Abstract
A new series of nanocomposite polymer electrolytes (NCPEs) was prepared using poly(vinylidene fluoride–co–hexafluoro propylene) P(VdF-HFP) as polymer, magnesium triflate (MgTr) as salt and nano-sized barium titanate (BaTiO3) (<100 nm) as nanofiller via traditional solution casting technique. Decrease in crystalline nature of the samples due to the incorporation of nano BaTiO3 was revealed through X-ray diffraction (XRD) analysis. From AC impedance spectroscopy, maximum conductivity of 4.11 × 10−4 Scm−1 was attained for the addition of 6 wt% of nano BaTiO3 to the P(VdF-HFP)/MgTr matrix. Dielectric studies were found to be in accordance with the ionic conductivity studies. For the most highly conducting sample, a greater number of mountain valley pattern was observed from Atomic Force Microscopy (AFM) analysis. Thermal stability of the sample, P(VdF-HFP)/MgTr/BaTiO3 (6 wt%) (which possessed maximum ionic conductivity) was observed through TG/differential thermal analysis studies. All these results suggested that these materials are favorable and find application in practical electrochemical devices.
Keywords
Introduction
Solid polymer electrolytes (SPEs) have been widely used because they possess unique combined properties such as ionic conductivity and good mechanical strength leads to use in solid-state devices including batteries, electrochromic displays, supercapacitors, and smart windows1, 2 Up-to-date researchers focused their attention in developing rechargeable batteries based on lithium ion, since it possess high specific energy capacity and cycling stability. 3 But, due to its high cost and less abundance in nature, it could not be used for widespread applications. 4 Hence, a search for alternative resources instead of Li ion is needed.
In the recent past, magnesium ion–based polymer electrolyte system has attained a better interest because the performance capabilities shown by them are similar to that of lithium based rechargeable batteries. 5 Also, the secondary magnesium batteries are quite fascinating compared with lithium batteries because of the following advantages: (i) the ionic radius of Li+ is 68 pm, whereas the ionic radius Mg2+ is 65 pm. Therefore, Li+ can be replaced by Mg2+; 6 (ii) the magnesium metal is more stable to oxygen and humid atmospheres, and thus, it can be handled more easily; (iii) plenty in nature; (iv) lower cost than that of lithium; (v) considerable negative electrode potential; (vi) the charge density is high; (vii) melting point is high (649 °C); and (vii) less toxic, and hence, it is easy to handle and dispose.7–10 But, the development of polymer electrolyte for the rechargeable magnesium batteries is still in search. Due to the above said appealing characteristics of magnesium, magnesium triflate (MgTr) is chosen as an electrolyte for the present work.
For the present work, poly(vinylidene fluoride–co–hexafluoro propylene) (P(Vdf-HFP)) is chosen as a polymer host, as it possess less crystallinity when compared to PVdF. 11 In addition to this, the value of dielectric constant of P(Vdf-HFP) is (ε = 8.4); hence, it could solvate more salt which result in an augmentation of ionic conductivity of the polymer electrolyte. 12
Earlier investigators suggested that the addition of nanofillers such as SrTiO3, TiO2, MgO, yttria-stabilized zirconia, sepolite, and BaTiO3 to polymer electrolytes can improve (i) its ionic conductivity and (ii) its mechanical stability.13,14,15 Based on these facts, to enhance the ionic conductivity of polymer electrolyte, nano-sized barium titanate (BaTiO3) is used here. The filler, BaTiO3 is used as a material for capacitor applications, as it has a high dielectric constant (103–105). Further, it possesses ferroelectric domain. The presence of ferroelectric domain and high dielectric nature of BaTiO3 facilitate the electrolyte dissociation into charged species. Also, more amorphous phase is produced; therefore, the conductivity of the samples can be boosted. 15
For the current work, an effort has been made to prepare P(Vdf-HFP)/MgTr samples with different concentrations of nano BaTiO3, and the influence of content of the BaTiO3 filler on the ionic conductivity, structural, dielectric, morphological, and thermal properties are examined which have not been yet explored. Thus, this work paves a way for more work in magnesium based polymer electrolytes in near future.
Experimental techniques
Materials
Polymer, poly(vinylidene fluoride–hexafluoro propylene) (P(VdF-HFP)) (Mw ∼ 130,000 gmol−1) and the electrolyte, magnesium triflate (MgTr) were purchased from Sigma-Aldrich, USA. Nano-sized barium titanate (BaTiO3) (<100 nm) was obtained from Alfa Aeson, India. Acetonitrile was used as a solvent, and the same was procured from Merck, India. These chemicals were used without any further purification.
