Abstract
The present study investigates the biodegradation of synthesized condensation nonisocyanate polyurethanes (NIPUs). It was prepared by reacting phenol sulphonic acid and oligooxypropylene diol and formaldehyde and contained different amounts of hard segments (HSs). Polyurethane samples were submitted to biodegradation with microorganisms R-14 and in garden soil. The tensile strength (TS), relative elongation at break and weight loss of the prepared samples were evaluated and their structure and morphology analysed. It was found that maximum decrease of the TS of all tested NIPU samples occurred only after 7 days of biodegradation. Maximum TS decrease attained was approximately 70% and 75%, respectively, for NIPU based on 0.8-mol and 1-mol HS. Moreover, significant decrease of relative elongation at break and weight loss values after biodegradation in culture R-14 for NIPU samples based on 0.9-mol HS and 1-mol HS was observed. Fourier transform infrared and scanning electron microscopic results confirmed that biodegradation occurred in urea or urethane groups. The glass transition of HSs decreased by at least 20°C due to biodegradation, suggesting that this later took place almost only in the crystalline region of NIPU samples.
Introduction
As for traditional plastics, the recycling of polyurethanes (PUs) can include the mechanical method, the chemical as well as the incineration or energy recovery methods. However, biodegradable processes seem to be an additional promising relatively cheaper method to be considered in plastics reconversion. They consist of the degradation of the material under the action of microorganisms such as bacteria, fungi, yeast or algae in conditions favourable to their development. 1–3 The microbial degradation of PUs against fungi was studied by Darby and Kaplan for the first time in 1968. 1 The obtained results showed that polyether-based PUs were moderately highly resistant to fungal attack, whereas all polyester PUs tested were highly affected depending on the number of adjacent methylene groups in the polymer chain. Numerous studies also confirmed that PUs undergo hydrolytic decomposition in the presence of isolated enzymes, 2 synthesized and investigated the biodegradation of segmented poly(ether-ester-urethanes). The effect of enzyme solutions on the urethanes degradation was investigated for 12 days. Scanning electron microscopic (SEM) and gravimetric results showed that significant erosion occurred in urethane systems in the presence of enzymes. In a separate study, Shi et al. 3 reviewed the degradability of chemically synthesized bioelastomers such as biodegradable PUs. They presented and discussed the in vitro and in vivo degradation mechanisms as well as the factors affecting the degradation behaviours of polymers. They also presented the mechanical properties, biocompatibility and potential applications of these bioelastomers.
Howard 4 demonstrated that PUs are susceptible to biodegradation by microorganisms. He showed that microbial degradation of polyester-based PU is mainly due to the hydrolysis of ester bonds by these esterase enzymes. Furthermore, microbial degradation of PUs is found to be dependent on the crystallinity, cross-linking and chemical groups of the polymer. The research conducted by Nakajima-Kamba et al. 5 showed that PUs are also subjected to bacterial degradation, especially in the presence of microorganisms belonging to the genera Bacillus and Pseudomonas (Pseudomonas chloraphis, P. fluorescens, P. cepacia, P. putida). It was found that the cells of Bacillus macromolecule complex with the PU. However, it was found that the exact mechanism of enzymatic degradation of PUs remains poorly defined. In this context, Duguay et al. 6 proposed a mathematical model to elucidate and simulate the mechanisms of PUs biodegradation. Their model was also used to describe the PU surface dynamics, its enzymatic degradation as well as enzyme absorption and inactivation.
As reported in the literature, polyester and polyether-based PUs are susceptible to the attack of fungi and bacteria, but the biodegradation mechanisms are still not fully elucidated. However, the published works were focused only on the degrading capability of bacterial enzymes PU. This review approaches PU biodegradation by focusing on the enzymes reported to date. 7
Nowadays, the main concern of scientists is also the production of polymers derived from non-petrochemical feedstocks and hence environmentally friendly polymers. Indeed, a lot of efforts have been made to use renewable resources such as vegetable oils for the synthesis of polyols that can be used for ‘greener’ PUs prepration. 8 -11 Diisocyanates, which are used as basic raw materials for the production of the conventional addition PUs, are expensive, toxic, unstable and harmful to the environment and human health. The elimination of these materials and their replacement by natural and chemically stable compounds lead to the preparation of biodegradable, safe and environmentally friendly polymers. The new nonisocyanate polyurethanes (NIPUs) are obtained via a safer and greener route or from raw materials of natural origin without the use of dangerous and toxic raw materials.
