Abstract
Lignosulfonate (LS)/mercerized cotton composite was prepared to study the LS suppressive possibility to control malignant cell activities to facilitate chemotherapy treatment. The composite was characterized by cross-polarized/magic angle spinning 13C nuclear magnetic resonance (NMR) spectrometry and scanning electron microscope (SEM). 13C NMR spectra and SEM indicated that the presence of LS increased crystallinity and porosity of mercerized cellulose, respectively. Furthermore, the composite efficacy against cancer cell activity was evaluated by cytotoxicity for malignant cell line HCT116, HepG2, and MCF-7. The cancer cells thrive in an acidic environment and they do not survive in normal or alkaline media. The release of LS was intended at acidic pH, in which, the acidity increased due to the activity of the cancer cells that activate the LS released from the mercerized cotton into the media. Therefore, this release process reduced the activity of cancer cells. The released LS kept cancer cells in a low activity via decreasing acidity. Hence, the composite may not eliminate the cancer cells, but it reduces the activity of cancer cells.
Introduction
The likely association between fiber admission and cancer risk has been considered in several researches, but the topic is disrupted till now. 1 –3 Fibers suffer from some weaknesses as microbial pollution and in addition batch-to-batch variability in the fabrication process; they are dependent on environmental and seasonal factors. 4 Malignancy is a devastating disease since it is responsible for 13% of all deaths worldwide. Cancer is considered the most difficult and common problem all over the world. 5 –7 The difficulties confronting the generation of anticancer treatment are as follows: first, the capacity of the disease treatments to extend the target without changing their nature; second, their capacity to cross the cell membrane; third, their capacity to achieve the specific cancer cells; and fourth, their capacity to enact particular systems in the cells such as apoptosis. 8 –10 Along these lines, considering the mentioned rules, the generation of new competitor anticancer medications ought to have highlights however not haphazardly amalgamation. Reality shows that, till now, existing molecules “natural or synthetic” are insufficient to control cancer cells proliferation or spread. Subsequently, specialists started to look at the internal mechanisms of cancer cells with the end goal to get in their regulatory mechanisms, as the most promising way to design new anti-malignant drugs. 11 –13 However, several researches have seen that apoptosis (programmed cell death) mechanisms can be enacted inside the malignancy cell like other ordinary cells. 14 –17 In the light of these data, numerous medications have been produced with the true objective to authorize apoptosis in malignancy cells as it is the most secure approach to monitor growth cells under control in addition to activate the immune system. 18 –20 From this start and rule, the synergistic effect of numerous anticancer medications has been examined to improve their cytotoxic effect or to beat the obstruction of malignant cells to chemotherapy by enacting apoptosis. The main problem with chemotherapy is their reaction with normal cells and other organs in the human body. So, they will upset the body homeostasis rather than help in control. 