Abstract
The effect of plasticizer on dielectric properties of poly(methyl methacrylate) (PMMA)/titanium dioxide (TiO2) composites was investigated. Propylene carbonate (PC) was used as plasticizer in the samples which were prepared with the conventional solvent casting technique. Scanning Electron Microscopy with Energy Dispersive X-Ray Analysis (SEM-EDX) and Differential scanning calorimetry (DSC) analyses and LCR Meter measurements (performed between 300 K and 400 K), were conducted to examine the properties of the composites. With the addition of plasticizer, the thermal properties have changed and the dielectric constant of the composite has increased significantly. The glass transition temperature of pure PMMA measured 121.7°C and this value did not change significantly with the addition of TiO2, however, 112°C was measured in the sample with the addition 4 ml of PC. While the dielectric constant of pure PMMA was 3.64, the ε′ value increased to 5.66 with the addition of TiO2 and reached 12.6 with the addition of 4 ml PC. These changes have been attributed to increase in amorphous ratio that facilitates polymer dipolar and segmental mobility.
Introduction
With its electrical, mechanical and host polymer properties, PMMA has been investigated in the literature for every aspect. Besides being easily processed, inexpensive, with ease of functionalization and a high carrier mobility, it has been the subject of research in many fields from aviation industry to microelectronics as well as basic science research.1-10 In addition to its optical properties, TiO2 has attracted considerable interest for researchers in transistors, capacitors, dye sensitive solar cells and microelectronic device applications because it is an inexpensive and nontoxic material.11-17 Although anatase, rutile and brookite phases are available for TiO2, anatase form which is metastable in a wide temperature range is the most used TiO2 types (antase phase is thermally unstable and turns into rutile phase above 600°C).11,12,18
Organic-inorganic composite dielectric materials provide many advantages with their mechanical and electrical properties. Polymer composites are mostly produced by mixing, blending, crosslinking polymeric materials and inorganic additives. Researches have shown that as a result of additives to the host polymer material, thermal, electrical and mechanical properties of polymer composites can be modified.19-24 In this context, composites obtained with PMMA and TiO2 are important for modifying their structural, chemical and electrical properties.
Another method for modifying the properties of polymer blend composites mentioned above is the addition of plasticizers to the structure. The addition of plasticizer has an effect on enhancing interfacial properties as well as increasing ionic conductivity in polymers.4,25-28 It is known that ionic conductivity is affected by factors such as anion and cation motion, ion pair formation, amorphous structure of the material, ambient viscosity and high dielectric constant. In this context, the plasticizer causes an effect that facilitates ionic conductivity among these factors. Plasticizers such as ethylene carbonate (EC), propylene carbonate (PC) and dimethyl carbonate (DMC) allow the ionic conductivity to be increased by increasing polymer chain flexibility and electrode/electrolyte interface stability. To put it briefly; besides the ion and polar groups mobility in the polymer chain, the most effective method of increasing the ion mobility in the structure is the addition of a plasticizer to the polymer composite structure. The addition of low molecular weight and high dielectric constant plasticizer produces effects such as lowering the glass transition temperature, increasing viscosity, elasticity, deformability as well as increasing conductivity. Increasing amorphous fraction and specific volume in polymers by adding plasticizer facilitates the movement of ions between polymer chains and thus increases ionic conductivity.4-6,25-27
In the present paper, the effects of varying concentrations of plasticizer (propylene carbonate (PC)) on the morphological, thermal and dielectric properties of PMMA/TiO2 composites have been investigated. The PMMA/TiO2 composite has already been well researched in the literature, but it is known that the addition of plasticizers such as propylene carbonate can result in different properties in materials without requiring too much effort and expense. For this reason, the properties of the composite obtained by adding PC to PMMA/TiO2 were examined. In order to characterize these features of the composites, SEM-EDX, DSC and dielectric measurements were used, respectively. It was observed that the addition of plasticizer had an effect on increasing ac electrical conductivity in PMMA/TiO2 composites and this effect was morphologically and thermally in relation to the results. Considering the study, it is observed that the results obtained are suitable for technological applications and academic interest purpose.
Experimental procedures
Materials
PMMA (Mw: 350000) was provided by Aldrich, TiO2 (anatase, 15 nm APS powder, 99.7%) was obtained from Alfa Aesar, acetonitrile (ACN, 99.9%) was bought from Merck and used without further purification. Propylene carbonate (PC, >99%) as a plasticizer purchased from Fluka.
