Abstract
Lignin–melamine–formaldehyde (LMF) resin was prepared by three steps: (i) tosylation of lignin, (ii) synthesis of lignin-melamine (LM) copolymer, and (iii) formation of methylol LM. The synthesized resins were characterized by Fourier transform infrared (FTIR) spectroscopy and phosphorous 31 nuclear magnetic resonance analysis. The curing parameters of LMF resin were determined by differential scanning calorimetry (DSC) and thermal gravimetric analysis. The yield of tosylation is 80%. The FTIR spectrum of tosylated lignin shows the presence of two new bands at 1171 and 1370 cm−1. The formation of the LM was demonstrated by the disappearance of both bands and appearance of the absorbances at 3115, 3312, 3415, and 3470 cm−1 corresponding to the stretching vibrations of primary and secondary amine. The peaks observed at 147.0 and 148.5 ppm are attributed to the new aliphatic hydroxyl groups formed by the methylolation of LM. One exothermic peak was observed in the DSC analysis indicating a one cross-linking reaction.
Keywords
Introduction
In the context of rarefaction of fossil fuel resources and protection of the environment, special attention has been given over the past few years to the development of processes that are more environmentally friendly. Lignocellulosic polymers represent an “inexhaustible” source of biomass-issued matter. Mainly exploited in the paper industry, these polymers show interesting physicochemical properties and high potential for development. Millions of tons of several lignins are produced by the paper industry every year. 1 Only a small portion of the lignin is marketed, which is a result of the uncertainties linked to its structure and high molecular weight. Lignin is a complex tridimensional polymer made up of phenylpropane. This tridimensional polymer forms from three phenolic precursors: guaiacyl, syringyl, and p-hydroxyphenyl. 2 Lignin has a lot of potential for applications as a biomaterial and many groups work on lignin valorization in this area, exploring their properties and characteristics in the making of composites and resins. 3,4 Monteil-Rivera et al. 5 discussed recently the applications of agricultural lignin in biomaterials, based on their physicochemical characteristics. The use of lignin as a reinforcement in composite materials has been largely explored. 6,7
Melamine–formaldehyde (MF) resins are widely used because of their low cost, versatility, and many applications such as decorative panels, leather tanning, coating, and materials. 8,9 MF is also used in the electric domain. 10 Several researchers have tried to replace a part of the melamine in the MF resins by a biodegradable material. Chai et al. 11 used the bark alkaline extractive from mountain pine to partially replace 30 wt% of melamine in formulating the bio-based bark extractive— MF resin without loss of mechanical properties. Bergmann et al. 12 successfully replaced 50 wt% of the melamine with wood. Melamine-modified lignin was prepared by Wang et al. 13 using grafting reaction from hydrolyzed lignin by enzyme, melamine, and formaldehyde, and its adsorption for silver ions was studied.
To the best knowledge of the authors, a study about the synthesis of the LMF resin using a tosylated lignin intermediate and polymer LM has not yet been published. Hence, the main goal of this study is to reduce the amount of the synthetic monomers by incorporating lignin in the synthetic process. These kinds of biomaterials be a new way of valorization for lignins, mainly kraft lignin. Therefore, the modified MF resin would constitute a greener alternative to existing resins.
In the present work, it was planned to prepare modified MF resin by incorporating kraft lignin. Kraft lignin was tosylated according to the method developed in our previous research. 14 We want to use this tosylated lignin to synthesize a LM copolymer. This resulting copolymer then reacted with formaldehyde in a basic medium to give methylol LM (LM-OH). The thermal curing of LM-OH gave the final LMF resin. The resin and intermediates will be characterized by FTIR, 31P NMR, thermogravimetric analysis (TGA), and differential scanning calorimetry (DSC).
Materials and methods
Materials
Kraft lignin (M w = 6175 g mol−1, polydispersity index = 2.8) was supplied by Dupont Inc. (Québec, Canada). Triethylamine (99.5%), p-toluenesulfonyl chloride (TsCl, 99%,), 2-chloro-4,4,5,5-tetramethyl-1,3,2-dioxaphospholane (TMDP, 95%,) and pyridine (99.8%) were purchased from Sigma-Aldrich (Québec, Canada). Dimethylsulfoxide (DMSO, 99.5%), melamine (99%), ethanol (≥99.8%), and formaldehyde solution in water (37% by weight) were also purchased from Sigma-Aldrich. All solvents and chemicals were commercially available and were used as received without further purification.
Methods
LMF resin synthesis
Tosylation of lignin
Lignin was tosylated according to the writing method by Diop et al. 14 Tosylated lignin (L-Ts) was prepared in aqueous medium. One gram (5.91 mmol of hydroxyl) of lignin was mixed with 2.25 g (2 equivalents; 11.82 mmol) of tosyl chloride in the presence of 2.4 mL (3 equivalents; 17.73 mmol) of triethylamine. The mixture was stirred for 24 h and was concentrated in a rotary evaporator, precipitated with ethanol, and then filtered and washed with deionized water (300 mL) and hot ethanol (150 mL). The product was dried at 60°C.
Synthesis of LM copolymer
LM copolymer was synthesized by nucleophilic reaction between L-Ts and melamine in DMSO. In a typical synthesis, L-Ts (1.00 g) and melamine (0.75 g) were dissolved in 50 mL of DMSO. The reaction mixture was heated up to 100°C. After 6 h, the reaction product was allowed to cool to room temperature. The product was precipitated with cold ethanol (0°C) and then filtered and washed with ethanol (100 mL).
