Abstract
In this work, various stabilizers have been introduced to prevent or delay degradation due to ultraviolet (UV) light exposure to prolong the service life of cotton fiber-reinforced composites. The effect of various additives like hindered amine light stabilizer, UV absorber (UVA), and antioxidant as photostabilizers of CF/low-density polyethylene (CF/LDPE) composites was compared. We showed how they influence to delay or eliminate the photodegradation of CF/LDPE subjected accelerated weathering. Surface analysis was performed by Fourier transform infrared spectroscopy and color measurements. The results showed us the insight of the photodegradation mechanism of weathered CF/LDPE composites undergoing photooxidative reactions which causes a loss surface quality such as micro-cracking and color change. Among the stabilizers, UVA was found to be the most effective to delay some color changes in long term.
Introduction
Nowadays, the utilization of natural fibers as reinforcement, instead of glass and carbon fibers and inorganic fillers, inside the thermoplastic industry has gained much acceptance. Even though synthetic fibers, such as glass or carbon, have a few drawbacks, such as biodegradability, recyclability, and initial process cost, they are still used in the production of composites because of the much appreciated properties such as rigidity and strength. 1 Natural fibers are desirable due to the high possibility to perform as a recyclable primary phase as a substitute to the use of inorganic fillers and fibers, such as glass and carbon. 2 Significant production conveniences, desired mechanical features, low cost, low density, biodegradability, and low energy usage are some of the many advantages which natural fibers provide. 2 -4 The hydrophilic property of natural fibers is believed to be the most important disadvantage because of the undesirable low adhesion with the polymer matrix which inhibits hydrophobic properties. 1 -5 This low adhesion results in undesired low physical and mechanical features of natural fiber-reinforced composite materials. 6
Applications varying from building, packaging materials, and automotive are providing a market for fiber-reinforced composites. Varying concerns for durability have risen when the fiber-reinforced composites are vulnerable to the outdoors. 7 -9 Durability in these types of environments may include stability to ultraviolet (UV) and thermal, with resistance to moisture, and fungal threats. UV exposure can be a reason for the composites to experience photodegradation which leads to unwanted effects that include a decrease in mechanical properties and surface quality.
The photodegradation mechanism of cotton fiber (CF) and low-density polyethylene (LDPE) separately is well reported in the literature. 7,10,11 Due to photodegradations mechanisms, photooxidation reactions of LDPE arise in the composite structure. Oxidation reactions can be divided into two groups which are thermal oxidation and photo oxidation. Thermal oxidation starts from the manufacturing of composites. Free radical generation is a result of oxidation degradation of polyethylene. The free radicals, which are assembled during the first phase, charge the polymer in the multiplication phase and produce brand-new free radicals. 11 During the oxidation reactions, hydroperoxides are produced as primary photoproducts. Due to the UV radiation, divisions in chains can occur by the breakage of the weak O–O bond, which results in a formation of a macro-alkoxy and a hydroxyl radical (OH). During this reaction, alkoxy macro radical is the key transitional. 12,13
