Abstract
Polymer blending is one of the most convenient methods to be used to overcome the limitations of some single properties of polymers and to achieve the combinations required for specific applications. Another feasible common practice is the incorporation of additives of low molecular weight such as plasticisers to impart flexibility, improve toughness and lower the glass transition temperature (Tg). This study focused on the effects of blending and plasticising on the crystallisation behaviour of poly(lactic acid) (PLA)/poly(ε-caprolactone) (PCL). PCL with longer degradation time compared with other polymers was blended with PLA to overcome the limitation of its brittleness and poor thermal stability. Acetyl tributyl citrate (ATBC) and acetyl triethyl citrate (TEC) were used as plasticiser in PLA/PCL blends. The rigid and plasticised blends at various ratios were analysed by differential scanning calorimetry, thermogravimetric analysis and X-ray diffraction. The results revealed a slight increase in the degree of crystallinity and a significant increase in the Tg of PLA due to the addition of PCL. The addition of ATBC has promoted a decrease in thermal stability of the blends. The slight increase in the degree of crystallinity suggested that PCL acted as a nucleating agent. The citrate plasticisers were shown to lower the Tg and have much more enhanced the crystallisation of PLA. Moreover, the rigid and plasticised blends were shown to be partially miscible.
Introduction
In recent years, there has been an increased interest in the use of polymers made of renewable raw materials. The advantage over traditional materials is due to the huge environmental problems mainly created by petrochemical-based polymers. 1 -3 Biodegradable materials are expected to overcome the sustainability issues and environmental challenges in the near future. This is due to their low cost, extraordinary versatility and compostability, which allows for easier waste management. 3,4 These properties have targeted applications in various fields specifically such as packaging, agricultural, textile, pharmaceutical, electronic or medical domains. 5
Poly(lactic acid) (PLA) is a highly transparent and rigid material with a relatively low crystallisation rate and is produced from renewable resources such as corn, sugar beets and so on. Products with high molar mass are usually produced from lactides and the lactic acid monomers via ring-opening polymerisation route. It is one of the most promising alternatives to conventional plastics due to its biodegradability, sustainability, high melting temperature (Tm) and mechanical strength compared to those of many petroleum-based plastics. 6,7 However, under practical processing conditions, it is well known to show slow nucleation and crystallisation rates and would be nearly 100% amorphous, which limits wider applications in sectors, such as automotive and packaging fields. Moreover, several other drawbacks limit its widespread use such as brittleness, hydrolysis under high humidity conditions, hydrophobicity, poor moisture and gas barrier, low heat resistance and high cost. 8,9 -13
Among the several approaches that are believed to be effective in enhancing the crystallisation process of semi-crystalline polymers are blending with polymers with high crystallinity, nucleating agents such as inorganic compounds, low-molecular-weight organic compounds and polymeric nucleating agents. Plasticisation with low-molecular-weight compounds has been widely explored to keep the best attributes of these biopolymers. 4,13,14 The nucleating agent will lower the surface free energy barrier towards nucleation and thus initiates crystallisation at higher temperature upon cooling. 8 According to recent studies, the biodegradable poly(ε-caprolactone) (PCL) is reported to be an effective polymeric nucleating agent for PLA. 15,16 Many research groups have reported on biodegradable PLA/PCL blends, revealing that these blends were thermodynamically immiscible, which results in a multiphase structure with poor dispersion and some interfacial adhesion when the majority phase is PCL but not when it is PLA . 17 -19 In contrast, some studies revealed partial miscibility, first of all, since the lack of specific interactions between PCL and PLA leads to partial miscibility of their blends, and the PLA -rich phase seems to partly dissolve the PCL. 16,20 Similarly, the solubility parameters of both materials are close to each other, that is, 9.2 and 10.1 cal cm−3, respectively. 14
