated for each sample, to determine its average weight. The swelling ratio was calculated using the following equation
where W2 and W1 are the weights of the water-swollen sample and the dry sample, respectively.
FTIR spectrum and SEM analysis
Infrared absorption spectra of samples were studied using IR Prestige-21 Shimadzu (Shimadzu Co., Japan). The SAPs were mixed with KBr and pressed to a plate for spectrometric measurements from 4000 cm−1 to 400 cm−1 at room temperature. A background spectrum containing no sample was subtracted from all spectra. The SEM images were recorded on a Zeiss Supra 40 (Germany) at an acceleration voltage of 5 kV.
Results and discussion
FTIR spectroscopic analysis
FTIR spectroscopic analysis was used to determine the nature of the bond formation. Pure CMC had absorption bands related to O–H stretching at 3498 cm−1, carbonyl stretching in the anhydroglucose unit of the cellulose at 1591 cm−1, –CH2– stretching at 2951 cm−1, C–OH at 1408 cm−1, and C=O stretching from an asymmetric oxygen bridge at 1178 cm−1. As shown in Figure 2, the absorption bands of CMC were obviously weakened after the reaction in the CMC/AAc blend hydrogel. In spectrum (b), the new band at 1759 cm−1 (carbonyl stretching of –COOH groups) showed the carbonyl group of AAc, which was absent in spectrum (a). The band shift apparent at 1599 cm−1 (at 1591 cm−1) for CMC/AAc SAPs indicates the formation of SAPs.

Infrared spectroscopy of (a) pure CMC powder and (b) CMC/AAc SAP.
Grafting of AAc and PVA was confirmed by FTIR studies. In pure PVA (Figure 3(a)), characteristic peaks appear at 3379 and 2943 cm−1, respectively, for the O–H and C–H stretching vibrations, whereas the bands appearing at 1441 and 1097 cm−1 are due to C–H and O–H bending vibrations, respectively. Appearance of a new band at 1724 cm−1 for PVA/AAc along with other bands confirms the formation of a graft copolymer.
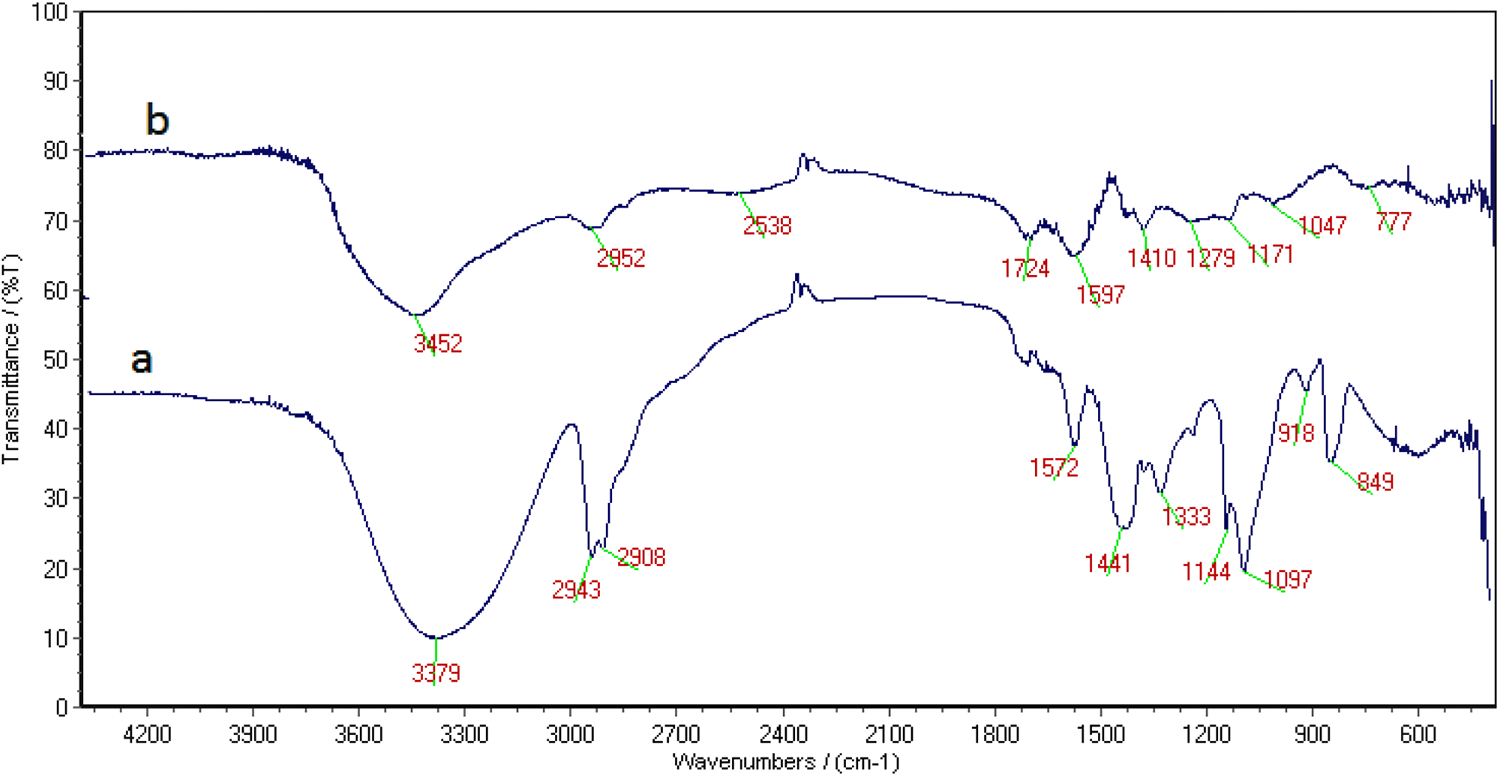
Infrared spectroscopy of (a) pure PVA powder and (b) PVA/AAc SAP.
SEM analysis
Two samples were chosen with different amounts of initiators, and their morphologies were examined by SEM. Figure 4 shows morphological images of CMC/AAc with initiator amounts of (a) 0.1 g and (b) 0.7 g. The result showed that the morphology in Figure 4(a) was less porous and thus more compact than that in Figure 4(b) which confirms the results of swelling analysis showing that sample (a) has less value than the other. The initiator, therefore, affected both the length of the polymer chains and the morphology and molecule weight. Excessive overgrowth of the initiator causes very high branching as a result of small cavities.