Synthesis of LM-OH
LM copolymer was mixed with formaldehyde to produce a LM-OH. One gram of LM and 15 mL tetrahydrofuran were taken in a round-bottomed flask equipped with a magnetic stirrer. The reaction bath was kept at 65°C. Sixteen milliliter formaldehyde (37% w/w) maintained at pH 9.0 using sodium hydroxide solution (1 N) was added to the above mixture and stirred during 1 h. The solution was cooled down. The excess of formaldehyde was removed by a rotary evaporator at 50°C. The yield of tosylation is calculated relative to the hydroxyls. The reaction mechanism of these three steps is summarized in Figure 1.
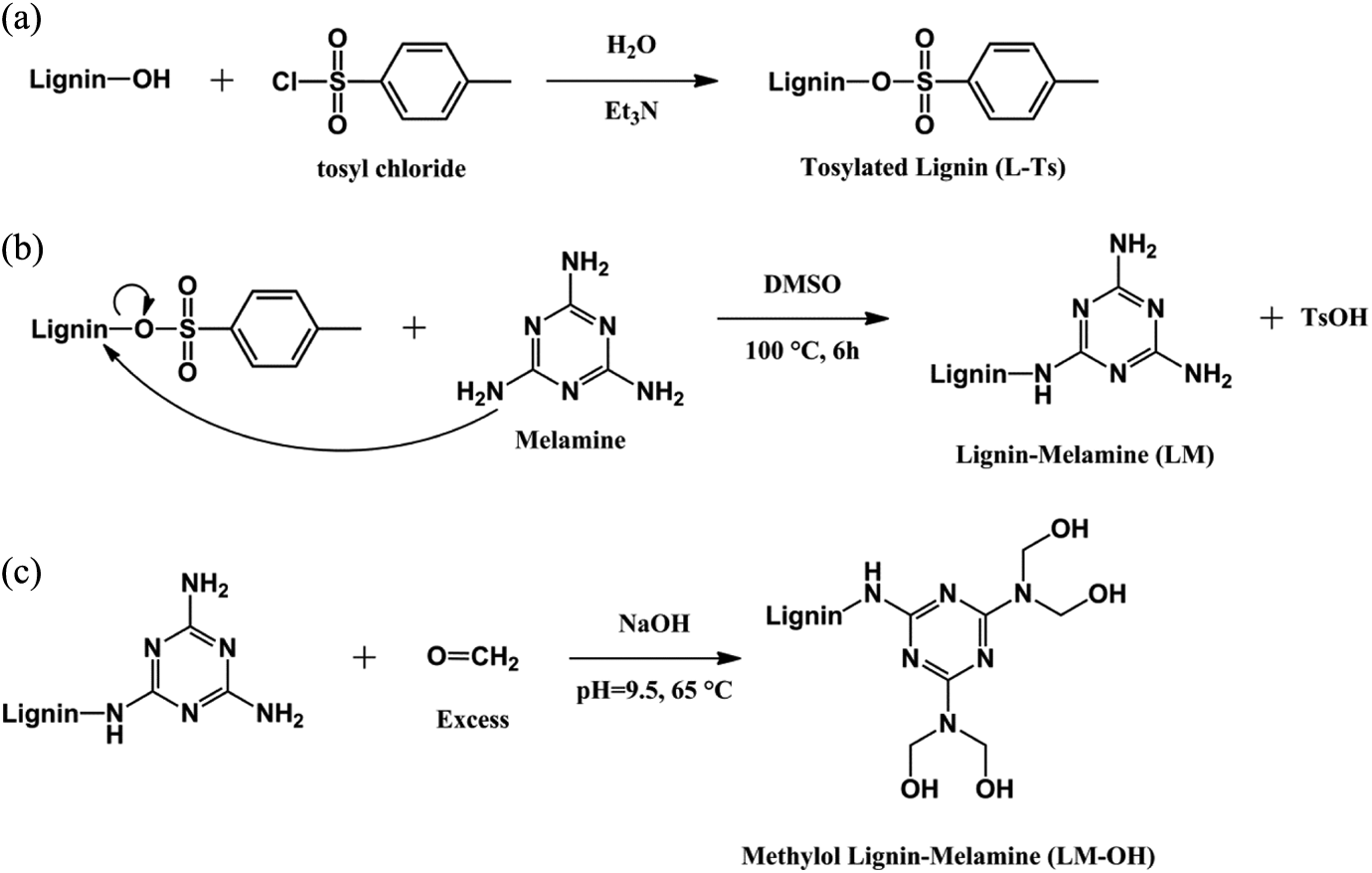
Formation of (a) (L-Ts), (b) LM, and (c) LM-OH. L-Ts: tosylated lignin; LM: lignin–melamine; LM-OH: methylol lignin–melamine.
Curing of resin
Five grams of LM-OH sample was heated in a convection oven from room temperature to 200°C at a heating rate of 20°C min−1. Then, the sample was taken out from the oven, cooled in air, and used for the TGA study.
Analytical method
The FTIR spectra were obtained with a Perkin Elmer instrument, System 2000 (Waltham, Massachusetts, USA) using pellets of lignin mixed with potassium bromide (lignin content of 1%). Spectra were recorded between 400 cm−1 and 4000 cm−1 with a resolution of 4 cm−1.