Norrish type I reactions advance during the existence of UV light, which approves the construction of free radicals. This reaction may cause abortion via cross-linking or chain breakage. Groups, such as carbonyl and vinyl, are formed and chain breakdowns are developed if the degradation proceeds according to Norrish type II. During the UV exposure of polyethylene, generally chain scission and cross-linking reactions are happening simultaneously. 7 -9,11
The UV exposure is accountable for the degradation of lignin, which is also associated with the photodegradation of cotton. Researchers stated that the main reason for yellowish color seen in CF is due to the degradation of lignin, first to hydrophilic products, then to the production of chromophoric functional groups. Lignin experiences photodegradation in many ways. Phenoxy quinone redox cycle is one of the most common reaction ways. In this reaction, hydroquinones and paraquinones are a redox couple. Under UV radiation, hydroquinones experience oxidation to form paraquinones (chromophoric structures). This reaction is called photo-yellowing. The reverse cycle which is paraquinones to hydroquinone groups is the reason for photobleaching. 10,11
UV absorber (UVA), hindered amine light stabilizer (HALS)-free radical scavengers, and antioxidant (AOx) are some of the photostabilizers which can be used on polyethylene-based composites to minimize the effects of UV radiation. In the literature, many cases are reported that features of the fiber-reinforced composite, such as surface chemistry, mechanical property, and appearance, are affected by weather. 7 -9,14,15 HALS have been proven to be very effective in constraining photooxidation, but no significant effects on color stability have been shown. 16,17 In polyolefins, in addition to HALS, UVA is commonly used. The photostabilizers’ working principle is based on the intake of unwanted UV exposure and the wastage of heat. Stark et al. 9 applied color measurement tests to analyze the change in color of wood-flour (WF)-filled high density polyethylene (HDPE) composites. According to their studies, by adding UVA to WF/HDPE composites, the lightness effect is reduced. 7 Due to the cheap and non-discoloring properties of AOxs, they are still used for stabilization purposes regardless of the fact that AOxs are generally less effective than other UV additives. 18
In this research, accelerated weathering was done on composites with HALS, UVA, and AOx compared to the control sample without a photostabilizer. Both lightness of unstabilized and stabilized CF/LDPE composites and initial changes in surface characteristics were resolved and compared with each other. The main aim of this work is to compare the performance of HALS, UVA, and AOx on the matter of preventing the physical and chemical changes for CF/LDPE composites with respect to accelerated weathering.
Materials and methods
Composite production
In the production of unstabilized composites, only 25 wt% CF and 75 wt% LDPE were used. Chopped, rib knitted, 30 Ne yarn, and 100% cotton was supplied by Linens Marketing Inc., Turkey. LDPE was supplied by Petkim Inc., Turkey. Based on molecular structure, three UV additives were chosen for the research. Chimassorb 944, Tinuvin 326, and Irganox 1010 were used as HALS, UVA, and AOx, respectively. In order to study an acceptable concentration for UV protections, two distinct combinations of the mentioned additives were used. In Table 1, all the used additive’s formulations can be seen.
Composite material formulations.
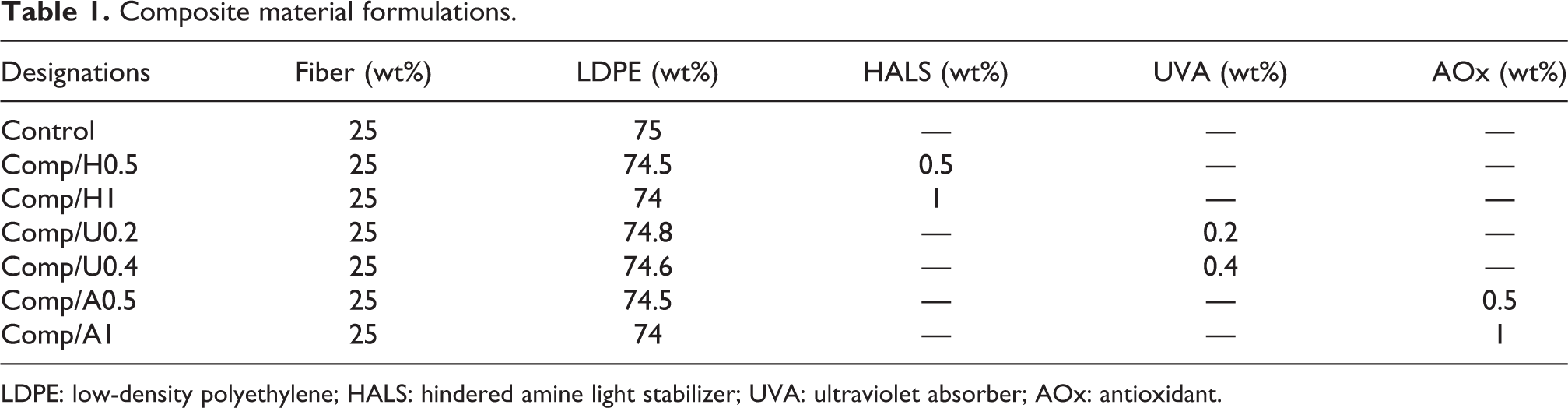
LDPE: low-density polyethylene; HALS: hindered amine light stabilizer; UVA: ultraviolet absorber; AOx: antioxidant.