Jalilian and Yeganeh 8 prepared a series of renewable resource-based NIPU using carbonated soybean oil (CSBO), 3-aminopropyl-terminated poly (ethylene glycol) (ATPEG) and ethylene diamine (ED) at different weight ratios. Obtaind resuelts showed that hydrolytic degradation of NIPU increased with increasing ATPEG content as well as good cytocompatibility of the prepared PUs. Two chemically recyclable PUs namely diol–diacid-based poly(diester-urethane) (PDEU) and hydroxy acid-based poly(diester-urethane) (PEU) were synthesized without hazardous diisocyanates. 10 Urethanediol that was obtained by reacting ethylene carbonate with amino alcohols and amino acids underwent polymerization with dimethyl adipate in the presence of lipase to yield PDEU. However, PEU was prepared by polycondensation of urethane-based on hydroxyl acid monomer and lipase as catalyst. It was shown that both PDEU and PEU were degradable in the presence of lipase.
To the best of our knowledge, no work was carried out so far on the biodegradation of this kind of NIPUs. Hence, the aim of the present study was to investigate the biodegradation of nonisocyanate condensation PUs prepared from phenol sulphonic acid, formaldehyde, urea and oligooxypropylene. The biodegradation of condensation NIPU was carried out using culture R-14 isolated from garden soil.
Experimental
Materials
The following ingredients were used for the synthesis of PU: Phenol sulphonic acid as 65% aqueous solution from Sigma Chemie GmbH Aldreich Riedest, Steinheim Germany; Oligooxypropylene diol (trade name Rokopol G1000) from Rokita (Brzeg Dolny, Poland), with an average molecular weight of 1000 g/mol and hydroxyl number of 118 mg/g potassium hydroxide; Ethyl urethane from POCh Company (Gliwice, Poland); Formaldehyde with concentration 34–37% (‘Zakłady Azotowe’, Tarnów, Poland); Tetrabutoxy titane (Schuchardt, München, Germany); and Toluene p.a. OBR Petroleum Industry Plock, BN-85/6193-89.
The reagents used in the study of PUs biodegradation of used with high purity level p.a., Polish Chemicals Reagents (POCH) in Gliwice Sp. z o. o and Enzymes and Peptones Plant in Lodz (BTL) Sp. z o. o.
Preparation of microorganisms
Moulds isolated from garden soil designated R-14 were used for the evaluation of PUs biodegradation. First, microorganisms were prepared as 0.8% suspension in NaCl. The liquid culture of mould composition of R-14 on substrate GP-1 was as follows: 0.50 g/ml of NaNO3, 0.75 g/l KH2PO4, 0.22 g/l MgSO4·7 H2O, 0.22 g/l CaCl2· 6 H2O and 0.20 g/l glucose. The pH of the substrate was 5.5 at 25°C. The substrate was sterilized for 15 min at a temperature of 121°C. Finally, the substrate components were dissolved in distilled water. Isolation and selection of microorganisms showing the ability to degrade condensation PUs were mainly carried out by surface spreading on agar plates with specified composition.
The plates were incubated for 24 h in medium with agar at 37°C and then inoculated with 5 ml of spore suspension of the fungus R-14. Then, the substrate was added to previously weighed sample of NIPU samples, which was immersed in alcohol solution and rinsed with distilled water.
Biodegradation process of PUs in culture media R-14 and garden soil
Conical flasks containing culture medium with grafted microorganisms were stirred in a shaker Elpin + type 357 at a speed of 170 r/min. Biodegradation processes were carried out on condensation NIPUs containing varying amounts of hard segments (HSs) and flexible segments (FSs). The molar ratio of the HS to the flexible segment (FS) for the samples designated P-0.8 was 0.8:1 and sample P-0.9 (0.9:1) while P-1 (1:1). The degradation of PU samples was also carried out in garden soil in plastic containers. In both cases, the biodegradation process was carried for 7 days. Then, the samples were washed with distilled water and dried to constant weight in a drying oven. The efficiency of the process was determined by the weight loss of samples, their appearance as well as structural changes and mechanical properties.
Preparation of PU films
Condensation segmented NIPU was prepared by a three-step reactions according to a previously developed procedure. 11 -13 In the first step, oligooxypropylene carbamate containing flexible segments was prepared by amidation of oligomerol hydroxyl groups with ethyl urethane (the molar ratio of hydroxyl groups to ethyl urethane was 1:1.14). In the second step, oligomer containing HSs was obtained by reacting urea, phenol sulphonic acid and formaldehyde (molar ratio = 1:1:2). Finally, PU is obtained by mixing at room temperature previously obtained: oligooxypropylene carbamate containing oligomer with HS and formaldehyde (as 37% formalin) to initiate the polycondensation reaction.
