Abstract
The aim of this study is synthesis of polyurethane (PU) composites from 4,4′-methylenebis(cyclohexyl isocyanate), 1,2-ethanediol and different amounts of copper-based polyoxometalate (Cu-POM) and investigation of their dielectric properties. Firstly, Cu-POM structure as reinforcement was synthesized with ammonium heptamolybdate and Cu metal ion salt by a hydrothermal synthesis method. Then, Cu-POM-PU composites were prepared by in situ polymerization process. The prepared Cu-POM-PU composites were characterized by means of various techniques for their structure, morphology and thermal behaviour. The chemical structure, surface and morphological properties of the obtained composites were studied by Fourier transform infrared spectroscopy, X-ray diffraction spectroscopy, scanning electron microscopy (SEM) and energy-dispersive X-ray spectroscopy (EDX) techniques. Thermal properties of these Cu-POM-PU composites were determined by thermogravimetric analysis, differential thermal analysis and differential scanning calorimetry analysis techniques. Dielectric properties of the Cu-POM-PU composites were also determined. The dielectric measured results showed that the dielectric constants decreased with the increase of the Cu-POMs content. The Cu-POM-PU composites showed relatively low dielectric constant compared to pure PUs.
Introduction
Polymeric composites are particularly important in the electronics industry, especially for the production of capacitors, transistors, inductors, resistors, diodes, insulators, conductors, interconnections, printed circuit boards, interlayer dielectrics, connectors, thermal interface materials and so on; these types of composites are widely used in the field of microelectronics. 1,2 Among these fields of application, it is very important to develop new dielectric materials. In the electronics industry, during the preparation of low dielectric materials, there are two important production strategies. 2 In the first production strategy, materials having a lower polarity than Si-O or materials having low density are used. 2 The second production strategy aims to reduce the number of dipoles by reducing the density of a material with different additives. 2,3 For this purpose, the dielectric constant of polymers is decreased by increasing the free volume between the polymer chains by using different additives such as polyhedral oligomeric silsesquioxane, silicon dioxide, zinc oxide and so on. 4 -6 The aim of this study is to reduce the dielectric constant of polyurethane (PU) structures using copper-based polyoxometalates (Cu-POMs) structures which are easily produced and have important electrical properties. PUs have high potential in many applications due to their properties such as high tensile strength, compression strength, tear strength, high adhesion, good water resistant properties, high impact and abrasion resistance. 7,8 There are many studies in literature to improve dielectric properties of PUs. 9,10 In these studies, additives such as graphite, graphene oxide and cadmium sulfide were used to adjust the free volume and dielectric constant of PU structure. 11 -13 For reducing the dielectric constants of polymeric structures, POMs structures as reinforcement with superior electrical properties have very high potential. 14,15
POMs are discrete, molecularly defined, transition metal oxide clusters with sizes ranging from approximately one to several nanometres that exhibit various topologies and diverse chemical and electronic properties. 16 POM macroanions have attracted considerable interest because of their widespread use in many fields including catalysis, magnetism, electronic material, medicinal chemistry and materials science. 17 -20 But, the applications of POMs are seriously restricted by their poor processability. Thus, practical POM-based devices are quite rare. 21 -24 The POM-related research field has been experiencing an unparalleled development of new compounds but only a slow growth in POM-based materials and devices. 25,26 Polymers are used to solve this problem. Polymers are viable options as matrices for assembling POMs and exhibiting their functionality. Through properly combining POMs with polymer matrices, the resultant composite materials that integrate the unique functionalities of POMs and the advantages of polymers could find extensive use. 27 -38
In this study, PU-POMs nanocomposites were prepared from 4,4′-methylenebis(cyclohexyl isocyanate) and 1,2-ethanediol by blending Cu-POMs. The prepared PU-POMs nanocomposite films were characterized for their structure, morphology and thermal behaviour employing Fourier transform infrared (FTIR) spectroscopy, scanning electron micrograph (SEM), X-ray diffraction (XRD) and thermal analysis (differential thermal analysis (DTA)/thermogravimetric analysis (TGA)/differential scanning calorimetry (DSC)) techniques. This study showed that the dielectric properties increased despite the partially decreased thermal stability by the homogenous dispersion of POM into the PU matrix. The dielectric measured results showed that the dielectric constants decreased with the increase of the POMs content and these films showed relatively low dielectric constant when the POMs content was over 1 wt% compared to pure PU.
