Abstract
New liquid crystalline thiadiazole dibenzaldehyde monomers labeled as THDB1–THDB3 were successfully synthesized by alkylation of thiadiazole’s potassium salt with 4-(bromomethyl) benzaldehyde. A number of polymers consisting of thiadiazole and azomethine coded PTDAZ1–PTDAZ5 were synthesized via condensation reaction of the presynthesized monomers THDB1–THDB3 with aromatic diamines. The chemical structures of the prepared materials were confirmed using Fourier-transform infrared spectroscopy and proton nuclear magnetic resonance techniques. The liquid crystalline behavior of the studied monomers and polymers was examined by differential scanning calorimetry and hot stage polarized optical microscopy (POM) techniques. All these compounds were found to demonstrate mesomorphic transitions belonging to smectic and nematic liquid crystals. The studied monomers exhibited fan-like texture of smectic mesophase under POM cooling investigation. Fan-like texture of smectic and nematic mesophases was observed under POM for PTDAZ1 and PTDAZ2, while PTDAZ3 and PTDAZ4 revealed clay and schlieren textures of the smectic and nematic mesophases, respectively, and nematic texture has been found for the polymer PTDAZ5.
Introduction
The liquid crystalline term describes a unique state of matter in which the molecules diffuse much like the molecules of a liquid but are able to maintain some degree of orientational and positional order like that of a crystal. Within this state of matter, aggregation of two opposite characteristics occur: isotropic fluidity and anisotropic order. Materials in this state behave like a liquid in view of one aspect, but still hold a crystal attitude on the other. Substances that have such a unique combination of phases are named as mesogens, and the different phases in which they could appear are called mesophases. The main requirement for mesomorphic behavior is the presence of a high geometrical anisotropic shape, like a rod or disc in a rigid core, that is, the presence of heterocyclic or aromatic rings. 1 Transition of this rigid conjugated core into isotropic liquid may pass through one or more mesophases depending on the geometrical structure. The appearance of these mesophases, however, may be enhanced by the influence of thermal process (thermotropic mesomorphism) or by the influence of a suitable isotropic solvent (lyotropic mesomorphism). Thermotropic liquid crystals can, on the other side, be classified into three molecular fashions, which are called calamitic for rod-like, discotic for disc-like, and sanidic for brick- or lath-like molecules. Moreover, the mesomorphic phases that arise from thermotropic liquid crystals as a result of temperature, constituent, concentration, and substituent changes are called nematic, cholesteric, and smectic. 2,3
On the other hand, incorporation of heteroatoms such as nitrogen or sulfur into the core unit of the thermotropic liquid crystal materials can impart a wide range of changes in their mesophases and in the physical properties due to polarization increases of these heteroatoms. 4,5 However, vast research investigations have been conducted on liquid crystal compounds containing heterocyclic rings, in particular, the 1,3,4-thiadiazole and/or oxadiazole moieties. The importance of these heterocyclic rings arise from their thermal stability, high quantum yield of luminescence, electron transport properties, and wide applications like optical, electrical, and medical fields. 6 -10
Incorporation of Schiff base (SB) linkages into 1,3,4-thiadiazole system as core is a modern class of liquid crystalline materials. Such linkage system gives suitable route to design bent- and/or banana-shaped liquid crystalline molecule. However, research related to the liquid crystalline properties of SBs were started with the discovery of N-(4-methoxybenzylidene)-4-butylaniline and its derivatives (Figure 1) which exhibits a nematic phase at room temperature. 11 -13