Finally, the mixture of homogenized components was casted onto glass plates and cured during 2 h at 50°C followed by 10 min at 135°C. The structure of such obtained NIPU was already confirmed and presented in our previous work. 11
Evaluation of PU samples biodegradability
The biodegradation of PU samples was estimated by the following parameters: The weight loss of samples submitted to biodegradation was determined. Tensile strength (TS) and elongation at break of polymer films were determined at room temperature according to DIN EN ISO 257 using an Instron tensile machine 5566 deformation speed of 100 mm/min. PU films had 10 cm in length and 1 cm in width. The structure of PUs samples determined by differential scanning calorimetry (DSC) method. The determination was performed using a differential scanning calorimeter Q2000 TA Instruments according to PN-EN ISO 11357 in an atmosphere of nitrogen (25 mL/min). Samples were tested PUs in the form of discs with a diameter of 5 mm. Measurements were taken during the heating/cooling of the sample at a constant rate of 10 K/min at the temperature range from −100°C to 200°C (173–453 K). The morphology was analysed with a scanning electron microscope (SEM) FEI Quanta 250 FEG under a low-pressure mode (pressure in the chamber of 130 Pa). The microanalysis quality was performed using analytical system EDAX Genesis APEX 2i with Apollo 10 SDD spectrometer at 10 kV (produced by Thermo Fisher Scientific Poland). The analysis by infrared spectroscopy was carried out using technique attenuated total reflection at the frequency range of 4000–700 cm−1 with a resolution of 4 cm−1. The determination was performed by IR spectrophotometer system 2000 Perkin Elmer (Germany).
Results and discussion
Effect of biodegradation on NIPU physical properties
