Abstract
A suspension of ultrasonically agitated Titanium dioxide micrometer-sized powder has been introduced into a poly(methyl methacrylate) (PMMA) matrix by solvent (CHCl3) mixing under slow mechanical stirring, and the resulting suspension was used to cast free-standing PMMA/TiO2 composite films. Filler contents from 0 wt% to 7.5 wt% were selected, since TiO2 particles could not be previously suspended in chloroform at higher concentrations by ultrasonic agitation. Optical and dielectric properties of composites were determined by ultraviolet (UV)-visible and dielectric spectrometry. In the UV region, 450% improvement of UV absorption was achieved in the TiO2/PMMA composite compared with neat PMMA. The structural properties and hole-free volume of the composites were determined by X-ray diffractometry and positron annihilation lifetime spectroscopy, respectively. The particle size and homogeneous dispersion of the particles in the polymer matrix were related to the hole-free volume of the PMMA.
Keywords
Introduction
Inorganic–organic hybrid composite and nanocomposite materials based on functional inorganic particles embedded into organic networks represent widely explored field of investigation. 1 Such hybrid materials join interesting characteristics of polymer such as flexibility, processability, and high thermal and mechanical stability with specific size-dependent properties of inorganic nanocrystalline oxides. Fabrication of hybrid materials can be accomplished by dispersing inorganic fillers in host polymer, “in-situ” synthesis of particles in polymer matrix or reaction of polymer precursor in the presence of inorganic fillers. 2 Poly(methyl methacrylate) (PMMA) is an amorphous thermoplastic and widely used as a host material in hybrid composites. 1 -9 PMMA has specific electrical properties with high resistance and low dielectric constants. Therefore, it is widely used in high-voltage applications as dielectric film. 1 Besides, it transmits light in the range of 360–1000 nm and resists oxidative photodegradation.
Inorganic oxides with high refractive indices are generally characterized by poor workability; however, lightweight, flexible, and good processable organic materials have lower refractive indices than inorganic materials. 3 TiO2 has attracted a high research interest due to photochemical activity and optical properties. 4 It has also high dielectric constant. As an inorganic filler in the polymer matrix, it can increase the dielectric constant of composite compared with the neat polymer. Because of wide variety applications of research interests, many researchers have studied PMMA/TiO2 hybrid materials to improve their thermal and mechanical 5,6 surface properties, 7,8 sensor and battery application, 2,9 and optical and dielectric properties. 3,10 -14
Agglomeration of inorganic fillers and particle-size distribution in polymer matrix prevent the improvements of composite properties. The agglomeration problem can be handled by using different techniques such as ultrasonic agitation, polymerization, and solution mixing condition. Ultrasound-assisted emulsion polymerization and ultrasonic agitation of inorganic fillers were well described.
15
One can also change the solution mixing conditions such as temperature, mixing duration, or speed. Agglomeration of inorganic fillers can also cause a change in the “free volume” of the polymer matrix. For this reason, it is important to determine the free volume in the polymer matrix. The free volume model in polymers may give an explanation about some significant physical quantities such as viscosity, ionic conductivity, glass transition temperature, and mechanical and thermodynamic properties. Bamford et al.
16
and Cohen and Turnbull
17
interpret the transport phenomena in polymers in terms of the free volume arisen from structural disorders in amorphous polymers. On the other hand, the size of the hole-free volume and its intensity are measured by ortho-positronium (o-Ps) lifetime (
The aim of this study is to obtain the structural and dielectric properties of PMMA/TiO2 composites prepared by ultrasonic agitation and low mixing speeds. The structural and dielectric properties of PMMA/TiO2 composites were determined and free volume of PMMA with TiO2 in different weights (10, 30, 50, and 75 mg) was studied under vacuum in the temperature range from 293 K to 373 K. Furthermore, a relation between optical and dielectric properties as well as the hole-free volume and free volume fraction of the samples was presented for each TiO2 proportion.
Experimental
Materials
PMMA, from Alfa Aesar (molecular weight = 120,000), was dissolved in 25 mL chloroform (Merck) for 3 h. TiO2 powders (anatase phase), selected in micrometer size (<33 µm), in 25 mL chloroform were subjected to ultrasonic agitation for 4 h. Then, TiO2 solutions were added to polymer solutions and mechanically stirred at a slow stirring rate (60 r min−1) for 30 min.
Slow stirring rate and duration were selected to prevent the agglomeration of TiO2 particles in the polymer matrix. In all samples, PMMA was 1 g and TiO2 masses were chosen to be 10, 30, 50, and 75 mg, respectively. Over the 100 mg of TiO2, TiO2 particles were not well dispersed in the ultrasonic agitator. Therefore, the maximum TiO2 mass was selected as 75 mg. All solutions were cast on Petri dishes to obtain composite films.
Characterization
Ultraviolet (UV)-visible transmittance spectra of pure PMMA and its composites were taken by PerkinElmer (United Kingdom) Lambda 35 (190–1100 nm) UV-visible spectrometer. Dielectric spectroscopy studies of composites were carried out by NovoControl (Germany) Alpha-A Dielectric, Conductivity and Impedance Analyzer (1 Hz to 1 MHz).