Experimental section
Instrumentation and reagents
4,4′-Methylenebis(cyclohexyl isocyanate) and 1,10-phenanthroline were ordered from Across Organics Co. (Illkirch, France) and dried in 4 Å molecular sieves before the experiment. Dibutyltin dilaurate was purchased from Sigma-Aldrich Corporation (Vienna, Austria). CuAc2.4H2O, ammonium heptamolybdate tetrahydrate, tetrahydrofuran (THF), trimethylamine, ethylene glycol, ethanol and KBr were supplied from Merck (Darmstadt, Germany). Dimethyl sulfoxide and dimethylformamide (DMF) were obtained from Carlo Erba (Milano, Italy). THF was also dried by vacuum distillation. Other organic solvents were purified by vacuum distillation. The other reagents used were of the highest purity and commercially available and solutions were prepared with distilled water.
FTIR (Mattson 1000) was used for detecting the functional groups of the materials in the range of 400–4000 cm−1 using KBr disks. Thermal properties of the Cu-POM and Cu-POM-PU composite materials were determined using TGA-50 (Shimadzu, Japan) and DTA-50 (Shimadzu) under a nitrogen (N2) flow at 50 mL min−1 and at a heating rate of 10°C min−1 in the temperature range from 30°C to 800°C. The initial sample mass was about 10 mg. Platinum crucibles were used for the TGA and DTA experiments. The DSC measurements were performed on a DSC-60 (Shimadzu) with a thermal analyser. All samples (about 5 mg) were placed in sealed aluminium pans before heating under N2 flow (20 mL min−1) at a scanning rate of 10°C min−1 in the temperature range from 35°C to 150°C. An empty aluminium crucible was used as reference. The structure and morphology of the composite were investigated with SEM (Scanning Electron Microscopy)/EDX (Energy-Dispersive X-ray Spectroscopy; LEO Evo-40 VPX). Chemical composition analysis of the synthesized Cu-POM structure was performed by EDX spectrum and elemental mapping with an EDX; Rönteck Xflash detector analyser associated with a scanning electron microscope (SEM; LEO Evo-40 VPX).
The dielectric measurements of the fabricated Cu-POM-PU composite in this study were measured using with the HP4294A Precision Impedance Analyzer at room temperature with ± 0.08% impedance sensitivity at 1 Hz to 1000 KHz. For dielectric measurements, 0.5 g sample was first converted into cylindrical discs under 10 tons pressure. Then, these discs were coated with 80 nm gold/palladium (Au/Pd) on both sides by using a sputter coater (Bal-Tec SCB 050). The dielectric constants of Cu-POM-PU composites with obtained sandwich structures of approximately 100 mm2 were determined between 1 Hz and 1000 kHz at room temperature.
Synthesis of Cu-POM
CuAc2.4H2O, (NH4)6Mo7O24.4H2O and o-phenanthroline in a ratio of 5:15:3 were dissolved in 90 ml of water. The pH value of the resulting solution was adjusted to 5–6 with 2 M NaOH. The reaction mixture was stirred at room temperature for 40 min. The temperature was adjusted to 200°C and obtained solution was magnetically stirred in a high-pressure Teflon container for 6 days. The reaction mixture was cooled to room temperature and purified by washing with ethanol. The obtained Cu-POM was dried in the vacuum oven at 40°C. 18,19
Preparation of the Cu-POM-PU composites
Cu-POM-PU nanocomposites were prepared with in situ synthesis methods by blending different amounts of Cu-POM and 4,4′-methylenebis(cyclohexyl isocyanate) and 1,2-ethanediol. Equimolar amounts of isocyanate and diol were added into a reaction flask with toluene as solvent, followed by heating up to 75°C. Then, the solution (solid content: 95 wt%) was reacted in the presence of dibutyltin dilaurate (0.02 wt%) at the same temperature for 3 h to synthesize the NCO-terminated prepolymer. After toluene was injected into flask to adjust the solid content (30 wt%), the solution was allowed to cool down to room temperature. Then, Cu-POM was dissolved in DMF and then added into flask under argon atmosphere. The whole system was kept at 80°C for 3 h to make it react completely. The obtained product was isolated by filtration and the resulting product was dried at room temperature. The prepared Cu-POM-PU nanocomposites were characterized for chemical structure, morphology and thermal behaviour employing FTIR, scanning electron micrograph (SEM) and thermal analysis (DTA/TGA/DSC) techniques. These studies showed the homogenous dispersion of Cu-POMs in the PU matrix with an increase in the thermal steadiness of the composite films on POMs loadings. The DSC measured results displayed that the glass transition temperature (T g) values increased with the increase of the POMs content and the films showed relatively high porous structure when the POMs content was over 1 wt% compared to pure PU.