The isolation and selection of microorganisms having the capacity to degrade condensation NIPU was carried out by surface dispersion method on plates with agar substrate. Figure 1 shows the effect of biodegradation using R-14 strain and soil garden on the weight loss of NIPU containing different amounts of HS. It can be observed that significant weight loss took place after 30 days of biodegradation in culture microorganisms R-14 on the substrate GP-1 for NIPU samples was based on 0.9 mole HS and that with 1 mole HS. Moreover, the least weight loss was observed for PU sample P-0.8 and two times higher for samples of PUs: P-0.9 and P-1.0. Similar results were obtained for biodegradation in garden soil with maximum weight loss showed by NIPU containing 0.9 mole HS and 1 mole HS.
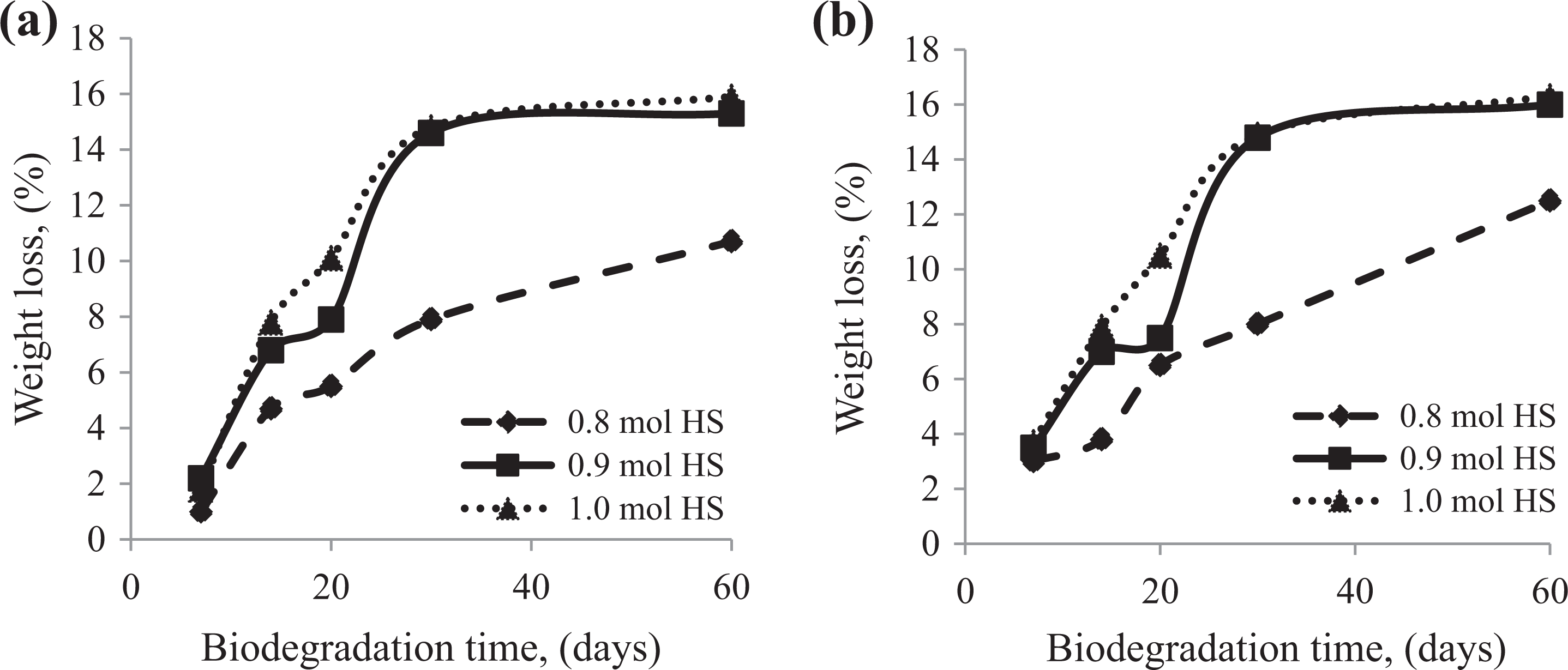
Effect of biodegradation time on the weight loss of (NIPU with R-14 strain in the GP-1 substrate (a) and garden soil (b).
Figure 2 illustrates the effect of HSs content and time of biological biodegradation (biodegradation in culture microorganisms R-14 on the substrate GP-1 as well as in garden soil) on the TS of NIPU. It can be noted that TS increased with increasing amount of HSs. Moreover, TS of all tested NIPU samples maximally decreased only after 7 days of biodegradation and then levelled off. Maximum TS decrease attaining approximately 70% and 75%, respectively, was exhibited by NIPU samples based on 0.8 mole and 1 mole HS. It was already reported in a literature that HSs contribute to improvement of TS of segmented nonisocyanate PUs 13 or diisocyanate-based PUs. 14,15

Effect of biodegradation time on the TS of NIPU by means of R-14 strain (a) and soil (b).
As in case of TS, the tensile relative elongation at break of nonisocyanate PU, as shown in figure 3, was affected by biodegradation as well as HSs content. In contrary to TS results, maximum decrease of elongation at break was observed after 20 days of ageing for all NIPU samples. A significant decrease of elongation at break of about 55% was noted for NIPU sample based on 0.8 mole HS, after only 7 days of biodegradation. However, the elongation at break of other samples decreased by approximately 70% in relation to non-aged samples.
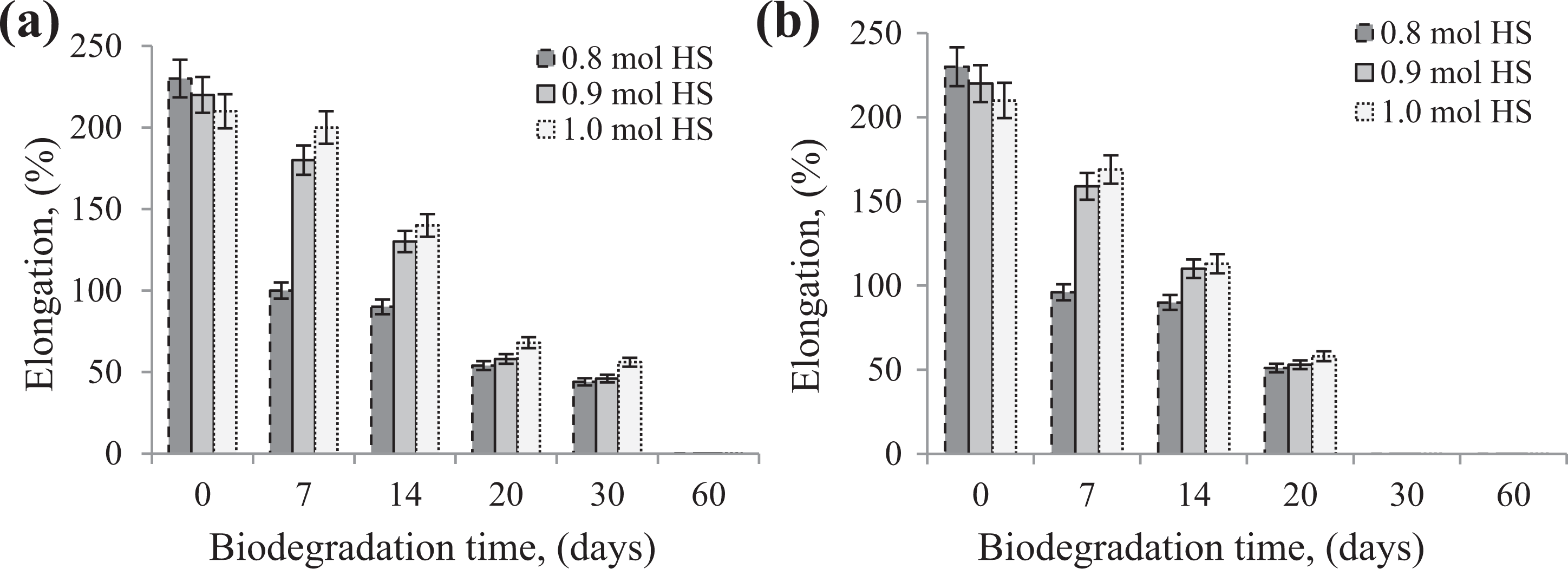