Real (∊′) and imaginary (∊″) parts of the dielectric constant were calculated by
where C 0 is the vacuum capacitance, C p is the capacitance, and tan δ is the loss factor. The real part (∊′) is related to the stored energy by the external field and the imaginary (∊″) part (dielectric loss) is related to loss of energy.
The PALS measurements were carried out using a fast–fast conventional coincidence system measuring the spectrum of time interval between prompt γ-ray, as a start signal of positrons accompanied by a γ-ray of 1274 keV, and subsequent γ-ray, as a stop signal due to the fact that positrons annihilated with electrons from the sample resulting in 511 keV of γ-rays. The positron source was prepared by depositing about 25 μCi of 22NaCl aqueous solution on a thin aluminum foil (5 µm thick) and sandwiched between two sets of samples. Each set of samples was kept about 2 mm thickness piling up three films of samples. Each film thickness is about 0.7 mm.
The plastic scintillators mounted onto Hamamatsu (Photonics Deutschland GmbH, Germany) 2059 photomultiplier tubes based by Ortec-265 (AMETEK GmbH, Meerbusch, Germany) operated at negative 2050 V have been employed for γ-ray detection. For windows settings of 1274 and 522 keV and timing signals, two constant fraction differential discriminators (Ortec CFDD 583B) were used. A time to amplitude converter used to convert pulses of different heights to a time-to-pulse-height signal was fed to a multichannel analyzer (Ortec Model 919E Ethernim MCA). The spectroscopic data yielded from Multi-Channel Analyzer were analyzed by using the code of Life Time-polymer 19 to obtain the lifetime and its intensity revealing the information about the free volume. The resolution of the system is about 350 ps and a million counts have been taken for each run.
Kobayashi purposed the free volume fraction to be proportional to the product of hole-free volume calculated from o-Ps lifetime and o-Ps intensity. It is supposed to be related with the hole fraction as a measure of free volume computed by the Simha–Somcynsky (SS) hole theory. 20 The SS theory, based on the lattice-hole model, is formulated in terms of reduced pressure, volume, and temperature variables with the reduced characteristic parameters of each variable. 20
Results and discussion
UV-visible study
Figure 1 shows the UV-visible absorbance spectrum of neat PMMA film. There was intense absorption below 260 nm in the UV region which is a typical characteristic of PMMA. Figure 2 shows the UV-visible absorbance spectra of TiO2/PMMA composites. The absorbance onset shifted to the higher wavelengths with an increase of TiO2 compared with neat PMMA. The absorbance was also increased with an increase of TiO2 content. Maximum absorbance (4.5%) was observed in 50 mg TiO2-loaded composite in the UV region. Interestingly, the absorbance of 75 mg TiO2 content which was found to be the limit of good dispersion in the solvent by ultrasonic agitation is lower than the absorption of 50 mg TiO2 content (UV region). In the UV region, 450% improvement of UV absorption was achieved in the TiO2/PMMA composite according to the neat PMMA. Similar results were observed in TiO2/PMMA nanocomposites. 5,21 Increase in the absorption was related to the chemical interaction between TiO2 and PMMA. 5 In this region, the reduction in the absorption value for the 75 mg TiO2 containing composite can be related to a decrease in the chemical interaction between TiO2 and PMMA. The reason for the loss of absorbance for this filler rate (7%, weight/weight (w/w)) may be that the TiO2 particles are agglomerated in the polymer matrix and thus the hole fraction in the polymer matrix may have increased. In the visible region (400–700 nm), the absorbance values were also increased by an increase in the TiO2 content. Low filler rates (7% w/w), low solution mixing speed (60 r/min−1), and ultrasonic agitation of micro-sized fillers before the composite preparation can improve the UV absorption properties of the TiO2/PMMA hybrid materials.

UV-visible spectrum of neat PMMA film.