Sample preparation technique
The samples P(VdF-HFP)/MgTr with different concentrations of BaTiO3(2 wt% to 10 wt%) in steps of 2 wt% were synthesized by conventional solution casting technique. The ratio of polymer and salt was used here was P(VdF-HFP) (70 wt%)/MgTr (30 wt%) as it was found to be an optimized one for getting stable SPEs. The polymer and the salt were dissolved by adding in sequence to the solvent acetonitrile and stirred well for 24 h. Now, the nanofiller BaTiO3 (<100 nm) was added to the polymer electrolyte solution and stirred well until the solution was homogenously mixed. Then, these NCPE solutions were poured in a Petri dish and kept inside a vacuum oven at 60 °C for 24 h. Using this technique, mechanically stable and free standing films were harvested. The sample preparation is illustrated in Figure 1. Schematic representation of sample preparation.
Characterization and measurement
The prepared samples were subjected to AC impedance measurement to determine ionic conductivity. AC impedance measurement was carried out using PSM 1735 impedance analyzer which was purchased from Newton’s 4th Ltd, UK. Here in, we have used two stainless steel (SS) blocking electrodes. The prepared samples were placed between these two electrodes. The experiment was performed in a wide frequency range of 1 Hz–10 MHz and with a signal amplitude of 2 mV.
The total ionic transference number of the prepared samples was analyzed by means of Wagner’s polarization technique. In this work, the cell was polarized by applying a constant potential to a cell configuration of SS electrode/sample/SS electrode. As a result, current is produced, which was then examined as a function of time with the impedance analyzer. Using this technique, the total ionic transference number was calculated using the following formula
X-ray diffractograms of the nanocomposite polymer electrolytes (NCPEs) were obtained using an X’pert PRO-PANalytical diffractometer with Cu-Kα radiation (wavelength, λ = 0.154 nm) with a scanning range between 2θ = 10ο and 80ο at ambient temperature.
The thermal stability of the sample was analyzed using thermo-gravimetric analyzer whose model is 2950 under N2 atmosphere. The temperature range used here was from room temperature to 700 C in steps of 20 C min−1. Atomic Force Microscopy (AFM) analysis was carried out using Agilent Pico LE microscope instrument.
Results and discussion
AC impedance analysis
Impedance spectra for the prepared P(VdF-HFP)/MgTr system and P(VdF-HFP)/MgTr with different contents of BaTiO3 are presented in Figure 2. It possesses a semicircle in the higher frequency side, which arises because of the bulk effect of the electrolyte and the linear region in the lower frequency range, ascribed to the effect of the blocking electrodes. The ionic conductivity of BaTiO3-added polymer electrolyte system is calculated with the help of the following equation AC impedance plot for the nano BaTiO3–incorporated P(VdF-HFP)/MgTr system.

Values of ionic conductivity for P(VdF-HFP)/MgTr/BaTiO3 samples.

The enhancement in ionic conductivity can be explicated via Lewis acid–base interactions as follows. There exists a competition between the strong acidic surface of nano BaTiO3 and Mg2+ cation, so as to interact with the fluorine atom of the polymer P(VdF-HFP). It in turn reduces the coupling of the fluorine and the cation (Mg2+), thus supporting the dissociation of the electrolyte and hence the ionic conductivity. Similar results were reported by earlier investigators.15,16
Earlier researchers15,16 reported that the augmentation of ionic conductivity is not only due to the Lewis acid–base interaction, but also due to the creation of conductive layer in the vicinity region of the filler particle. Such a conductive layer paves the way for the Mg2+ ion to get migrate. In addition, it is also to be noted that BaTiO3 is a ferroelectric filler, and hence, it possesses spontaneous polarization. This in turn also enables the solvation of the electrolyte MgTr, 15 and therefore, the ionic conductivity is improved.
The value of ionic conductivity gets decreased for the system with more than 6 wt% of nano BaTiO3. It is due to the fact that the higher concentration of filler blocks the ionic movement, and hence, the system does not conduct the ions. Such a blocking effect is due to the discontinuities in phases which overwhelm the polymer segmental motion and the ion mobility.17,18
Transference number analysis
Values of transport number for the prepared samples.