21 –23 Lignin is a standout among the most polymers in plants, simply behind cellulose. Lignin is basically situated in the center lamella, giving mechanical quality to the cell wall and to the entire plant. In the sulfide pulping process, the disintegration of lignin is acquired by the activity of bisulfite ions, the counter ion can be calcium, sodium, ammonium, or magnesium. 24 During the sulfide procedure, lignin ether bonds are broken haphazardly, and the sulfonic group makes the fragments (lignosulfonate (LS)) more water-soluble. This gives LS interesting properties as it is a combination of a hydrophobic aromatic skeleton with a hydrophilic sulfuric group. LS appears to be an effective antioxidant, hence opening the likelihood of new potential applications. 25 –27 Patients with malignancy in the skin suffer from open wounds due to deterioration of the tumor mass. The utilization of cotton is regular to repel pathogens from the bloodstream. 28,29 Hence, the idea of utilizing anti-malignant agents on cotton fiber will diminish the reactions and harms. Despite the wide utilization of LS in numerous industrial creations, this is the first run through to utilize it as malignancy hopeful medication. As it is known, the cancer mass urges microscopic organisms to develop inside it with the end goal to shield itself from chemotherapy. Our present work demonstrated that LS has decent cooperation and scattering onto mercerized cellulose owing to the breaking of the hydrogen bonds present in cellulose. 30 This study will be utilized the LS to upgrade the anticancer impact by creating an innovative design of cotton gauze that has three prominent features: first, the capacity to close the wounds by cotton; second, the capacity to eliminate microorganisms from cancer mass, permitting chemotherapy (presence of LS) to reach its target; and third, the capacity to keep malignant cells with stable concentration of LS released from the fibers of cotton under the acidic condition.
Materials and methods
Preparation of LS/cellulose composite
Mercerized cellulose (sodium cellulose; 0.64 g, 0.013 mole) was immersed in 5% NaOH (100 ml), followed by the addition of LS (0.1 wt%). At that point, the blend was refluxed for 24 h. 30,31
Magnetic resonance
The cross-polarized/magic angle spinning (CP/MAS) 13C nuclear magnetic resonance (NMR) spectrometry was performed utilizing a Bruker 500 MHz spectrometer (Karlsruhe, Germany) at a frequency of 250 MHz with an obtaining time of 0.011 s at room temperature. 32
Scanning electron microscopy
The morphologies of the distinctive cellulose tests were reported utilizing scanning electron microscope (SEM)—FEI Co Quanta 200 model SEM (USA). All examples were gold-covered preceding examination. 33
Release study
The absorbance of the examined medium was estimated utilizing an ultraviolet (UV)-Vis V-630 spectrophotometer (Jasco Inc., Tokyo, Japan). An initial scanning was performed to decide the wavelength at which the readings would be completed (200–450 nm).