The unmodified lignin, L-Ts, LM, and LM-OH were dried under vacuum for 48 h. A solvent mixture composed of pyridine and deuterated chloroform in a 1.6/1 (v/v) ratio was prepared. Twenty milligrams (20 mg) of lignins were accurately weighed into a 2 mL volumetric flask. The sample was then dissolved in 0.5 mL of the above solvent mixture. Quantitative 31P NMR spectra of the lignins were obtained using published procedures with TMDP as a phosphitylating reagent and N-hydroxynaphthalimide as an internal standard. 15 The 31P NMR spectra of the resulting mixtures were obtained using a Bruker 500 MHz spectrometer (Billerica, Massachusetts, USA) equipped with a broadband inverse probe dedicated to 31P. An acquisition time of 0.2 s, a delay time of 5.00 s, at number of scan of 512 were used in each analysis. The content of hydroxyl groups was obtained by integration of the following spectral regions: aliphatic hydroxyls (149.1−144.6 ppm), syringyl (S) phenolic hydroxyls (143.3–141.9 ppm), condensed phenolic units (difference between 144.3–141.3 ppm and 143.3–141.9 ppm, as previously reported by Cateto et al., 16 ) guaiacyl (G) phenolic hydroxyls (140.6–138.6 ppm), p-hydroxyphenyl (H) phenolic hydroxyls (138.4–137.2 ppm), and carboxylic acids (135.3–134.4 ppm).
The DSC experiments were conducted on a Mettler Toledo (DSC with a Mettler Toledo DSC822e, Columbus, Ohio, USA) to study the cure properties of LMF resin. Dynamic scans were made from 25°C to 300°C at a constant heating rate under nitrogen atmosphere (50 mL min−1). Heating rates of 5 and 20°C min−1 were used. A sample of approximately 5 mg was weighted and encapsulated into an aluminum pan. An empty aluminum pan was used as the reference.
The thermal stabilities of cured LMF were analyzed by TGA with a Mettler Toledo TGA system (Model TGA/SDTA851e). Approximately 4 mg of each sample was weighed and heated in alumina crucibles. Nitrogen was purged as an inert atmosphere at a flow of 50 mL min−1. Samples were heated in the temperature range from 25°C to 700°C at a heating rate of 10°C min−1.
Results and discussion
FTIR analysis
Figure 2 shows the FTIR spectra of lignin, L-Ts, LM, and LM-OH. The spectrum of lignin (Figure 2(a)) shows a large band at 3400 cm−1 corresponding to phenolic and aliphatic OH groups. The bands observed at 2933 and 2863 cm−1 are respectively related to the C–H vibrations and the C–H stretching in methoxy groups. The band at 1708 cm−1 is assigned to unconjugated C=O stretching. The most characteristic vibrations of lignin correspond to those of aromatic rings at approximately 1599 and 1509 cm−1. The typical bands assigned to guaiacyl (G) moieties were identified at 1271 and 1216 cm−1 (guaiacyl ring breathing) and at 853–858 cm−1 and 817 cm−1 (C–H out-of-plane in 2-, 5- and 6-positions of G groups). The bands assigned at 1030 and 1125–1128 cm−1, correspond to C–O(H) and C–O(C) stretching of first-order aliphatic OH and ether groups.
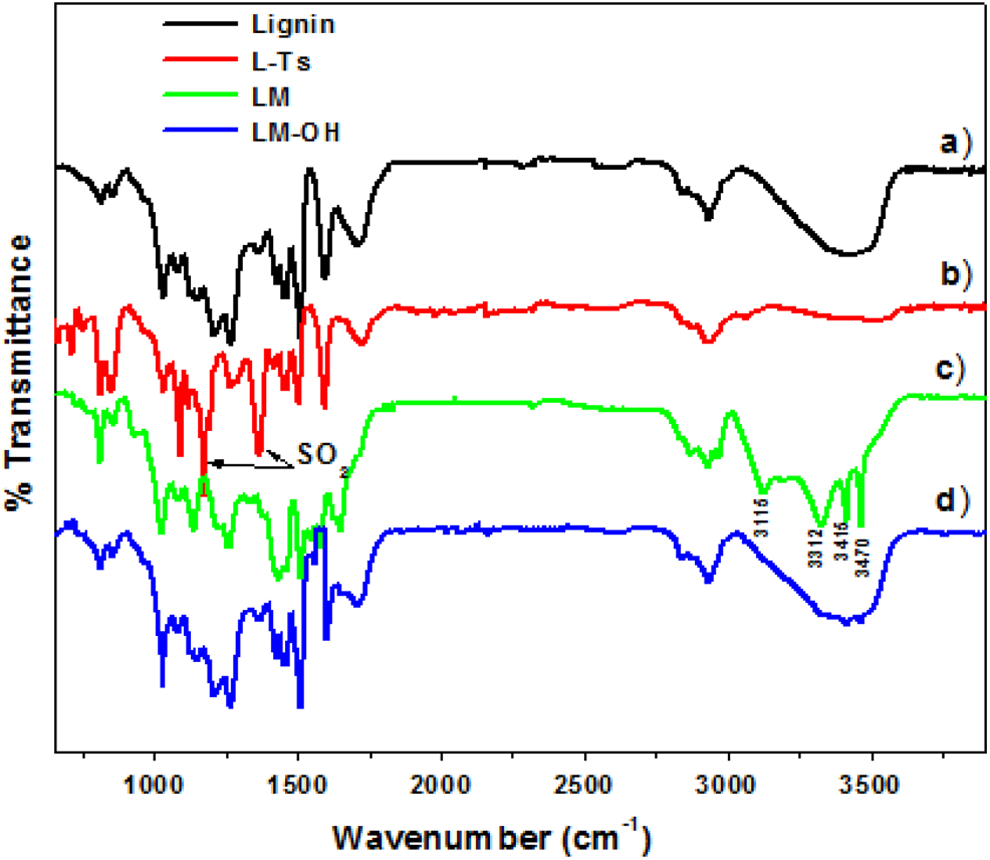
FTIR spectra of (a) lignin, (b) L-Ts, (c) LM, and (d) LM-OH. FTIR: Fourier transform infrared; L-Ts: tosylated lignin; LM: lignin–melamine; LM-OH: methylol lignin-melamine.
