Abstract
Polymer composite electrolyte based on polysulfone-sodium sulfonate (SPSU(Na)) blended with poly (polyethylene glycol methacrylate) (PPEGMA) using nano-sized hexagonal boron nitride (nano-hBN) as filler was fabricated using a solution casting technique for use in Na-ion batteries. Polysulfone was sulfonated by a post sulfonation method followed by ion exchange with sodium hydroxide. Fourier transform infrared spectroscopy was used to study SPSU(Na)/PEGMA blend and incorporation of hBN nanoparticle into the SPSU(Na)/PPEGMA blend. Thermal properties of the composites were studied with thermogravimetric analysis (TGA) and differential scanning calorimetry tests. X-ray diffraction was used to study phase change. The TGA curve showed two weight loss regions, where 30% weight loss occurred between 200°C and 350°C due to degradation of sulfonic acid groups, and the polymer backbone degradation occurs above 500°C. Surface morphology of the membranes was examined using scanning electron microscopy which reveals the homogeneous dispersion of the nano-hBN particles in the polymer matrix. Ionic conductivity was studied with impedance spectroscopy and the total ionic conductivity increases with increasing PPEGMA ratio. SPSU(Na)/PPEGMA(1:4) sample showed maximum ion conductivity of approximately 5.5 × 10−6 S cm−1 (5.5 × 10−4 S m−1) at 100°C because of the high content of PPEGMA.
Keywords
Introduction
Over the past decade, storage of energy has happened to an arising global worry due to a growth of smart grid’s needs in inexpensive and sustainable energy storage, 1 in conjunction with extreme rises for the cost of fuels and environmental aftermaths of using them. This raised the call for substitute sources for the generation as well as storage of energy. It is acknowledged that storage of energy technology such as a battery is going to be the key to future development of renewable energy. 2 Rechargeable lithium (Li)-ion batteries (LIBs), comprising of Li-ion conducting electrolyte and two Li insertion electrodes, have become prospering and sophisticated energy storage devices since the first commercialization of LIB (carbon/LiCoO2 cell) in 1991. 3 Li-based electrochemistry provides various attractive attributes: Li is the lightest metal with an exceptionally low redox potential (Li+/Li = −3:04 V vs standard hydrogen electrode), 4 this makes the cells having high energy density and voltage. Moreover, Li ion has a very small ionic radius which is advantageous for easy diffusion into materials. LIBs also have long cycle life with rate capability. These properties made Li-ion technology to confine the market of portable electronics. 5 Unfortunately, the scarceness of Li is a cause for concern. As Li is constantly mined from the natural deposits, the availability of it might likely exhaust to unsustainably depleted levels. This signifies that future LIBs may need backing up technologies to meet growing consumer need. Thus, it is crucial to seriously reckon the future political, economic, and social consequences of this global Li fixation. 6
Sodium (Na) is available in great abundance with a low cost: natural Na is more than 1000 times more abundant than Li, and it can be obtained from both deposits in Earth’s crust and salt water.
7
Na has very desirable redox potential (
In reality, the electrolyte that ionically links electrodes is an essential aspect that determines the battery’s performance. Liquid electrolytes are used for most of the research on battery in which usually metal salts (Li, Na, Mg, etc.) in different non-aqueous organic solvents are being used as the liquid electrolytes. 9 Conventional batteries (e.g. LIB) mostly comprise of liquid electrolyte that aids Li+ ion transport to and fro between anode and cathode. 10 This leads to the high probability of electrolyte leakage if at all holes are present, this is one of the foremost conventional batteries pulls back. Numerous research on Na-ion PEs has been reported, such as poly(ethylene oxide) (PEO) and polyvinyl alcohol-based polymer–salt complexes comprising NaPF6, NaClO4, NaTFSI, NaFSI, NaTf, Na2SO4, NaCF3SO3, and NaPO3 Na salts. 11 PEO is the most extensively studied polymer host for SPE applications. This is due to its high electrochemical stability in comparison with other polyethers, copolymers, or PEO-branched polymers. 8 SPEs for SIBs simply comprise an Na salt dissolved in a polymer matrix, the latter usually being PEO because of its effectiveness in dissolving alkali metal salts. Following the trend of sodium battery research, studies on sodium SPEs were performed since 1990s and have regained attention quite recently. 12 So far, different combinations of Na salts and PEO have been considered, mostly mimicking the much more researched Li-based SPEs.
Polymer nanocomposites have received interest for the past two decades because of their exciting bulk and surface properties. The hexagonal boron nitride (hBN) is basically an important material with a combination of unique properties. It has been used in the matrix of ceramic composites to decrease thermal expansion coefficient, improving thermal shock resistance of the composite and enhancing machinability. 13 A high performance and relatively low cost of hBN nanosheets, as compared to the very expensive graphene, give them great potential in serving as fillers for preparing high-conductivity composites. However, there is so far a lack of attempt to use hBN nanosheets in preparing composite thermal energy storage purpose. 14
In this work, polysulfone-sodium sulfonate (SPSU(Na)) was synthesized with a high degree of sulfonation using trimethylsilyl chlorosulfonate as sulfonating agent and blended with a different mole ratio of poly (polyethylene glycol methacrylate) (PPEGMA). The blended polymers were used as a matrix to prepare polymer composite electrolytes using nano-sized hBN (nano-hBN) as filler. Physical and chemical properties of the PEs were characterized using Fourier transform infrared (FTIR), X-ray diffraction (XRD), thermogravimetric analysis (TGA), differential scanning calorimetry (DSC), and scanning electron microscopy (SEM). Ionic conductivity of the polymer composite was investigated with an impedance analyzer at various temperatures.
Experimental
Materials
Polysulfone (PSU; 22,000 g mol−1) and trimethylsilyl chlorosulfonate (TMSCS) were purchased from Sigma-Aldrich (Japan). Dimethylformamide (DMF) and azobisisobutyronitrile (AIBN) were purchased from Merck (Canada). PEGMA (
Preparation of the samples
Preparation of PPEGMA
An amount of 4 g of PEGMA was dissolved in 10 ml of DMF and stirred for 10 min; 1% of AIBN initiator was introduced into the reaction flask to initiate the polymerization reaction. The mixture was stirred at 70°C under a nitrogen (N2) environment for 5 h. Then, the PPEGMA was washed with ethanol to remove the unreacted monomer and the remaining AIBN. Figure 1 shows the polymerization of PEGMA to PPEGMA.