Considerable efforts have been made to enhance the miscibility of PLA and PCL blends using generally known techniques. Prepared diblock PLA-PCL 21 and triblock PLA-PCL-PLA 22 were used as compatibiliser between PLA and PCL. The results showed that the elongation at break of the PLA/PCL blends with the diblock and triblock increased to 71.4% and 53%, respectively. In addition, some molecule compatibilisers have been used in PLA/PCL blends, such as glycidyl methacrylate, 23 dicumyl peroxide 24 and lysine triisocyanate. 25 These compatibilisers reduced the size of the dispersed phase and improved the toughness of the PLA matrix. However, these small molecule compatibilisers are unsuitable environmentally friendly material. Hence, maleic anhydride-grafted PLA was used in different blend systems, such as those based on polyethylene glycol. 26
Many researchers published works on the compatibilisation of immiscible PLA/PCL mixtures using new strategy in the development of composites and nanocomposites for improving the interfacial adhesion between the polymer and the filler/nanofiller. PLA/PCL (42/58) blend nanocomposites were prepared with 1.5 titanium dioxide (TiO2) nanoparticles. It was concluded that TiO2 nanoparticles were located at the interface of the two phases due to kinetic effects and nanoparticles geometry. 27 PLA/PCL was also used by combining organomontmorillonites (OMt; Cloisite 15A and Cloisite 30B) with epoxy-functionalized graphene (Gr). They concluded that the compatibility of PLA/PCL is improved in the presence of organoclays and OMt/Gr mixtures. 28 Recently, Stoja et al. 29 have employed for the first time to impregnate PLA/PCL films with natural antibacterial agents such as thymol and thyme extract for bacterial growth control in food packaging.
Plasticisers are frequently used, not only to improve the transformation but mainly to increase the polymer chain mobility and to enhance the crystallisation rate of polymers by reducing the energy required for its crystallisation for the chain folding process. 8 Citrate esters are investigated as plasticisers for PLA. At least 20% of plasticiser needs to be introduced to lower its glass transition temperature (Tg), increase ductility and improve processability. 13,18 -20,30 Recently, much attention has been turned to the study of PLA crystallisation behaviour because crystal structure and arrangement of crystallites have been reported to have a profound effect on the physical and mechanical properties of biodegradable polymers. 8
Only few researchers have studied the influence of the addition of plasticisers on the miscibility and crystallisation of PLA/PCL blends. The effect of PCL contents and triethyl 2-acetyl citrate (TEC) and tributyl O-acetyl citrate (ATBC) as plasticiser at various compositions on PLA/PCL blends were investigated. In this study, a series of rigid and plasticised PLA (2002D) and PCL (CAPA6250) were prepared by melt mixing at various concentrations. A slight increase in the degree of crystallinity suggested that PCL acted as a nucleating agent. TEC and ATBC are biodegradable plasticisers that have been blended with PLA/PCL. By adding up to 30%, these citrate plasticisers were shown to lower the Tg. Moreover, the rigid and plasticised blends were shown to be partially miscible. Therefore, the effects of blending and plasticising on the crystallisation behaviour of PLA/PCL were investigated.
Experiment
Materials
The selected commercial PLA grade was PLA 2002D purchased from NatureWorks LLC (Minnetonka, MN, USA). It is a semi-crystalline extrusion material with approximately 4%

Chemical structure of (a) PLA, (b) PCL, (c) TEC and (d) ATBC.
Preparation method
To prepare rigid and plasticised blends, PLA pellets were dried in an oven at 60°C overnight. PCL, ATBC and TEC were used as received. Appropriate quantities were measured and poured in a container and then were manually mixed by shaking. The melt mixing process of PLA/PCL blends was carried out using a plastograph Brabender LP2100 (Duisburg, Germany) at a temperature of 180°C during 8-min cycle with 40 r min−1 screw speed. The obtained samples were cut into small pieces and then compression moulded into 2-mm-thick sheets and thin films at the same temperature (180°C) for 5 min using a hydraulic press at a pressure of 75 Pa. Subsequently, the composition of rigid and plasticised PLA/PCL blends with TEC and ATBC are summarised in Table 1.