Sample preparation
Films of PMMA, filled with TiO2 were prepared by the casting method.4-6 The concentration of PMMA (1.05 g) and TiO2 (0.45 g) in the solution was kept constant in all samples, which corresponds to 10% (by weight) of the total solution. The remaining 90% (wt) is shared between PC/ACN according to the changing PC ratio. An exemplary sample preparation procedure of casting method is given as follows: 1.05 g PMMA was dissolved in acetonitrile (15.2, 12.1, 11.66, 9.8 and 7.98 ml according to the composition) at 40°C and stirred until highly transparent and viscous gel obtained. Then, different amounts of propylene carbonate were added to the mixture (1, 2, 3, 4 and 5 ml). After obtaining a homogeneous mixture, TiO2 (0.45 g) was added to the mixture and the entire mixture continued to be stirred at 70°C. The resulting mixture was cast onto a petri dish and kept in a vacuum oven at 60°C for 96 h to remove the solvent traces.
Characterizations
DSC measurements were performed using a Seiko Electronics (Tokyo) DSC 7020 differential scanning calorimeter. All thermograms were performed in the temperature range from 0°C to 250°C at a heating rate of 10°C/min under nitrogen flux. Samples were heated then cooled and heated again to the determined temperature. Tg was recorded as the middle temperature in the curves of heat flow at the transition region (half height technique).
The surface morphology of the fabricated samples was investigated by scanning electron microscopy (SEM) using ESEM-FEG and EDAX Philips XL-30 microscope (Philips, Eindhoven, the Netherlands).
For dielectric measurements, the sample surfaces were covered with silver paste to form electrodes. The dielectric measurements were performed between 20 Hz and 1 MHz frequency range at different temperatures (between 300 K and 400 K at 10 K intervals) by using Agilent 4284A LCR Meter. The temperatures were controlled by using Oxford ITC 502 temperature controller. The real part of dielectric constant was calculated by using the following equation:
where, Cp is the capacitance of the sample, ε0 (dielectric permittivity in vacuum) is equal to 8.85 × 10−14 F.cm, A is the effective surface area, and d is the thickness of the samples. Ac conductivities (σac) of the samples were calculated with the following equation:
where
Results and discussion
Morphological analysis
SEM images obtained for all examples are given in Figure 1. Figure 1(a), consisting of pure PMMA, exhibits a smooth and porous film structure. On the other hand, when TiO2 was added to PMMA, it was observed that particles of TiO2 did not disperse between molecule chains and even discontinuities occurred in the bulk structure of the film (Figure 1(b)). However, when PMMA/TiO2/PC composites are examined (Figure 1(b) to (g)), it is understood from SEM micrographs that a uniform distribution occurs in PMMA.

SEM micrographs of (a) pure PMMA, (b) PMMA/TiO2, and (c to g) PMMA/TiO2/PC (1 ml, 2 ml, 3 ml, 4 ml, 5 ml, respectively).
In Figure 1(b), the roughness of the sample obtained by adding TiO2 to PMMA can be attributed to the increase in the amorphous ratio.13,29,30 This change becomes important when analyzed together with other analyses.
The addition of plasticizer caused the change of morphologies compared to pure PMMA and PMMA/TiO2 structures (Figure 1(c) to (g)). Since plasticizers are thought to act as a spacer by forming connections between the molecules in the polymer chain, it is common to obtain a smooth morphology in the resulting structures with increased amorphous ratio and correspondingly increased grain.4,25,26 When the morphologies of PMMA/TiO2/PC composites are examined, depending on the amount of PC in the sample (Figure 1(c) to (g)), it is observed that the PC content has the same effect on all samples. In samples containing 1, 2, 3 and 5 ml of PC, aggregate sizes are up to approximately 25 μm. However, it can be seen that TiO2 particles are dispersed more smoothly and there is a lower level of aggregation in 4 ml PC added sample.
EDX analysis was used to confirm the presence of TiO2 elements in the composites. Unlike Figure 2(a), Ti and O peaks are observed in the EDX analysis for PMMA/TiO2/PC (4 ml) sample (Figure 2(b)) and which proves the presence of TiO2 in the composites.31,32

EDX analysis of the sample: (a) pure PMMA and (b) PMMA/TiO2/PC (4 ml).