At that point, different LS concentration levels were performed. These LS solutions of various concentration levels were taken to the UV spectrophotometer at wavelength 360 nm; the absorbance of the solutions at various concentrations was then decided, and afterward the adjustment bend was produced. The LS-loaded fibers were gauged (0.0030 g for each sample) and put in a glass vessel containing 10 ml of phosphate-buffered saline buffer solution (pH 5), and the analysis was accomplished every half an hour for 4 h, and then, data were taken at 24 and 48 h. The experiments were directed twice.
Antitumor activity chemicals and drugs
Sulforhodamine B (SRB), methanol, and ethanol were bought from Sigma Chemical Co. (St Louis, Missouri, USA). Growth supplements and culture media were bought from Gibco/Life Technologies Co. (Carlsbad, California, USA). Cell culture vessels were bought from Nunc Co. (Roskilde, Denmark).
Cell culture
Colon cancer (HCT116), hepatic cancer (HepG2), and a breast (MCF-7) cells line were obtained from Viscera (Giza, Egypt). Cells were preserved in Le Roswell Park Memorial Institute medium media supplemented with 100 U/ml penicillin, 100 µg/ml streptomycin, and 10% heat-inactivated fetal bovine serum in a humidified, at 37°C with 5% (v/v) carbon dioxide atmosphere, the cells were subcultured two times in 1 week.
Cytotoxicity assays against malignant cells
The extracts cytotoxicity was measured against the HepG2 tumor cell line by using SRB. Exponentially developing cells were gathered utilizing 0.25% trypsin–ethylenediaminetetraacetic acid and plated in 96-well plates at 1000 cells/well. Before fixing cells with trichloroacetic acid (10%) for 1 h at 4°C, the cells were exposed to the extracts. After repeated washing, in a dark field, cells were exposed to 0.4% SRB solution for 10 min and therefore washed with glacial acetic acid (1%). After drying overnight, Tris–HCl was used to dissolve the SRB-stained cells and the color intensity was measured at 540 nm. 34
Statistical analysis
Treatments were orchestrated in a totally randomized outline. Analysis of variance was performed on the data, using the SPSS V23 software package, to determine the minimum significant difference among treatment at p < 0.05. 35
Results
Figures 1 and 2 showed solid-state 13C NMR spectra of mercerized cellulose and LS/mercerized cellulose composite. The chemical moves (δ ppm) of resonance lines of each sample are reported in Table 1. However, LS has no resonance line in the composite overlapped with the resonance line of mercerized cellulose. It is therefore possible to discuss the structure of mercerized cellulose component in the composite using CP/MAS 13C NMR spectra. As shown in Figures 1 and 2 and Table 1, the resonance line of each carbon of cellulose component in LS/mercerized cellulose composite was sharper and clearer than that of mercerized cellulose. Moreover, the amorphous resonance lines of both C4 and C6 at 88.9 and 69.9 ppm of the mercerized cellulose, respectively, were disappeared in the LS/mercerized cellulose composite. Furthermore, the C6 and C2, 3, 5 resonance lines of the composite were shifted downfield compared to that of mercerized cellulose. So, CP/MAS 13C NMR spectra indicate that the mercerized cellulose components in the presence of LS are in a crystalline state.
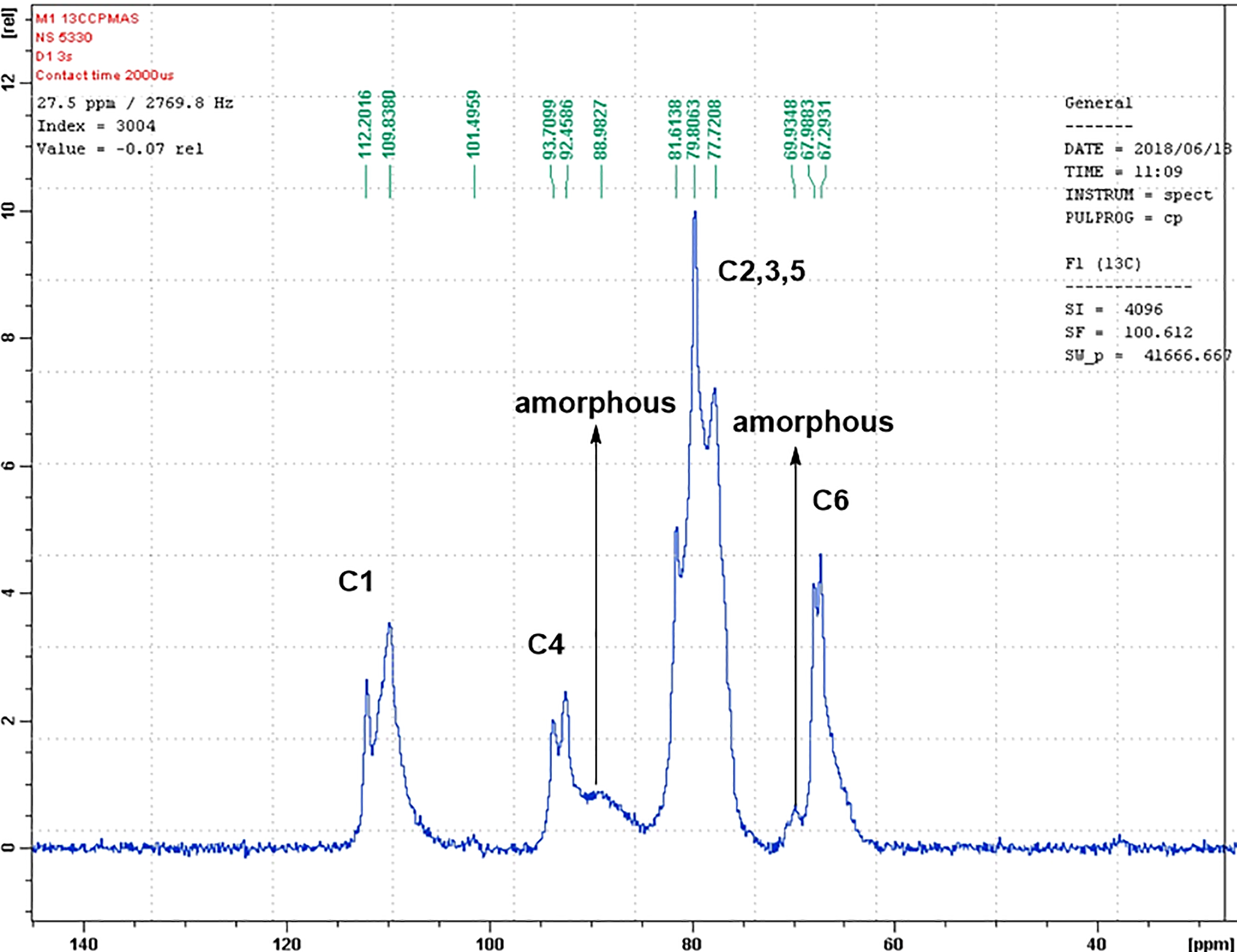
13C NMR of mercerized cellulose. NMR: nuclear magnetic resonance.