In the FTIR spectrum of the L-Ts (Figure 3(b)), the absorption bands at 1370 cm−1 (SO2), 1172 cm−1 (SO2), 811 cm−1 (SO), and 663 cm−1 (SO) are assigned to the tosyl (Ts) groups. 14,17 The spectrum shows also the decreasing of the characteristic band of the hydroxyl groups (3400 cm−1). These observations show that lignin was tosylated. FTIR of LM (Figure 3(c)) shows the disappearance of bands of the tosyl groups (1370 and 1172 cm−1) and the appearance of the absorbances at 3115, 3312, 3415 and 3470 cm−1 corresponding to the stretching vibrations of primary and secondary amines. The combination of these informations shows clearly the substitution of the tosyl groups by the amine group, confirming the synthesis of the copolymer LM. The FTIR spectrum obtained for LM-OH is characterized by a broad OH band at 3400 cm−1 and another at 1030 cm−1 assigned to primary alcohol. The intensity of these bands significantly increased for the LM-OH relative to those of the LM. In addition, bands corresponding to amino functions are almost completely disappeared. These results show that the LM has reacted with the formaldehyde.
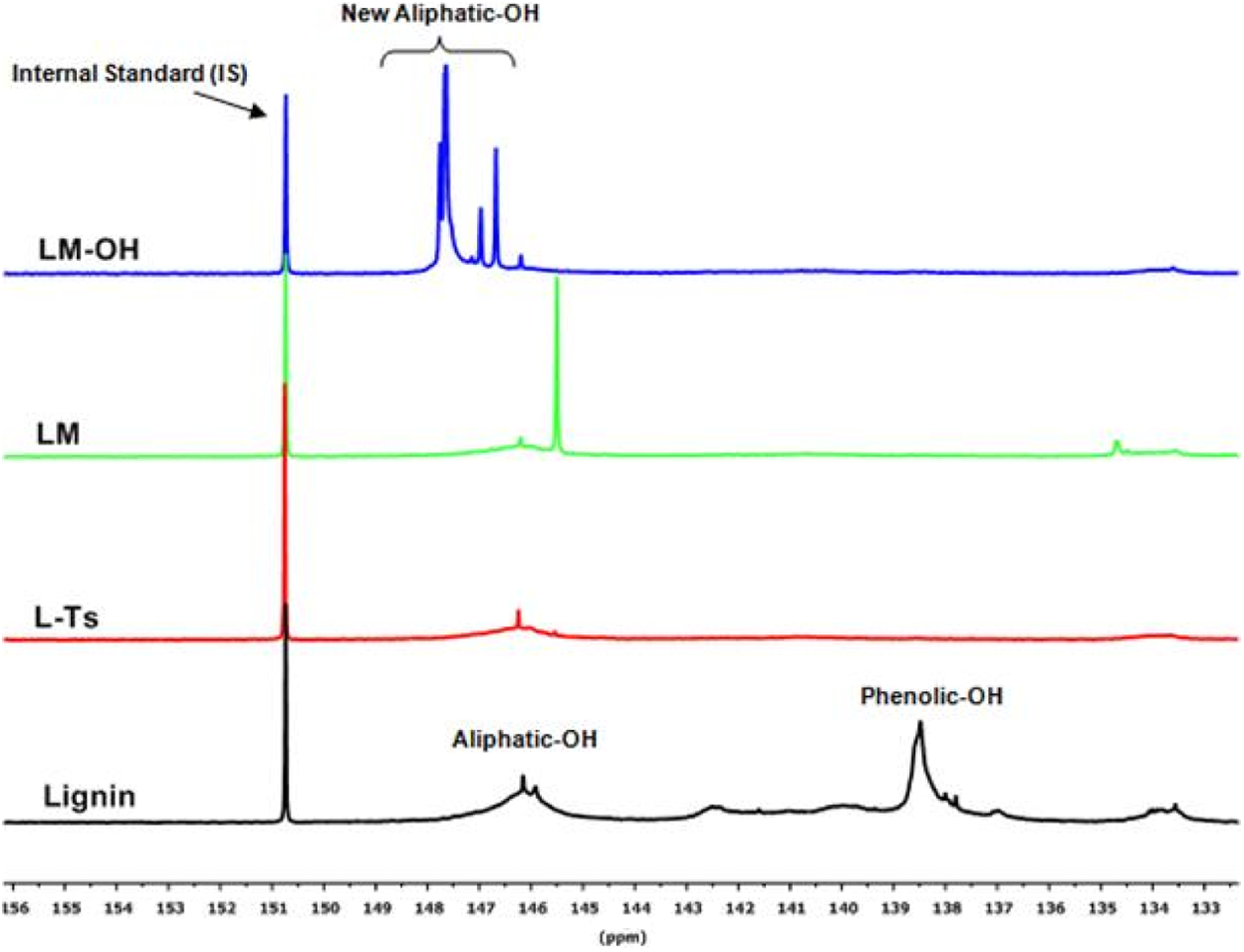
31P NMR spectrum of the unmodified lignin, L-Ts, LM, and LM-OH. 31P NMR: phosphorous-31 nuclear magnetic resonance; L-Ts: tosylated lignin; LM: lignin-melamine; LM-OH: methylol lignin–melamine.
