Abstract
Abietic acid (Aba) and ethylene-vinyl acetate (EVA) were blended by mixing and dissolving in toluene. The copolymer blend (Aba/EVA 85:15 wt%) was subjected to varying doses of gamma irradiation (10, 20, 30, 40, 50, 60, and 90 kGy). The crystallinity and the chemical structure of the polymeric blends were investigated by Fourier transform infrared, X-ray diffraction, and scanning electron microscopy and their adhesive properties were verified through utilizing the tensile strength testing. According to the findings, the gamma irradiation was found to increase the adhesive power of the blend polymer (Aba/EVA). To achieve the optimal adhesive force, 30 kGy was used as the irradiation dose. The adhesive forces of both irradiated and unirradiated samples were 1084 and 411 kPa, respectively. For this reason, adhesive force went up to 263%. Moreover, the process also entailed an examination of the antibacterial activity of the aforementioned copolymeric adhesive blend for irradiated (30 kGy) and unirradiated samples. The outcome from the antibacterial study pointed out that this polymer blend does entail antibacterial activity, something that is safe and helpful in the context of food packaging.
Introduction
In the current day, adhesive bonding is known as an established technology that is used extensively to join similar dissimilar materials across an increasing number of modern industries such as aerospace or automotive. On the other hand, adhesives can be defined as social substances capable to join permanently to surfaces, by an adhesive process. 1,2 It may be worthwhile to note that wafer bonding facilitates the packaging as well as fabrication of intricate three-dimensional microcomponents. 3 -5 The wide broad range of wafer bonding techniques includes anodic bonding, 6 direct bonding, 7 solder bonding, 8 thermocompression bonding, eutectic bonding, 9 ultrasonic bonding, 10 low-temperature melting glass bonding, adhesive bonding, and direct metal to metal bonding. 11
Abietic acid (Aba) is a natural polymer, solid and brittle obtained from pine trees, and its derivatives have attracted much interest in the field of pharmaceutical applications due to their characteristic properties such as biocompatibility, biodegradability, low cost, and capable of chemical modifications due to their special structure. 12 They have also been used in the fields of cosmetics, adhesives, medicines, chewing gum, and dental varnishes. 13 Moreover, rosin can be proposed in order to prepare a matrix for silver nanoparticles to apply as antibacterial filler for wooden furniture or air filter for indoors, a catalyst carrier with potential application as counter electrode for dye-sensitized solar cells, 14 a coating for bentonite particles and support for ferric oxide nanoparticles for chromium ions adsorption, 15 and activated carbons. 16 On the other hand, Aba, an abietane diterpenoid, has shown antiallergic, 17 anti-inflammatory, 18 phytoalexin-like, 19 antimicrobial, 20 and anticonvulsant activities. 21
Ethylene-vinyl acetate (EVA) copolymer produced by the copolymerization of ethylene and vinyl acetate is readily available in the form of rubber, thermoplastic elastomers, and plastic and is widely used in the wire and cable industry due to its desirable physicochemical properties and its easy acceptance of additives. 22 On the other hand, EVA has become one of the most useful copolymers in the transportation industry as an insulator, in the electric industry as a cable insulator, in the shoe industry as soles, and in many other industries as a hot melt adhesive, a coating and so on. 23
Polymeric materials are widely used for packaging and especially for active packaging. “Active” refers to the ability of the packaging to interact with the content, for example, by releasing or scavenging specific molecules, with the goal of increasing the shelf life of the product. One of the most interesting applications of active packaging is food preservation. Polymeric materials with antibacterial properties suitable for food packaging, medical packaging, medical devices, and pharmaceutical fields have been suitable for many applications. 24
Importantly, the adhesive polymer comprised of natural polymers like Aba and synthetic polymer including EVA was deeply explored as part of blend formulations. Three distinct attributes of copolymeric blend (Aba/EVA) are the following: mucoadhesive, biodegradable, and nontoxic. Notably, the copolymeric blend does not necessitate further modifications to render them biodegradable. The experiments investigated the impact of irradiation doses on the antifungal/antibacterial activities as well as the adhesive properties of the blend. Moreover, according to early experiments, the impact of ionizing radiation on the microorganisms was revealed through their indirect effects. 25 This study aims to prepare a film of the adhesive polymer whose elasticity can be utilized antibacterial applications. Bioadhesives feature a broad range of applications, including buccal drug delivery systems, 26 safe-food packaging adhesives, tissue bioadhesives, and associated biomedical applications. 27 The adhesive blend of Aba/EVA has applications on the painting of surgical, clinical rooms, surgical clothes, and also used as food packaging, so the resistance of Aba/EVA to pathogenic bacteria is very important. Meanwhile, another reported application of bioadhesives is the prevention of seroma, a frequently occurring postsurgical complication. The interruption of vasculatures and lymphatic system during surgery leads to accumulation and drainage of serous fluids within the space caused by surgery. 28 If left untreated, seroma can lead to massive complications. 29 The objective of the existing study is to synthesize the polymeric blend of Aba/EVA at varying ranges of dose from 10 kGy to 90 kGy. The copolymeric blend from Aba/EVA was analyzed by the following: X-ray diffraction (XRD), Fourier transform infrared (FTIR) and mechanical tools. After the investigation of the adhesive force, the antibacterial activity of several bacteria was studied on the aforementioned prepared copolymeric blend.