Figure 1 shows the basic flow chart of this study. A single screw extruder was used to mix the CF, LDPE matrix, and the UV additives. A unique die located at the end of the extruder, acted as a plate former to produce composites with a thickness of 6 mm. In addition to turning the waste cotton fabrics to fibers and also to provide better fiber homogeneity in the LDPE matrix, the reprocessing process was executed three times. 19 Once the manufacturing process had ended, the plates were kept under pressure to keep them at a constant flatness and thickness.
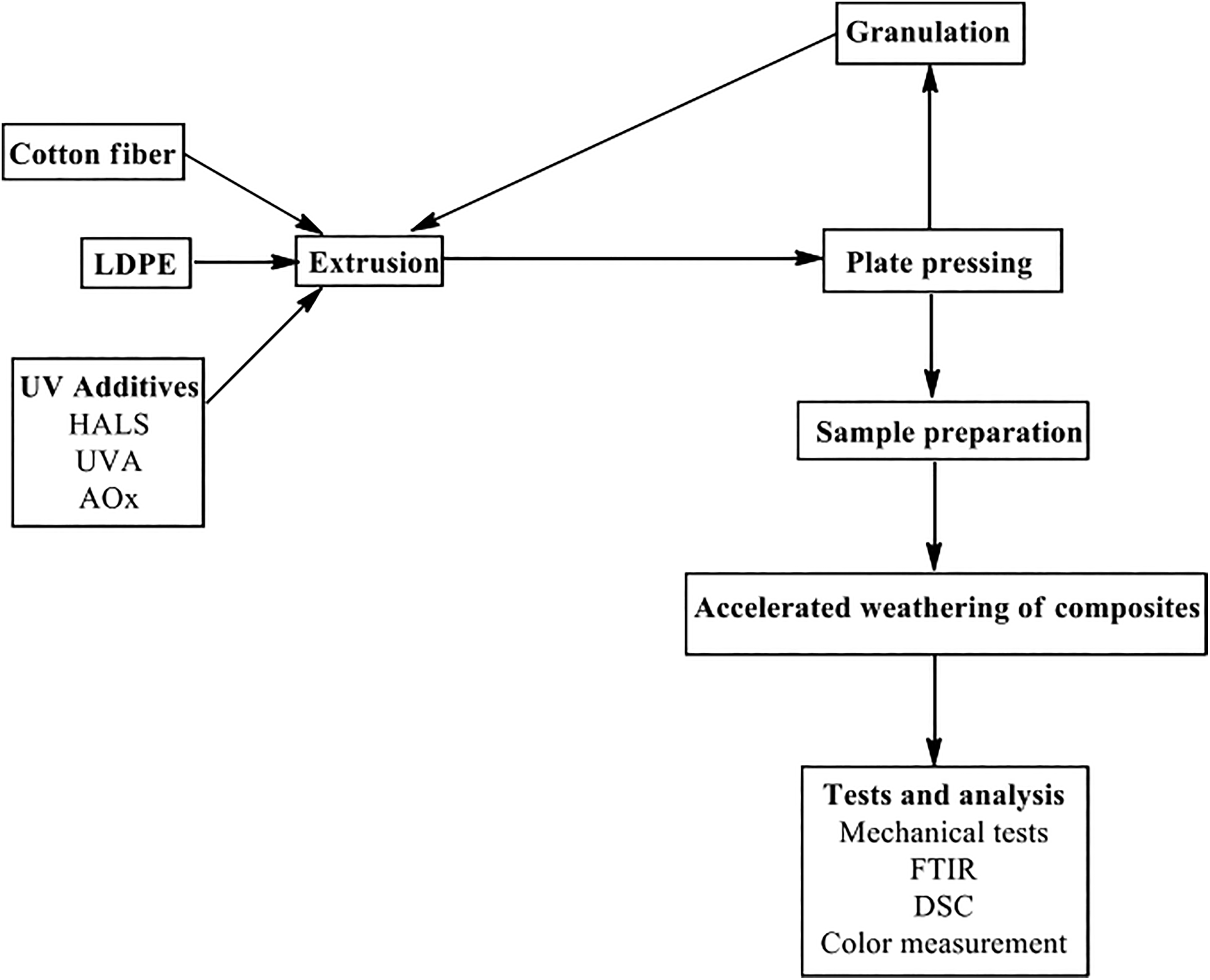
Flowchart of the study.