SEM of CMC/AAc copolymer with initiator amounts of (a) 0.1 g and (b) 0.7 g.
Effect of initiator contents on the water absorbency
With the aim of investigating the effect of initiator contents on the water absorbency, samples obtained from five different amounts of benzoyl peroxide were subjected to equilibrium swelling measurements in distilled water. Figure 5 shows that the water absorbency for samples increased with the increasing benzoyl peroxide content. The initiator, therefore, affected the length of the polymer chains and influenced the molecule weight. The increase of initiator amounts will shorten the chains, thus the produced SAPs will be less hard. As shown in Figure 5(c), maximum value was obtained from the CMC/AAc SAP sample.

Effect of initiator content on the water absorbency in distilled water at room temperature: (a) polyacrylic acid, (b) PVA/AAc copolymer, and (c) CMC/AAc copolymer.
Effect of cross-linker content on the water absorbency
Figure 6 shows the effects of cross-linker dosage on the swelling capacity of samples. It is seen that 0.15 g of MBA provided the highest swelling capacity, with an overdose giving much poorer swelling. Since the MBA served as a cross-linker, it increased the cross-linking, which later decreased the ability of water swelling. The increase of network density also led to increased physical entanglements and decreased swelling property.

Effect of MBA content on the water absorbency of (a) polyacrylic acid, (b) PVA/AAc SAP, and (c) CMC/AAc SAP at room temperature.
Effect of reaction temperature on the water absorbency
The influence of the reaction temperature on the swelling capacity was investigated at varying range of 25–90°C and the results are depicted in Figure 7. The highest water swelling capacity was observed at 75°C. Since a low temperature reduces the activity of radicals, slows down the decomposition rate of the initiator agent, and impedes the delivery of polymerization reaction chain, the prepared SAP with a low reaction temperature shows relatively low molecular weight and water absorbency. As the reaction temperature increased, the conversion ratio of the monomer raised, leading to increased ability for the absorption of water.