Experimental
Materials
EVA with melt flow index 19 g 10 min−1, melting point of 67°C, and containing 18% vinyl acetate was supplied by Arkema Inc., North America. Aba with melting point 173°C and molar mass 302.458 g mol−1 was supplied by HAB Co., Delhi, India. Toluene (99.9%) was purchased from Sigma Aldrich Co., St. Louis, USA. Figure 1 depicts the structures of EVA and Aba.

The structure of Aba and EVA.
Sample preparation
Copolymeric adhesive blend comprised one synthetic plastic component (EVA) in the form of elastomer within the adhesive formulation. Meanwhile, the natural polymer Aba was used as a tackifier that was compatible with EVA. Notably, the EVA and Aba were evaluated in order to determine the optimum miscibility of both components. Subsequently, each blend was stirred and dissolved evenly in toluene, after which it was left undisturbed for a period of 24 h so as to ensure absolute dissolution. According to the findings, the optimum miscibility and elastic film formation was found to be at 85/15 wt/wt of Aba/EVA. The resulting products were viscous solutions.
Gamma irradiation
Gamma irradiation of samples was carried out in the cobalt-60 source of gamma Chamber-4000 A, manufactured by Atomic Research Center, Delhi, India, installed at National Center for Radiation Research and Technology, NCRRT, Cairo, Egypt. The samples of copolymeric adhesive blend (eight glass vials were filled with the viscous blend and tightly sealed) were subsequently exposed to gamma irradiation at a fixed dose rate 1.66 kGy h−1 from 10 kGy to 90 kGy. Then, the extracted adhesive was stored at room temperature for testing and characterization.
Characterization
The prepared copolymeric blend (Aba/EVA) adhesive was examined by XRD by Shimadzu apparatus using nickel filter, copper K target, voltage = 40 (kV), current = 30 (mA), and scan speed = 8 (deg min−1) (Shimadzu Scientific Instruments (SSI), Kyoto, Japan). FTIR spectroscopy (a Nicolet Avatar 320 FT-IR Spectrophotometer, Cambridge, UK) with wave numbers ranging from 4000 cm−1 to 400 cm−1 was used. The surface of the samples was examined by scanning electron microscopy (SEM) (Jeol, JSM, Japan) at a voltage of 30 kV. The surfaces were pre-coated with a thin gold layer in order to reduce charging in the SEM.
Adhesive force test
The two glued slides of carbon steel bonded by a uniform layer of either irradiated and unirradiated copolymeric blend (Aba/EVA) samples are illustrated in Figure 2. After the collection of slides of carbon steel, they were kept for 24 h prior to adhesion testing of eight samples at 0, 10, 20, 30, 40, 50, 60, and 90 kGy. Figure 2 depicts the procedure of adhesion testing in the following manner: Figure 2(a) indicates the gradual filling of the gallon bucket with water. Figure 2(b) denotes a tested sample involving carbon steel and Figure 2(c) shows the digital balance. Furthermore, the balance here indicates the weight (kg) at which both steel slides will get collapsed. It is these points that will become the adhesive force.