Determination of the dielectric properties of POM-PU composites
The dielectric properties (dielectric constant) of Cu-POM-PU composites prepared by in situ polymerization from Cu-POM structure, 4,4′-methylenebis(cyclohexyl isocyanate) and 1,2-ethanediol, were tested by impedance analyser device at room temperature without additive at 1 Hz to 1000 KHz frequency range. During these analyses, firstly 0.5 g of each sample was weighed; 10 tons of pressure was applied to make a cylindrical pellet with a diameter of 10 mm. The prepared pellets were coated with Au/Pd (80 nm) on both sides to produce conductive surfaces. The dielectric constant measurements of each sample were performed with the HP4294A Precision Impedance Analyzer at room temperature with ± 0.08% impedance sensitivity.
Result and discussion
Cu-POM structures were successfully prepared by hydrothermal synthesis method. The microstructures and morphology of the as-obtained Cu-POM samples were studied by XRD, IR spectra, scanning electron microscopy (SEM) equipped with an energy-dispersive X-ray spectrometer (EDX) and TGA. Cu-POM-PU composites were synthesized by in situ synthesis method with this Cu-POM structure; 1%, 3%, 5% and 10% of Cu-POM were incorporated into PU matrix. Synthesized polymeric composites were analysed by FTIR, elemental analysis and X-ray spectra in comparison with Cu-POM structure and pure PU structure. SEM and thermal analyses were also carried out in comparison with pure PU structures, depending on the amount of additive added. Thermal analyses were performed with DTA, TGA and DSC techniques to determine the thermal stability, weight losses, char yield and T gs of the polymeric composites.
Characterization of Cu-POM
The Cu-POM structure obtained in the hydrothermal synthesis conditions using the Cu acetate salt has the closed formula and the charge balance is obtained using the phenanthroline ligand. It is a very bulky molecule in the structure of Cu-POM structure. Structural and chemical characterizations were performed by elemental analysis, IR spectroscopy and X-ray diffractometry. The IR spectrum of Cu-POM is given in Figure 1. Firstly, the peaks of the phenanthroline ligand at 1428–1623 cm−1 indicate the presence of this ligand. The IR spectrum of Cu-POM sample displays a symmetrical stretching vibration at 1058 cm−1, a Mo–O stretching vibration at 953 cm−1 and asymmetric stretching vibration at 878 cm−1 and Mo–O–Mo and Mo–O at 813 cm−1 for the metal oxygen bonds at the bridge. There are two important peaks at approximately 640 and 460 cm−1 in IR spectrum of Cu-POM structure and these peaks stem from the stretching Cu–O vibration. In conclusion, we have successfully synthesized Cu-POM structure.