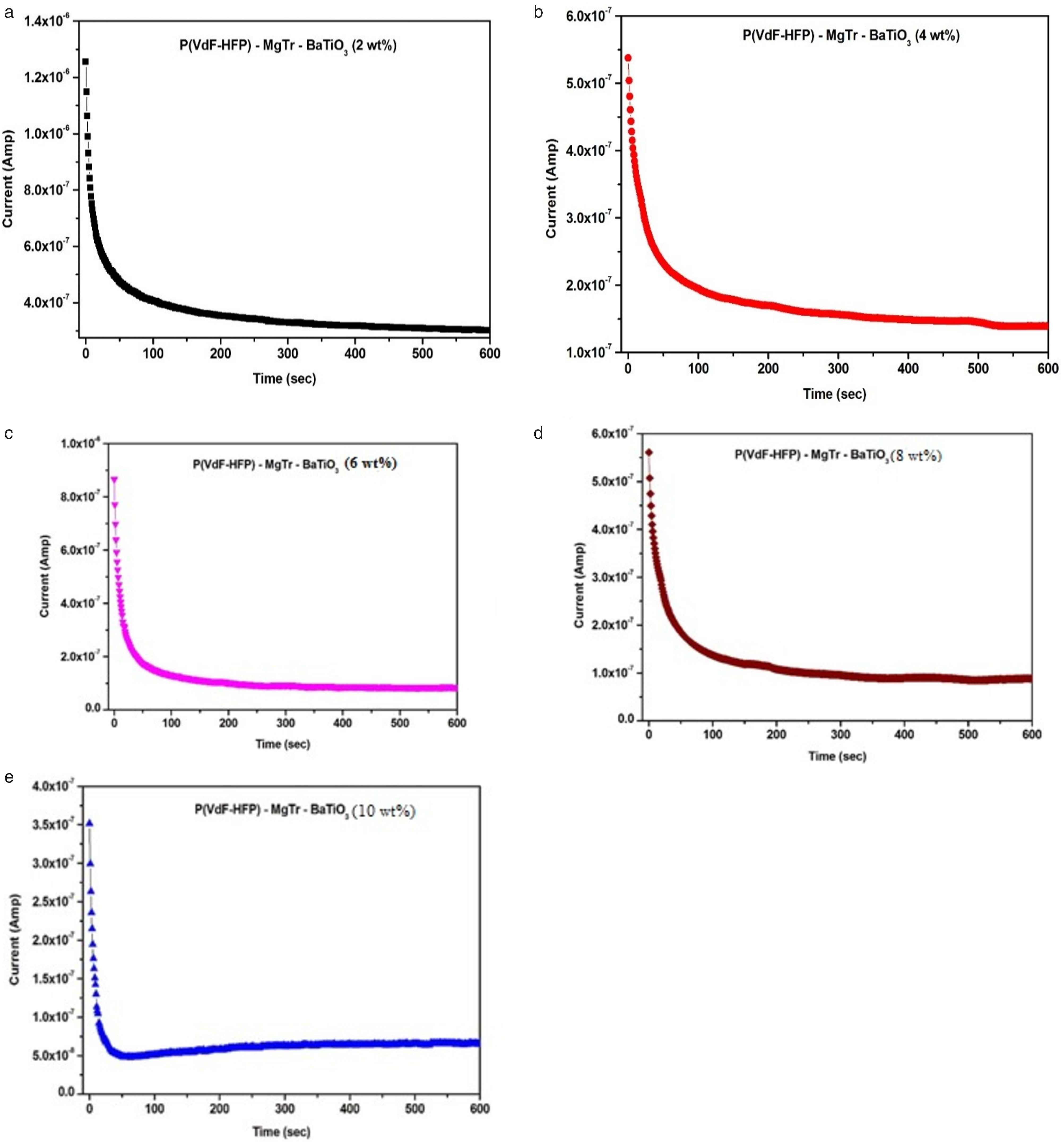
Variation of polarization current as a function of time under a constant voltage for a) 2wt % of BaTiO3, b) 4wt % of BaTiO3, c) 6wt % of BaTiO3, d) 8wt % of BaTiO3, e) 10wt % of BaTiO3-added P(VdF-HFP)/MgTr systems.