Thermal properties
DSC analysis was done to get thermal information about the changes in the composites during loading TiO2 and PC to the PMMA. While the DSC thermograms of the samples are given in Figure 3, the Tg value of each sample is listed in Table 1. Observing a single Tg curve for all samples shows that the composites exhibit a thermally homogeneous behavior.33,34

DSC thermograms for pure PMMA, PMMA/TiO2 and PMMA/TiO2/PC composites.
Values of glass transition temperatures (Tg) for pure PMMA, PMMA/TiO2 and PMMA/TiO2/PC composites.

When the obtained results are examined; in the PMMA/TiO2 sample, the Tg value remains almost the same as that of pure PMMA, whereas in the materials with PC loading, it can be seen that the glass transition temperature decreases. The slight change in Tg temperature with the addition of TiO2 results in the conclusion that TiO2 does not have a major impact on the mobility of the chain segments of PMMA. 35 On the other hand, the decrease in Tg values with the addition of PC indicates the increase of segmental chain mobility in PMMA/TiO2/PC composites. The addition of PC changes the chain kinetics, resulting in decreased Tg values, and thus increases polymer chain and dipole segment mobility, which facilitates charge transport and dipole orientation.4,6,25-27,36 However, the lowest Tg value was observed in the sample of 4 ml PC and the effect of this result on dielectric properties should be considered together with changes in morphological properties.
Dielectric properties
When thermal and morphological analyses are examined, it is observed that PC additive has an effect on the mentioned properties. In this context comparison of 4 ml sample was made in dielectric measurements due to the lowest Tg value and TiO2 distribution being more homogeneous. Figure 4 shows the frequency dependency of the dielectric loss factor (tanδ) for 300 K and 380 K. When the graphics are examined, it can be seen that the lowest dielectric loss factor is obtained for the pure PMMA. However, with the addition of TiO2, the tanδ value almost doubled (at 100 Hz). In sample containing 4 ml PC, it was observed that tanδ value increased to higher values (three times increased). It is known that the dielectric loss factor represents the ratio of energy lost in a period to stored energy, on the applied external electric field.10,37-39 In this context, the increase in tanδ values with the addition of PC can be attributed to factors such as an increase in the number of dipoles directed by the external electric field, easier polymer chain segmental movement and increased ionic mobility. In addition, when the frequency dependence of the tanδ values is examined, it is observed that dielectric loss values decrease with the increase in frequency. This decrease is attributed to the decrease in the number of dipoles oriented toward the outer field.

Variation of dielectric loss factor (tanδ) of samples depending on frequency: (a) 300 K and (b) 380 K.
The frequency dependence of the real part of the dielectric constant (ε′) is given in Figure 5. As observed in the tanδ behavior, the lowest ε′ value was measured in the sample of pure PMMA sample. While the ε′ value of TiO2/PMMA composite increased approximately twice as compared to pure PMMA, the ε′ value of composites increased almost five times with the addition of 4 ml PC (compared to pure PMMA) at 1 kHz. Since ε′ is a function of the number of dipoles oriented per unit volume with the effect of external electric field the addition of TiO2 and/or PC has an increasing effect in this respect.40-42 As can be remembered from thermal and morphological analyses, adding PC to the structure has effects such as increasing amorphous ratio and decreasing Tg value. In this context, increases in ε′ values are the results of these effects. The decrease of the real part of the dielectric constant (ε′) with the increasing frequency is an expected and typical property for polymers. As mentioned above, ε′ is proportional to the number of dipoles per unit volume in the direction of the external electric field. At low frequencies, since the number of dipoles aligned in the direction of the external electric field is high, ε′ value is high. When the frequency of the external electric field increases; The dipoles cannot keep up with the change of the external electric field and the ε′ value decreases because of the number of the oriented dipoles decreases.

Variation of the real part of the dielectric constant (∊′) of samples depending on frequency: (a) 300 K and (b) 380 K.
Figure 6 shows the frequency dependence of σac values calculated for PMMA and its composites.

Variation of ac conductivity (σac) of samples depending on frequency: (a) 300 K and (b) 380 K.