Effect of biodegradation time on the TS of nonisocyanate polyurethane by means of R-14 strain (a) and soil (b).
Very similar mechanical properties (TS and relative elongation at break) results were obtained with NIPU samples containing different amounts of HSs (0.8 mole, 0.9 mole and 1.0 mole HS) and subjected to garden soil biodegradation.
Based on the drastic decrease of the TS after only 7 days of biological degradation, further analysis was devoted to samples subjected to 7 days ageing time.
The obtained results suggest that the atoms C, N, S and O present in the specific PUs systems can constitute adequate medium for microorganisms. The above assumptions were supported by elemental analysis of NIPU compositions that were subjected to biodegradation. Figure 4 shows the results of elemental analysis for NIPU samples obtained from equimolar amounts of rigid (or HS) and flexible segments (sample designated P-1). Similar results were obtained for other samples (P-0.8 and P-0.9).
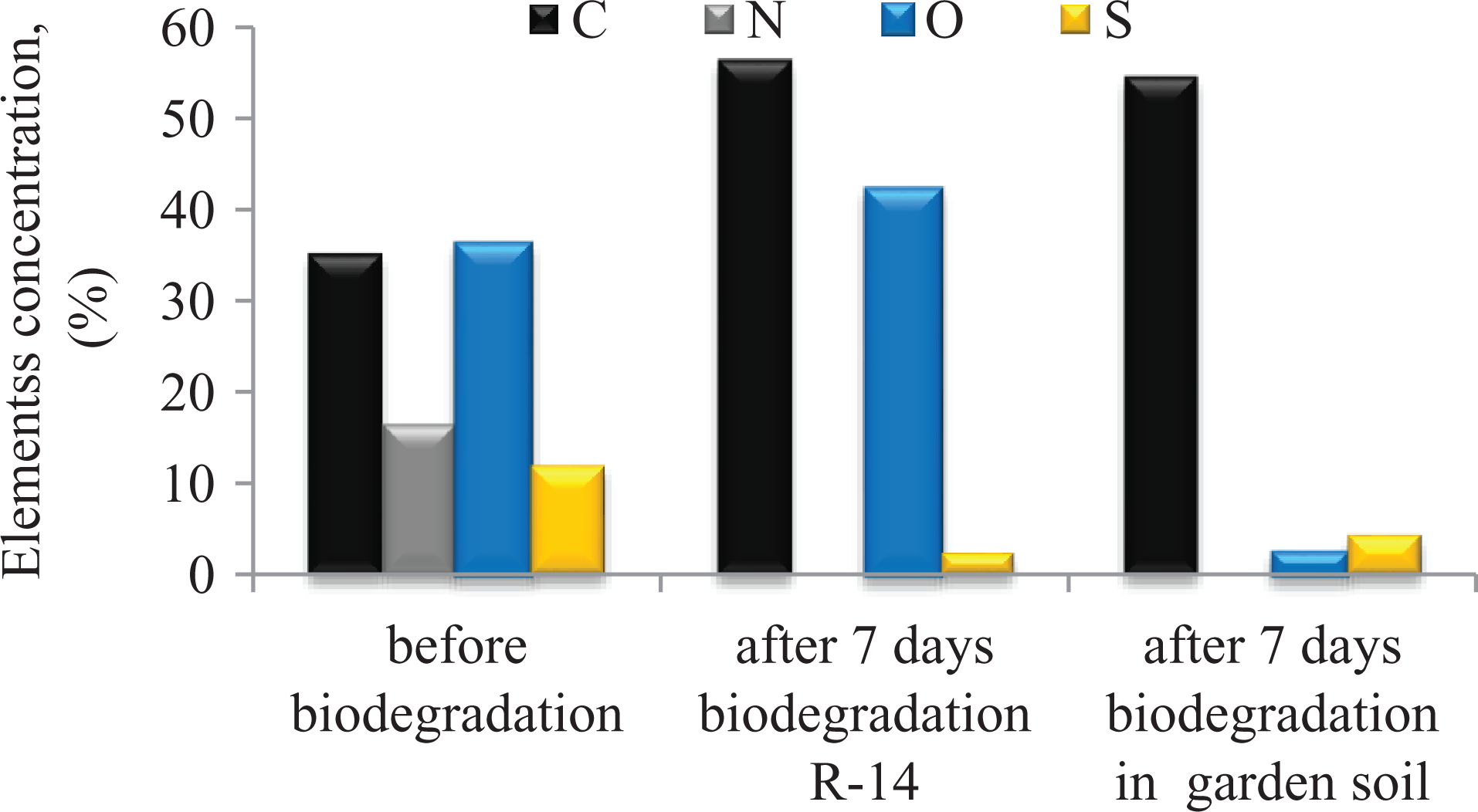
Variations of PU composition after biodegradation.
PU compositions underwent a distinct change due to 7 days of microorganisms effect. We could not identify N and observed a reduced percentage of the other elements, that is, C, O, and S in the case of the samples subjected to a 7-day biodegradation. Hence, it appears that the main microorganism attack the systems containing N and, to a lesser extent, C-, O- and S-based substances.
Effect of biodegradation on NIPU structure and morphology
The effect of biological degradation on the structure and properties of condensation NIPU was also investigated. Analysis of DSC and infrared spectroscopy (FTIR) was carried on NIPU films obtained from equimolar amount of hard to flexible segments (sample designated PU-0 before biodegradation, sample PU-1 after 7 days of degradation in culture R-14 and sample PU-2 after 7 days after degradation in soil).