X-ray diffraction analysis
Figure 4(a)–(e) represents the X-ray diffraction (XRD) pattern of P(VDF-HFP), MgTr, P(VDF-HFP)/MgTr, nano BaTiO3 and P(VDF-HFP)/MgTr/BaTiO3(6 wt%) system, respectively. Figure 4(a) shows the X-ray diffractogram of P(VDF-HFP). The appearance of diffraction peaks at 2 X-ray diffractograms of (a) P(VDF-HFP), (b) MgTr, (c) P(VDF-HFP)/MgTr, (d) nano BaTiO3, (e) P(VDF-HFP)/MgTr/BaTiO3 (6 wt%)–added system.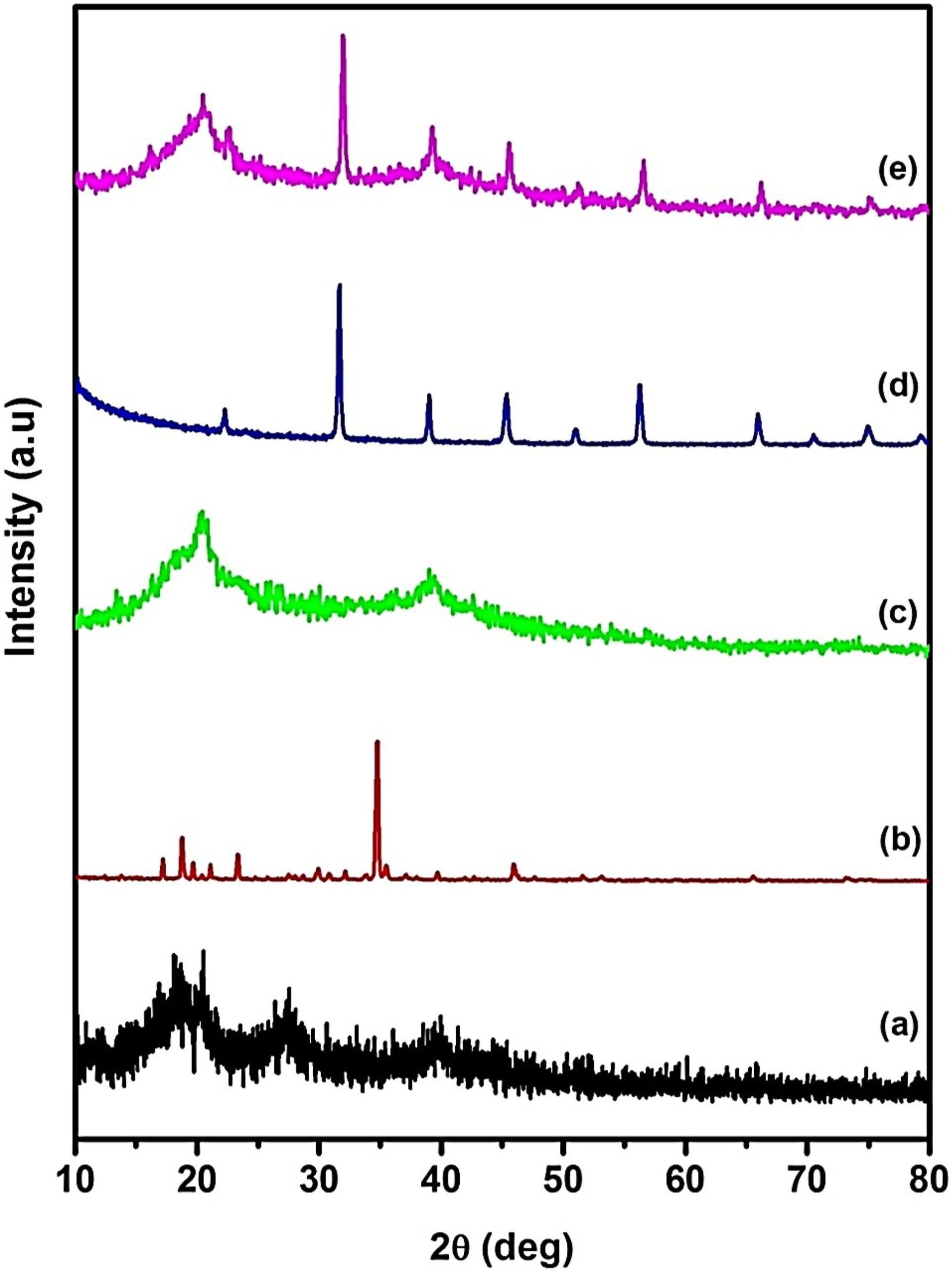
The appearance of diffraction peaks in Figure 4(b) and (d) suggests that the salt and the filler BaTiO3 used here are of highly crystalline. It is observed from Figure 4(c) (P(VDF-HFP)/Mg(CF3SO3)2 system) that the amorphous nature of the polymer P(VDF-HFP) is greatly enhanced after the addition of Mg(CF3SO3)2. In addition, the characteristics diffraction peaks of Mg(CF3SO3)2 are found to be absent. This implies that the salt Mg(CF3SO3)2 is solvated well in P(VDF-HFP) matrix, and hence, the P(VdF-HFP)/MgTr complex is formed.