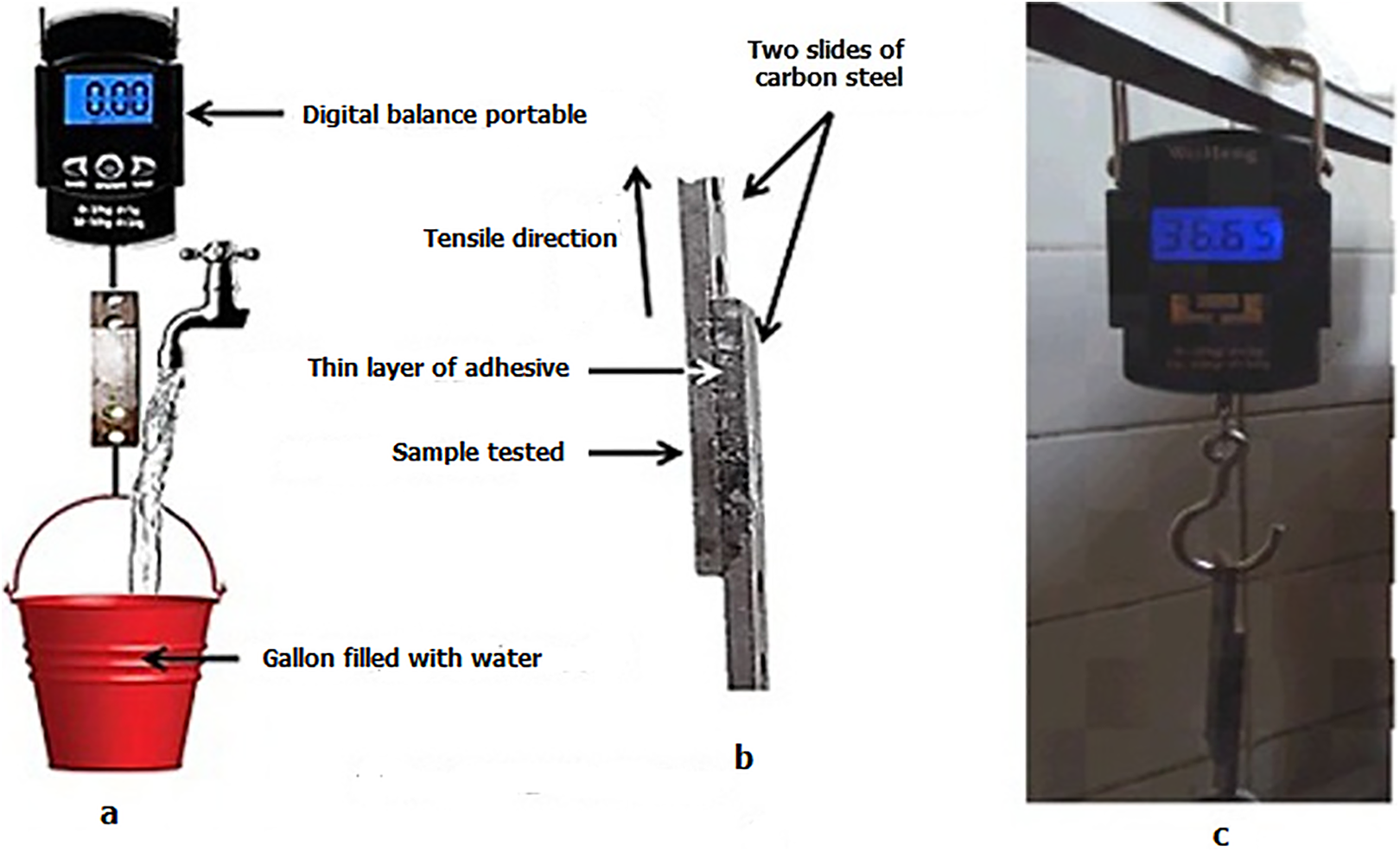
Illustration of a practical method of measuring adhesion strength test machine.