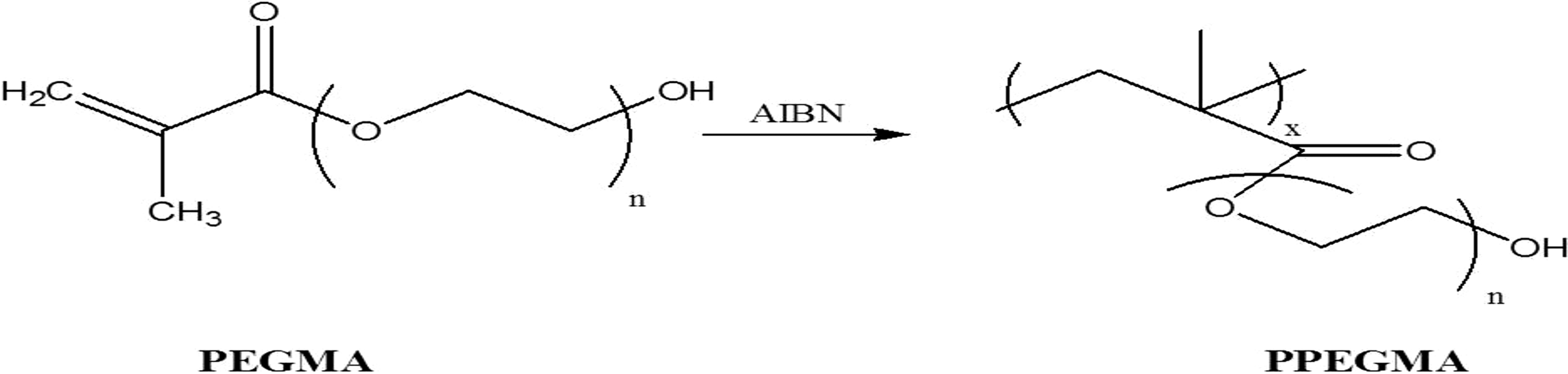
Polymerization of PEGMA.
Preparation of SPSU
Sulfonated polysulfone (SPSU) was prepared in accordance with the literature. 15 PSU was placed in 1,2-dichloroethane at 25°C and stirred for 5 h under an N2 environment. TMSCS, the sulfonation agent, was introduced into the reaction flask at 25°C. During the reaction, N2 was continuously purged through the reaction flask, and hydrochloric acid (HCl) was continuously produced and released out of the reaction flask together with N2. After 36 h, methanol was added to quench the reaction and also to cleave the silyl sulfonate moieties yielding SPSU. Dichloroethane, methanol, and water were removed by evaporation. Sulfonation of PSU is shown in Figure 2.
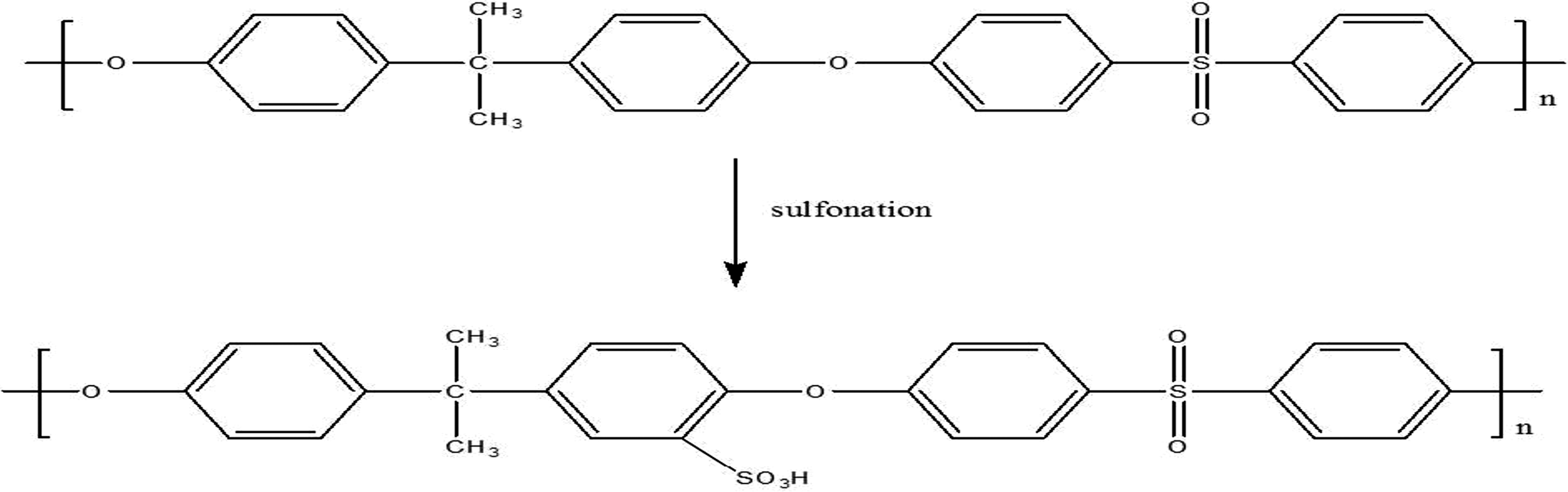
Sulfonation of PSU.
The degree of sulfonation was determined by titration: the sulfonated polymer was dissolved in DMF and titrated with 0.05 M sodium hydroxide (NaOH). The degree of sulfonation was found to be 150.14% (it is 100% if there is one sulfonate group per each repeating unit of PSU monomer).