It is seen that the intensities of the diffraction peaks of BaTiO3 get broadened and decreased (as in Figure 4(e)), when it is incorporated into the polymer electrolyte matrix at a concentration of 6 wt% (possessed maximum ionic conductivity). This proposes that the inert filler BaTiO3 promotes a change in the re-organization of the polymer chain and assists higher ionic conduction. 20 Moreover, the establishment of amorphous phase through Lewis acid–base interaction is also acknowledged through the XRD pattern. It in turn releases of Mg2+ ion and hence enhanced the ionic conductivity. These results divulge that the nanofiller added acted as a “Solid Plasticizer.”
Dielectric analysis
The real and imaginary parts of the dielectrics (ε′,ε″) can be expressed by

Figures 5(a) and (b) show the frequency dependence of the dielectric constants ε′ and ε″ for different concentrations of nano BaTiO3–added P(VdF-HFP)/MgTr system at ambient temperature. Variation of ε′ and ε″ values against the concentration of BaTiO3 corroborates the variation trend of ionic conductivity. Variation of the (a) dielectric permittivity ?′ versus logω, (b) dielectric loss ?′′ versus log ω for P(VdF-HFP)/MgTr/BaTiO3 system at room temperature.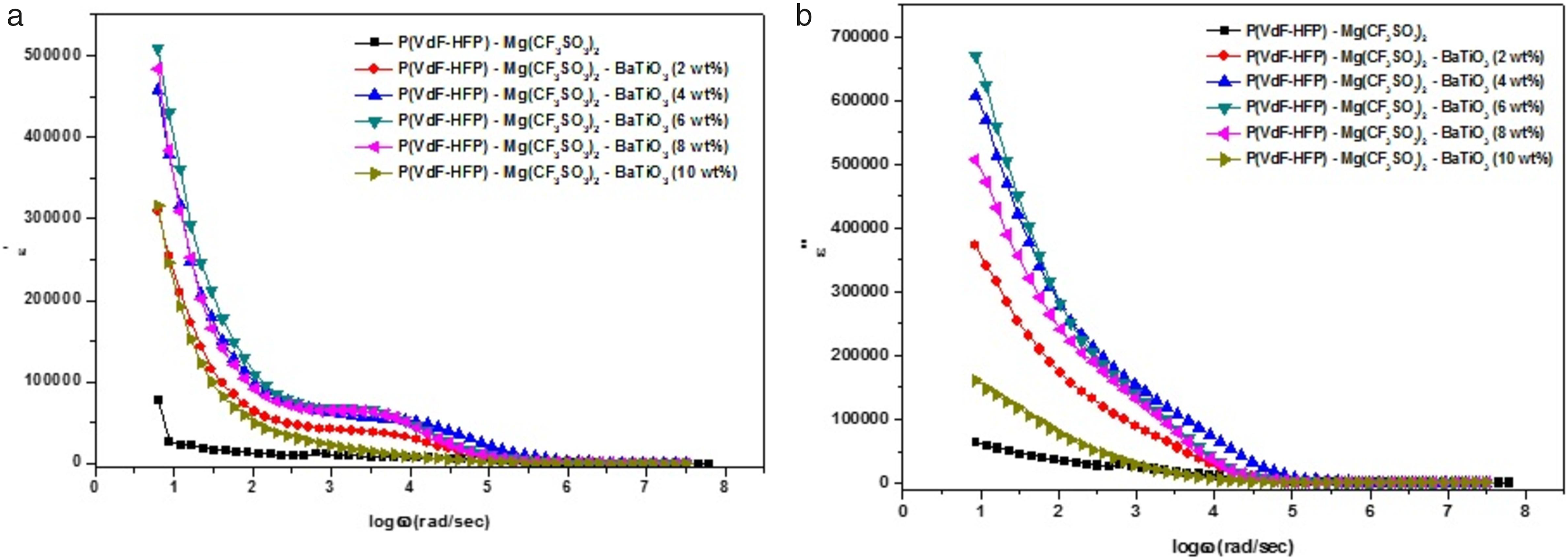
From these figures, the magnitude of dielectric constant (ε′) and dielectric loss (ε″) are found to be large in the lower frequency region. However, these values get decreased with increase of frequency and reached saturation in the higher frequency region. This is because, in the lower frequency region, dipoles in the P(VdF-HFP) may align more easily in a faster manner toward the applied fields direction. This event does not happen in the higher frequency side as there is no enough time for the dipoles to align along the direction of the field applied; 20 hence, ε′ and ε″ values are low.