13C NMR of LS/mercerized cellulose composite. NMR: nuclear magnetic resonance; LS: lignosulfonate.
13C-Chemical shifts (ppm) of mercerized cellulose and its composite.

LS: lignosulfonate.
Figure 3 presents the surface microstructures of cellulose, mercerized cellulose, and LS/mercerized cellulose under various magnifications. Figure 3(a) to (c) clearly shows the homogenous, fibrous distribution of size and shape of the cellulose (cotton fiber). The cellulose fiber surfaces are generally smooth in appearance with diameters of about 20 µm. Mercerized cellulose fibers, Figure 3(d) to (f), exhibit a very similar appearance to the cellulose fibers but with higher roughness. Significant changes occurred to the cellulose morphology after treatment with LS, as illustrated by Figure 3(g) to (i), in terms of appearance, porosity, size, and level of roughness. The LS/mercerized cellulose particles are sponge-like with high porosity.

SEM images under different magnifications for (a)–(c) cellulose, (d)–(f) mercerized cellulose, and (g)–(i) LS/mercerized cellulose. SEM: scanning electron microscope; LS: lignosulfonate.
Table 2 presented the energy-dispersive X-ray (EDX) analysis results for unmodified cellulose and LS/cellulose composite. The results recorded the sulfur at 11.6%. Effects in a biologic system are not produced by a chemical agent unless the agent or its metabolites interact with appropriate receptors in the system at enough time and concentration.
The EDX analysis (weight %) of cellulose, mercerized cellulose, and LS/cellulose composite.

EDX: energy-dispersive X-ray; LS: lignosulfonate.
In the light of these convictions, colon cancer (HCT116), hepatic cancer (HepG2), and a breast (MCF-7) cells line were incubated for 72 h, which is accepted as enough time for cytotoxicity in vitro studies. Results revealed that LS presents moderate cytotoxic and moderate antiproliferative activities (Table 3). The results have been confirmed by the cell viability curves (Figures 4 to 6).
The IC50 (µg/ml) of LS against different solid tumor cell lines.

LS: lignosulfonate.

Dose-response of LS against HCT116 cells by SRB assay. Cells were treated with various concentrations of compound and incubated for 48 h. LS: lignosulfonate; SRB: sulphorhodamine B.

Dose-response of LS against MCF-7 cells by SRB assay. Cells were treated with various concentrations of compound and incubated for 48 h. LS: lignosulfonate; SRB: sulphorhodamine B.