Characterization
An equipment by Suntest CPS (Heraeus Industrietechnik GmbH, Germany) was used for weathering of the composite samples. A xenon light source, which emitted an energy density of 300 W m−2, was used for a period and the manufactured composites were exposed to wavelengths light higher than 340 nm at 30°C for 120 and 240 h.
Thermo Scientific Nicolet IS 10 Fourier transform infrared (FTIR) spectroscopy was used for the evaluation of the surface chemistry of the composites. IR spectra were collected by attenuated total reflectance method. The data were collected at room temperature in the spectral range of 450–4000 cm−1 with a signal resolution of 4 cm−1. For FTIR characterization, at least three samples were used and the graphs present the average results for each sample.
To measure color, a Datacolor 3890 spectrophotometer containing L*, a*, and b* coordinates was used in correlation with ASTM 2244. L* represents the lightness coordinate, a* represents the red (+a*) to green (−a*) coordinate, and b* represents the yellow (+b*) to blue (−b*) coordinate. To achieve the average values of color, at least five replicate samples were measured. Carbonyl index of composites and discoloration (ΔE) of the weathered samples were calculated by equations (1) and (2), respectively


where I 1712 and I 2915 refer to the C=O stretching and C–H stretching in methyl and methylene groups in equation (1), respectively. However, ΔL*, Δa*, and Δb* are the difference of weathered and unweathered values of L*, a*, and b* for all composite materials, respectively.
Results
For all composite samples, FTIR spectroscopy was utilized to analyze the structural differences in the surface chemistry during accelerated weather conditions. In Table 2, the different important bands are shown according to the IR spectra of the composites and distinct functional groups. 20 -22
FTIR: Fourier transform infrared.
In Figure 2, FTIR spectra of the surface layer of control composite sample show a series of bands associated with both cotton and polyethylene with respect to UV exposure time. In the FTIR spectrum of the surface layer, some distinctive bands, for example, 2916 and 1510 cm−1, for polyethylene and lignin components, respectively, were detected.

FTIR results for control composite samples due to UV exposure time.
In Figure 3, the carbonyl index for all composite materials is shown. For the unweathered composites, due to the chemical structure of hemicellulose and lignin, the carbonyl index is analyzed for composite samples. 23 Al-Malaika 18 expressed that because of the existence of oxygen in the development stages, LDPE experiences molecular entanglement. Due to UV radiation, the formation of carbonyl groups is decisively the outcome of both the alkoxyl and alkyl radicals which are formed in the backbone chain of the polymer. In any case, it is well known that all thermoplastic polymers exhibit reasonably high mechanical degradation during the beginning of the process. This high mechanical degradation has exhibited characteristics which affect the consecutive aging performance of the polymer because of the introduction of macromolecular irregularities. With the addition of photostabilizers, for CF/LDPE composites, the radicals produced by extrusion or fracture operations have wider domains. Therefore, as we have a look at Figure 3, higher values of carbonyl index were obtained for stabilized composites compared to control (unstabilized) composite for unweathered (0 h) condition. On the contrary, the carbonyl index value of composites containing AOx additives included was seen to have an increase compared to the carbonyl index of other composites. The probable reason for this is the extra carbonyl groups which emerge from the chemical structure of the AOx.