31P NMR analysis
Tosylation of lignin, LM, and methylolation of LM were monitored and analyzed by quantitative 31P NMR. The 31P NMR spectrum, with the signal assignment, of the unmodified lignin, L-Ts, LM, and LM-OH are presented in Figure 3. The hydroxyl content of the analysis data of each product is shown in Table 1.
Phenolic, aliphatic, and carboxylic hydroxyl groups (in mmol g−1), present in lignin, L-Ts, LM, and LM-OH using 31P NMR in pyridine/CDCl3 after phosphitylation.
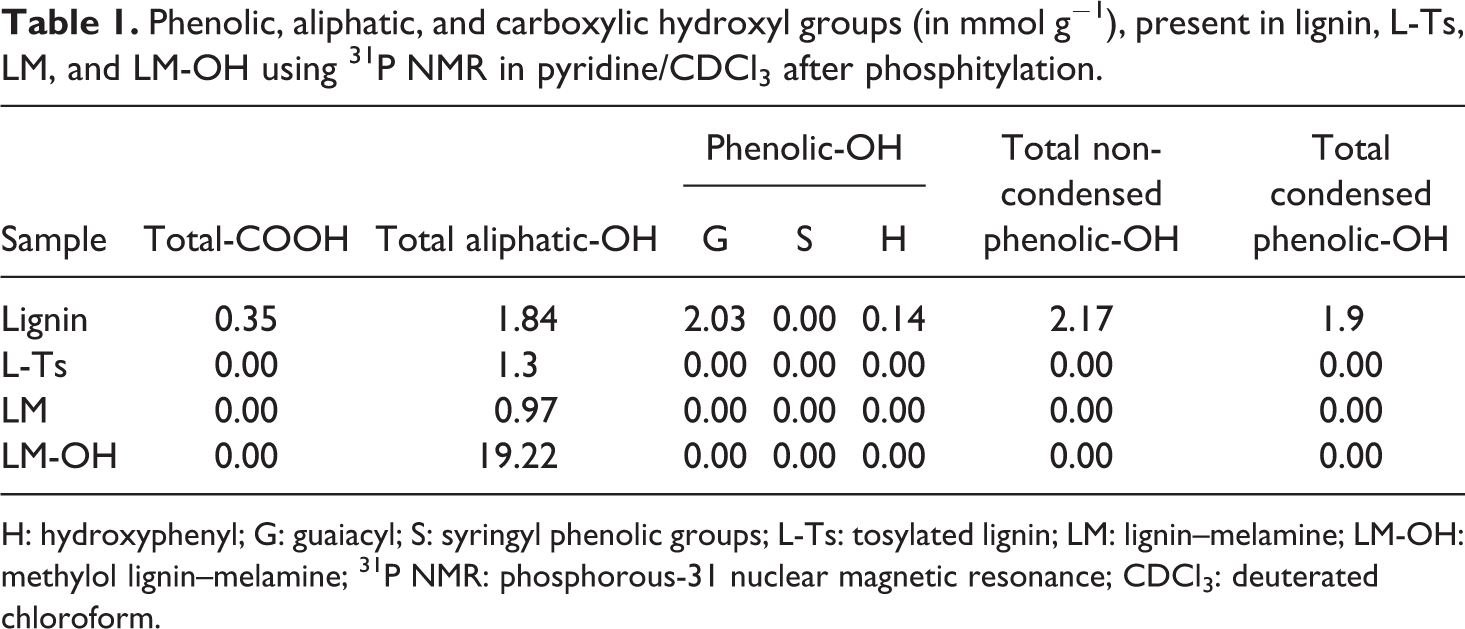
H: hydroxyphenyl; G: guaiacyl; S: syringyl phenolic groups; L-Ts: tosylated lignin; LM: lignin–melamine; LM-OH: methylol lignin–melamine; 31P NMR: phosphorous-31 nuclear magnetic resonance; CDCl3: deuterated chloroform.
Examination of the spectra reveals significant differences between the OH group distributions of the four samples.
The unmodified lignin contains several functional groups, of which phenolic hydroxyl and aliphatic hydroxyl groups are more important for the reactivity of the lignin. After tosylation, L-Ts contains almost aliphatic hydroxyl group (1.30 mmol g−1). One-third of aliphatic hydroxyl groups reacted with TsCl and all the phenolic hydroxyl groups reacted. The observed reactions of lignin can be rationalized on the basis of the large nucleophilicity differential of the aliphatic hydroxyl groups of lignin compared to their phenolic counterparts. The yield of tosylation is 80%.
The 31P NMR spectrum of L-Ts is very similar to that of LM. This resemblance is due to absence of any new hydroxyl after substitution of tosyl groups with melamine. Indeed melamine simply substituted the tosyl group to give the LM copolymer.