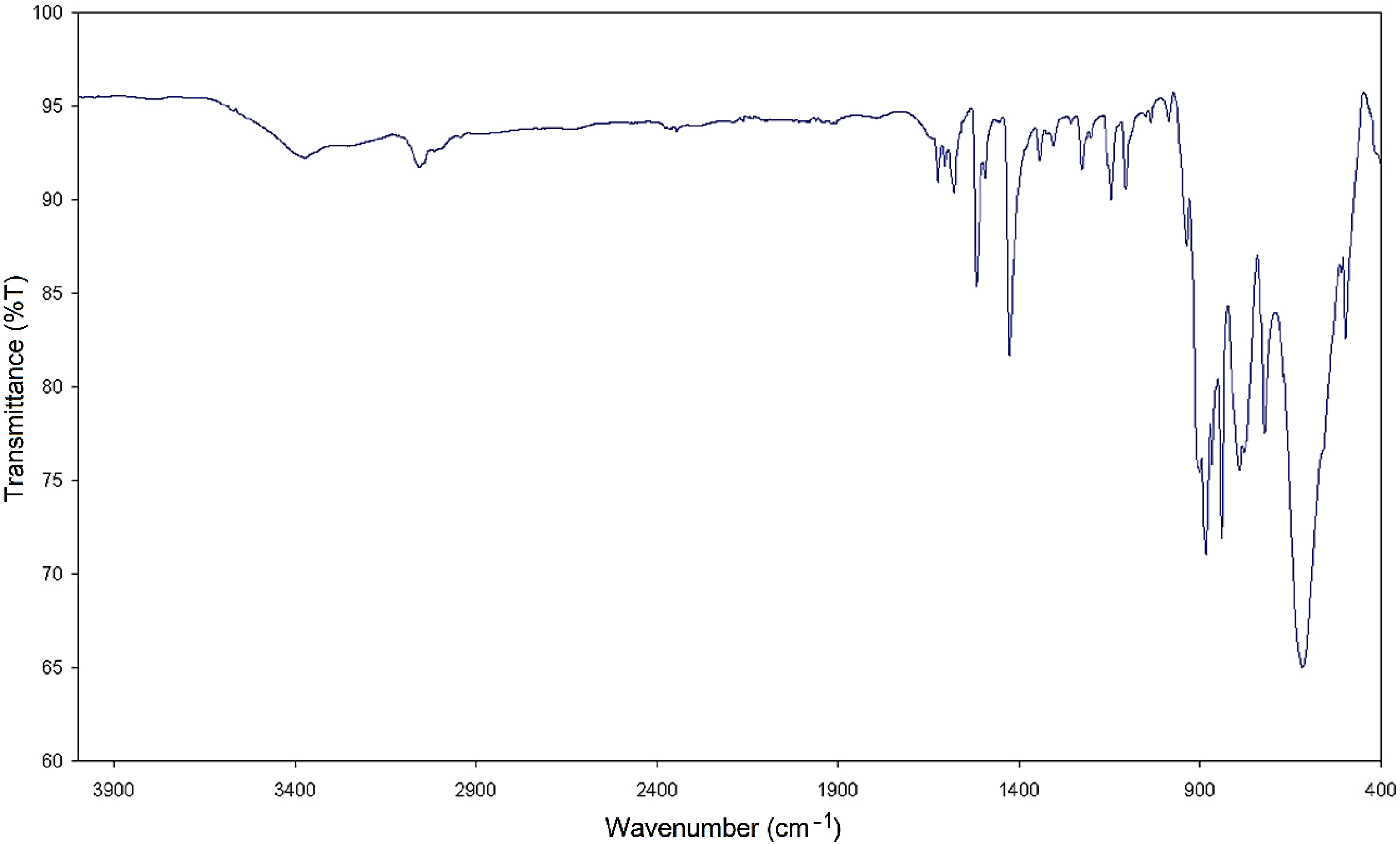
FTIR spectrum of Cu-POM structure.
The synthesized Cu-POM structure was also confirmed by X-ray diffractometry technique. Figure 2 shows the X-ray diffractogram of the prepared Cu-POM structure. In the X-ray diffractogram, there are four basic peaks at 7.5, 8.9, 11.3 and 20.1 Å. These peaks confirm that the structure is POM structure.35–37SEM images of the Cu-POM compound are given (Figure 3). As seen from these images, it is seen that there is a very homogeneous and uniform distribution. It appears that this morphology is preserved in the corresponding POM composites.