Preparation of SPSU(Na)
SPSU(Na) was prepared by ion exchange: SPSU was soaked in 0.1 M NaOH for 24 h followed by filtration. The filtrate was titrated with 0.1 M HCl using phenolphthalein as indicator. Figure 3 shows the ion exchange between SPSU and NaOH, and ion exchange capacity (IEC) was calculated using the equation below

where
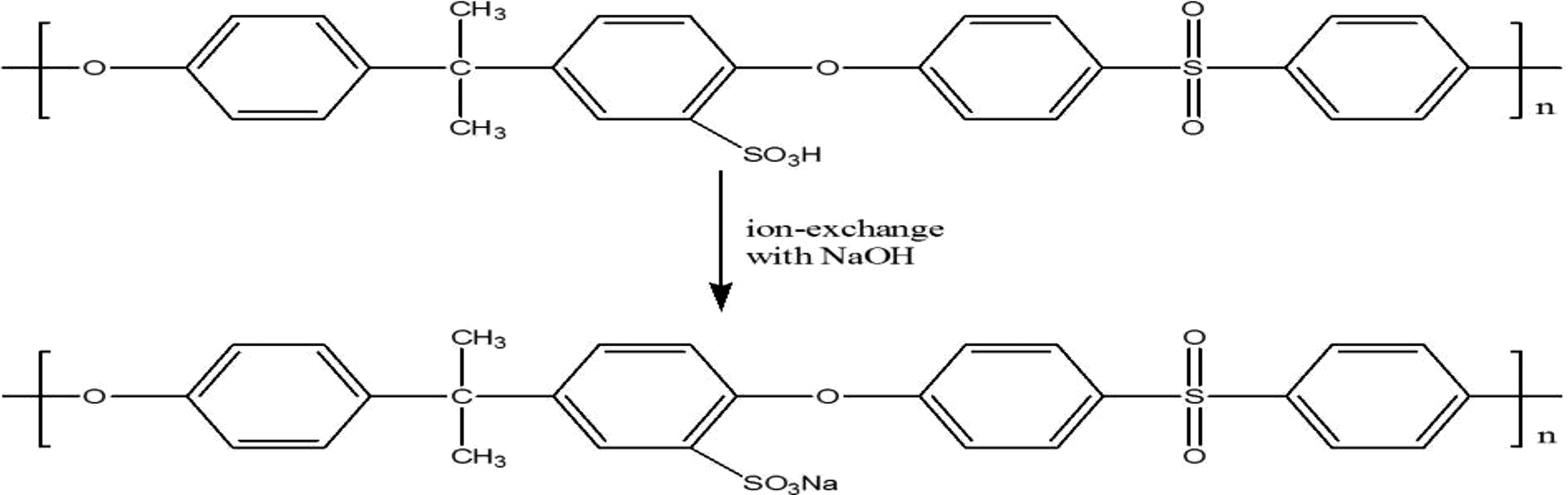
Preparation of SPSU(Na).
Preparation of the polymer composite electrolytes
The polymer composite electrolytes were obtained by blending SPSU(Na) with PPEGMA and 5% hBN (w/w) followed by solution casting at room temperature then placed in vacuum at 55°C for 24 h. Final composite electrolytes were named hBN-SPSU(Na)/PPEGMA(X:Y) and SPSU(Na)/PPEGMA(X:Y) for sample with hBN and without hBN, respectively, where (X:Y) denote the SPSU(Na) to PPEGMA ratio (i.e. 1:1, 1:2, and 1:4)
Characterization
A Bruker Alpha-P in attenuated total reflectance (ATR) was used to record FTIR spectra in the range of 4000–400 cm−1 in order to observe the functional group and their interaction in the system.
The PEs thermal stabilities were analyzed with Perkin Elmer STA 6000 Thermal Analyzer (USA). The samples were heated from 30°C to 750°C under an N2 atmosphere at a scanning rate of 10°C min−1.
In order to investigate thermal transitions of the PEs, Perkin Elmer JADE DSC (USA) was used. The samples (approximately 10 mg) were placed into aluminum pans. The sample and reference are heated in heat flux instruments and the temperature difference is measured. During the measurements, at first, the samples were heated from 0°C to 150°C and then cooled from 150°C to 0°C. Finally, the second heating was performed from 0°C to 250°C at a rate of 10°C min−1 under an N2 atmosphere.
Surface morphology of the samples was examined by SEM, JEOL-7001 FESEM (Tokyo, Japan). Prior to the SEM measurements, all of the samples were coated with gold for 150 s in a sputtering device.
XRD patterns of the PEs and pure hBN were obtained by XRD instrument, Rigaku Smart Lab Diffractometer operated at 40 kV and 35 mA using Cu-Kα radiation having a wavelength (
The ionic conductivity measurements were carried out using a Novocontrol dielectric-impedance analyzer. The membranes were sandwiched between platinum blocking electrodes, and the conductivities were measured in frequency range of 1 Hz and 3 MHz at 10°C intervals. The temperature was controlled with a Novocontrol cryosystem.
Result and discussion
Several polymer composite electrolytes based on SPSU(Na) with and without hBN were prepared by varying the PPEGMA ratio (1:1, 1:2, and 1:4). The PPEGMA was used to soften the SPSU to improve conductivity. For all composites, homogeneous and flexible films were obtained.