Dose-response of LS against HepG2 cells by SRB assay. Cells were treated with various concentrations of compound and incubated for 48 h. LS: lignosulfonate; SRB: sulphorhodamine B.
Discussion
In 13C NMR, the C1, C4, and C6 chemical shifts of cellulose have long been known to be diagnostic of cellulose crystallinity and allomorphs. 36 The C4 and C6 chemical shifts of 93.7 and 67.9 ppm were assigned to interior crystalline glucan chains and the 88.9 and 69.9 ppm peaks to surface chains with partial disorder. The 93/67 ppm interior glucan signals are well resolved in LS/mercerized cellulose signals; thus, they are unambiguous indicators of crystallinity enhancement of cellulose in the presence of LS. The possibility that the 93/67 ppm chains may reside inside the microfibril instead of on the surface is considered low because the 93/67 ppm peaks have strong cross peaks with cellulose matrix and exhibit large amplitude dynamics. 36 Molecular weight of LS is high (2700) and its solubility toward sodium hydroxide is low so its affinity to mercerized cellulose is higher as our previous reported data. 37 So, LS enhances the crystal structure of mercerized cellulose fiber, especially the conformation of cellulose chains.
Interestingly, it is noticed that the composite film showed smooth and homogeneous surfaces with the incorporation of CaLS (Figure 3). Thus, CaLS is homogeneously dispersed in the sodium cellulose matrix. The CaLS particles enhanced the compatibility due to strong interactions between CaLS and sodium cellulose. Nevertheless, the presence of CaLS particle increased the aggregation and consequently also the width of the fiber, as shown in Figure 3. So, CaLS is improving composite compatibility that attributed to the intermolecular hydrogen bonding. The elemental analysis conducted by EDX confirmed the presence of sulfur with 11.6% which confirms the presence of LS.
From the reported study, the cancer mass encourages bacteria to grow inside it to protect themselves from chemotherapy. 37 However, the relationship between cancer cells and some pathogens constitute a symbiotic relationship for their survival. 38 Cancer cells provide pathogens with nutrition; on the other side, bacteria deactivate cancer cells from chemotherapy. 39 This study aims to enhance the anticancer impact by creating new designs. On the other side, cancer thrives in an acidic environment and does not survive in normal or alkaline environment. 40 Cancer cells make the body even more acidic as they produce lactic acid. 41 Depending on that, this study has been designed to control the release of LS at acidic pH value. 42 Owing to the activity of the cancer cells, the pH will decrease and activate the LS released from the cotton to the plasma (Figure 7). Therefore, this process will minimize the activity of cancer cells. The ease of releasing LS from cotton owing to the physical bond between LS and cellulose is affected by an acidic condition. 43 Hence, the LS/cellulose composite is not designed to eliminate cancer cells because of the weakness of LS cytotoxicity, but it contributes to the activity reduction of cancer cells that are associated with the regulation of pH. 44,45 Therefore, this technique will keep cancer cells in low activities, thus the chemo or radiotherapies will be more effective. Hence, the cotton LS may not contribute to eliminate cancer cells, but it reduces the activity of cancer cells.

Morphological changes of HCT-116, MCF-7, and HepG2 cells induced by IC50 concentration of LS, stained with AO/EB. The images were taken using fluorescence microscopy at ×20. A: apoptotic body; N: necrosis; LS: lignosulfonate; AO/EB: acridine orange/ethidium bromide.
Conclusion
In this study, a CaLS/sodium cellulose composite was successfully synthesized. CaLS showed a good interaction and dispersion onto sodium cellulose owing to the breaking of the hydrogen bonds present in cellulose. CaLS has ability to control cancer cell division (antiproliferative agent) via open wound covering. Consequently, the composite may be considered as innovative design for a novel cotton bandage possesses both antibacterial and antiproliferative features. Therefore, the bandage will provide multiple functions like: first, close the wounds by cotton; second, eliminate bacteria from the cancer mass allowing chemotherapy (presence of LS) to reach their target; and third, keep the cancer cells with stable concentration of LS released from cotton under the acidic condition.
Footnotes
Author’s note
Sherif MAS Keshk is now affiliated with Nanomaterials and Systems for Renewable Energy Laboratory, Research and Technology Center of Energy, Technoparc borje cedria, BP 095 Hammam Lif, Tunisia.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by King Abdelaziz City for Science and Technology under grant no. 1–17–01–0–010–0001.