As observed in ionic conductivity, the values of ε′ and ε″ are seen to be high for 6 wt% nano BaTiO3–added P(VdF-HFP)/MgTr system while compared with other concentrations of BaTiO3 which implies there exists only a small electrostatic attraction between the mobile ions, and hence, the probability of mobile ions in the free states becomes greater. 20
Modulus analysis
Figure 6(a) and (b) indicate the variation of M′ and M″ versus logω for nano BaTiO3–added P(VdF-HFP)/MgTr system. Variation of the (a) real part of electric modulus M′ versus log ω, (b) imaginary part of electric modulus M′’ versus log ω for P(VdF-HFP)/MgTr/BaTiO3 system at room temperature.
In these plots, at low frequency side, a long tail is observed. It is due to the presence of large capacitance which are accompanied by the electrodes. This substantiates that the materials are of non-Debye type. It is seen from these figures that the spectrum shape is found to be same for all BaTiO3 contents. But, the position of the peak is shifted to higher frequency side for the addition of 6 wt% of BaTiO3 system, whereas the position of the peak is found to be in lower frequency side for the other concentrations of BaTiO3. It assures that the charge carrier hopping is higher for the said BaTiO3 concentration than any other concentrations. 13
Loss tangent studies
Loss tangent spectra for P(VdF-HFP)/MgTr system with different concentrations of BaTiO3 are given in Figure 7. These spectra show a broad peak at a distinctive frequency which confirms the existence of dipole relaxation in the prepared system. The peak maxima of loss tangent for 6 wt% of BaTiO3-added system exhibited a small shift toward the higher frequency, whereas the others in the low frequency side. Such shifting of peak toward the high frequency side is a consequence of fast segmental motion of the free ions, whereas the peak shift toward low frequency side is a result of constrained segmental motion coupled with the ion.