FTIR study
FTIR analysis was conducted in order to study SPSU(Na)/PEGMA blend and incorporation of hBN nanoparticle into the SPSU(Na)/PPEGMA blend. The FTIR results of the composite electrolytes both with and without hBN (i.e. hBN-SPSU(Na)/PPEGMA(X:Y) and SPSU(Na)/PPEGMA(X:Y)) were shown in Figure 4. The peaks observed at 1144, 1015, 1236, and 1476 cm−1 are assigned to asymmetric stretching of S–O bond of SO3Na groups, Ph–O–Ph, C–O, and C=C (aromatic), respectively. The peaks at 1728 and 1088 cm−1 represent C=O and C–O stretching vibrations, respectively, of the carboxylic ester from PEGMA and the wide band at 3441 cm−1 represents the O–H stretching vibration. The band at 2898 cm−1 is attributed to stretching vibrations of the aliphatic C–H groups and bands at 1452 and 1347 cm−1 are attributed to symmetrical and asymmetrical bending vibration C–H groups in CH and CH2, respectively. 16
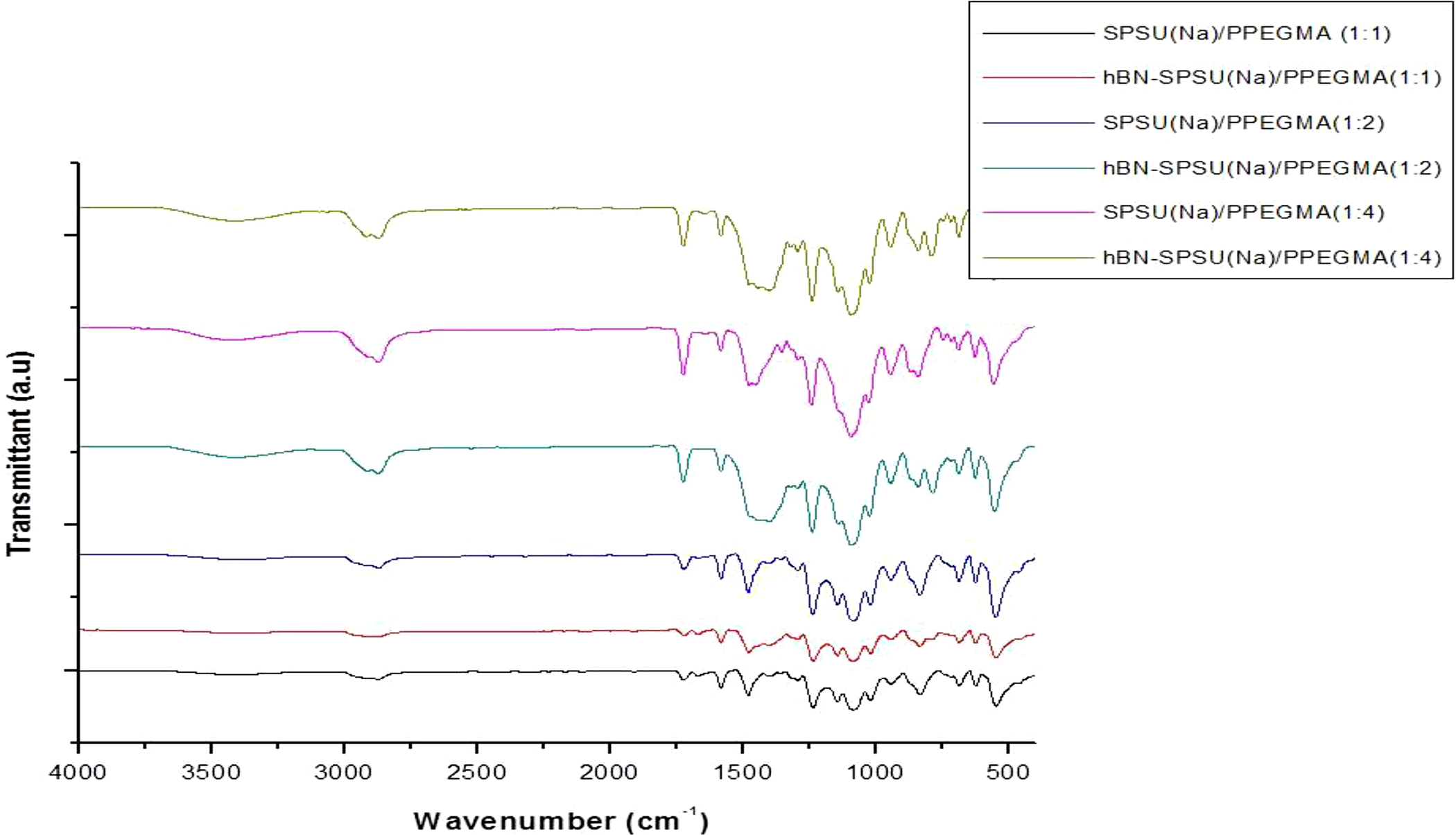
FTIR spectra of SPSU(Na)/PPEGMA(X:Y) and hBN-SPSU(Na)/PPEGMA(X:Y).
The SPSU(Na) to PPEGMA ratio can be estimated by comparing the C–O ether peak (1088 cm−1) intensity of the PEGMA. It can be seen from Figure 4 that the intensity of the peak at 1088 cm−1 increases as the PPEGMA content increased. And also, as the content of the PPEGMA increases, the O–H band becomes broader. Peak around 1336–1481 cm−1 broadens for samples with nano-hBN, which indicates the dispersion of the nano-hBN in the polymer blend because hBN has a characteristic broad band around 1336–1481 cm−1. 15
DSC analysis
DSC analysis was performed under an inert atmosphere at a scan rate of 10°C min−1 by a heating–cooling–heating cycle and the second heating curves were evaluated. The second heating curves of SPSU(Na)/PPEGMA(X: ) and hBN-SPSU(Na)/PPEGMA(X:Y) samples are given in Figure 5. From the literature, neat PSU is reported to have
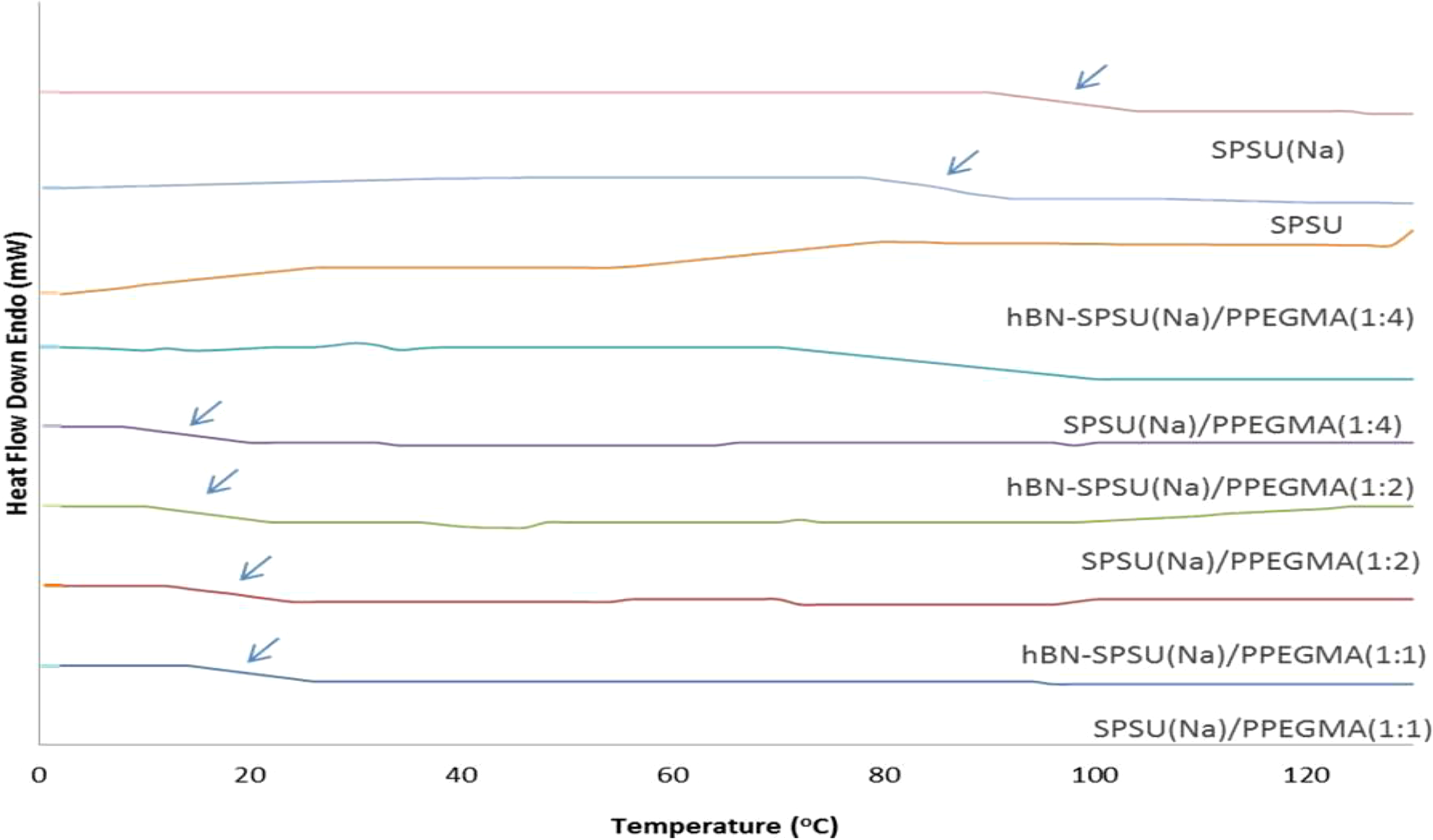
DSC result for hBN-SPSU(Na)/PPEGMA(X:Y) and SPSU(Na)/PPEGMA(X:Y).