Figure 5 shows three DSC thermograms of analysed NIPU films that were recorded during the samples cooling. Each thermal image shows the extremes of peak energy, and the areas under the peaks correspond to the energies of transformations endo- and exothermic. Temperatures of these changes are determined by the tangent to the front line of the peak at the inflection point.

DSC thermograms of PU samples with equimolar content of hard and flexible segments (PU-0: without degradation, PU-1: after 7 days of degradation in R-14 and PU-2: after 7 days of degradation in soil).
A two-phase system was found in the polymer for all tested samples. This was confirmed by the occurrence of glass transition temperature flexible segments (T gFS) and melting temperature of HSs (T mHS). The T gFS of condensation PUs before and after degradation was in the range from –30.4°C to –29.8°C. The slight differences in the values of T gFS may imply little or even lack of changes (differences within the test accuracy) occurring in the PU flexible (soft) segments.
Therefore, it is assumed that no change occurs in the structure of oligomerol hydrocarbon chain during the biodegradation process. However, there was a distinct variation in the structure of HS-based samples. The difference in the glass transition temperature of HSs were already clearly visible.
It was confirmed that the decrease of temperature T mHS of NIPU samples at 109°C immediately after sample preparation to 86.2°C and 90.1°C, respectively, in samples biodegraded with R-14 (sample designated PU-1) and sample biodegraded in soil (sample designated PU-2), suggests that the degradation processes occur almost only in the HS region. A slight reduction in the melting points for HS areas may indicate that biodegradation decreases the size of the domain phase, for example, due to degradation of biodegradable urea groups. A greater degree of degradation was observed in sample placed in a garden soil (T mHS = 86.2°C) rather than samples treated in culture R-14 (T mHS = 90.10°C). A similar conclusion can be done based on the values of phase transition energies.