Carbonyl index of all composites.
According to Figure 3, unstabilized samples (CF/LDPE) presented an increase from 0.0080 to 0.023 and 0.05 in carbonyl index after 120 h and 240 h of weathering, respectively. These increases are due to the photooxidative reactions in the composites. 24 Furthermore, from the results, it can be stated that the chain scissions increase immediately after UV exposure and exposure time. However, the increase of the carbonyl index value was less for the stabilized composites compared to the unstabilized composites. For the most part, after 240 h of exposure, the largest increase was seen for the unstabilized composites, yet composites which contained 5% and 1% HALS showed a decrease in the carbonyl index value. A decrease of 77% in the carbonyl index value after 240 h exposure in composites with 1% HALS is notable. This result points out that 1% HALS is more effective than other photostabilizers due to its radical scavenging feature. Figure 4 supports the theory of the significant protection of HALS by reducing the photooxidative property which is held accountable for the formed ketone groups. The mentioned theory was also supported by Kaci et al. in his report. 17 Stating that the passivizing effects of the ketonic carbonyls on the Norrish types I and II photochemical reactions, with radical or molecular groups, are present as polyolefins during the oxidative deterioration by inhibiting amine light stabilizers located in the LDPE. Thus, changes in carbonyl concentration for composites with HALS during all exposure times can be linked with the removal of ketonic carbonyls. However, as expected, the results showed that the addition of AOx to CF/LDPE composite has no positive effect on the carbonyl index compared to other photostabilizers except UVA (0.2%) after 240 h of weathering. Irganox 1010, which is an AOx where its role is as a chain-breaking donor (hindered phenols), is usually less effective than other UV additives. Yet they are still used for stabilization purposes due to their cheap and non-discoloring features. 20
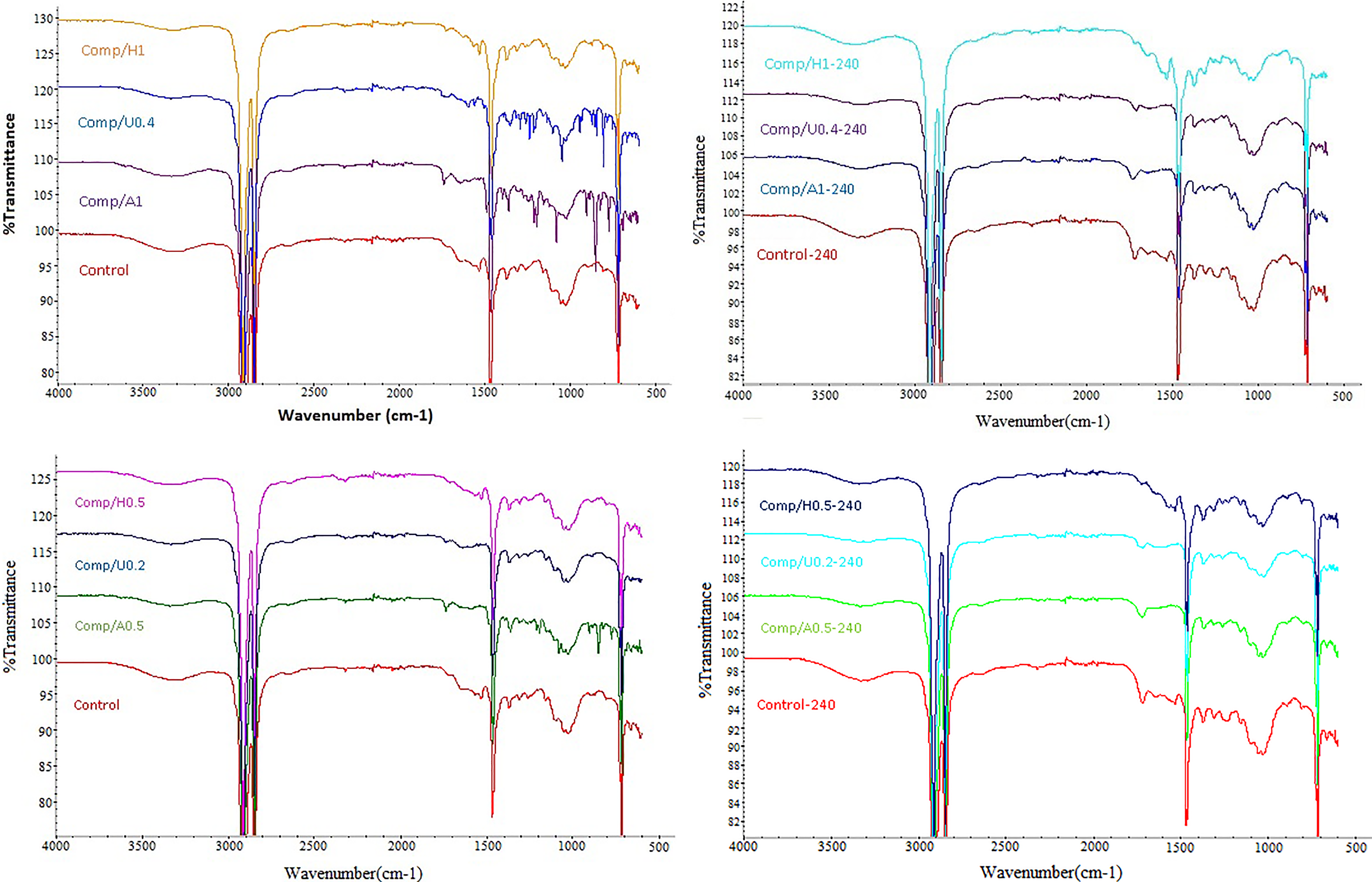
FTIR results for unweathered and 240h of exposed composites.