21
Loss tangent spectra for P(VdF-HFP)/MgTr system with different concentrations of BaTiO3.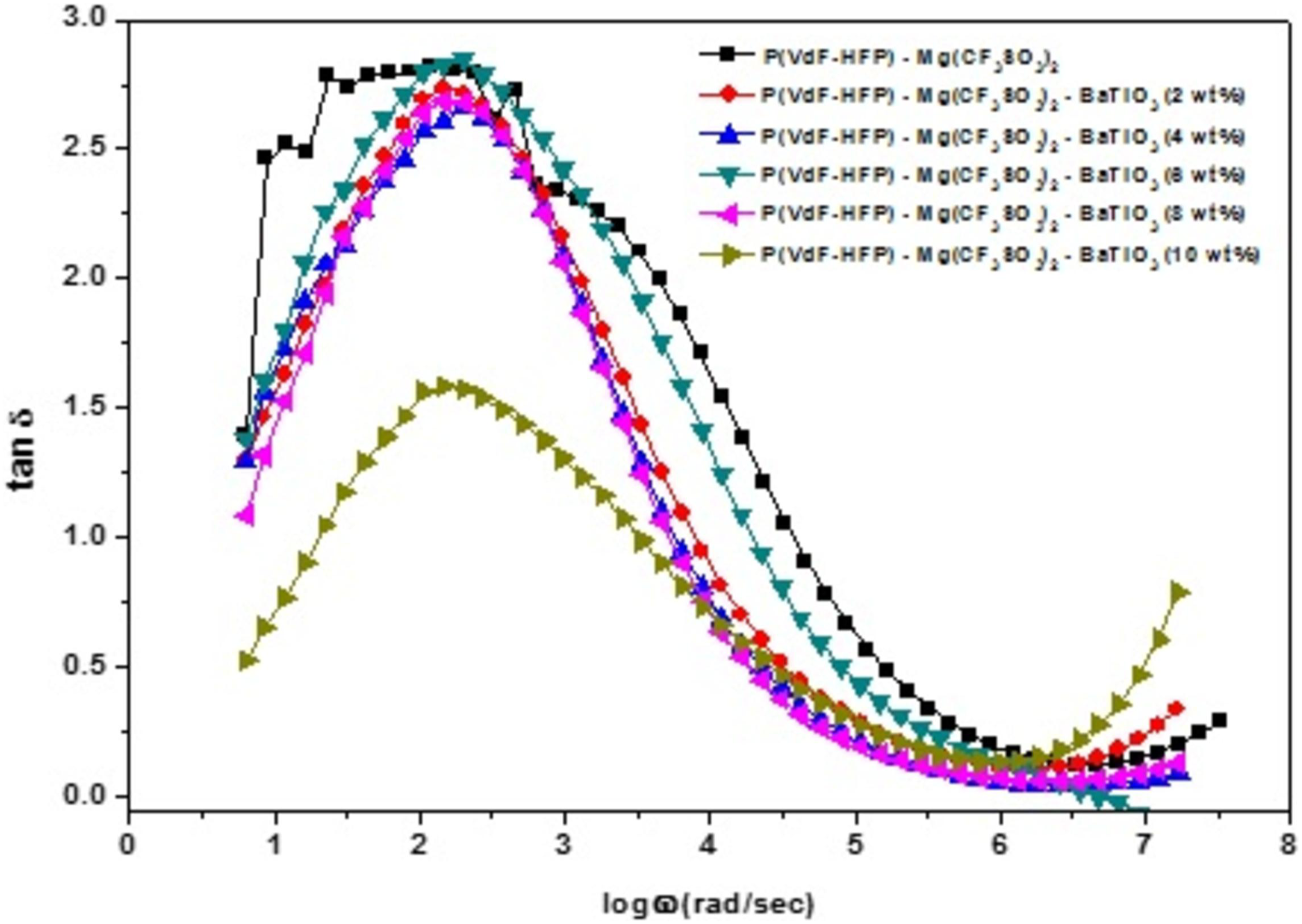
Tangent loss can be expressed by the following equation
The values of relaxation time (τ) are obtained by plotting tan δ as a function of logω and using the relation ωτ = 1, where τ is the relaxation time and ω is the angular frequency of the applied signal.
For the long-chain polymers, the bond rotation is favorable only at low frequencies, that is, higher is the relaxation time. But the presence of filler increases the disorder within the polymer network, and thus, the rotation becomes feasible; making a shift toward higher frequency side means a shorter relaxation time. The presence of the appropriate concentration of filler particles makes the polymer chain very flexible to allow any conformational changes, and hence, the relaxation occurs at a higher frequency in conjunction with smaller relaxation time. Therefore, it can be concluded that the doping of BaTiO3 changes the amorphicity and produces relatively fast segmental motion coupled with mobile ion.
Values of relaxation time (τ) for the prepared samples.

Atomic force microscopy analysis
Figures 8(a) and (b) illustrate the 3D-AFM images of P(VdF-HFP)/MgTr and P(VdF-HFP)/MgTr with 6 wt% of nano BaTiO3 systems. The system P(VdF-HFP)/MgTr shows a mountain valley pattern with a roughness (root mean square (RMS)) value of 85.80 nm. Similar mountain valley pattern also appeared for the nano BaTiO3 (6 wt%)–added P(VdF-HFP)/MgTr system (Figure 8(b)), but the number of mountains and valleys is more compared with filler-free one. Further, the roughness value is increased to 116 nm Atomic force microscopy images of (a) P(VdF-HFP)/MgTr system, (b) P(VdF-HFP)/MgTr system/BaTiO3 (6 wt%) system.