Antibacterial study
The nutrient agar (medium) contained 3 g of yeast extract, 1 g beef extract, 5 g peptone, and 5 g sodium chloride, and distilled water up to 1000 mL, and pH was adjusted at 7.0–7.4. The medium used for purification and antagonistic effect of clinical bacterial strains study the antibacterial effect of irradiated or unirradiated Aba/EVA. The antibacterial activity of Aba/EVA was tested by using the classical diffusion methods against Enterobacter cancerogenus m2, Pseudomonas aeruginosa m3, Citrobacter diversus m4, Enterobacter dissolvens m7, Staphylococcus aureus m11, S. aureus m14, Serratia fonticola m8, Escherichia coli m 9, and Bacillus cereus m 15, which were isolated from patients suffering from cancer from the previous study. Generally, this method is based on the observation and measurement of inhibition zones of bacterial growth. The plates were incubated at 37°C for 18–24 h and examined. The diameter of inhibition zone was measured in millimeters. Moreover examination effect of Aba/EVA on the morphological shape and growth of bacterial strains was carried out by SEM.
Results and discussion
The gamma irradiation of (Aba/EVA) adhesive blend for use as an adhesive
Figure 3(a) to (d) illustrates the various photos of stringiness during the involvement of two plates used for tack test. The photos in Figure 3(a) and (b) represent the tack of unirradiated and irradiated samples, respectively. The adhesive blend (Aba/EVA), which was irradiated at 30 kGy, demonstrated the highest tack. Meanwhile, the interfacial adhesion between these two plates at a 45° angle showed a greater elongation of the irradiated adhesive blend. On the other hand, Figure 3(c) and (d) identified the length of both samples’ stringiness sun irradiated and irradiated (30 kGy). The elongation of both unirradiated and irradiated samples was observed to appear at 10 and 15 mm, respectively. Furthermore, it was observed that there was an increase of 150% in the elongation of 30 kGy sample in comparison to the unirradiated sample.
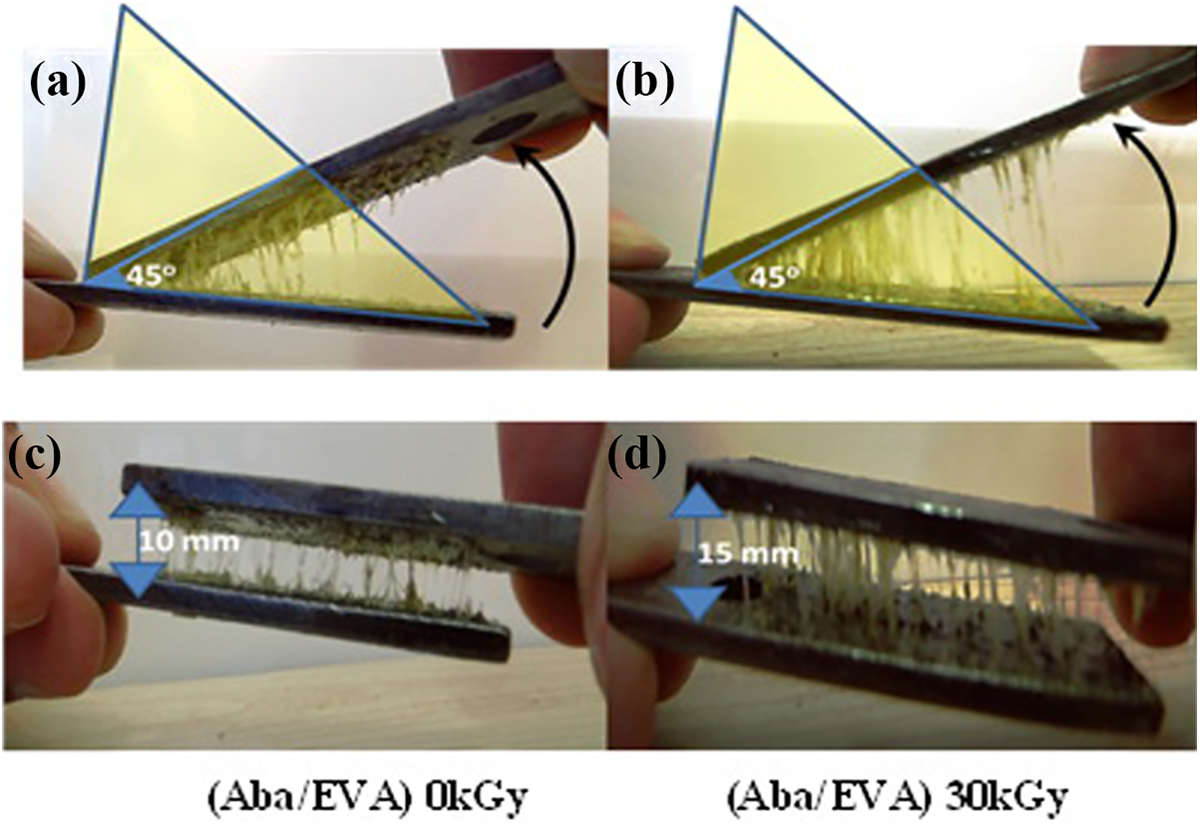
Optical photos of unirradiated ((a) and (c)) for sticky and elongation, respectively and irradiated samples ((b) and (d)) for tack and elongation, respectively.