As shown in Figure 3, two distinct broad signals appeared in the aliphatic region of the 31P NMR spectra between 147.0 ppm and 148.5 ppm. While the peak at around 146.0 ppm is responsible for the aliphatic hydroxyl groups originally present in lignin, the relatively sharper peak observed at around 147.0 ppm and 148.5 ppm are attributed to the new aliphatic hydroxyls groups formed by the methylolation of LM. Indeed, these new hydroxyl are obtained by addition reaction between the amino group of melamine fixed on the lignin and the carbony1 group of formaldehyde at the pH value of 9.0. A molecule of melamine fixed on lignin has potentially five active sites on its chemical structure and can react with up to five molecules of formaldehyde to form pentamethylol LM. The amount of the aliphatic hydroxyls of LM-OH (19.22 mmol g−1) shows that at least four sites of melamine reacted with formaldehyde (Figure 1(c)).
Differential scanning calorimetry
Figure 4 shows the DSC thermograms of an LMF resin at two heating rates (5 and 20°C min−1). The initial cured temperature (T i), peak temperature (T p), final temperature (T f), and activation energy (E a) for LMF resin at 5 and 20°C min−1 are summarized in Table 2. The DSC thermograms exhibit one exotherm, indication of one step of reaction occurring during the thermal curing process. This result is different from most of the data found in the literature. Merline et al., 18 Anderson et al., 19 and Manfred et al. have shown that cross-linking of the MF resins is done in two steps (two exotherms). The reversible demethylolation reaction, which releases free amine was also observed to be dominant at the cure temperature range of 140–160°C. When the cure temperature was higher than 160°C, the cross-linking reaction dominated the curing process. This difference could be explained by a stabilization of methylol groups by the lignin.
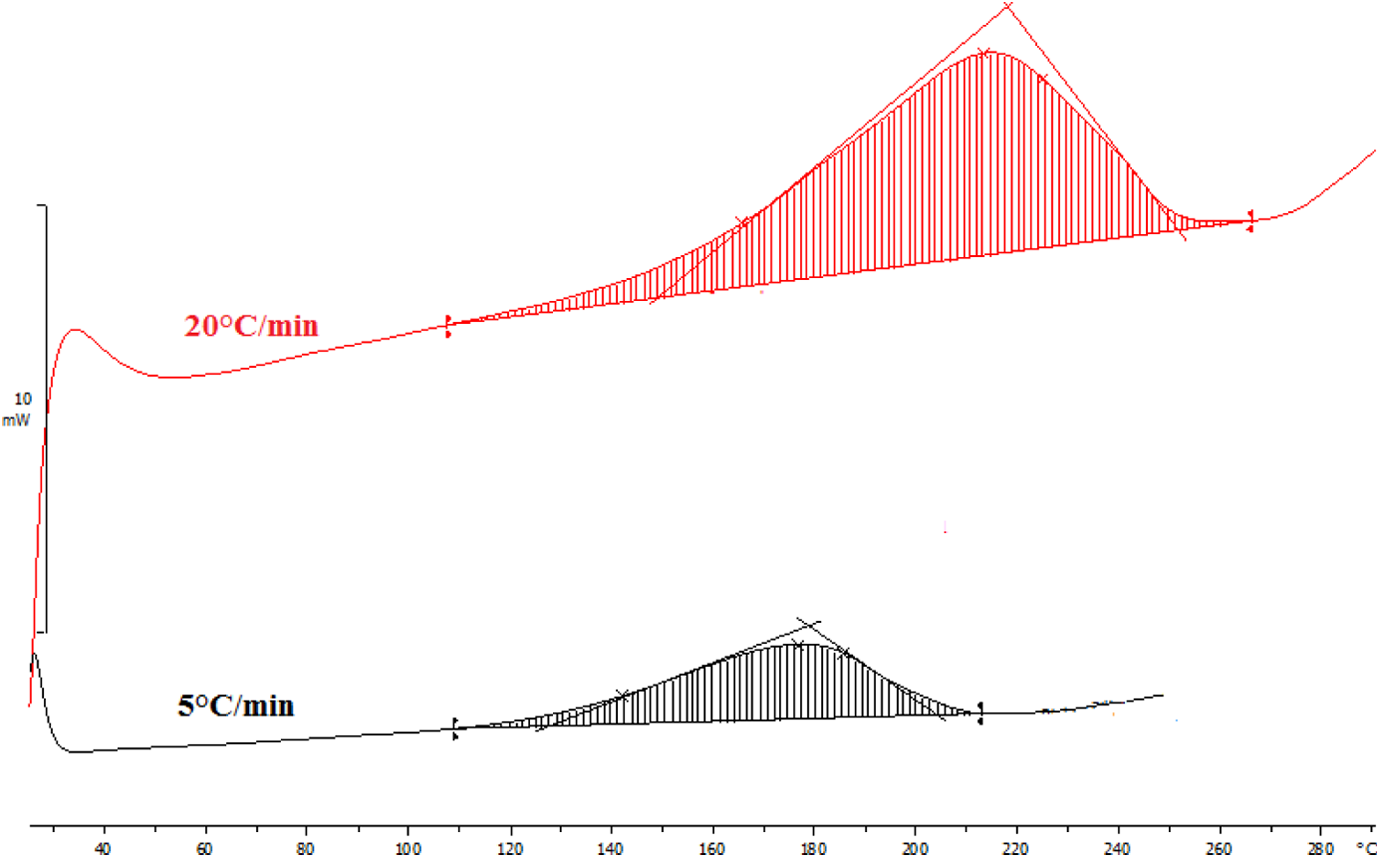
DSC thermograms of LMF resin at different heating rates (5 and 20°C min−1). DSC: differential scanning calorimetry; LMF: lignin–melamine–formaldehyde.
Variation of T i, T p, T f, and E a for LMF resin at different heating rates.