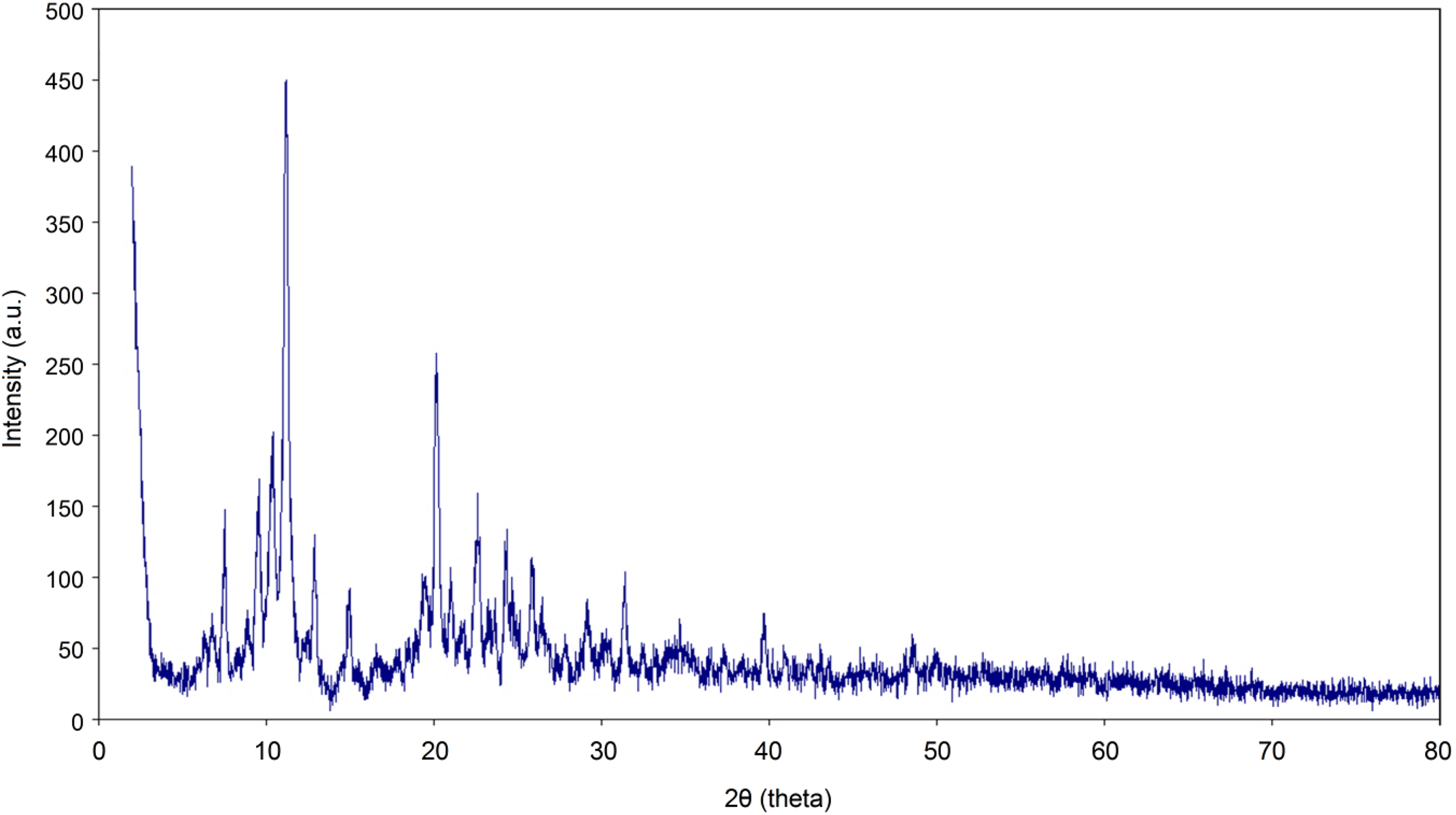
X-ray diffractogram of Cu-POM structure.

SEM images of the Cu-POM structure at different magnifications.
In addition, the EDX spectrum was taken by SEM analysis and the elemental distribution of the whole molecule was obtained by homogeneous distribution of Cu and Mo atoms of Cu-POM structure. The results of this analysis are given in Figure 4. According to these results, the POM structure consists of carbon (C), N, Cu, molybdenum (Mo) and oxygen (O) atoms. Cu and Mo atoms are distributed homogeneously in the structure. The results of this analysis prove that the desired structure is obtained homogeneously and purely.There are three weight losses in the TGA thermogram (Figure 5) of the Cu-POM structure. The first weight loss at between 100°C and 150°C corresponds to the removal of water molecules (about 1.98%). The second weight loss between approximately 300°C and 390°C is due to the loss of organic groups. The last weight loss starts at 430°C and is consistent with the DTA thermogram. When the TGA thermograms of the POM structures are examined, the prepared Cu-POM structure is thermally stable up to 308°C.