Adhesion testing
Figure 4 illustrates the relationship between adhesive force and irradiation dose. From the results, it is noticed that there was variation in the value of adhesive force in comparison to the exposed irradiation dose. At 30 kGy, the highest adhesive force of 1084 kPa is demonstrated by the irradiation effect. The feature of this dose is that it shows good miscibility, adhesive force, elastic film formation, cross-linking, and a uniform solution of the blend, while other doses cause lowering cross-linking and degradation. Moreover, it is essential to determine that the irradiation of gamma prevents the effect of radiation degradation and enhancement of radiation cross-linking in compatible Aba/EVA blend. 30

The relationship between the adhesive force and irradiation dose of adhesive mixture of Aba/EVA.
FTIR analysis
The FTIR spectra provide information about structural aspects of polymers, such as chemical composition, conformation, and structural configuration. The positions and relative intensities of the absorption bands may give information concerning the interaction between the blend components. Figure 5 illustrates the FTIR spectra of Aba, EVA, unirradiated, and irradiated blend of Aba/EVA. FTIR spectrum of Aba shows a characteristic peak at 3995 cm−1 which was attributed to stretching vibration of OH in COOH. The absorbance at 1690 cm−1 for the carboxylic acid C=O group. EVA shows a characteristic peak at 2850–2960 cm−1 caused by C–H stretching vibrations and C–H antisymmetric deforming in CH2 and CH3 at 1467 cm−1. These general peaks can be found in the literature. 31 For EVA, C=O stretching vibration lies at 1739 cm−1, and the antisymmetric and symmetric stretching vibrations of C=O in =C–O–C cause the peaks at 1245 and 1023 cm−1, respectively. 32 In general, it is observed that the intensity of characteristic peaks for the irradiated sample is higher in comparison to that of unirradiated Aba/EVA. In the situation of unirradiated and irradiated samples, the stretching peak at 3406 cm−1 was attributed to the stretching vibration of OH in COOH. In the case of an unirradiated adhesive