While a frequency-independent behavior is observed in the low frequency region σac values of all samples increase with increasing frequency. The reason for the increase in σac values with the increase in frequency is the frequency dependence of the equation (2). While the decrease observed in ε′ values (Figure 5) with the increase in frequency is attributed to the decrease in the orientation of dipoles that cannot respond to the external electric field with the increase in frequency, the same situation causes an increase in σac values.43,44 When the samples are compared within themselves in the constant frequency, it can be seen that the lowest σac value is obtained in the pure PMMA, the σac values increase with the addition of TiO2 and/or PC and these results are compatible with other analyses.
The graph of the σac and ε′ values depending on the temperature obtained at 1 kHz frequency is given in Figure 7.

Temperature dependency of (a) σac and (b) ∊′ values of pure PMMA and its composites at 1 kHz.
With the increasing temperature (up to 350 K): As the specific volume will increase with the increase in temperature, dipolar orientation becomes easier and dipolar polarization increases. Since this increase in polarization increases the real dielectric constant, the ac conductivity also increases. In both values, a decrease after 350 K is observed. This decrease may be due to improvement in thermal vibration of molecules with increasing temperature; therefore orientation of dipoles parallel to external electric field can be restrained. 45 In addition, the decrease in density as a result of thermal expansion of the polymer may have caused a decrease in conductivity values. On the other hand, when examining the behavior of the samples, the lowest values, similar to the previous analysis, are obtained for pure PMMA.
The variation of ε′ values obtained in PMMA and its composites depending on the filler at 330 K are given in Figure 8.
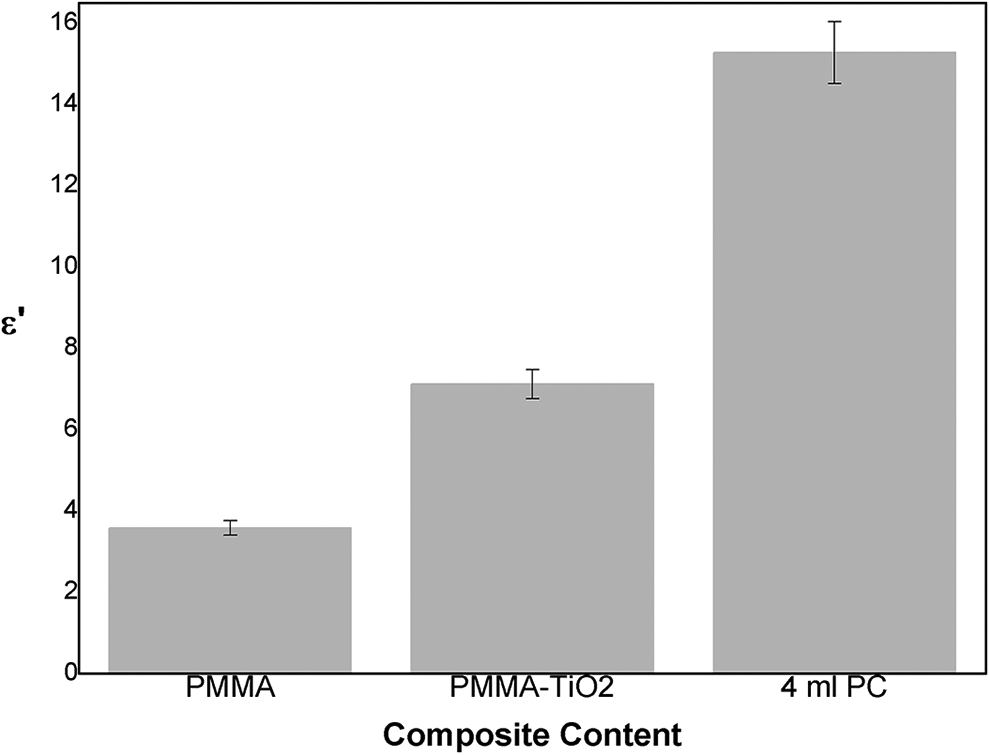
Variation of ∊′ values in 330 K depending on the filler and the filler rate (5% error bar).
Starting from pure PMMA, ε′ values increases with TiO2 addition and reaches its highest values with the addition of 4 ml PC. As seen in the thermal analysis, TiO2 did not change the glass transition temperature much, but with the addition of PC, Tg values decreased. These results explain the behavior obtained from Figure 8. The addition of PC facilitates dipole and segmental mobility by increasing amorphous ratio of PMMA and as a result ε′ values increase.4,25 The increase observed with the addition of TiO2 is due to TiO2 particles dispersed in the structure and having higher dielectric constant than PMMA.