Thermogravimetric analysis
hBN-TGA gives information about the possible physical and chemical changes that may happen during a thermal excitation in a PE when it is applied to working systems. 17 The electrolyte should possess good thermal stability at high temperature to meet the criteria for application. Thermal stability of neat pure PPEGMA, SPSU(Na)/PPEGMA(X:Y), and hBN-SPSU(Na)/PPEGMA(X:Y) was evaluated by TGA. Prior to the measurement, the samples were dried under vacuum at 55°C for 24 h.
Figure 6 presents the TGA of pure PEGMA, SPSU(Na)/PEGMA(xy), and hBN-SPSU(Na)/PEGMA composites. Pure PSU is reported to be thermally stable up to 500°C than major weight loss occurs due to degradation of the polymer backbone. 15 TGA curve of SPSU has two weight loss regions where 30% weight loss occurs around 200°C and 350°C due to degradation of sulfonic acid groups and the polymer backbone degradation occurs above 500°C. 15 SPSU(Na)/PPEGMA and hBN-SPSU(Na)/PPEGMA composites can be said to be thermally stable up to at least 190°C. The decomposition temperature increases a little with hBN content and it decreases a bit with increasing PPEGMA ratio.
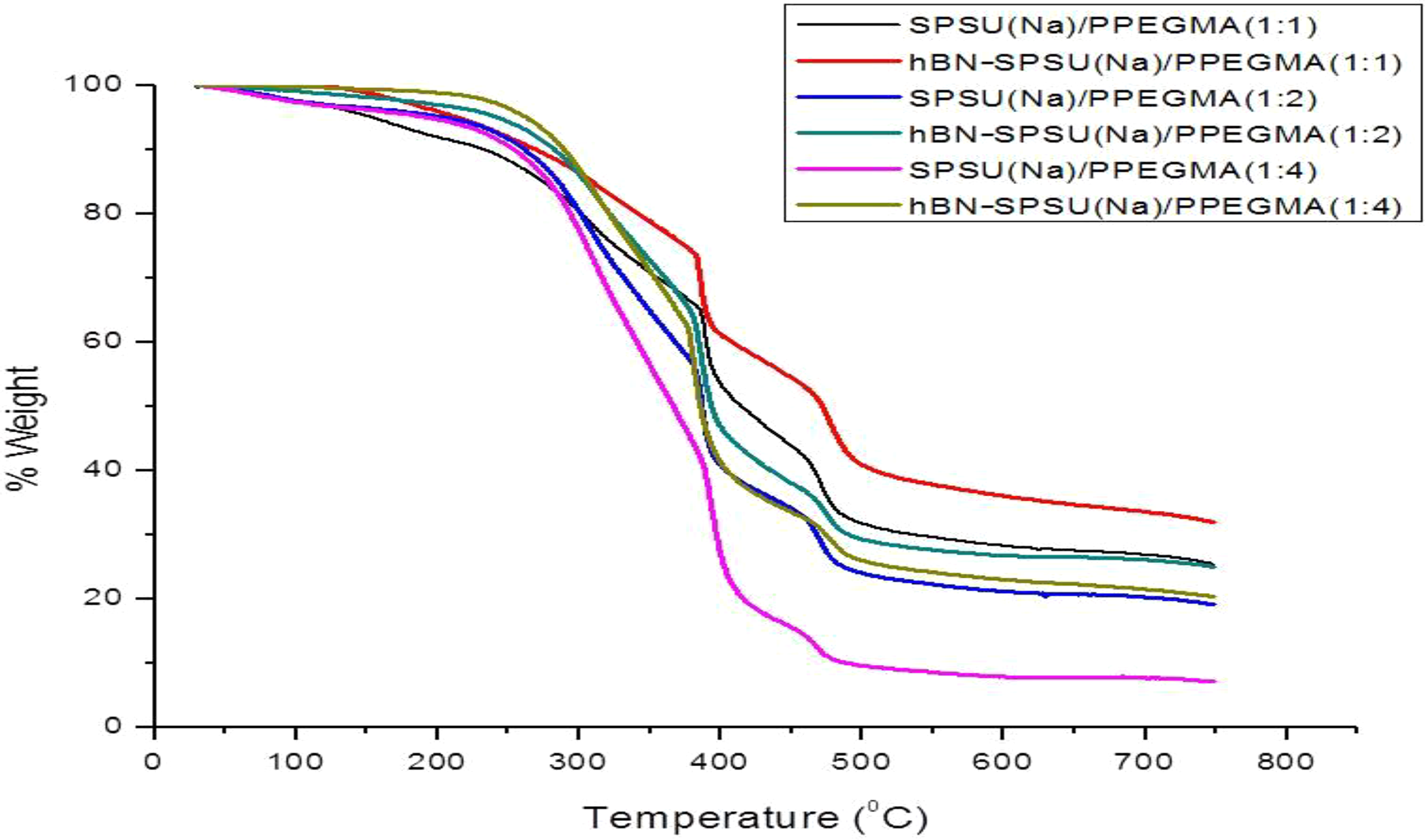
TGA curves of SPSU(Na)/PPEGMA(X:Y) and hBN-SPSU(Na)/PPEGMA(X:Y).