blend, the peaks at 2861 and 2925 cm−1 were attributed to the asymmetric and symmetric vibrations of stretching C–H in the pyranoid ring. 33 At 1692 cm−1, the broad peak centers are associated with stretching C=O in both Aba and EVA. There is a good correlation between these data based on the fact that the gamma irradiation causes cross-linking of an adhesive blend of Aba and EVA. At this point, the peaks at 1141, 1183, and 890 cm−1 are associated to the widening of the vibrations of the alkyl peroxides stretching (C–O–O–C), the vibration of the stretching (C–O) of carboxyl group, and out of plane bending vibration of CH(CH2=CH–H). Moreover, with the increased intensity of these peaks, the cross-linking of Aba/EVA got confirmed, especially the peak at 890 cm−1 that got increased for the irradiated sample. This is another characteristic of the absent auto-oxidation of Aba. Hence, the results obtained demonstrate that there is an increase in the methylene motion and cross-linking of molecules of an adhesive blend of Aba/EVA which is induced by the gamma irradiation. 15 On the other hand, the gamma rays are capable to be absorbed even by C–C and/or C–H bonds. Thus, formed radicals led to the formation of high amount of cross-links in an adhesive blend of Aba/EVA. 34
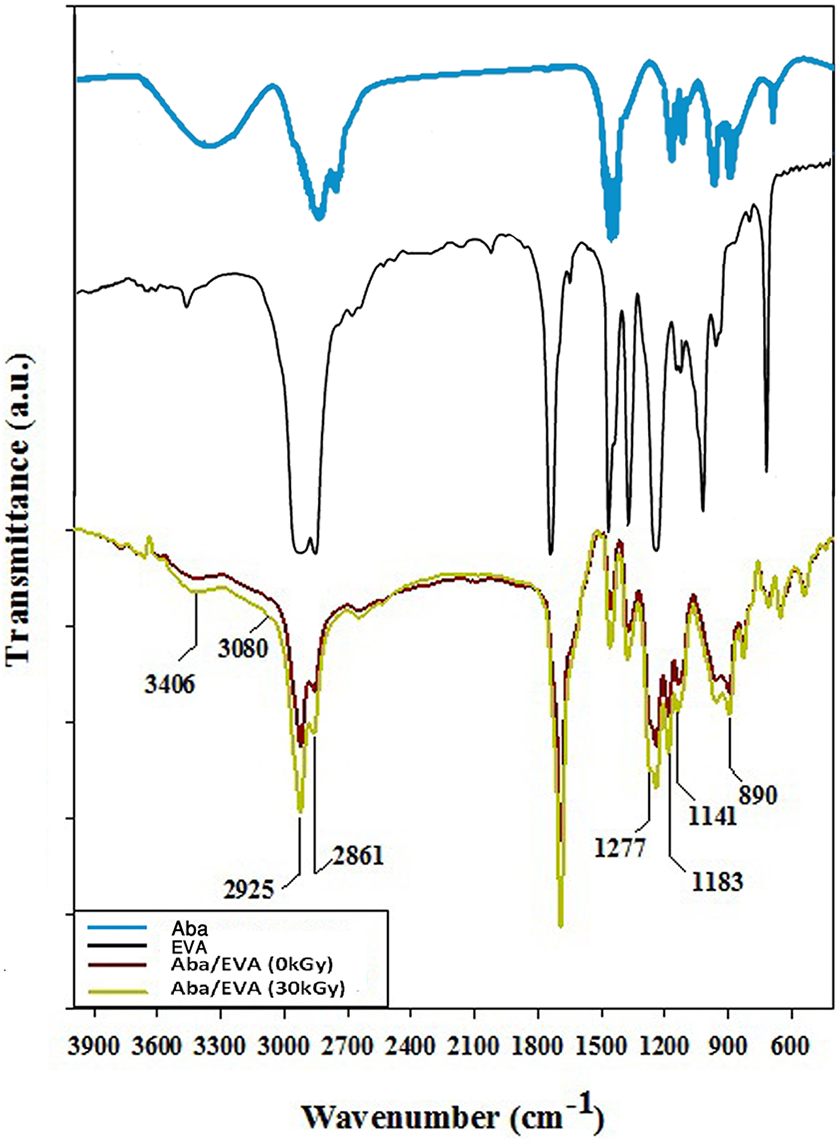
ATR-FTIR spectra of Aba, EVA, unirradiated, and irradiated (30 kGy) adhesives of blend of Aba/EVA samples.
XRD diffraction
Figure 6 shows the XRD patterns of Aba, EVA, unirradiated, and irradiated (Aba/EVA) blend. In the case of Aba, a broad characteristic peak observed at 15.1° reflecting the amorphous nature of Aba, while the XRD pattern of EVA is characterized by a diffraction peak at 21.2° reflecting its semicrystalline nature. For unirradiated and irradiated Aba/EVA, there is a characteristic peak at 15.46°. However, in the situation of unirradiated Aba/EVA, there was a high intensity in the peak at 15.46°. This could be used for revealing the presence of cross-linking structure of the irradiated blend. 35

XRD patterns of Aba, EVA, unirradiated, and irradiated (30 kGy) samples of adhesive blend of Aba/EVA.