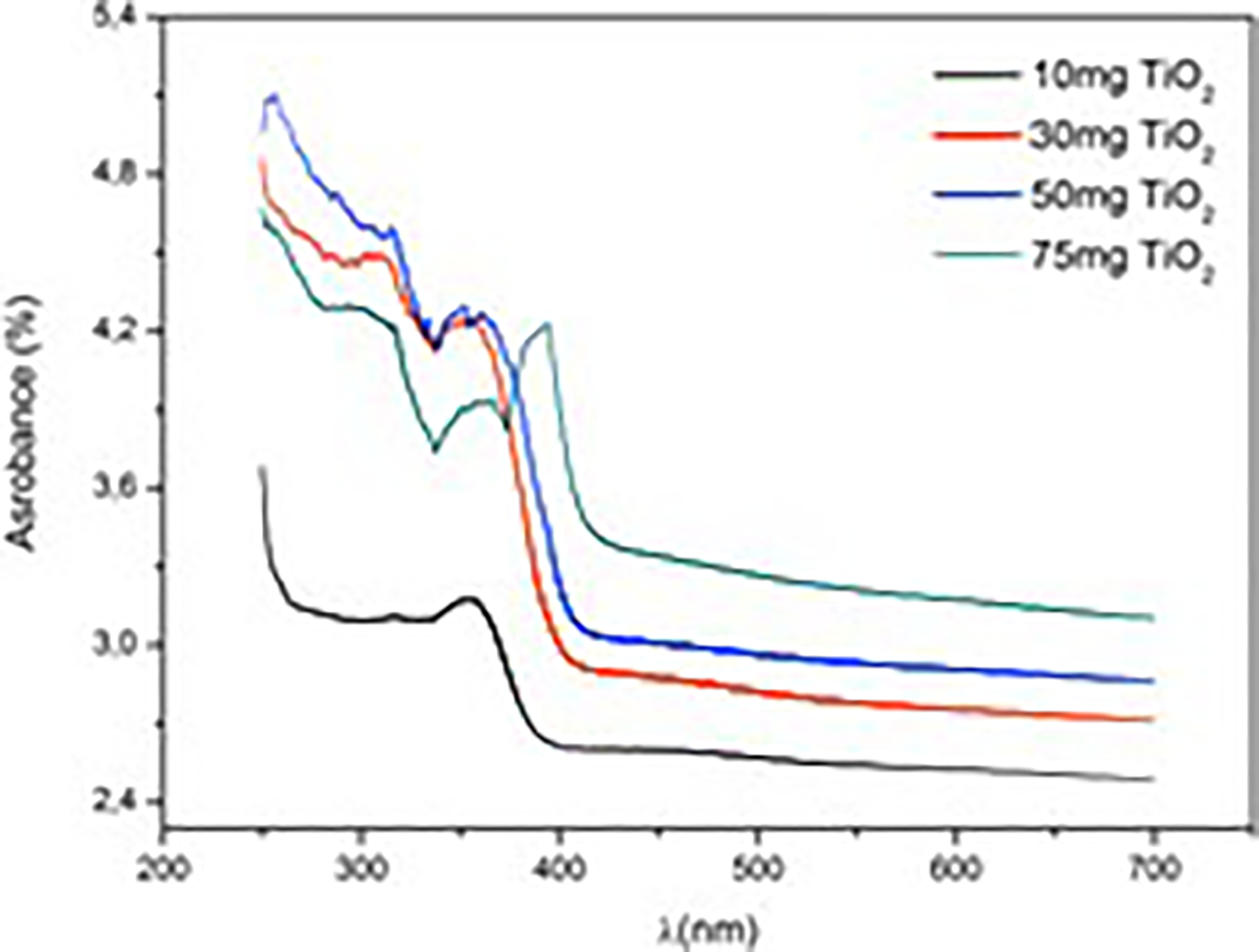
UV-visible spectra of TiO2/PMMA composites.
X-ray Diffractometer
Figure 3 shows X-ray Diffractometer spectra of TiO2 and PMMA. In this figure, sharp peaks, especially at high-angle values, represent crystalline regions. At small angles, broadened peaks with low amplitude represent scattering from amorphous regions of PMMA. In Figure 3, a typical amorphous polymer behavior is also observed. The observed peak at 13.7° indicates the packing order of the polymer chains. 22 The observed peak at 29.5° represents the arrangement within the main chains, and this arrangement is observed to be rather limited due to its high level of broadening and low amplitude. 22 Similarly, X-ray diffraction of TiO2 is shown in Figure 3. The values of 2θ of the peaks and corresponding planes they are scattered in are given in Table 1. The peaks observed at 25° and 55° are an indication that TiO2 is in anatase form. The notable difference for the rutile form is the shift from 25° to 27–28°. Similar results for TiO2 have been pointed out in the literature. 10,12,23,24

X-ray diffraction of natural PMMA and TiO2.
X-ray diffraction maxima and corresponding planes of TiO2 (anatase structure).

Figure 4 shows the X-ray diffractograms of TiO2/PMMA composites. TiO2 peak intensities are very strong in 30 and 50 mg TiO2-doped composites. In 10 and 75 mg TiO2-doped composites, these peaks are weakened. The peak at 13.7° (Figure 4) in powder samples of pure PMMA has shifted to 17° in the film sample. This behavior can be explained by the formation of the crystalline phase, which is observed in very small amounts in the powder sample. In the 30 and 50 mg TiO2-added composites, the TiO2 peak (d101), observed at 22° compared to the other composites, is due to the amorphous behavior of PMMA. 12 The traces of the amorphous behavior of the polymer are explained by the shift of the peak (17°) seen in the PMMA film sample to the lower 2θ values in 10 and 75 mg TiO2-loaded composites. In the 10 and 75 mg cases, this peak was weakened and broadened. The particle size and homogeneous dispersion of the particle in the polymer matrix can also be related to peak intensities and peak areas. 25 In the 30 and 50 mg TiO2-added samples, the peak observed at 25° (d101 plane of TiO2) is sharp. Thus, it can be concluded that the filler grain size is low and the homogeneity is high in the 30 and 50 mg TiO2-added composites where severe peaks are observed compared to other composites.