Old and famous Schiff bases in the history of liquid crystals. 13
In this regard, SBs are state of the art for building banana-shaped or bent-shaped liquid crystalline molecules. It is a new theme of liquid crystals science owing to their ability to self-organize and show self-assembly properties. 14 , 15 Nonetheless, their inherent chemical structure and thermal and photophysical instability restrain its use in electro-optical displays. This challenge has encouraged researchers to design new molecular architecture calamitic SB mesogens containing 1,3,4-thiadiazole in their chemical structures. Therefore, the aim of this work is to design novel polymers which contain both moieties, 1,3,4-thiadiazole ring and SB linkage. Such a polymer system can be synthesized through condensation polymerization of dialdehyde monomers containing 1,3,4-thiadiazole rings with aromatic diamines. The chemical structures of all prepared polymers were examined by Fourier-transform infrared (FTIR) spectroscopy and proton nuclear magnetic resonance (1H NMR) techniques. The liquid crystalline behavior of both monomers and polymers were tested by the use of hot stage polarized optical microscopy (POM) and differential scanning calorimetric (DSC) techniques.
Experiment
Chemicals and reagents
Chemicals, reagents, and solvents used in this work were purchased from Sigma-Aldrich (Sigma-Aldrich, Taufkirchen, Germany), Fisher Scientific (Fischer Scientific, Hampton, New Hampshire, USA), Merck (Merck KGaA, Darmstadt, Germany), and Fluka companies (Fluka, Industriestrass 25, Buchs, Switzerland). All the materials, anhydrous solvents, and reagents were used without further purification, unless specified.
Instrumental design and techniques
Infrared spectroscopy was performed on all samples using FTIR spectrophotometer, Shimadzu 8400 (FTIR: Shimadzu 8400 spectrophotometer Nishinokyo Kuwabara-cho, Nakagyo-ku, Kyoto 604-8511, Japan), by using KBr discs in the range of 400–4000 cm−1. Bruker (Billerica, Massachusetts, USA) 400 MHz spectroscopy instrument was used for measuring the 1H NMR. Phase transition temperature measurements were carried out by DSC and hot stage POM. The DSC equipment was a Mettler Toledo (Columbus, Ohio, USA) with thermal analysis processor Mettler TC 10A based on a TA 3000 system with GraphWare TA 72 software. The DSC measurements were examined under N2 atmosphere with a heating rate of 10°C min−1 in case of polymers and 5°C min−1 in case of monomers. POM (Linkam HFS 91, Nikon) fitted with a Linkam (Epsom Downs Metro Centre Tadworth, Surrey, KT20 5LR UK) THMS-600 heating stage was used to confirm the liquid crystal mesophases of the prepared monomers and polymers. All these measurements were performed kindly at the Faculty of Science, Engineering and Computing Materials Research Centre, Kingston University, London.
Organic synthesis
Synthesis of thiadiazole monomers
The chemical synthesis of the liquid crystalline monomers containing 1,3,4-thiadiazole moiety is illustrated in Figure 2. The conducted method is the same for all of the prepared monomers following a typical procedure: Synthesis of 4,4′-[1,3,4-thiadiazole-2,5-diylbis (sulfanediylmethanediyl)] dibenzaldehyde (THDB1): 1,3,4-Thiadiazole-2,5-dithiol (0.3 g, 2 mmol) was dissolved in anhydrous Dimethyl Formamide (DMF) (10 mL) and placed in a three-necked flask provided with a condenser and N2 inlet–outlet tube. The reaction was run under N2 gas by evacuation and purging the reaction vessel with N2. A known quantity of potassium carbonate was added with stirring followed by dropwise addition of 4-(bromomethyl) benzaldehyde (0.796 g, 4 mmol) dissolved in anhydrous DMF. The reaction mixture was heated at 150°C for 8 h. Then, the mixture was cooled to room temperature and poured into 250 mL of distilled water. The light brown precipitate was collected by filtration and washed, thoroughly, with 5% aqueous NaHCO3 solution and distilled water. The crude product was recrystallized from ethanol. The yield was 82% and the melting point was 250°C. The 1H NMR (400 MHz, CDCl3) indicate 10.52 ppm (s, CHO), 7.18–7.55 ppm (d, aromatic protons of benzene rings), and 4.25 ppm (s, S–CH2). The compound of 4,4′-[2,2′-bi-1,3,4-thiadiazole-5,5′ diylbis (sulfanediyl-methanediyl)] dibenzaldehyde (THDB2) was prepared from the reaction of 2,2′-bi-1,3,4-thiadiazole-5,5′-dithiol (0.4686 g, 2 mmol) and 4-(bromomethyl) benzaldehyde (0.796 g, 4 mmol) according to the same procedure for THDB1. The dark brown yield was 74% and the m.p. was 178°C. The 1H NMR (400 MHz, CDCl3) indicate 9.97 ppm (s, CHO), 7.57–7.46 ppm (d, aromatic protons of benzene rings), and 4.14 ppm (s, S–CH2). The compound of 4,4′-[ethane-1,2 diylbis (sulfanediyl methanediyl))] dibenzaldehyde (THDB3) was synthesized from the reaction of ethane-1,2-dithiol (0.18840 g, 2 mmol) and 4-(bromomethyl) benzaldehyde (0.796 g, 4 mmol) following the same reaction conditions as mentioned for THDB1. The yield of the brown precipitate was 79% and the m.p. was 67°C. The 1H NMR (400 MHz, CDCl3) indicate 9.98 ppm (s, CHO), 7.77–7.43 ppm (d, aromatic protons), 3.81 ppm (s, S–CH2), and 2.25 ppm (s, aliphatic CH2).