Scanning electron microscopy
Figure 7(a) to (d) shows the SEM images of EVA, Aba, and both unirradiated and irradiated Aba/EVA blend. The SEM pattern of EVA is shown in Figure 7(a). The surface of EVA is dense, tabular, and smooth, and no sign of porous structure is visible in the samples displaying a regular layered structure perpendicular to the tensile direction. In Figure 7(b), the SEM of the Aba observed that the surface was rock shape, rough, and with sharp edges. Unirradiated polymeric blend containing Aba/EVA shows rough surface, holes, and with a few elongated shapes. The EVA particles dispersion shows that a small interfacial tension exists between Aba particles. For this blend, small holes and a network of canals have appeared as shown in Figure 7(c).The enhanced compatibility of the Aba/EVA blend at 30 kGy was observed in Figure 7(d). The image of the Aba/EVA blends at 30 kGy shows a more homogenous structure compared to the unirradiated blend. The EVA content in the blend causes the formation of an interpenetrating polymer network and makes the samples more flexible. Irradiation changes the polymer network; therefore, it changes the morphological state of the polymer. The radiation dose of gamma irradiation leads to an increase in cross-linking degree in the amorphous area and leads to increased molecular weight. 35

SEM photographs of (a) EVA, (b) Aba, (c) unirradiated Aba/EVA, and (d) irradiated Aba/EVA at 30 kGy.
Antibacterial activity
Figure 8 and Table 1 show that three samples were used for evaluating the antibacterial activity, which are toluene solvent, unirradiated sample, and irradiated sample of Aba/EVA at 30 kGy. The results confirmed that there were three samples of blend showing no antibacterial activity against Gram-positive S. aureus m14 (a), while samples of b and c showed antibacterial activity against S. aureus m11 (1.5 and 1.1 mm) inhibition zone, respectively. Furthermore, the sample of (a) exhibited antibacterial activity against Gram-negative P. aeruginosa m3, Klebsiella oxytoca m6, and S. fonticola m8 (1.1, 0.8, and 0.7 mm) inhibition zone, respectively. Also, sample (b) shows antibacterial activity against Garm-negative E. coli m12, E. coli m9, S. fonticola m8, and K. oxytoca m6 (0.9, 1.2,1.2, and 1.2 mm) inhibition zone, respectively. Moreover sample (c) shows antibacterial against Gram-negative S. fonticola m8, K. oxytoca m6, E.coli m12, E. coli m9, and P. aeruginosa m3 (0.8, 1.0, 0.8, 