Summary of compositions of rigid and plasticised PLA/PCL blends with ATBC and TEC.

PLA: poly(lactic acid); PCL: poly(ε-caprolactone); TEC: acetyl triethyl citrate; ATBC: acetyl tributyl citrate.
Characterisation methods
Differential scanning calorimetry
The main thermal transitions were investigated using a differential scanning calorimeter (DSC/Q10; TA Instruments (New Castle, Delaware, USA)), under a nitrogen flow of 50 ml min−1. To eliminate the material’s thermal record, samples were first heated to 180°C at a heating rate of 10°C min−1 by keeping this temperature for 3 min. The samples were then cooled down to −70°C at a cooling rate of 10°C min−1 by keeping this temperature for 3 min. Finally, they were reheated to 180°C at a heating rate of 10°C min−1 by keeping this temperature again for 3 min and then jumping down to 25°C. All thermal properties were obtained from the second heating scan curves. The degree of crystallinity of PCL (Xc, PCL) in the blends was calculated from equation (1) with consideration of the amount of PCL in the blend. 18 For PCL, a 100% crystalline melting enthalpy (ΔH0, PCL) of 156.8 J g−1 was assumed. 31 The degree of crystallinity of PLA (Xc, PLA) was calculated from equation (2), and for PLA, the 100% crystalline melting enthalpy (ΔH0, PLA) was taken to be 93 J g−1. 32
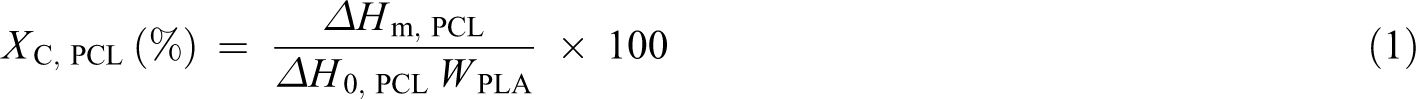
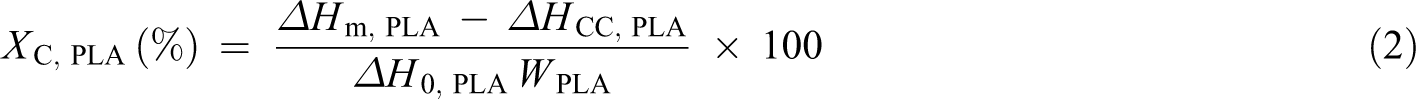
where WPCL and WPLA are the weight fractions of PCL and PLA in the blends, respectively; ΔHm, PCL and ΔHm, PLA are the melting enthalpies of PCL and PLA in the blends, respectively and ΔHcc, PLA is the cold crystallisation enthalpy (ΔHcc) of PLA in the blends.
X-Ray diffraction
The X-ray diffraction (XRD) of PLA/PCL and PLA/PCL/plasticiser films studied was carried out using a Bruker D8 Advance diffractometer (Billerica, Massachusetts, USA). Scans were recorded in reflection using α radiation with a wave length (λ) of 0.1542 nm at room temperature. The angle of incidence was varied from 8°C to 50°C in steps of 3° min−1. The accelerating voltage was 40 kV, and the tube current was 150 mA. A computer-aided curve fitting technique was used to separate crystalline and amorphous phases of diffracted graphs. The crystallinity was calculated by separating intensities using Gaussian and Lorentzian functions due to amorphous and crystalline phase on diffraction phase. The degree of crystallinity (Xc; %) was measured as the ratio of crystalline area to the total area as shown in equation (3):

where Ac and Aa are the areas of crystalline and amorphous phases, respectively.
Thermogravimetric analysis
Thermogravimetric analysis (TGA) of the PLA/PCL/plasticiser samples was studied using a TGA Q500 analyser (TA Instruments) (New Castle, Delaware, USA). The samples were heated from 10°C to 550°C at a heating rate of 10°C min−1 under nitrogen atmosphere at the flow rate of 10 ml min−1. The onset temperature (onset) was determined from the thermogravimetric curve as being the temperature at which the weight loss begins.