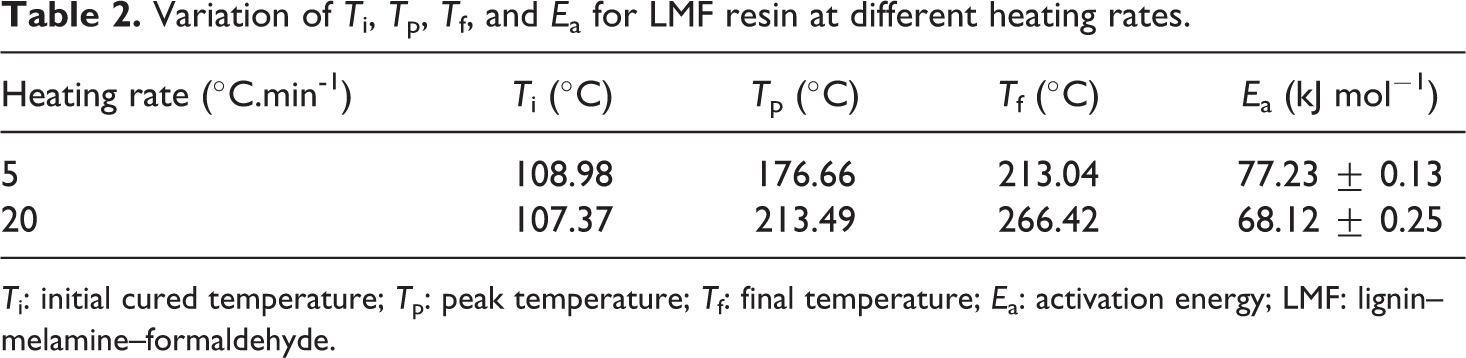
T i: initial cured temperature; T p: peak temperature; T f: final temperature; E a: activation energy; LMF: lignin–melamine–formaldehyde.
The start of crosslinking (T i) is near to 110°C. The curing reaction can be carried out at 110°C over long periods of time. At this temperature, the reaction can be incomplete and can give a low glass transition temperature of cured LMF resin. The recording performed at high-speed heating provides complete cross-linking enthalpy slightly lower than that obtained at 5°C min−1. The E a decreased from 77 kJ mol−1 to 68 kJ mol−1 as the heat rates increase. This is attributed to the fact that the viscosity of the resin decreases, which enhances the mobility of reactive groups. The peak was attributed to the condensation reaction of LM-OH. In addition, with the formation of LM-OH, methylol groups may interact with one another and form dimethylene ether links with liberation of water (Figure 5).
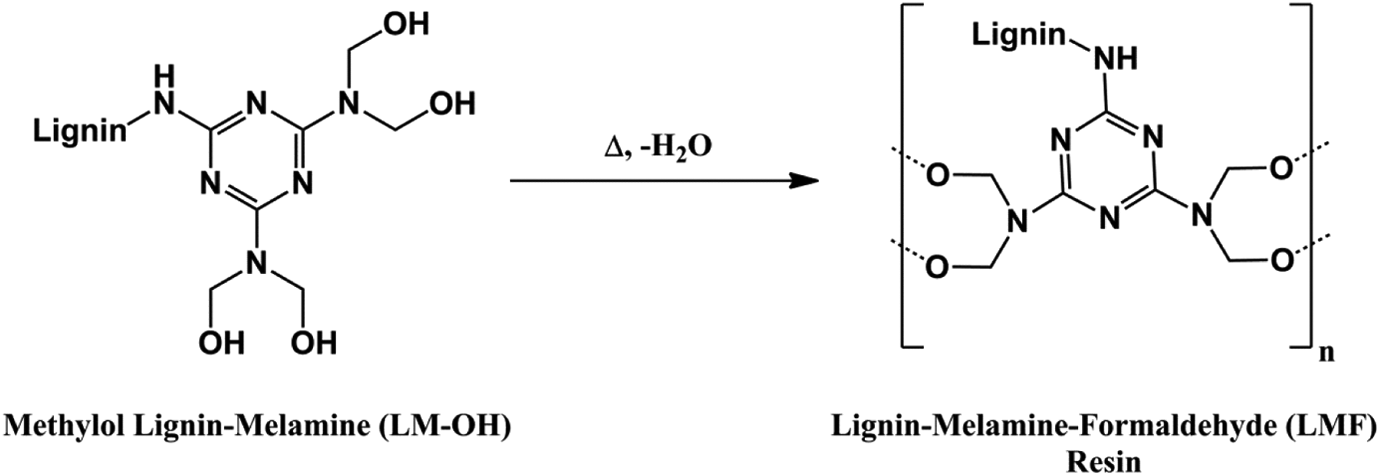
Cross-linking of the LMF resin. LMF: lignin–melamine–formaldehyde.
Thermogravimetric analysis
The heat stability of LM-OH and LMF cross-linked at 200°C are presented in Figure 6.