The chemical reaction for synthesis of the monomers and the polymers.
Synthesis of thiadiazole azomethine polymers
Synthesis of PTDAZ1: An equimolar of THDB1 monomer (0.3865 g, 1 mmol) and 4,4′-sulfonyldianiline (0.2483 g, 1 mmol) dissolved in isopropyl alcohol: chloroform 1:1 (v/v) was placed in a three-necked round bottom flask fitted with Dean–Stark and condenser. A few drops of trifluoroacetic acid (TFA), as catalyst, were added and the reaction mixture was heated under slight vacuum (section pump vacuum) at 100–120°C for 48 h. Then the cooled mixture was poured into methanol (50 mL), and the precipitate was filtered. The product was washed with hot methanol: acetone for 24 h by using Soxhlet device to remove the traces of low molecular weight polymers and unreacted starting materials. Finally, the brown product (PTDAZ1) was dried under vacuum for 24 h. Yield: 78%; IR ν cm−1: (3018, 2824, 1618, 1600, 1517, 1208, 844); 1H NMR (dimethyl sulfoxide (DMSO) d6, ppm): 8.74 (s, CH=N), 7.79–7.13 (aromatic protons), 3.98 (CH2–S–Ar); m.p.: decompose.
The polymer PTDAZ2 was prepared by the reaction of an equimolar of THDB1 and biphenyl-4,4′-diamine following the same strategy for PTDAZ1. The yellow product (PTDAZ2) was dried under high vacuum for 24 h. Yield: 70%; IR ν cm−1: (3009, 2902, 1639, 1596, 1514, 1196, 844); 1H NMR (DMSO d6, ppm): 8.23 (s, CH=N), 7.97–7.01 (aromatic protons), 4.25 (CH2–S–Ar); m.p.: decompose.
The polymer PTDAZ3 was prepared by the reaction of an equimolar of THDB2 monomer and biphenyl-4,4′-diamine following the same strategy for PTDAZ1. The deep brown product (PTDAZ3) was dried under high vacuum for 24 h. Yield: 75%; IR ν cm−1: (3060, 2868, 1634, 1600, 1517, 1140, 844); 1H NMR (DMSO d6, ppm): 8.50 (s, CH=N), 7.78–7.13 (aromatic protons), 4.51 (CH2–S–Ar); m.p.: decompose.
The polymer PTDAZ4 was prepared by the reaction of an equimolar of THDB2 monomer and 4,4′-methanediyldianiline following the same strategy for PTDAZ1. The yellow product (PTDAZ4) was dried under high vacuum for 24 h. Yield: 78%; IR ν cm−1: (3020, 2856, 1654, 1600, 1517, 1116, 844); 1H NMR (DMSO d6, ppm): 8.44 (s, CH=N), 7.79–7.2 (aromatic protons), 4.30 (CH2–S–Ar); m.p.: decompose.
The polymer PTDAZ5 was prepared by the reaction of an equimolar of THDB3 monomer and biphenyl-4,4′-diamine following the same strategy for PTDAZ1. The brown product (PTDAZ5) was dried under high vacuum for 24 h. Yield: 78%; IR ν cm−1: (3014, 2893, 1652, 1600, 1517, 1168, 844); 1H NMR (DMSO d6, ppm): 8.68 (s, CH=N), 7.01–7.68 (aromatic protons), 3.57 (CH2–S–Ar); m.p.: 234–236°C.
Results and discussion
Organic synthesis and characterization
The monomers (THDB1–THDB3) were prepared by a two-step mechanism. First one is the formation of thiadiazole’s potassium salt via the reaction of potassium carbonate with thiadiazoles. The second step is the nucleophilic substitution of this salt with 4-(bromomethyl) benzaldehyde whereby the bromine group is a good leaving group giving the final products (Figure 2). The FTIR investigations for the prepared monomers (Figure 3 and Table 1) demonstrate the absence of the bands belonging to C–SH and C–Br for the reactant which should appear at 763 and 1043 cm−1, respectively. Vice versa, a new strong to medium intensity bands in the range of 1164–1174 cm−1 correlated to C–S vibrations for the produced monomers were emerged instead. Table 1 lists the important FTIR bands for the prepared monomers.