Results and discussion
Differential scanning calorimetry
DSC thermograms of neat PLA, neat PCL and rigid and plasticised PLA/PCL blends with TEC and ATBC at various concentrations are shown in Figure 2. Parameters such as Tg, Tm, cold crystallisation temperature (Tcc), ΔHm, ΔHcc and Xc are presented in Table 2.
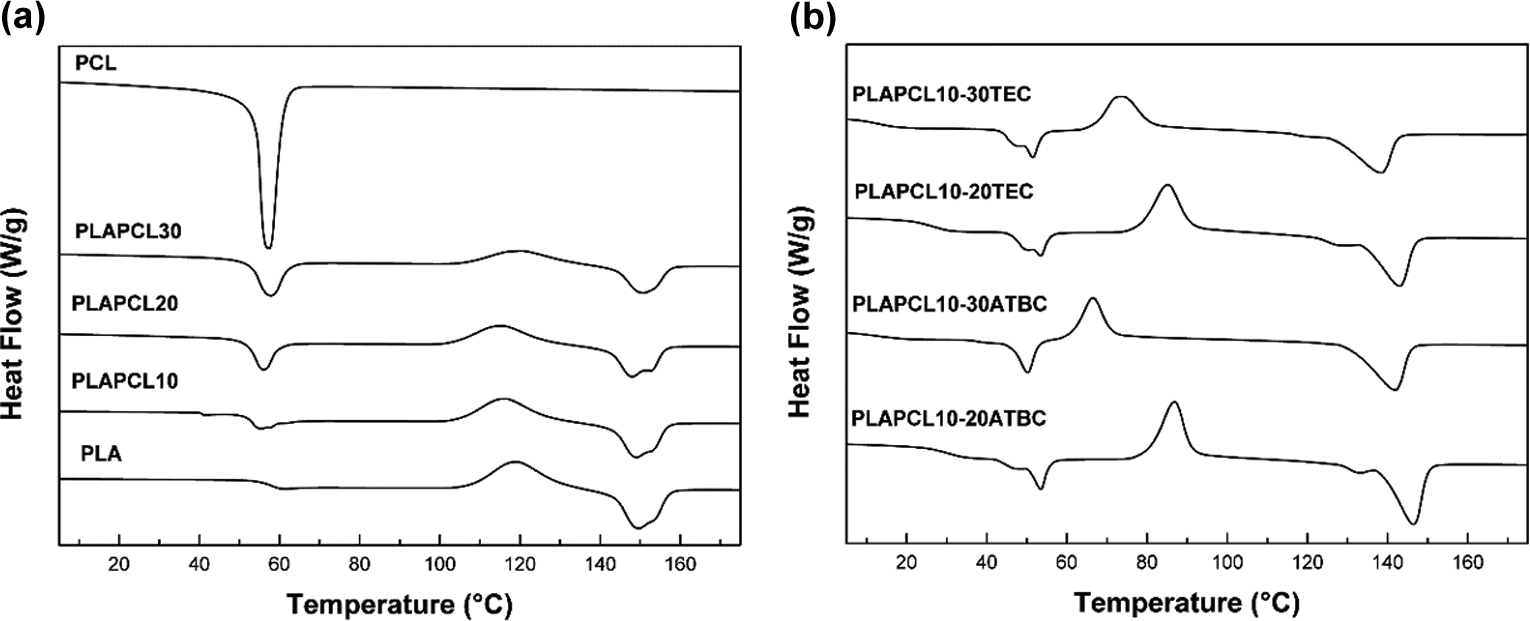
DSC thermograms of (a) rigid PLA/PCL blends and (b) plasticised PLA/PCL blends at various concentrations.
Thermal properties and crystallinity of neat PLA, neat PCL and rigid and plasticised PLA/PCL blends with ATBC and TEC at various concentrations.