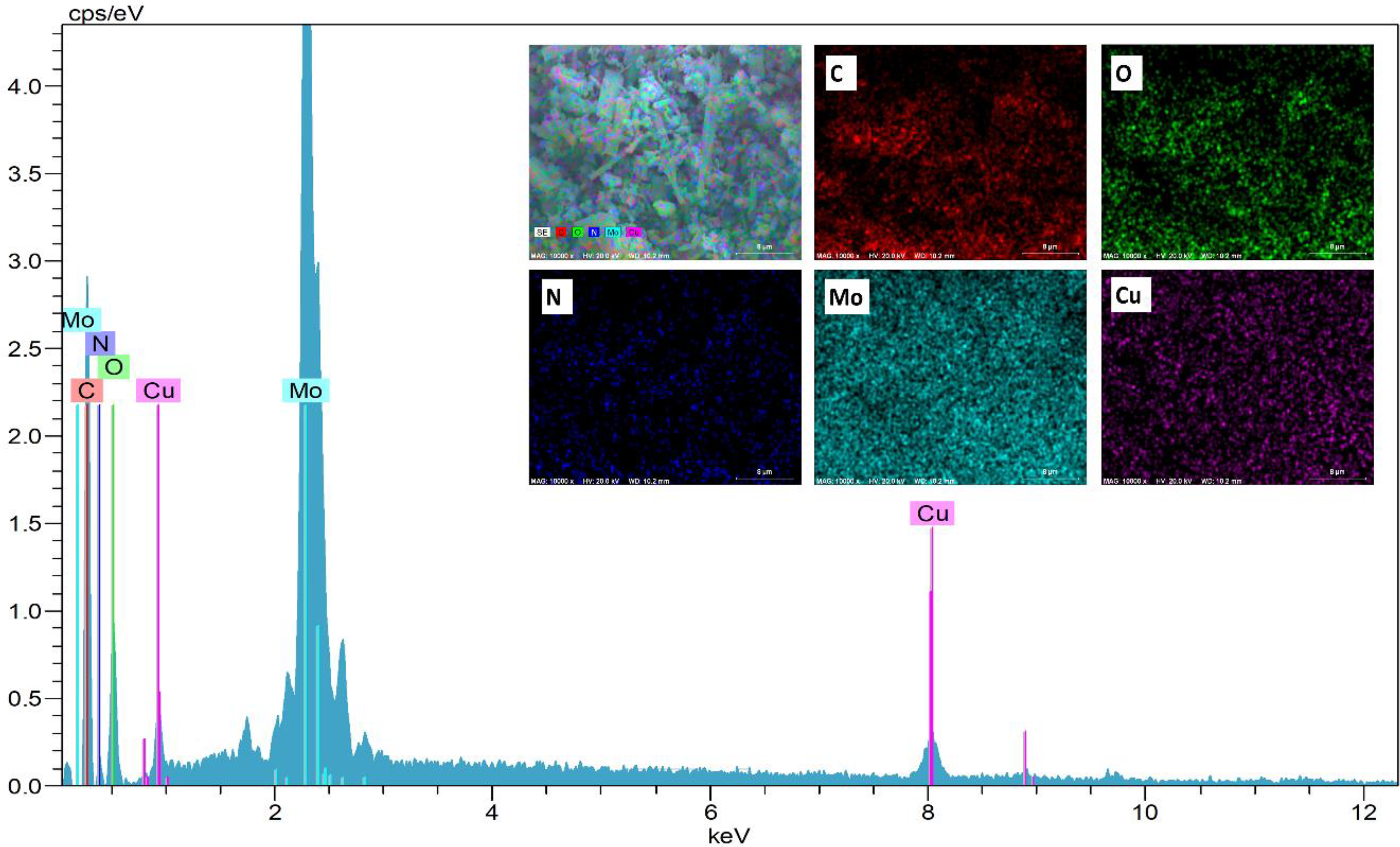
EDX spectrum of Cu-POM structure and mapping of C, O, N, Mo and Cu atoms.

TGA and DTA thermograms for Cu-POM structures under an N2 atmosphere (a flow rate of 50 mL min−1) at a heating rate of 10°C min−1.
Characterization of Cu-POM-PU composites
FTIR spectra of pure PU and PU composites synthesized using Cu-POM are given in Figure 6. When these spectra were examined, it was observed that the formation of urethane bond and the monomeric-free isocyanate peak disappeared. The absence of any peak at around 2280 cm−1 at all ratios is an indication of the formation of the PU structure and the absence of free isocyanate groups in the environment. The etheric bonds indicating the formation of the polymeric bond are clearly visible at 1274 cm−1. Except for these peaks, the carbonyl stretching vibration at 1716 cm−1, the bond N–H stretching vibration at 3200–3400 cm−1 and the C–N–O stretching vibration at 1584 cm−1 are clearly seen.SEM images of pure PU structure and Cu-POM composites are shown in Figure 7. When these SEM images were examined, it was observed that the surface properties of the composites change depending on the amount of Cu-POM additive in the composite structure. As the amount of Cu-POM in the composite structure increases, the surface properties and surface roughness of the composite increase. While the surface of the pure PU is smoother, Cu-POM-PU composites have a more fractal and rough structure.