FTIR spectra of monomers (a) THDB2 and (b) THDB3.
FTIR bands for the prepared monomers.
FTIR: Fourier-transform infrared.
The 1H NMR profile for the prepared monomers (THDB1–THDB3) is exhibited in Figure 4. The spectrum for monomer THDB1 (Figure 4(a)) indicated a singlet band at 10.52 ppm belonging to the CHO terminal proton, a doublet at 7.18–7.55 ppm corresponding to aromatic protons of benzene ring, and a singlet at 4.25 ppm due to CH2 adjacent to sulfur atom, while the 1H NMR for monomer THDB2 (Figure 4(b)) presented a singlet band at 9.97 ppm assigned as CHO terminal proton, a doublet at 7.57–7.46 ppm for the aromatic protons, and a singlet at 4.14 ppm due to S–CH2 segments. In case of monomer THDB3, the 1H NMR spectra (Figure 4(c)) display a singlet peak at 9.98 ppm presented by CHO group and a doublet in the range of 7.77–7.43 ppm for the aromatic protons. Whereas, the protons of the group S−CH2 appear as a singlet at 3.81 ppm, and the aliphatic −CH2− is sited as singlet at 2.25 ppm.

1H NMR spectra of the prepared monomers: (a) THDB1, (b) THDB2, and (c) THDB3).
The poly-SBs, on the other hand, were produced by condensation polymerization in the presence of isopropyl alcohol: CHCl3 solvent mixture and few drops of TFA as catalyst. The polymerization mechanism is thought to proceed via protonation of the terminal CHO groups (Figure 2). Protonation, however, makes the aldehyde group more susceptible to nucleophilic attacks by the nitrogen atoms to form azomethine linkages and subsequently release water molecules as a by-product. Since the polymerization reaction is a reversible process, therefore, a Dean–Stark device was used to eliminate the accumulated water and direct the reaction toward the products. The chemical structures for the prepared polymers were confirmed by FTIR (Figure 5 and Table 2) and 1H NMR techniques (Figure 6).

FTIR spectra of prepared polymers (a) PTDAZ4 and (b) PTDAZ5.
FTIR bands for the poly-SBs.
FTIR: Fourier-transform infrared; SB: Schiff base.

1H NMR spectra of the prepared polymers: (a) PTDAZ1, (b) PTDAZ2, (c) PTDAZ3, (d) PTDAZ4, and (e) PTDAZ5.
The FTIR data (Figure 5) confirmed the absence of the stretching vibrations for the amine and CHO groups, for the monomers, at the range of 3308–3246 cm−1 and 1708–1688 cm−1, respectively. On the other hand, a strong SB band for the polymers at the range of 1654–1618 cm−1 was detected. Table 2 indicates the most important FTIR bands for the prepared polymers. The 1H NMR spectra for the prepared polymers are further confirmation to their chemical structures. However, a singlet in the range of 8.23–8.74 ppm for all polymers is ascribed to the azomethinic protons, which displaced the aldehyde protons at 10.52–9.98 ppm (Figure 6). The other proton signals appeared at their usual chemical shifts. These results are clear indication that the condensation reaction was carried out successfully. Table 3 transcribes the characteristics of 1H NMR signs for the prepared poly-SBs (PTDAZ1–PTDAZ5).
The 1H NMR signs for the poly-SBs.

1H NMR: proton nuclear magnetic resonance; SB: Schiff base; DMSO: dimethyl sulfoxide.
Liquid crystalline properties
The DSC technique is a very useful tool to probe mesophases in liquid crystalline materials through heating and cooling scans. However, the liquid crystalline textures and phase transition of the synthesized monomers and the corresponding polymers were monitored by DSC and hot stage POM. The results are summarized in Table 4 and the DSC thermograms (for THDB1, PTDAZ1, PTDAZ3, and PTDAZ4) are shown in Figures 7 to 10. The DSC thermograms for the monomers indicated endothermic peaks correlated to the mesomorphic transitions. For instance, monomer THDB1 (Figure 7) indicates endothermic transition at 199°C during heating scan which is assigned as a transition from crystalline state into smectic SB mesophase, whereas the peak at 257°C corresponds to a transition into isotropic phase. Monomer THDB2 (Table 4) reveals three endothermic transitions at 154°C that probably belonged to smectic SA mesophase, second peak at 162°C corresponds to smectic SC, and third peak at 175°C which is assigned as a transition to an isotropic phase.
Phase transition temperatures and visual observations of thiadiazole dibenzaldehyde monomers (THDB1–THDB3) and polymers (PTDAZ1–PTDAZ5) from DSC and POM.