PLA: poly(lactic acid); PCL: poly(ε-caprolactone); TEC: acetyl triethyl citrate; ATBC: acetyl tributyl citrate; Tg: glass transition temperature; ΔHm: melting enthalpy; Tm: melting temperature; ΔHcc: cold crystallisation enthalpy; Tcc: cold crystallisation temperature; Xc: degree of crystallinity.
X-Ray diffraction
XRD is also used to determine the nature of polymer by measuring the extent of crystallinity with respect to the amorphous content. The XRD patterns of PLA, PCL and rigid and plasticised PLA/PCL blends with TEC and ATBC are shown in Figure 3.

XRD spectra of (a) rigid PLA/PCL and (b) plasticised PLA/PCL blends at 30 wt%.
The crystallinity obtained by this technique is calculated using equation (3). The values of Xc of rigid and plasticised PLA/PCL blends are shown in Table 3 and Figure 3.
Crystallinity of rigid and plasticised PLA/PCL blends.
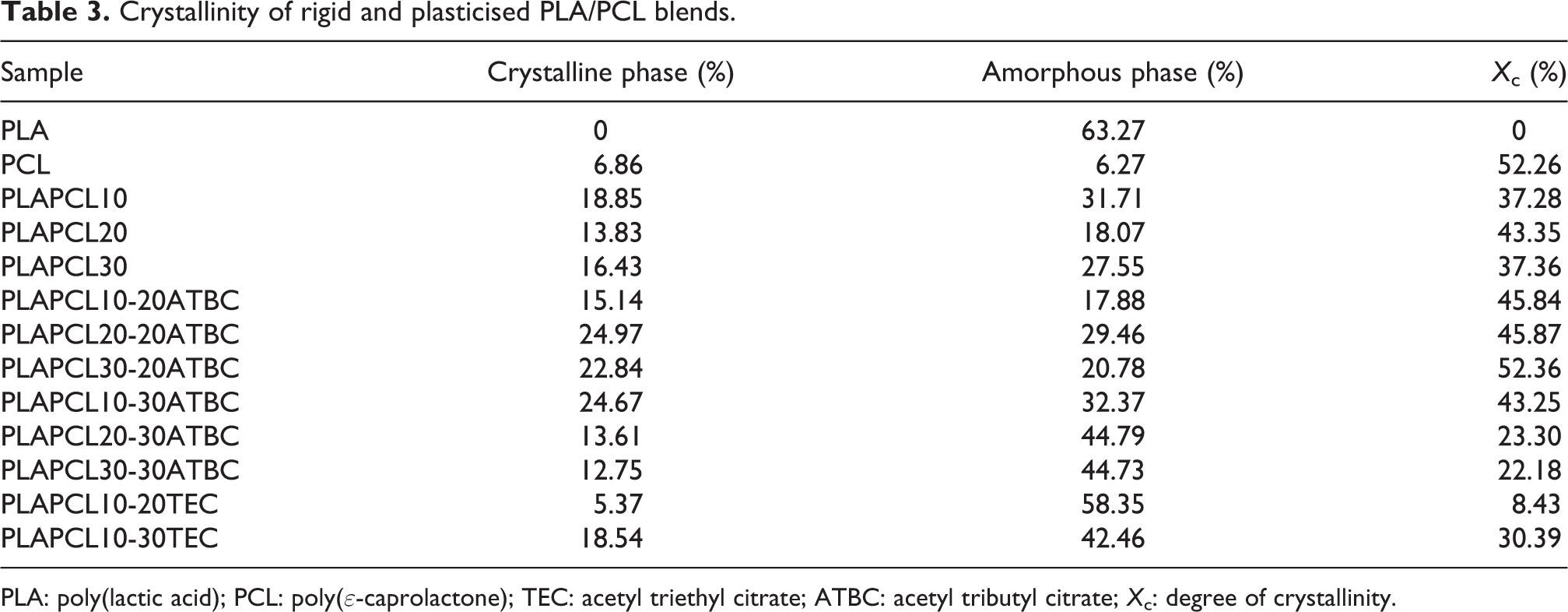
PLA: poly(lactic acid); PCL: poly(ε-caprolactone); TEC: acetyl triethyl citrate; ATBC: acetyl tributyl citrate; Xc: degree of crystallinity.