1.0, and 0.7 mm) inhibition zone, respectively. In the situation where the unirradiated sample can be compared with the irradiated sample, it could be exhibited the antibacterial activity is higher in the irradiated sample than unirradiated samples. In addition, Figure 9(a) to (n) observes the SEM of bacterial cells loaded with films of unirradiated and irradiated at 30 kGy of Aba/EVA against Gram-negative bacteria and Gram-positive bacteria (E. cancerogenus m2, P. aeruginosa m3, C. diversus m4, E. dissolvens m7, S. fonticola m8, E. coli m9, and B. cereus m15). The SEM images explained inhibition and disfiguration in bacterial growth with irradiated Aba/EVA at 30 kGy more than unirradiated Aba/EVA. On the other hand, the increase in disfiguration was noticed in the irradiated samples in comparison to the unirradiated samples, which indicated that the antibacterial activity of copolymeric material was increased with gamma irradiation dose. 36
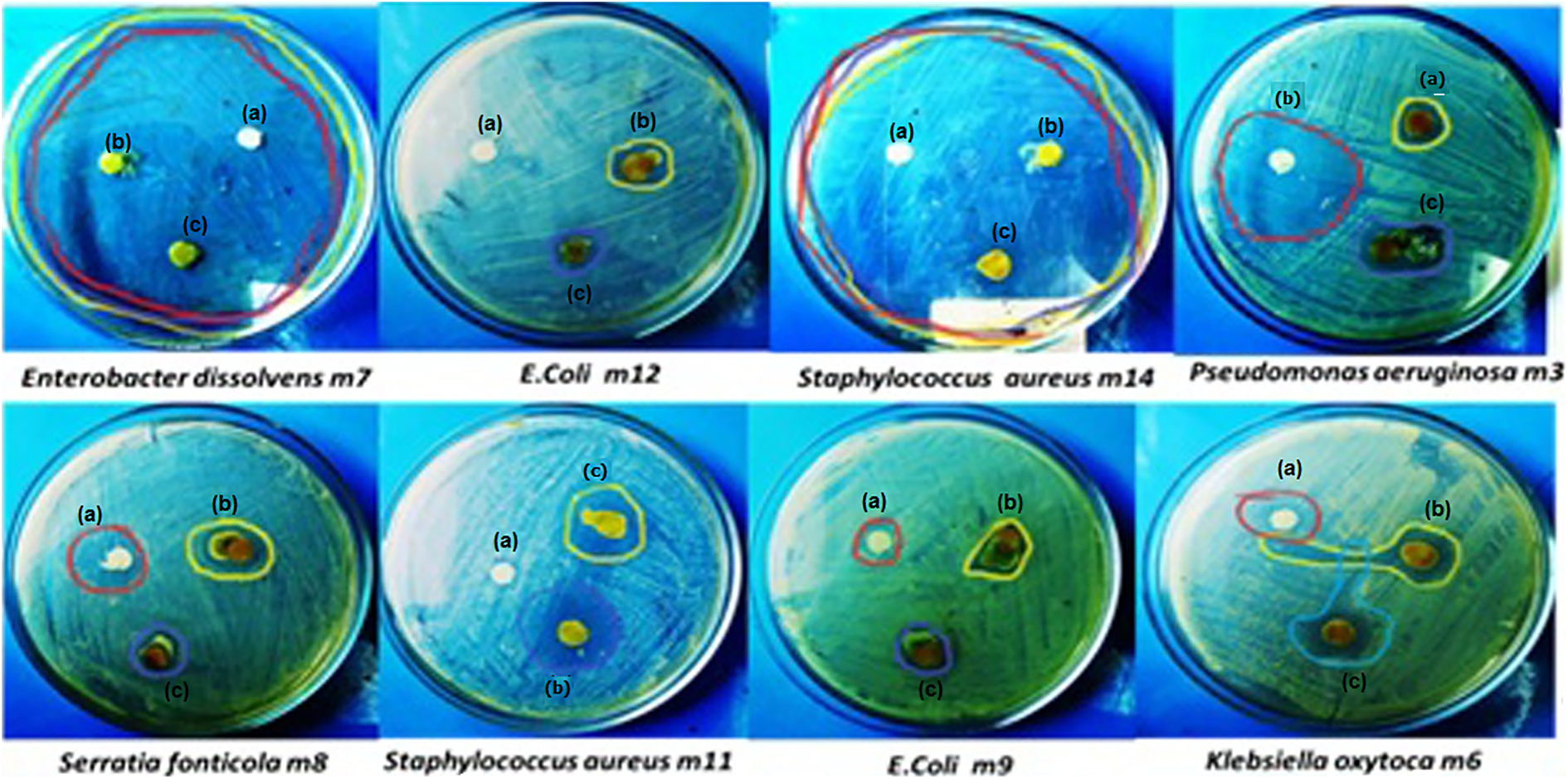
Zone of antibacterial action of (a) toluene solvent, (b) irradiated Aba/EVA at 30 kGy, and (c) unirradiated Aba/EVA.
Antibacterial activity of (a) toluene solvent, (b) irradiated Aba/EVA at 30 kGy, and (c) unirradiated Aba/EVA by disk diffusion method.

Aba: abietic acid; EVA: ethylene-vinyl acetate.

(a) to (n) SEM photographs of antibacterial activity of Aba/EVA before and after gamma irradiation.
Conclusions
The current study aimed to prepare adhesive polymer blends (Aba/EVA 85:15 wt%) by mixing and dissolving their polymers in toluene, then the solution was subjected to varying doses of gamma irradiation (10, 20, 30, 40, 50, 60, and 90 kGy).
The experimental study led to the following conclusions: 30 kGy is the optimal irradiation dose concerning the optimal adhesive force. The feature of this dose is that it shows good miscibility, adhesive force, elastic film formation, highest tack, cross-linking, and a uniform solution of the blend. Verification that a gamma irradiation dose of 30 kGy induces cross-linking and crystallinity and chemical structures of the polymeric blend were done through XRD and FTIR analysis. The SEM image of the Aba/EVA blends at 30 kGy shows a more homogenous structure compared to the unirradiated blend. The outcome from the antibacterial study pointed out that this polymeric blend has antibacterial activity, something that is safe and helpful in the context of food packaging.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