SEM analysis
Study of the morphology is important in order to confirm the homogeneity of polymer blends. SEM analysis provides detailed photographs that give important information about the surface structure which affirms the polymer blends homogeneity. 18 Several studies have reported the use of functionalized hBN; functionalization helps prevent agglomeration in various polymer matrices. 18,19 In this study, we used pure hBN directly and just sonication is enough in providing adequate blending. The morphology of hBN-SPSU(Na)/PPEGMA(X:Y) composites was investigated by SEM and the results are shown in Figures 7 to 9. As observed from the images, the composite films are homogeneous and display single-phase formation which indicates that hBN was uniformly distributed into the SPSU(Na)/PPEGMA matrix. It was also observed that the composites are becoming rougher with the increasing PPEGMA content.

SEM images of hBN-SPSU(Na)/PPEGMA (1:1).

SEM images of hBN-SPSU(Na)/PPEGMA(1:2).

SEM images of hBN-SPSU(Na)/PPEGMA(1:4).
XRD analysis
XRD analysis is a non-damaging technique which is used to analyze the existence of any phase change or crystallinity changes after blending two different materials with different crystallinity.
15
Figure 10 shows the XRD pattern of SPSU(Na)/PEGMA(X:Y), hBN-SPSU(Na)/PEGMA(X:Y), and pure hBN. SPSU is an amorphous polymer without any crystalline phase.
15
All the peaks observed are due to PPEGMA and hBN. Around 2
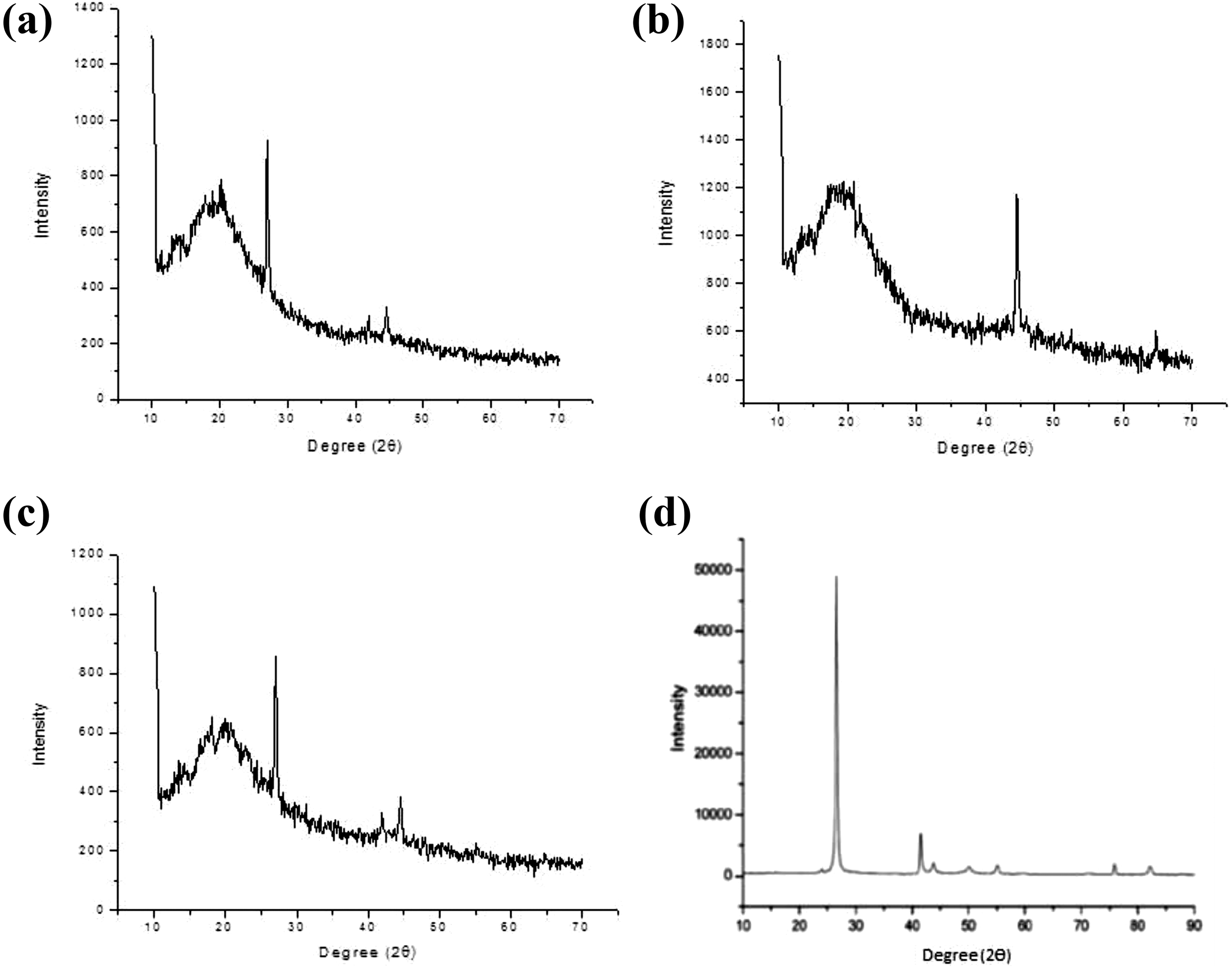
XRD pattern of (a) hBN-SPSU(Na)/PPEGMA(1:2), (b) SPSU(Na)/PPEGMA(1:2), (c) hBN-SPSU(Na)/PPEGMA(1:4), and (d) pure hBN.