DSC: differential scanning calorimetry; POM: polarized optical microscopy; Cr: crystal; I: isotropic; SA: smectic A; SB: smectic B; SC: smectic C.

DSC thermogram of THDB1 during heating scan and its microphotograph.

DSC thermogram of PTDAZ1 during heating scan and its photomicrographs.

DSC thermogram of PTDAZ3 during heating scan and its photomicrograph.

DSC thermogram of PTDAZ4 during heating scan and its photomicrographs.
Heating scan for the polymer PTDAZ1 (Figure 8) revealed endothermic transitions at 239, 280, and 306°C which are respectively ascribed to smectic and nematic mesomorphic forms. DSC tracing values for the polymer PTDAZ2 (Table 4) elucidated endothermic peak at 302°C during heating which is assigned as nematic phase transition. DSC thermogram for PTDAZ3 (Figure 9) showed endotherm characteristics in the range of 271–292°C, probably belonging to nematic liquid crystal phase transition. The small sharp endothermic peak located at 121°C in the case of PTDAZ4 (Figure 10 and Table 4) and the small peaks at 229 and 259°C were correlated to the smectic to nematic and to isotropic phase transitions, respectively.
Hot stage POM was investigated to confirm the transition temperatures obtained by DSC thermograms and to demonstrate the type of mesophases (texture) present in the studied materials. All the monomers and polymers in this work were heated (5°C min−1) on glass slides up to melt on the hot stage microscope. The phase changes were observed during cooling (3°C min−1) in the case of monomers, whereas polymers were examined during heating scan only. The microphotographs acquired by POM are shown in Figures 11 to 13. The POM monitoring during cooling scan at 190°C of monomer THDB1 (Figure 11(a)) elucidates a fan texture of a smectic phase, whereas the THDB2 microphotographs (Figure 11(b) and (c)) demonstrate the presence of smectic phases SC and SA during cooling scan at 163 and 152°C, respectively. The POM observations for the monomer THDB3 indicated no liquid crystalline mesophases which is probably due to the presence of ethyl groups that impart a kind of flexibility in the core of the monomer structure.

POM microphotographs observed from isotropic on cooling: (a) smectic SB fan texture of THDB1, (b) smectic SC fan texture of THDB2, and (c) smectic SA fan texture of THDB2.
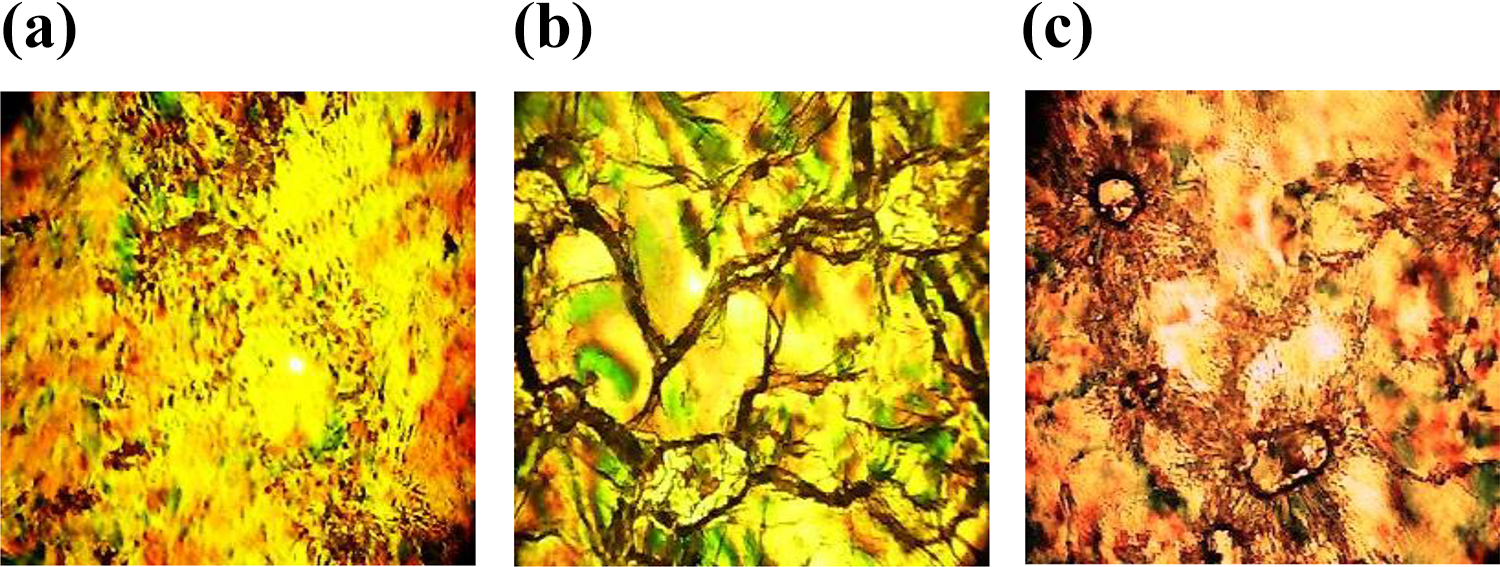
POM microphotographs observed during heating scan: (a) smectic SB fan texture of PTDAZ1, (b) nematic phase fan texture of PTDAZ1, and (c) nematic phase fan texture of PTDAZ2.