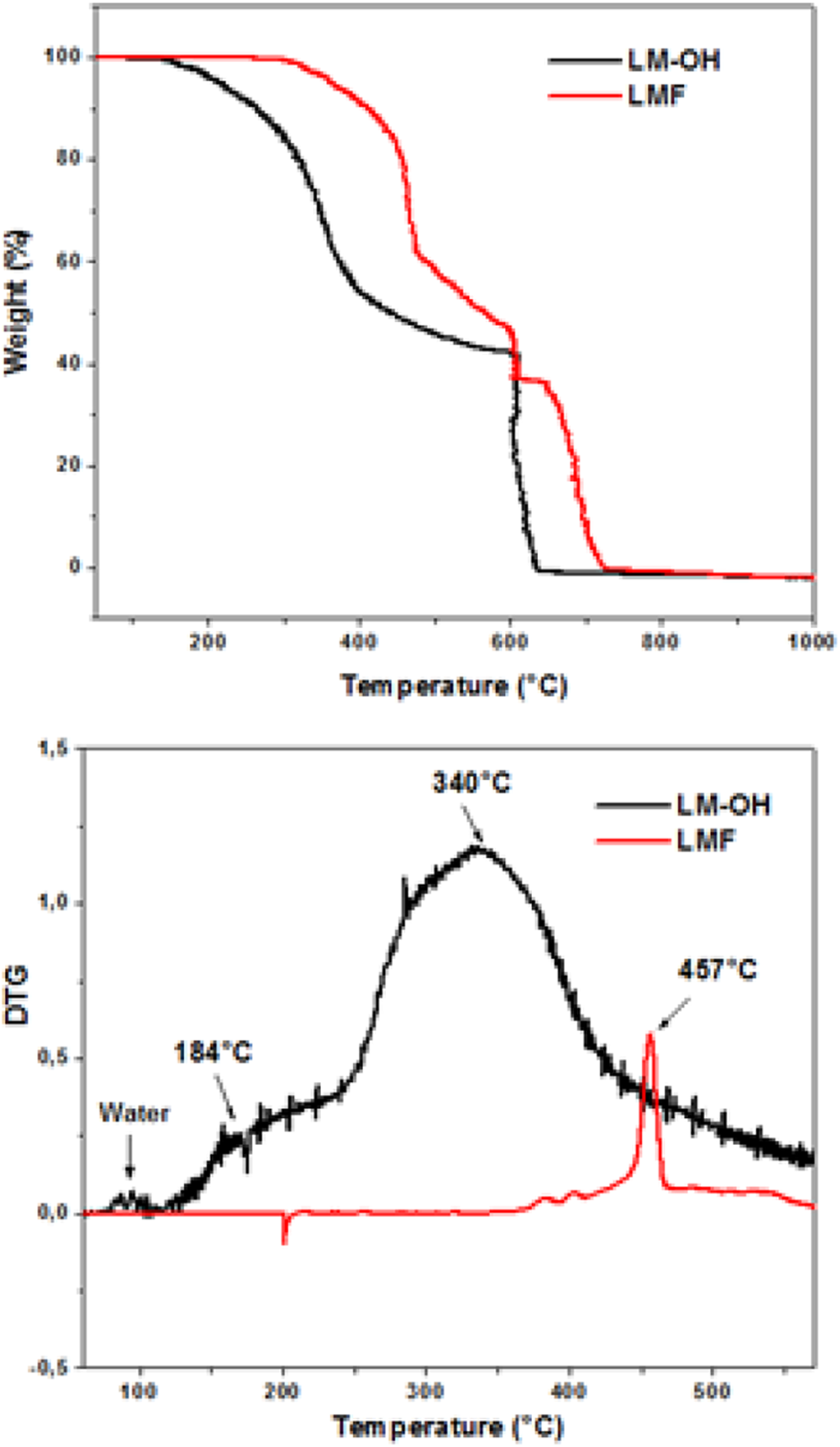
TGA and DTG curves of LM-OH and cross-linked LMF. TGA: thermogravimetry; DTG: derivative thermogravimetry; LM-OH: methylol lignin–melamine; LMF: lignin–melamine–formaldehyde.
The thermogram of LM-OH depicts the two steps of decomposition in the temperature range 25–700°C. The first step of decomposition starts from 125°C to 250°C, which represents the degradation of the small fragments of lignin and their disappearance in the thermogram of LMF is explained by their degradation during curing. The second step of decomposition of LM-OH starts from 300°C to 380°C. The mass loss in this second part is attributed to the degradation of the structure of LM-OH.
The TGA of the curves shows that the LMF is more stable than the LM-OH. Indeed the TG curve of cross-linked LMF is largely shifted to higher temperatures relative to that of the LM-OH. This increase in thermal stability is due to cross-linking of the methylolated melamine, which are fixed to the lignin. The derivative thermogravimetry (DTG) curves defined the rate of weight loss, while the DTGmax represented the maximum rate of degradation and can be used to compare the heat stability of the samples. The DTGmax of LM-OH and cross-linked LMF, respectively, are equal to 340°C and 457°C. These DTG values confirm that LMF cross-linked is more stable than the LM-OH.
Conclusion
The synthesis of a LMF resin from the copolymer LM and formaldehyde was completed successfully. The disappearance of the bands at 1370 and 1172 cm−1 and the appearance of bands at 3115, 3312, 3415, and 3470 cm−1 show that the lignin is linked with melamine. The LMF resin solidified upon heating with one exothermic peak at 160–210°C. DSC analysis showed that the LMF resin cross-link without demethylolation. The synthesis of LMF resin from the copolymer LM thus stabilizes the methylol groups during cross-linking. The TG curve of cross-linked LMF is largely shifted to higher temperatures relative to that of the LM-OH. This result confirms the cross-linking of the resin LMF. The kraft lignin is promising as a partial replacement for melamine suitable in MF resin. In addition, copolymer LM could be used as a fire retardant material or to capture the metal ions.
Footnotes
Acknowledgements
We thank Simon Langlois of Dupont Industrial Biosciences (Canada) for providing the kraft lignin sample.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