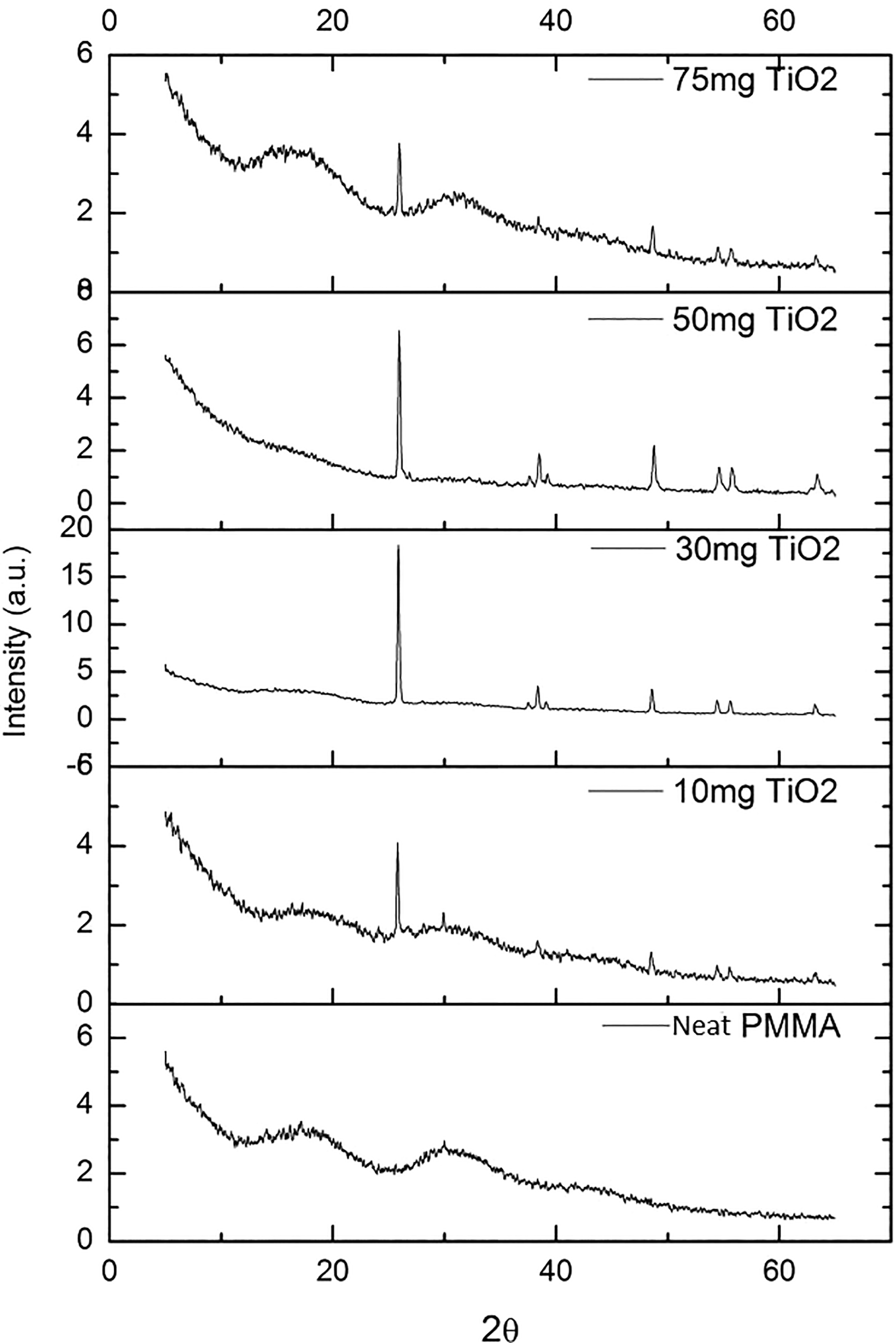
XRD diffractograms of PMMA/TiO2 composites.
Dielectric study
Figure 5 shows the dielectric permittivity of neat PMMA and TiO2/PMMA composites. It can be seen that dielectric permittivity was increased with an increase of TiO2 content in the low-frequency region. In the high-frequency region, an increase in dielectric permittivity was also observed, but 50 mg TiO2-filled composite has the lower dielectric permittivity. Maximum dielectric permittivity which is 25% higher than neat PMMA was observed in 75 mg TiO2-filled composite.