For all CF/LDPE composites, the color analysis tests were performed with UV radiations for about 240 h in accelerated weathering conditions. Chromacity coordinates (a* and b*) and lightness (L*) were both determined. Positive ΔL* increment indicates that the color of composites is lighter, while negative ΔL* indicates that the color composites are darker after weathering.
During UV exposure, due to the lignin properties of CF, the addition of CF to polyethylene is delayed with respect to the degradation of the polymer matrix. Furthermore, the lignin in the CFs acts as a natural AOx. Moreover, Abu-Shark and Hamid 25 proved that lignin is capable to stabilize natural fibers due to its AOx property. Discoloration and brown colors can be seen in the composite samples during the processing of natural fiber composites. It is also possible that the surface layer of fibers provides protection to the polymeric matrices from UV exposure to prevent degradation in the bulk. Therefore, for color measurements of CF/LDPE composites, the decay or degradation of lignin during UV exposure is important.
In Figure 5, Δa* and Δb* values change for both unstabilized and stabilized CF/LDPE composites can be seen. Results indicate that both Δb* and Δa* showed a decrease for all composites after the UV exposure of 240 h. In addition, yellow discoloration is lower in stabilized composites. This is interpreted as the chromophores undergoing deterioration which results in photobleaching. Muasher and Sain 11 reported the decrease of redox reactions of lignin. They support their claim with the redox reactions of the hydroquinone and paraquinone structures which are influenced by the Δb* behavior. Under UV radiation, the reactions first begin with the oxidation of the hydroquinones, which form paraquinones (chromophoric structures). Furthermore, quinones, carboxylic acid, and hydroperoxyl radicals, which are chromophoric groups, are known as the main reason for discolorations, like yellowing, of fibers. Consequently, photobleaching which is the result of the reduction of the paraquinone to hydroquinone groups could possibly result in a decrease of discolorations like yellowing by increasing exposure period. Reactions of which paraquinone turns to hydroquinone are unwanted because of the possibility of reoxidizing and propagating the formation of paraquinone which reinitiates the redox reaction. 2 In this matter, the lowest Δb* values of the unstabilized composites indicate that they are affected more from the lignin photodegradation reaction.