Figure 6 shows three FTIR spectra of NIPU films subjected to biodegradation. The FTIR spectra analysis indicated that bands were observed for specific groups at almost the same wavenumber for all tested samples. We can observe in all samples before and after biodegradation distinct and with similar intensities bands corresponding to urethane groups (approx. 2350 cm−1), C−H groups (2900 cm−1) within methylene bridges and −CH3, C−O in C−O−C from oligomerol as well as C=C of aromatic systems (approximately 1400 cm−1). This may indicate that no significant changes occur in the chain embedded in the PU oligomerol. However, spectra of analysed samples indicate distinct differences in the bands intensity corresponding to the urea groups, that is, NH (around 1600 cm−1 and 900 cm−1) and C=O (at 1700 cm−1). The differences are also observed in overlapping bands arising from the stretch vibration of urea N−H and OH groups combined with aromatic systems (∼3490 cm−1). Slight differences were also observed in the band intensities corresponding to the vibration of −S=O group within −RSO3.
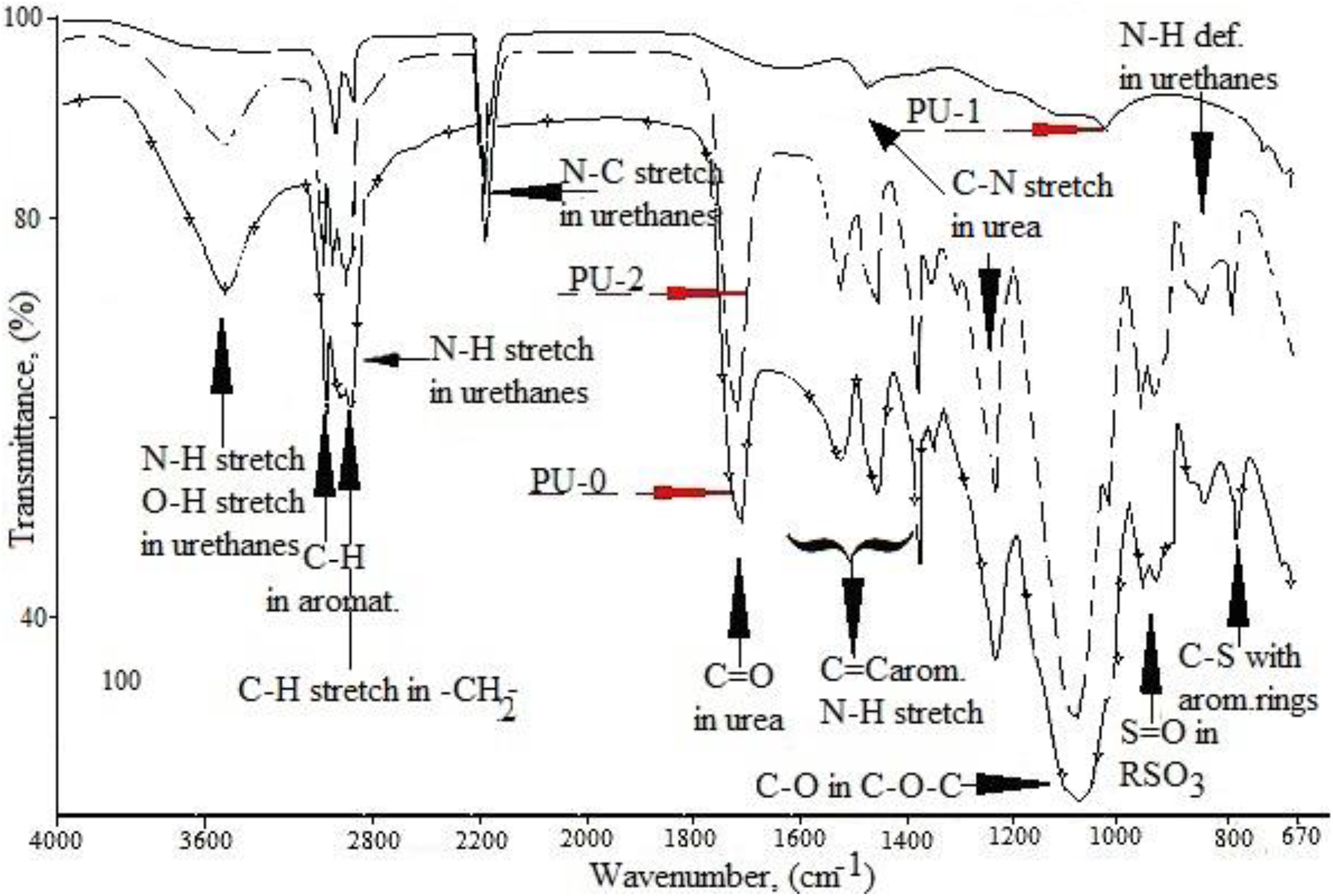
FTIR spectra of PU with equimolar of HSs and FSs before and after biodegradation (PU-0: without degradation, PU-1: after 7 days of degradation in R-14 and PU-2: after 7 days of degradation in soil).