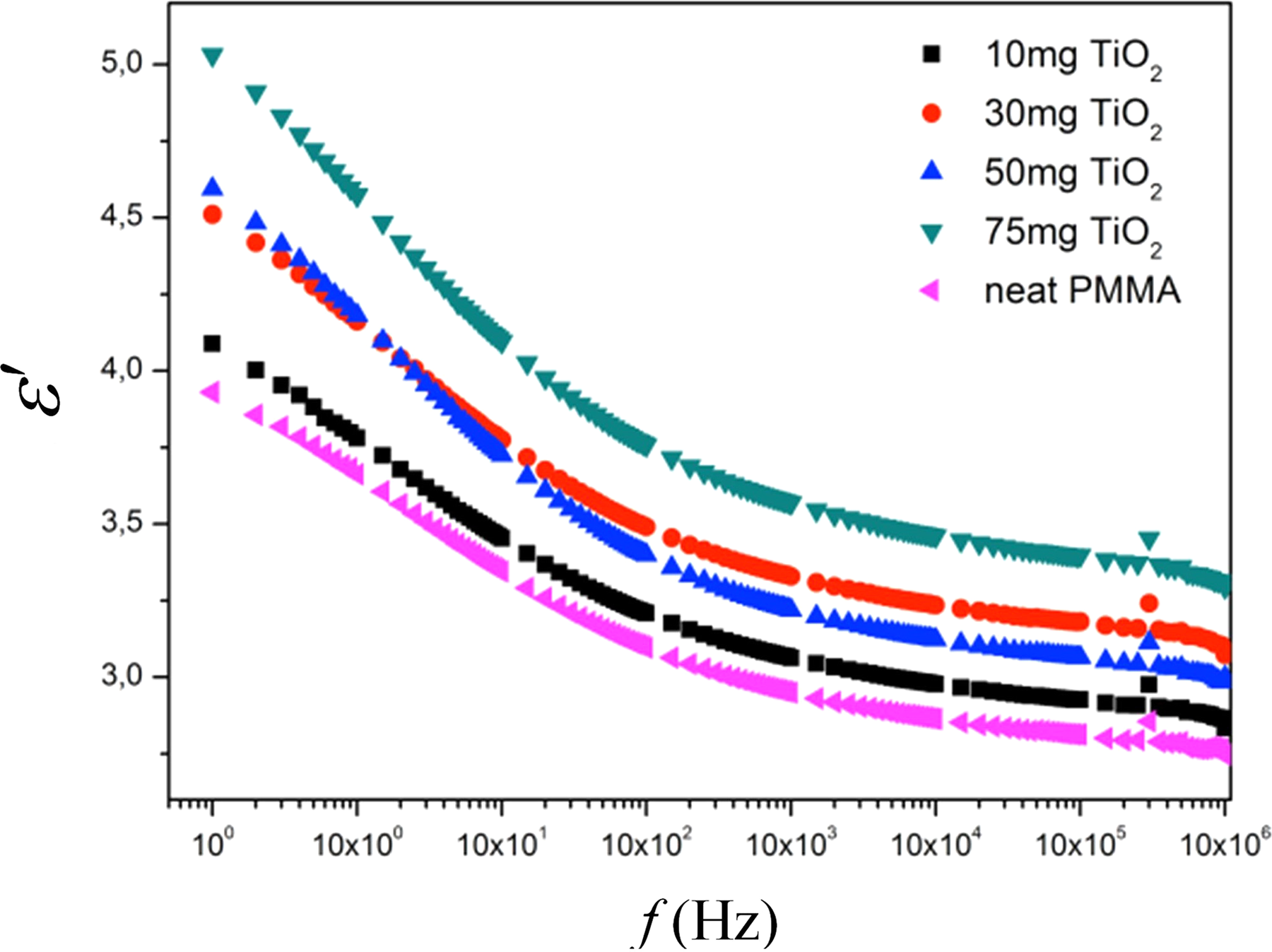
Dielectric permittivity neat PMMA and TiO2/PMMA composites.
Figure 6 shows the loss factor (tangent loss) of neat PMMA and TiO2/PMMA composites. Relaxation peaks were observed in the low-frequency region (<40 Hz). The relaxations were slightly shifted to lower frequencies with an increase of TiO2 concentration. Loss factors were increased with an increase of TiO2 concentration and interestingly maximum loss factor values were observed in 50 mg TiO2-filled composite. Similar behaviors were observed in some studies. 10,11,26

Loss factor of neat PMMA and TiO2/PMMA composites.
It was reported that both dielectric permittivity and loss factor increase with an increase of oxide fillers. Higher tan (δ) at low frequencies is generally attributed to the higher current leakage in the system. In the case of TiO2 nanocomposites, an increase in tan (δ) was observed below 10 Hz that could be a sign of an increase in electrical conductivity because of the higher possibility of charge carriers to reach the surface of the electrodes. 26 In our study, similar results were observed.
TiO2 particles having high surface energy cause a high particle–particle attraction resulting in the aggregation of TiO2 particles. This aggregation can cause the incompatibility between the TiO2 particle and polymer chain. Free volume increases at the interface between particle and polymer. 26 The decrease in dielectric constants with increasing free volume is the origin of electronic polarizations. 27 On the other hand, the increase in the dielectric constants with increasing free volume is the origin of the dipole orientation. 27,28 Anomaly in a decrease of dielectric permittivity for 75 mg TiO2 content can be explained by this phenomenon; 75 mg TiO2-added composites have low free volume value with respect to 30 and 50 mg TiO2-filled composites. In composites filled with 30 and 50 mg TiO2, the homogeneity of the particles is stable. With the increase of the TiO2 content in the PMMA matrix (75 mg), the particles are aggregated in the matrix and the homogeneity is degraded. The contribution of the metal oxide to the composite dielectric constants is limited by the agglomeration of the oxide particles in the polymer matrix. Thus, the maximum filler rate of TiO2 to PMMA is around 5% (w/w).