Color measurement results for samples after 240h UV exposure.
Because all of the stabilized composites exhibit a decrease in the negative value, a decrease in chromophoric formation can also be elucidated. 11 After 240-h UV exposure, HALS-stabilized composites show the least amount of change in the Δb* value compared to all stabilized composites. As shown in Figure 5, the total decrease of Δb* value for HALS (0.5%) is quite similar to the unstabilized composites value. This amount of decrease supports the ability of maintaining scavenge free radicals which increase the amount of formation of hydroquinones. Furthermore, the inclusion of UVAs in the composites resulted in a stoppage of photobleaching. Du et al. 24 showed that regardless of molecular weight, UVAs are more effective than HALS. They further proved that the UVAs start to take an active role before the fiber is exposed which results in bleaching. Therefore, after 240 h of UV exposure, the inclusion of both UVAs in the composites prevented any further photobleaching of composites. 26 Figure 5 also indicates that composites stabilized by AOx had the least amount of change in photobleaching. Although the effect of chain-breaking AOxs as a photostabilizer is small, some of their oxidative transformation products are photosensitizing. They are also called non-discoloring additives. 18
In Figure 6, the change in the ΔL* and ΔE values with respect to exposure time can be seen. Furthermore, after 240 h of exposure, the increase in lightness factor (ΔL*) can additionally be observed. This increase can be associated partly with the chain scission of LDPE and its movement to the surface and partly with the photobleaching of lignin. This situation is explained by the produced radicals that influence the LDPE chains as a result of photooxidation of lignin in the composites. 27 At the end of 240 h of UV radiation, for most of the stabilized composites, ΔL* values were lower compared to the value of the unstabilized composites.
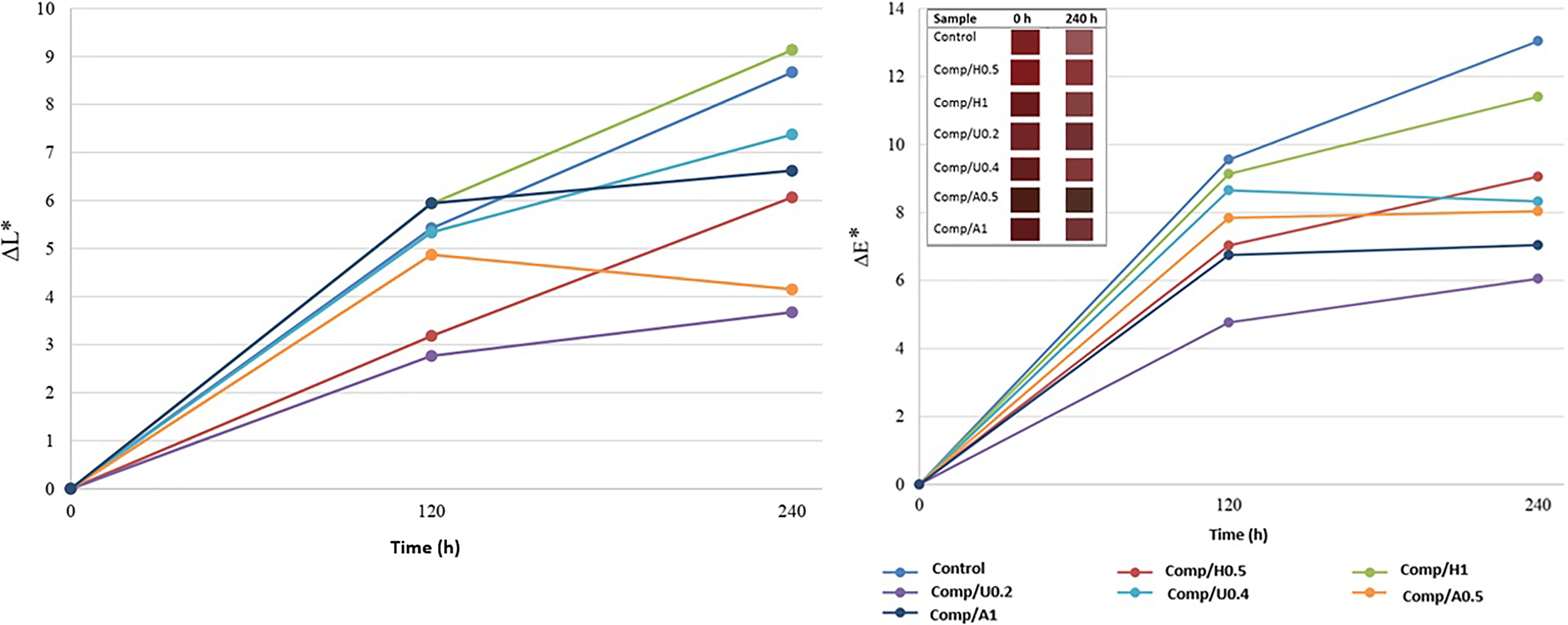
ΔL* and ΔE* values for all samples with respect to weathering time.
Figure 6 also shows that the effectiveness of both UVA (0.2%) and HALS (0.5%) on controlling lightning is comparable in the first 120 h of radiation. Furthermore, ΔL* value of UVA (0.2%) displayed an increase at a slower rate compared to HALS (0.5%) after the first 120 h of radiation. This study showed that UVA could slow down the fading by decreasing UV radiation available to bleach the