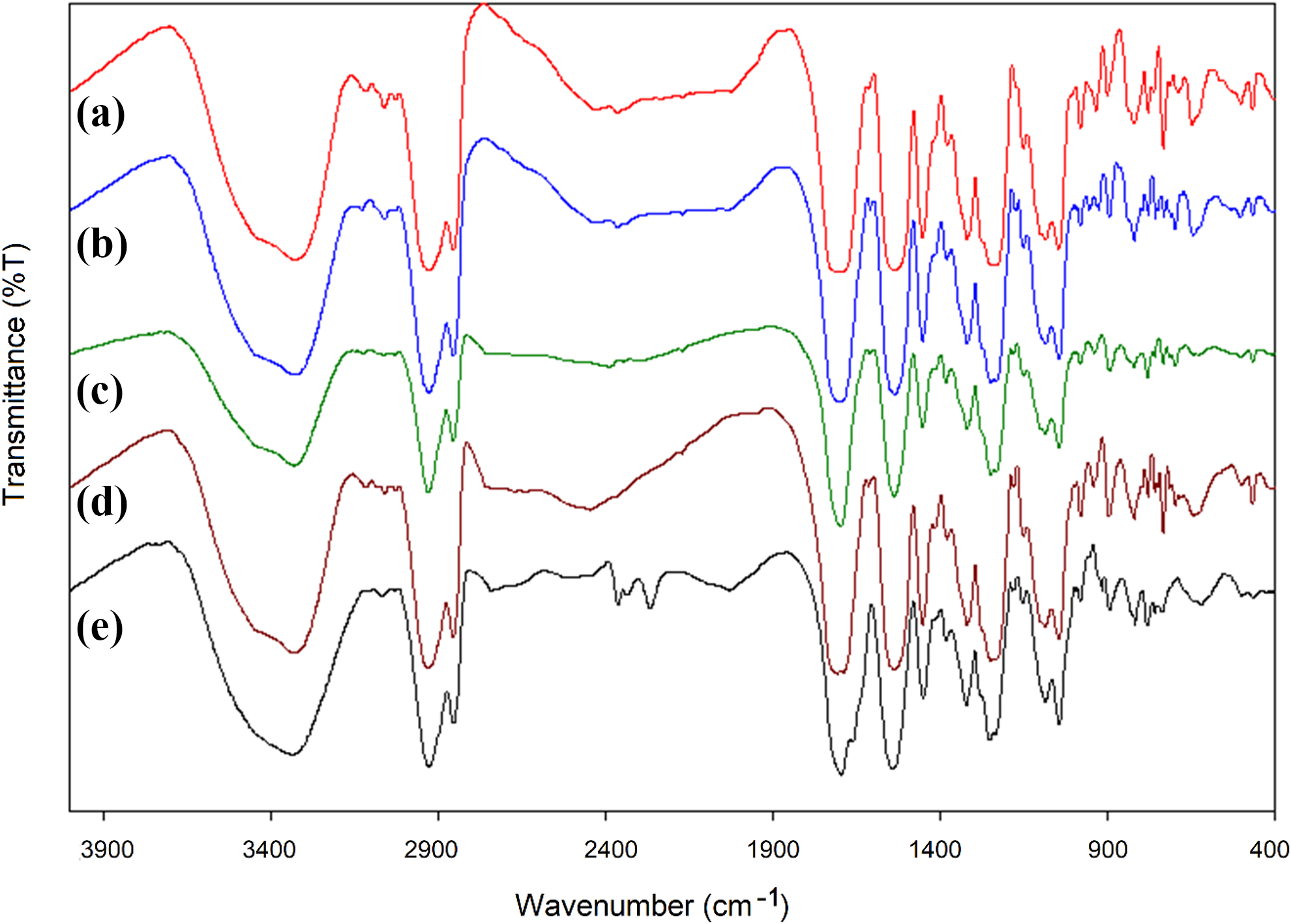
FTIR spectra of Cu-POM-PUs. (a) Pure PU, (b) Cu-POM-PU-1%, (c) Cu-POM-PU-3%, (d) Cu-POM-PU-5% and (e) Cu-POM-PU-10%.