The scaling parameters of the SS theory and the hole fraction
The experimental Presure-Volume-Temperature data for PMMA polymer for about 100 kg molar masses are taken from 29.7°C to 230.4°C temperature and from ambient to 200 MPa pressure by Zoller and Walsh.
29
The scaling parameters P*, V*, and T*, of the SS theory in the melt state from about 121°C to 220°C temperature and from ambient to 200 MPa pressure ranges are calculated as
PALS results
The longest-lived component, o-Ps lifetime (
o-Ps lifetime (

o-Ps: ortho-positronium.
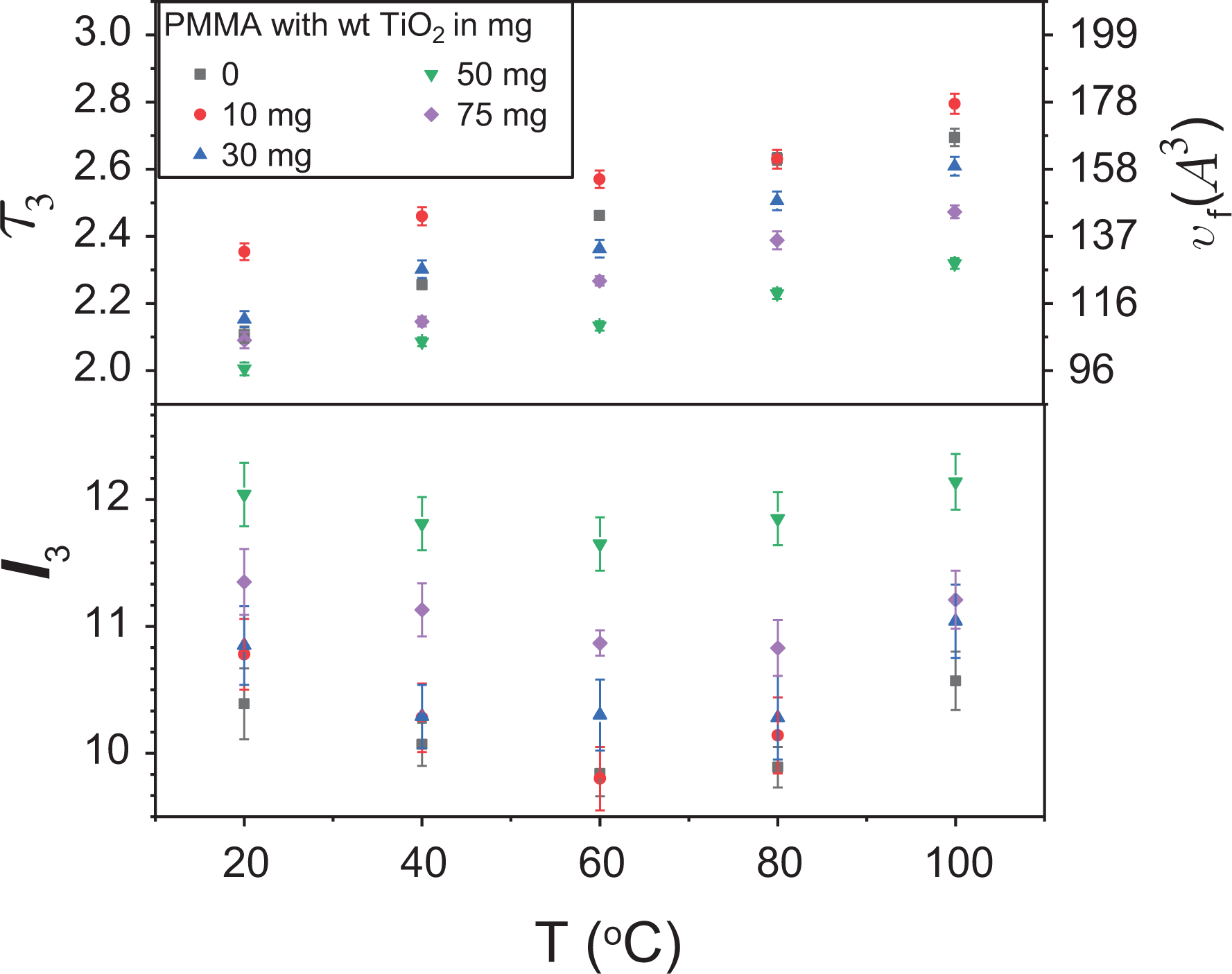
The o-Ps lifetime with the corresponding hole-free volume and intensity versus temperature for some different weight of TiO2 added to PMMA.