Conductivity measurement
The ionic conductivity of hBN-SPSU(Na)/PPEGMA(X:Y) and SPSU(Na)/PPEGMA(X:Y) was examined with impedance spectroscopy. To eliminate the effect of humidity on the ionic conductivity, the measurements were carried out in a fully water-free system. The ionic conductivities were measured in the temperature range of 20–100°C. The frequency-dependent AC conductivities,

where
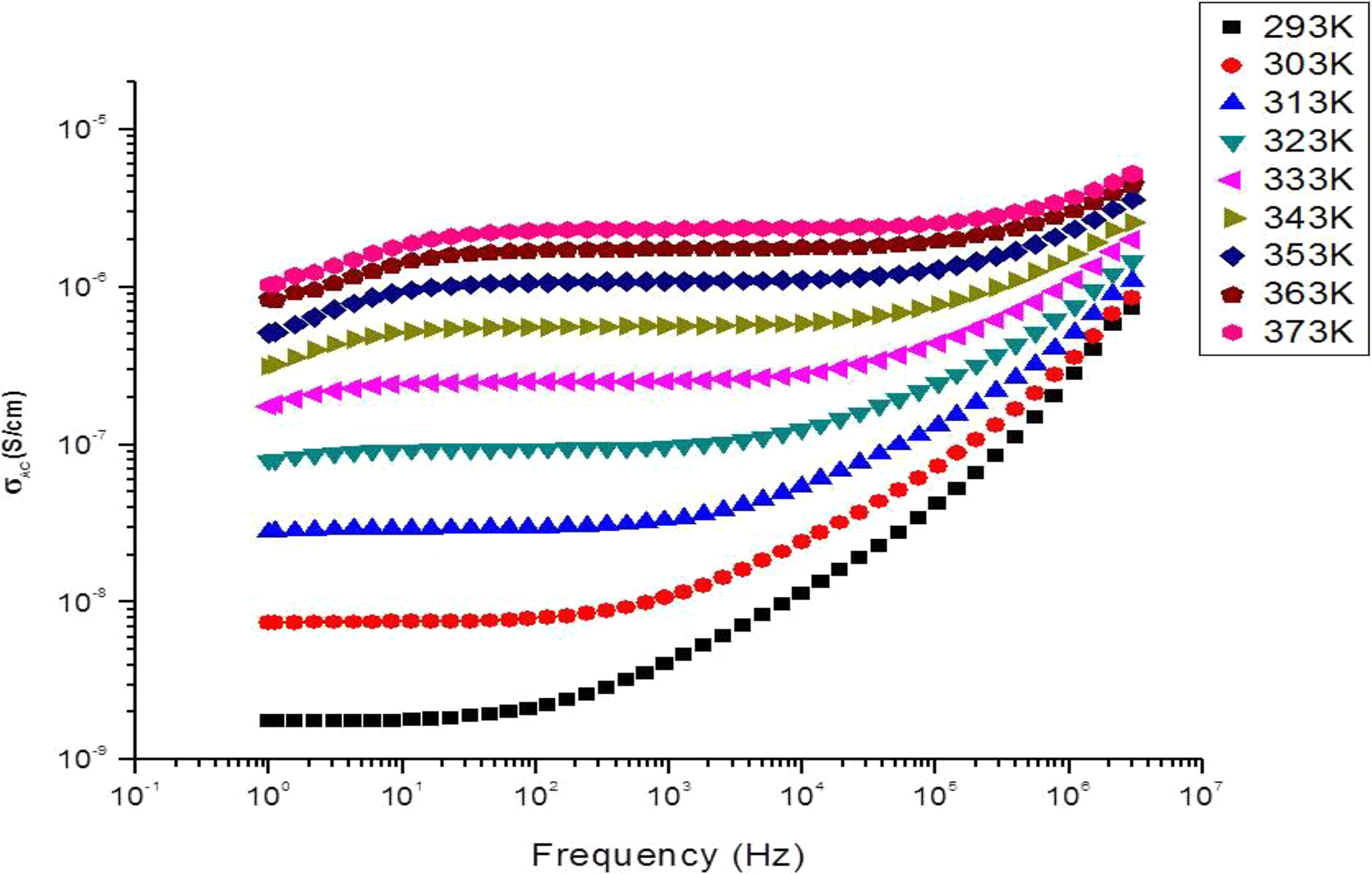
AC conductivity of SPSU(Na)/PPEGMA(1:4) versus log frequency (Hz) at various temperatures.
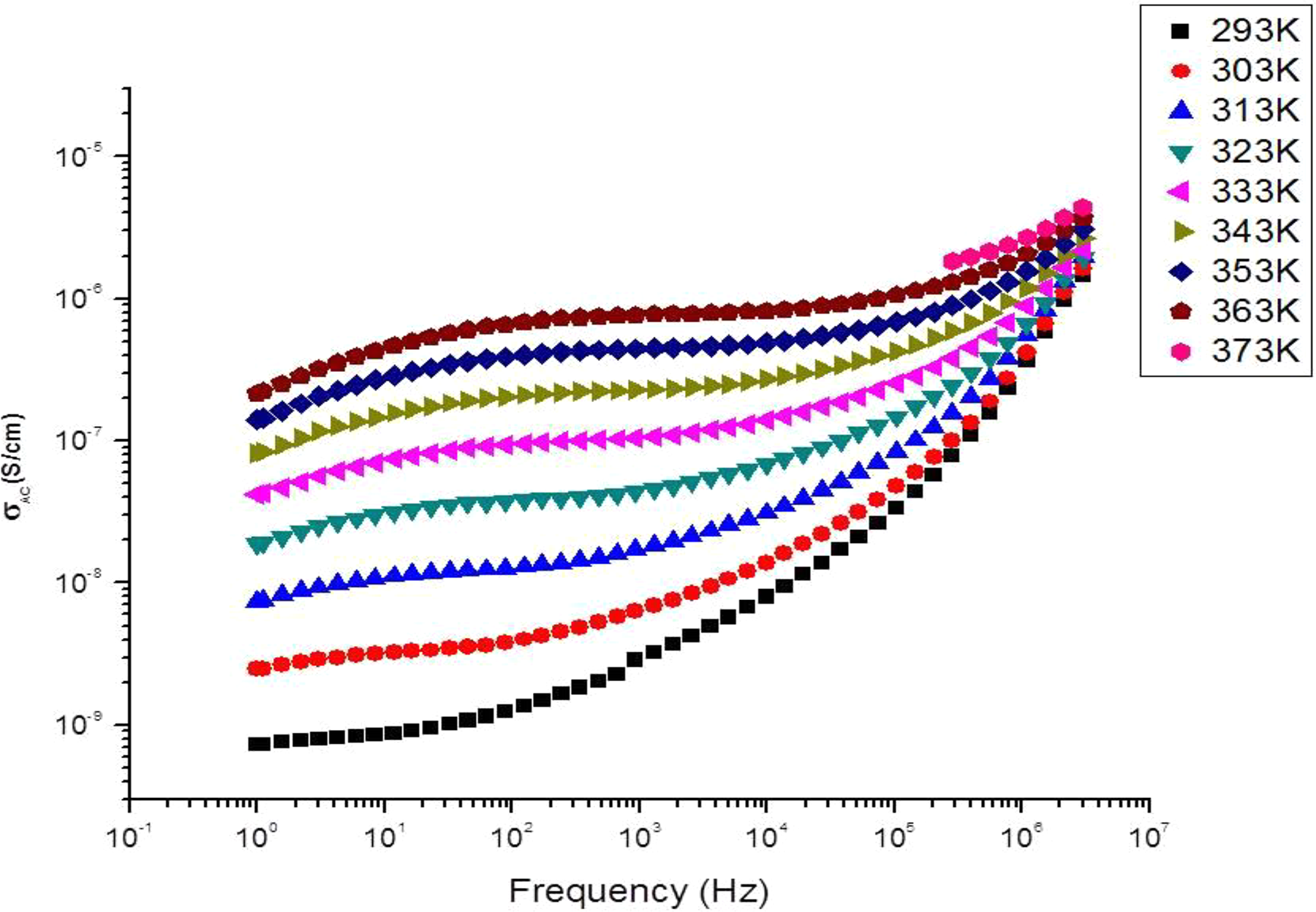