One should also pay attention to the varying intensity of these bands in the samples submitted to different biodegradation processes. The band intensity is the largest for PU samples before biodegradation (PU-0), slightly lower for samples degraded in culture (PU-2) and the smallest in the samples degraded strains (PU-1). Hence, it is believed that mainly urea and to a lesser extent sulphonic systems constitute the proper medium for microorganisms for this kind of condensation NIPU. The greater decrease of the mentioned bands height were shown by the samples treated under the influence of biodegradation under strain than under the mould and strains.
However, it was reported that the biodegradation of poly(ester urethanes) and poly(ester-ether-urethanes) take place by hydrolysis of the ester linkages in flexible segments. 14,16 In addition, Umare and Chandure indicated that biodegradation occurs within urethane groups. 16 Unlike the addition of PUs, the biodegradation of condensation NIPUs occurs within the HSs. The FTIR analysis suggested that biodegradation occurred in urea or urethane groups as confirmed by the pronounced reduction in the bands intensity or disappearance of N–H groups in urea and N–H of urethane. Moreover, spectra of both samples subjected to biodegradation did not show a peak derived from C–N vibrations from urea at the wavenumber of 1230 cm−1. In addition, the presence of nitrogen atoms has not been detected in biodegradable samples. In fact, the spectrograms of both biodegradable samples showed no peak resulting from C–N vibrations from urea at the wavenumber of 1230 cm−1 and a clear reduction in the C–N peak at 1530 cm−1 for the PU-1 sample after 7 days of degradation in R-14. Moreover, the presence of nitrogen atoms was not detected in samples subjected to biodegradation.
Figure 7 shows SEM micrographs of NIPU samples surfaces before and after biodegradation. It is seen that, before the biodegradation process, sample containing 0.8 mol of HS (P-0.8) were characterized by a two-phase structure based on crystalline and amorphous phase. The crystalline regions were formed from HSs and are clearly shown in Figure 7(a) and (b). After 7 days of the bioprocess, we noted that the PU samples undergone biodegradation characterized by a loss of crystalline regions (Figure 7(c) and (d)). After biodegradation, PU samples both in liquid cultures (Figure 7(c)) and in soil (Figure 7(d)) were characterized by the presence of numerous cavities and of micro-cracks that might indicate the action of microorganisms in this phase. However, no structural changes were observed in the amorphous phase.

SEM micrographs of NIPU samples surfaces: (a and b) sample PU-0 before biodegradation; (c) sample PU-1 after 7 days biodegradation (mould culture in liquid R-14); (d) sample PU-2, after 7 days biodegradation in soil.
The relevant SEM observations are in agreement with FTIR results. In fact, the crystalline regions as associated with HSs domains, which are visible in nondegraded samples, disappear in PUs subjected to biodegradation. Moreover, it can be observed that several micro-cracks are formed in place of crystalline regions. These findings were also reported by other researchers in case of addition PUs biodegradation. 17,18
Conclusions
The following conclusions can be drawn from the obtained results:
Only 1 week is sufficient to biodegrade nonisocyanate condensation PUs prepared by reacting phenol sulphonic acid, oligooxypropylene diol and formaldehyde. The factor determining the degree of biodegradation are the duration and the conditions of the biodegradation process as well as the content of hard and flexible segments in the PU structure. The biodegradation of condensation NIPU depends on the structure of the polymer and takes place much better in the soil than in the culture media of bacteria and fungi.
The mechanical properties of the tested nonisocyanate condensation PU samples considerably decreased due to microbial degradation. In all tested samples, the TS decreased by about 70% for NIPU containing 0.8-mol HS as a result of 7 days of biological biodegradation process, and the more affected sample contained 0.9-mol HS (approximately 90% decrease).
The FTIR and SEM results confirmed that the biodegradation occurred in urea or urethane groups. The decrease in glass transition of HSs by at least 20°C due to biodegradation suggests that the biodegradation processes occur almost only in the HSs region.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