Similarly, at 20°C, 60°C, and 100°C, the o-Ps lifetime and intensity are plotted with respect to TiO2 content in Figure 8. Increasing the TiO2 content, the o-Ps lifetime (or the hole-free volume) increases at 10 mg TiO2 additive, and then it has a decrease up to 50 mg and a slight increase at 75 mg. Unlike the lifetime, the change of intensity (I 3) behaves opposite to the lifetime. The decrease in the intensity may suggest a decrease in the holes’ numbers with increasing TiO2 additive since the additives occupy the preexisting free volume holes in the PMMA structure. On the other hand, the free volume hole size increases since the size of the TiO2 is much larger than the holes.
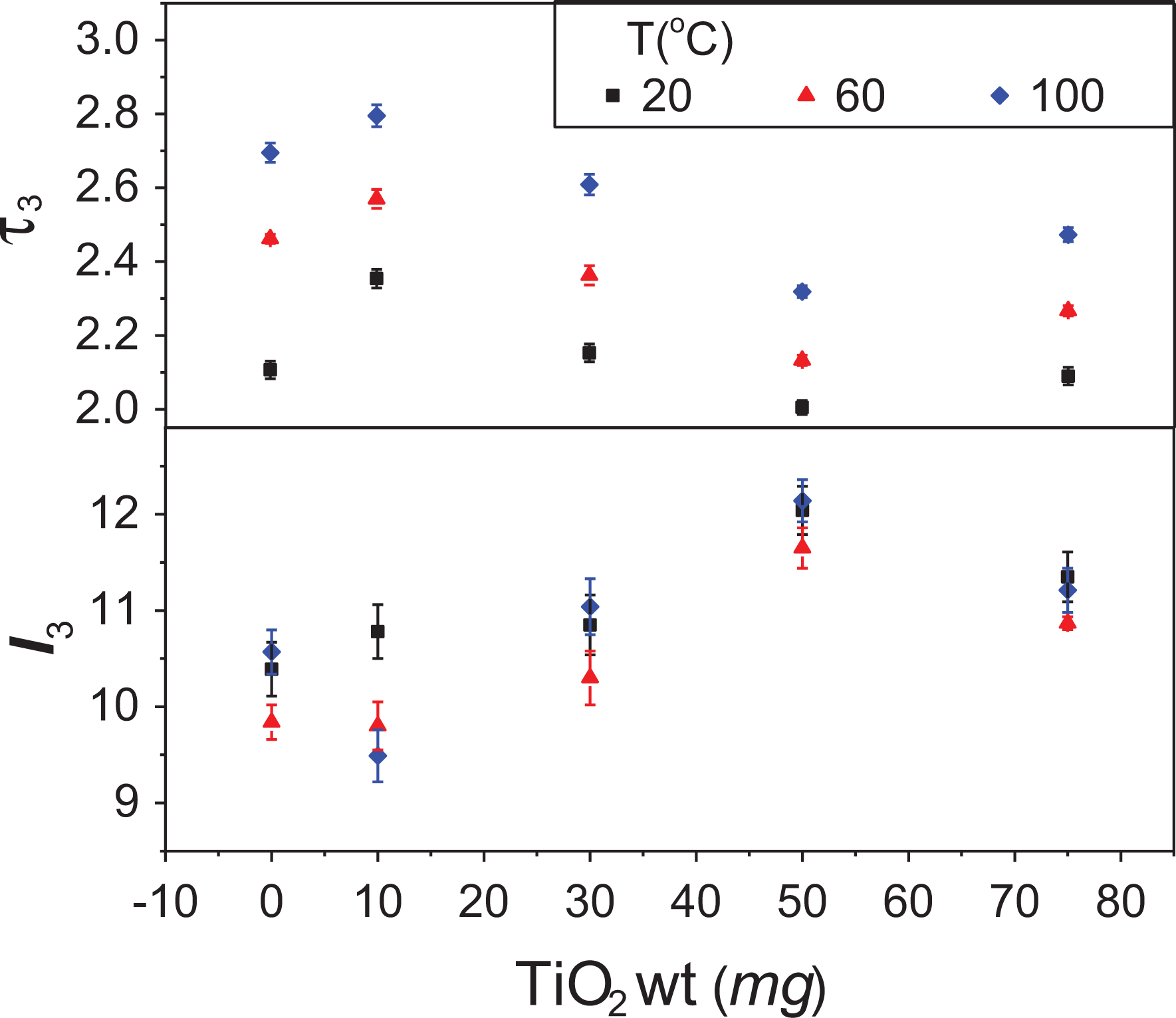
The o-Ps lifetime and intensity versus weight percentages of TiO2 added to PMMA for some temperatures of 20°C, 60°C, and 100°C.