samples. Furthermore, it can be seen that the values of ΔL* corresponding to composites stabilized with AOx (1%) and HALS (1%) showed an increase with the same rate as in the first 120 h period. Moreover, some changes in ΔL* values are observed for AOx (1%) between exposure periods of 120 and 240 h, whereas the ΔL* values of HALS (1%) maintained an increase. Due to this situation, it may be proposed that on the topic of color fading, AOx (1%) seems to be more effective compared to HALS (1%). Finally, an increase in total bleaching/discoloration of UVA (0.2%) is visible after 240 h of UV radiation, whereas HALS (1%) has the highest value of ΔE* among the stabilized CF/LDPE composites. After the color change analysis, a comprehensive conclusion can be that stabilized composites showed a total change in a lower degree compared to the unstabilized composites. The UVAs can considerably be efficient to inhibit bleaching/discoloration by absorbing UV radiation and can be proposed for usage.
Conclusion
This study is aiming to produce CF/LDPE composites which are UV stabilized. In this manner, during the study, AOx, UVA, and HALS were used to delay or diminish the effects of UV radiation on the composites. All composites were exposed to UV light radiation for periods of time up to 240 h. Carbonyl index values were determined to analyze the effect of photodegradation as a result of UV weathering. Results indicate that for control samples, the highest value of carbonyl index occurred after 240 h of exposure. In the case of composites with additives, results state that UVA offers important resistance to weathering. Results further show that the composite samples stabilized with HALS proved more effective compared to the other stabilized composites. This study also conducted color measurement analysis. After 240 h of UV exposure, a significant decrease is visible in the Δb* value which indicates the yellowing of samples. Furthermore, inhibition of photobleaching is a result of the inclusion of UVA to composites. After 240 h of UV exposure, an increase is seen in the lightness factor ΔL* for all samples. Notably, after the 240-h UV exposure, control samples exhibited higher ΔL* compared to the stabilized composites. Ultimately, for the lightness factor (ΔL*), AOx stabilized composites proved to be the most effective. Finally, among all samples, total discoloration (ΔE*) of HALS stabilized composites resulted in the highest value.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