AC conductivity of hBN-SPSU(Na)/PPEGMA(1:4) versus log frequency (Hz) at various temperatures.
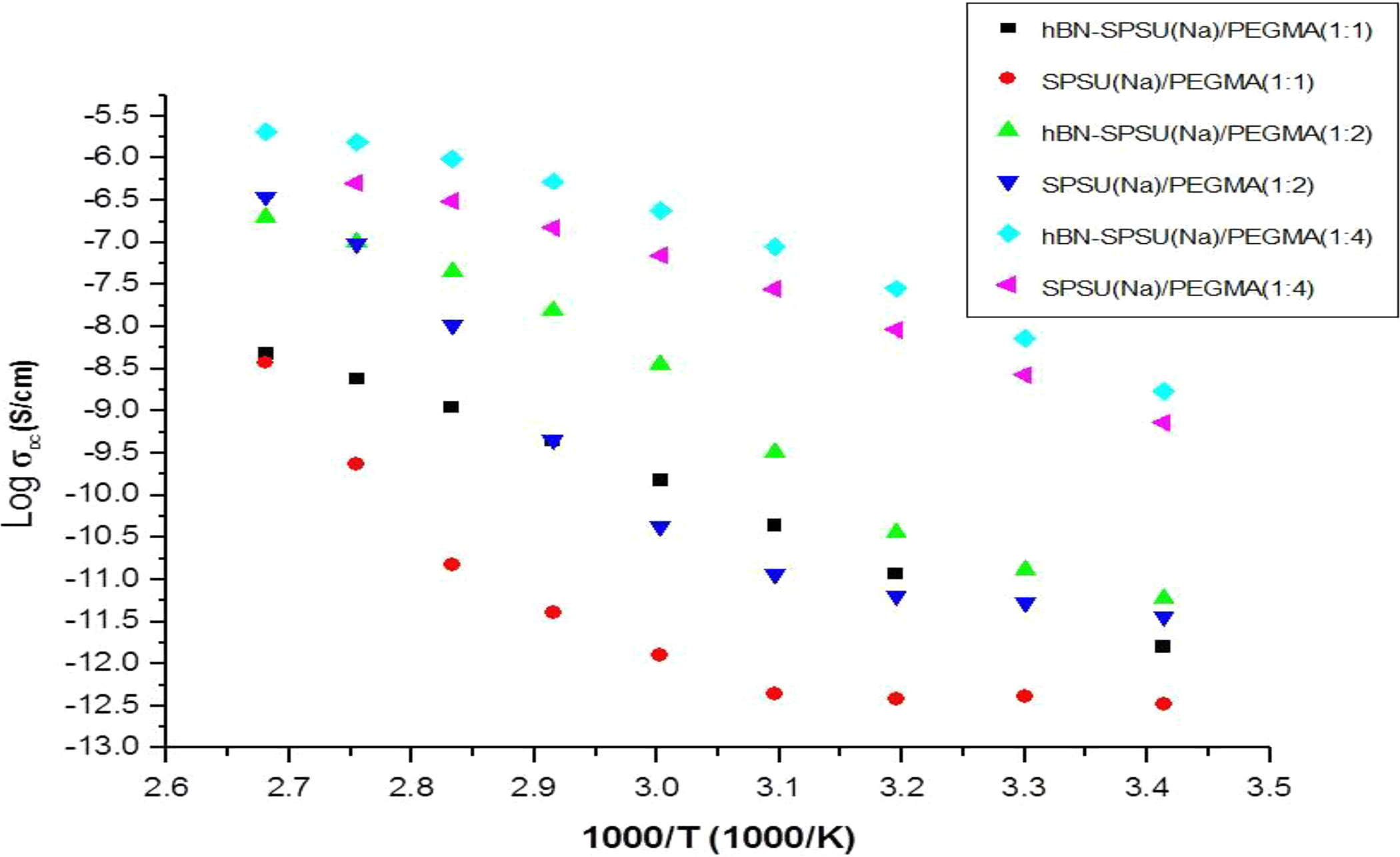
Conductivity of SPSU(Na)/PPEGMA(X:Y) and hBN-SPSU(Na)/PPEGMA(X:Y) composites as a function of reciprocal temperature.
The DC conductivity (
Conclusion
Novel hBN-SPSU(Na)/PPEGMA(X:Y) and SPSU(Na)/PPEGMA(X:Y) composite electrolytes were successfully prepared. Sulfonation of PSU and ion exchange with NaOH were verified with FTIR results. The composite electrolytes were thermally stable up to at least 190°C. Increasing the PPEGMA ratio decreased
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