TG/differential thermal analysis Studies
Figure 9 represents the TG/differential thermal analysis (DTA) curves of the sample P(VdF-HFP)/MgTr with 6 wt% of nano BaTiO3. It is noticed from DTA curve, that the sample shows an endothermic peak at low temperature at around 127 °C. This is due to the moisture’s removal or solvent residues present in the film. Further, the DTA curve shows a broad endothermic peak at 340 °C. At this point, the theromogravimetric analysis traces exhibited a weight loss of about 61%. At this stage TG/DTA thermogram of P(VdF-HFP)/MgTr system/BaTiO3 (6 wt%) system.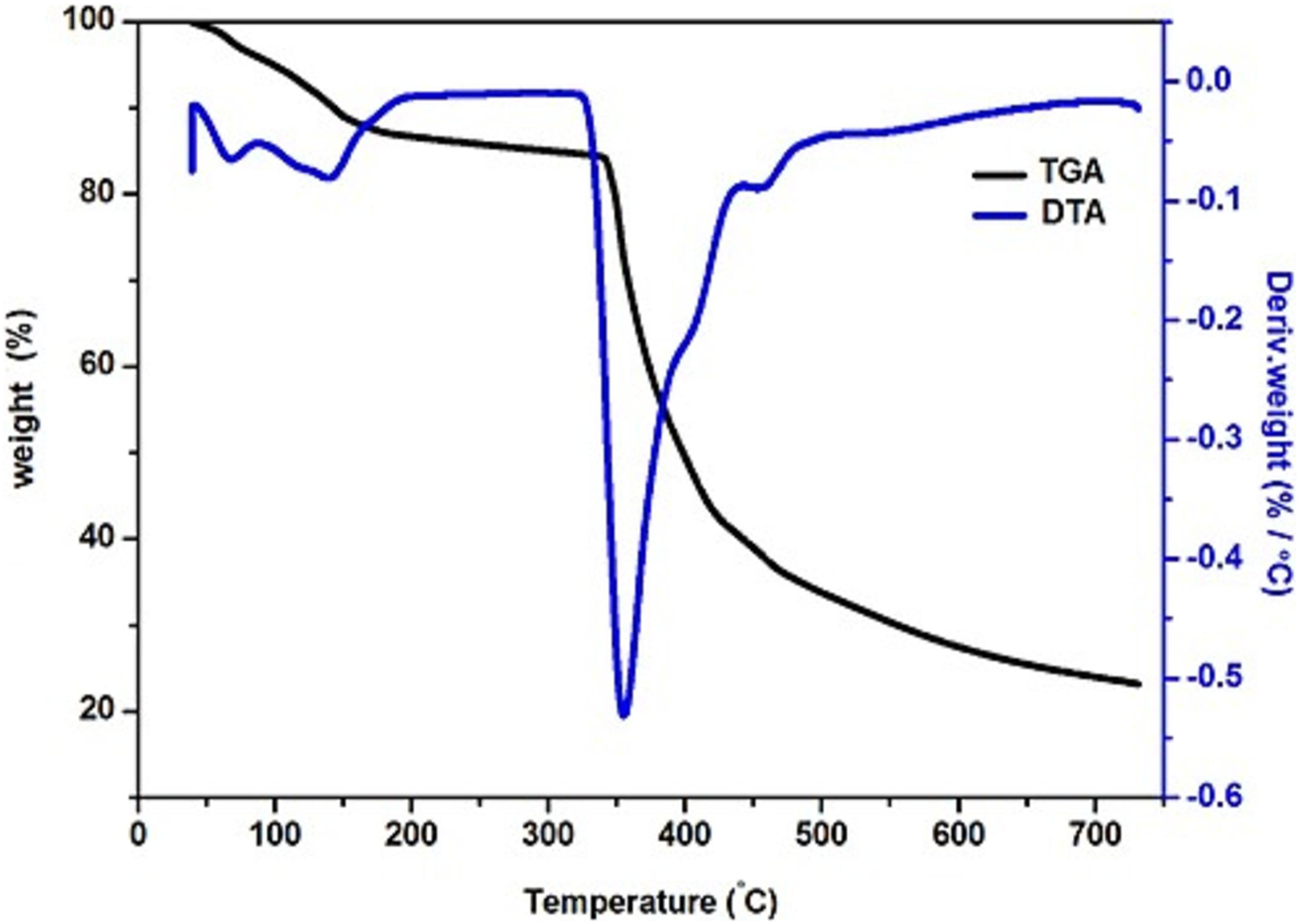
Conclusions
Nanocomposite polymer electrolytes (NCPEs) composed of P(VdF-HFP)/MgTr with various concentrations of BaTiO3 nanofiller were successfully prepared by conventional solvent casting technique. XRD studies revealed that the nano BaTiO3 promotes the structural modification in the P(VdF-HFP)/MgTr matrix. Values of ionic conductivity increased with the rise in filler concentration, and the system with 6 wt% of BaTiO3 showed a maximum ionic conductivity of 4.110 × 10−4 Scm−1 at room temperature. A low relaxation time was obtained for P(VdF-HFP)/MgTr with 6 wt% of nano BaTiO3 system which possessed maximum ionic conductivity. A significant change in RMS values was attained through AFM studies. From TG/DTA analyses, the maximum ionic conducting sample was found to be stable up to 340 °C. All these results clearly illustrate that the prepared NCPE met the prerequisite properties; therefore, it can be used as a separator in energy storage devices.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