Different magnification SEM images of pure PU (a) and Cu-POM composites. (b) Cu-POM-PU-1%, (c) Cu-POM-PU-3%, (d) Cu-POM-PU-5% and (e) Cu-POM-PU-10%.

General elemental topography and C, Mo, Cu mapping of Cu-POM composites. (a) Cu-POM-PU-1%, (b) Cu-POM-PU-3%, (c) Cu-POM-PU-5% and (d) Cu-POM-PU-10%.
The DSC thermograms of polymers containing Cu-POM at 1%, 3%, 5% and 10% are examined (Figure 9). There is a significant increase in the T g values. The T g value of the pure polymer structure is 57.30°C, but all composites have a T g above this value. The 1% composite has 62.42°C, the 3% composite has 66.32°C and the 5% composite has 72.89°C T g, while the 10% composite has a T g value of 75.04°C.

DSC thermograms and T g values of pure PU and Cu-POM-PU composites.
DTA and TGA spectra given in Figures 10 and 11 are examined, and two basic mass losses are observed. DTGA thermograms of pure PU and Cu-POM-PU composites at N2 atmosphere are also given in Figure 11. The first shows the cleavage of the CH2 groups and the etheric units attached to these groups in the polymer structure. The second fundamental mass loss is the degradation of the polymeric structure. The thermal stability of the polymeric composites is lower than that of the pure polymer. This is because the POM molecules are structurally oxidation catalysts and facilitate the oxidation of the polymer structure. Dielectric properties of Cu-POM/PU composites
The frequency dependence of the dielectric properties of the PU composites prepared at different additive ratios prepared in the study was measured and compared with pure PU, as shown in Figure 12. When these curves are examined, it is seen that the dielectric properties of PU composites are lower than those of pure PUs.

DTA thermograms of pure PU and Cu-POM-PU composites under N2 atmosphere (a flow rate of 50 mL min−1) at a heating rate of 10°C min−1.

TGA and DTGA thermograms of pure PU and Cu-POM-PU composites under N2 atmosphere (a flow rate of 50 mL min−1) at a heating rate of 10°C min−1.

Dielectric changes of Cu-POM-PU composites prepared using 4,4′-methylenebis(cyclohexyl isocyanate).
In this study, Cu-POM-PU composites were prepared by adding Cu-POM additives in pure PU matrix. These composites were used as materials that can be used to prepare capacitors for PU structure with proper energy storage capability. The purpose of the incorporation of additive materials is to examine the extent to which these materials have changed their dielectric properties. The three important factors that influence the dielectric properties in polymeric structures are the cemented density of the polymeric structure resulting from this effect, the polarizability and the free volume.
In the PU composites synthesized, dielectric constants ranged from 9.1 to 10.1 according to their polarizability and H-bonding ability of PUs prepared from diisocyanates. With the structural POM doping, the morphologies and free volume values of the polymers changed and this change led to the change of the dielectric constants of the polymeric composites. This change has become even more intense with the increasing amount of additives, leading to a reduction in the dielectric constant relative to the pure polymer. In PU composites prepared, there is no good adhesion between the additive and matrix. When an evaluation between prepared composites is made, the free volume ratio is very large as seen in SEM images of composites prepared using Cu-POM additive. Therefore, the dielectric constants of these composites are lower than that of the pure PU structure.
Conclusion
In this study, Cu-POM structures were synthesized in the presence of phenanthroline ligand using Cu salts. When these syntheses are examined, it can be seen that the POMs obtained are very thermally stable up to 300–350°C. Structural characterizations of Cu-POM structure are carried out by FTIR, elemental analysis and XRD, and this structure has a very bulky structure. PU composites were obtained by using this Cu-POM structure as a reinforcement (1%, 3%, 5% and 10% weight ratio). The synthesized composites were characterized in detail by different techniques. T g values of composites were increased according to pure PU structure. In addition, dielectric constants of these composites were decreased with the addition of Cu-POM groups. As a result, the obtained Cu-POM compound can be used as additives in reducing the dielectric constants of PUs due to their volumetric compatibility and thermal stability.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