The free volume fraction,

where A is a constant. Using the values of hole fraction at 20°C, 40°C, 60°C, 80°C, and 100°C calculated by means of the SS theory and then equating them to equation (2) with
In Figure 10, the free volume fraction (
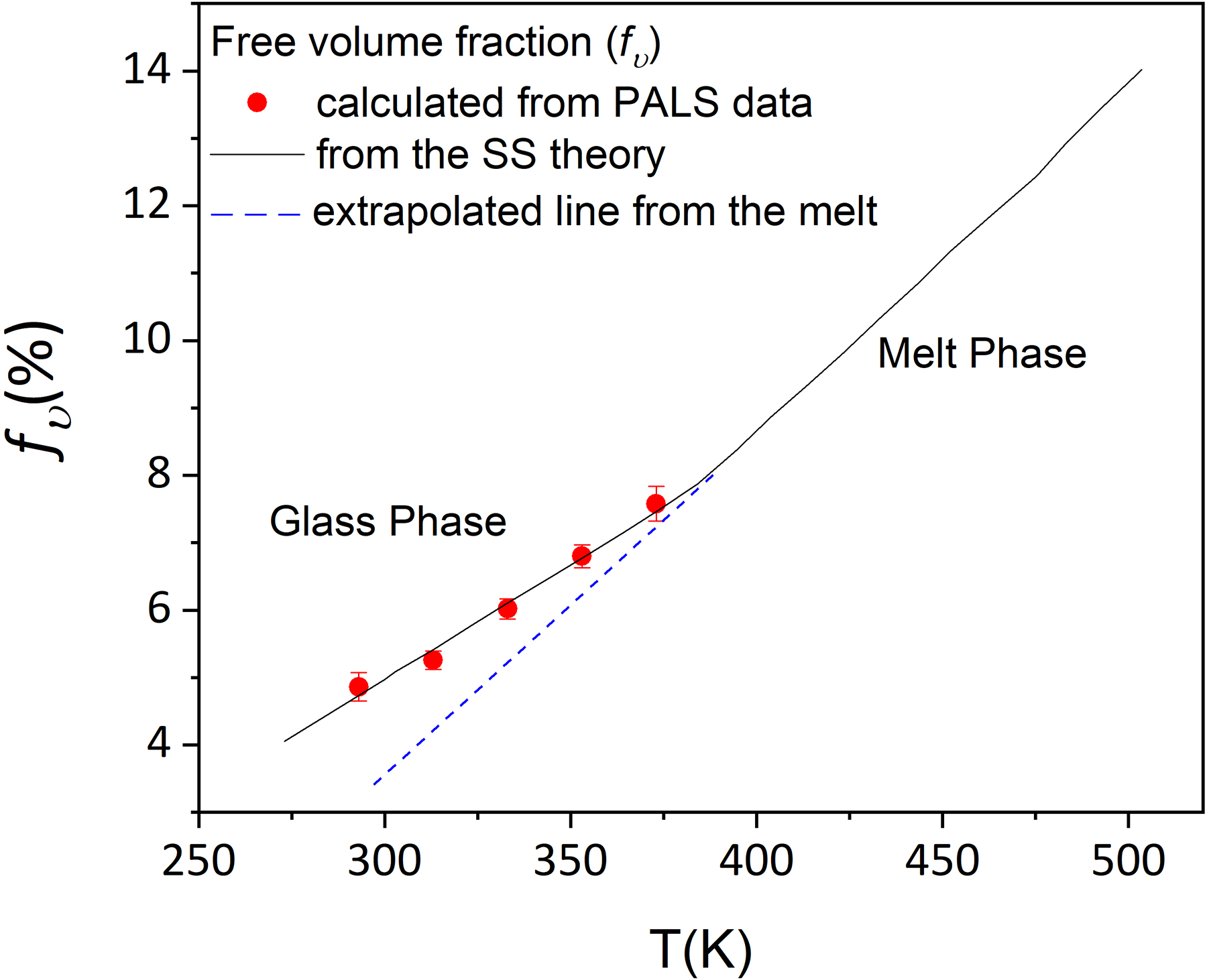
Free volume fraction of PMMA with respect to temperature is plotted. The solid line is the free volume fraction calculated from the SS theory. The red dots are calculated from experimental PALS data using equation (2).

The free volume fraction versus TiO2 added to PMMA for all the temperatures considered.
Conclusions
In this study, optical and dielectric properties of PMMA/TiO2 composites were investigated by selecting filler contents to be as 10, 30, 50, and 75 mg (<7 wt%). Since aggregation of fillers prevents the contributions to the composites, ultrasonic agitation was used for the TiO2 particles in chloroform. Four hours was selected as the best time for this filler masses. It was also selected low mixing duration (30 min) and speed (60 r min−1). Although the thick films were selected to investigate the optical properties of composites, in the UV region, 450% improvement of UV absorption was achieved in the PMMA/TiO2 composite according to neat PMMA. Dielectric permittivity was increased with an increase of filler concentrations. It was observed 25% improvement for dielectric permittivity with respect to neat PMMA.
Low free volume values in composites were associated with the doping rate and particle agglomeration and no contribution to dielectric constants was observed above 5% TiO2 doping.
As a result, desired improvements in optical and dielectric properties of the PMMA/TiO2 composites can be achieved by appropriate ultrasonic agitation process, low fillers contents, and low mixing speeds. Therefore, such hybrid materials can be used as a gate dielectric in organic field-effect transistors with desired dielectric permittivity or UV shielding materials with high absorbance in UV region and low transmission loss in the visible region.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