POM microphotographs observed during heating scan: (a) nematic phase clay texture of PTDAZ3, (b) smectic SA phase of PTDAZ4, (c) nematic phase schlieren texture of PTDAZ4, and (d) nematic phase needle texture of PTDAZ5.
Polymer PTDAZ1 under POM observations showed similar mesomorphic smectic SB fan texture at 278°C similar to that displayed by monomer THDB1 (Figure 12(a)). Interestingly, PTDAZ1 and PTDAZ2 exhibit nematic phase at 302°C (Figure 12(b) and (c)). On the other side, POM observations of PTDAZ3 confirmed clay texture of nematic mesophases (Figure 13(a)) at 290°C, while for PTDAZ4 (Figure 13(b) and (c)), the liquid crystal phases indicate smectic SA and nematic phase (schlieren texture) at 235 and 258°C, respectively. A nematic texture with long-range directional order was confirmed under POM for PTDAZ5 (Figure 13(d)). This behavior, however, might be attributed to the formation of a new rigid mesogenic core in the polymer PTDAZ5 backbone (Figure 14).

Rigid mesogenic core of PTDAZ5.
Generally, these mesophases for polymers and monomers are arising due to the specific constituents in terms of polarization, hardness, and bending capability. The presence of carbonyl groups and the heterocyclic system are predominantly responsible for the polarization features of the monomer molecules, while the planar conjugated system contributes to its rigidity or elucidate the hardness merits. Bending shape, on the other hand, is due to the SB groups and methylene sulfide spacers that are more inclined to be bent rather than arrange linear. The other factors such as attraction forces between molecules as well as hydrogen bonding are significantly contributed in exhibit smectic phases. In fact, monomers and the corresponding polymer molecules are arranged in a way to be positional along one direction within or between the layers which form the smectic phases.
By contrast, the occurrence of the nematic phase in the studied polymers is basically explained in terms of temperature increment, whereby the energy of the molecules increases and overcomes the attraction forces between them. Hence, the polymer molecules are losing its positional order and self-align to have long-range directional order with their axes to elucidate the nematic mesophases transitions.
Conclusions
New bent-shaped poly-SBs containing 1,3,4-thiadiazole were successfully synthesized by polycondensation reaction of their presynthesized monomers. The polymers and monomers were characterized by FTIR and 1H NMR techniques. All of the prepared monomers and polymers presented mesomorphic characteristics under DSC and POM techniques. The existence of these mesophases was attributed to the heterocyclic (thiadiazole rings) core. In addition, the factors such as attraction forces between molecules and hydrogen bonding have significantly contributed to the emerging smectic and nematic phases. The existence of the thiadiazole core and the carbonyl terminal groups in the backbone of the prepared materials impart their polarization characters, while the phenyl groups enhance the stiffness. SB linkages and methylene sulfide spacers render the polymer materials to favor bent conformation over linear one. The bent alignment of the monomers and polymers within layers conferred the banana- or bent-shaped liquid crystal (LC) of a smectic phase. The absence of heterocyclic system in the middle of the monomer THDB3 structure, which replaced by ethyl group, made this monomer highly flexible. Therefore, no mesomorphsim was affirmed for this monomer. Whereas the polymer PTDAZ5 produced from the reaction of monomer THDB3 and aromatic diamine presented an LC phase of nematic texture. The emergence of this mesomorphsim in the case of PTDAZ5 is attributed to the rigid mesogenic core in the polymer backbone.
Footnotes
Acknowledgment
The authors thank Professor Peter Foot, Kingston University, London, England, for his help in H1 NMR, DSC, and POM measurements and appreciate the support provided by the Chemistry Department, College of Science, Tikrit University, Iraq.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