The change of the σac values of PMMA and its composites depending on the filler (at 330 K) is given in Figure 9.
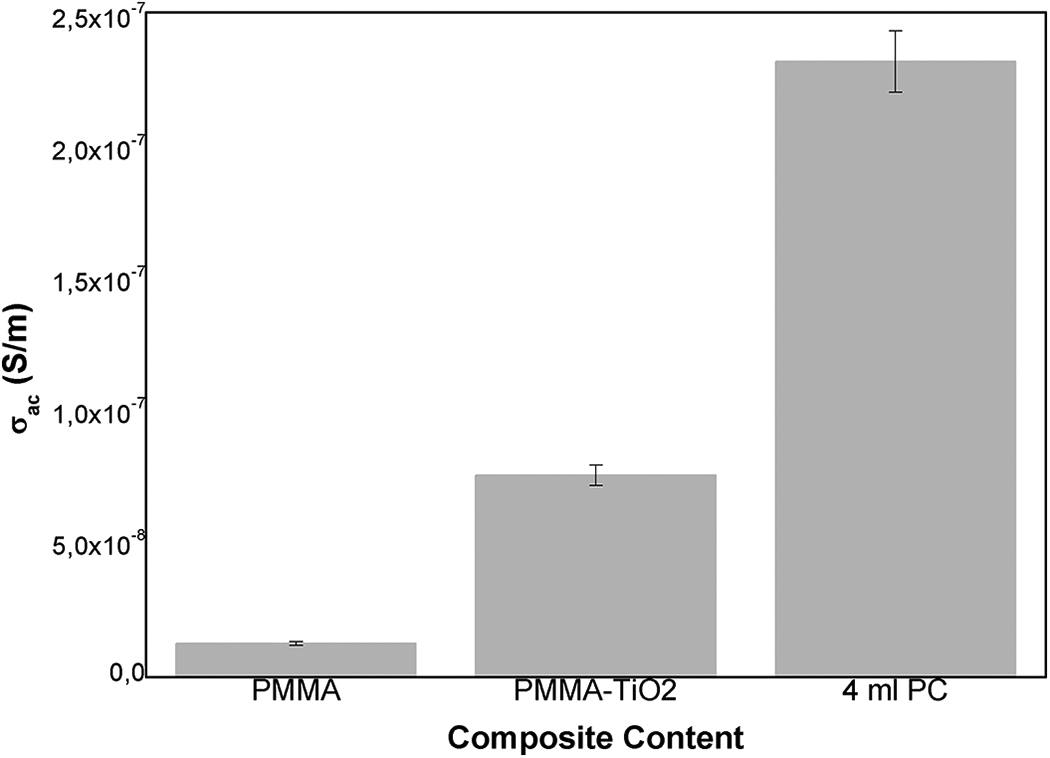
Variation of σac values in 330 K depending on filler and the filler rate (5% error bar).
While the lowest σac value is observed for pure PMMA, the value increases with the addition of TiO2, and furthermore, the highest values are achieved by adding the 4 ml PC to the composites. It is clear that adding plasticizer to the structure has an effect on increasing dipole orientation, which is the main actor of the ac-conductivity mechanism. This effect is supported by both morphological and thermal analyses and is clearly can be seen in other dielectric analysis terms.
Conclusions
In the current study, the effect of adding propylene carbonate (PC) as a plasticizer to PMMA/TiO2 composites on dielectric, thermal and morphological properties was investigated. Compared to pure PMMA, although PMMA/TiO2 composite did not change in terms of thermal properties, the change in morphology caused the dielectric constant to increase approximately twice. In the study where the main factor that caused the change of dielectric properties as well as thermal and morphological properties was the addition of PC; segmental, dipolar mobility were facilitated with the addition of PC and consequently while Tg values decreased, an increase was observed in σac and ε′ values. In the analysis for 100 Hz, while the ε′ value increased two times compared to PMMA with the addition of TiO2, this value increased almost five times with the addition of PC. The results of this study conducted in the light of academic interest will contribute to the fields of science and technology with observations that dielectric properties and especially σac values are improved without any major loss in thermal and mechanical properties.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