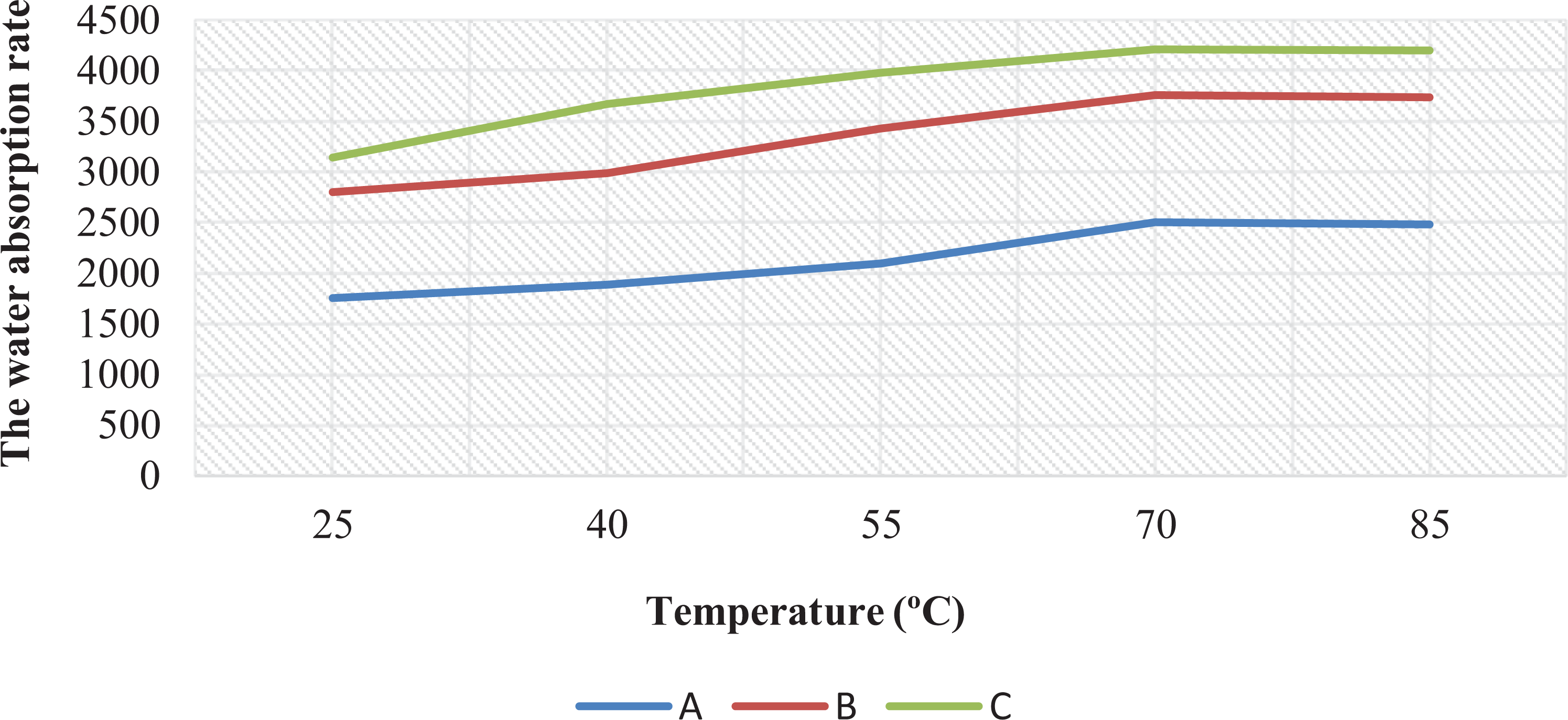
Effect of temperature on the water absorption of (a) polyacrylic acid, (b) PVA/AAc, and (c) CMC/AAc SAPs.
Conclusions
In summary, a series of superabsorbent composites was prepared by graft copolymerization based on AAc to investigate the effect of reaction variables on the swelling capacity. The highest swelling capacity was obtained for CMC/AAc superabsorbent composite. It was found that the contents of various factors, such as cross-linker amounts, temperature, and initiator content, had significant influences on the water absorbency. The water absorbency of samples increased with increasing benzoyl peroxide content as initiator and temperature. The increased cross-linking also decreased the ability of water swelling. The successful synthesis of SAPs was confirmed by FTIR spectra through comparing absorption peaks and the morphology of superabsorbent composites by SEM.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Islamic Azad University, Qom Branch, Iran.