Thermogravimetric analysis
TGA is the second most commonly used technique studying thermal degradation and stability of polymer materials. The thermogravimetric and derivative thermogravimetric experiments under nitrogen atmosphere were carried out to study the thermal degradation behaviour of PLA, PCL and their blends and to determine the impact of ATBC on the thermal stability of PLA. Curves of all samples are shown in Figure 4, and their thermal properties are summarised in Table 4.
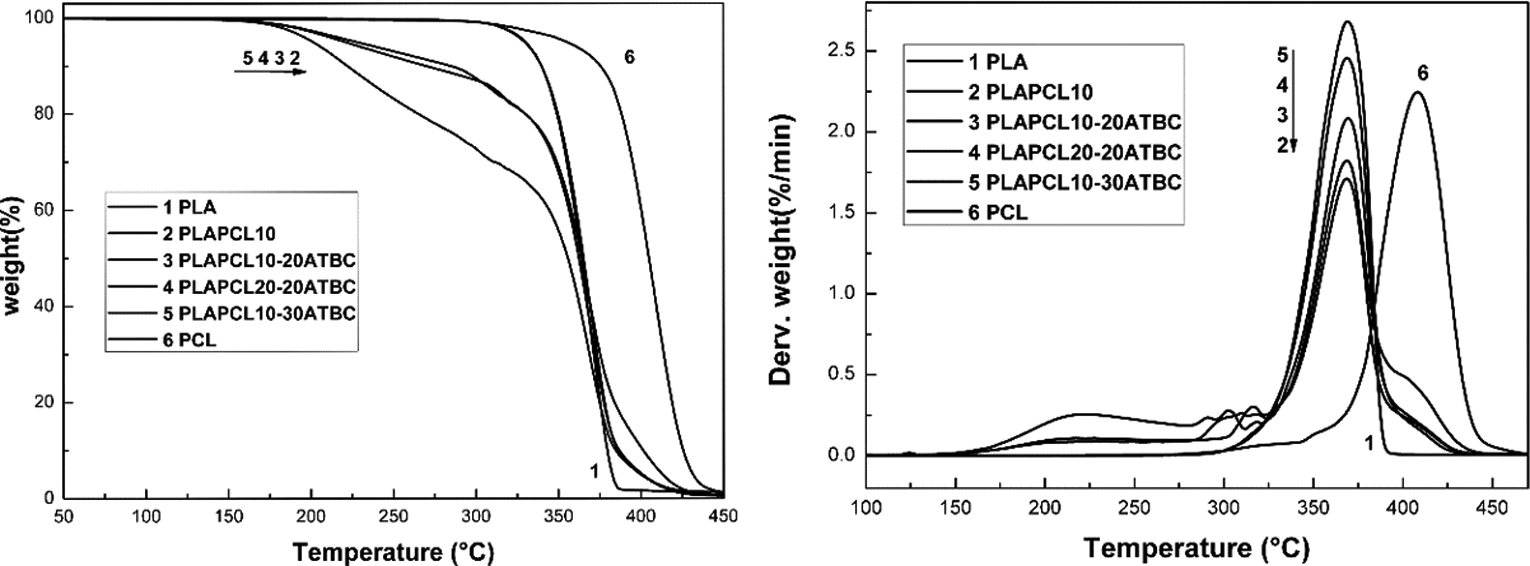
TG/DTG thermograms of rigid and plasticised PLA/PCL blends at various concentrations.
Thermal properties of PLA/PCL blends at various concentrations determined by TG/DTG.

PLA: poly(lactic acid); PCL: poly(ε-caprolactone); TG: thermogravimetric; DTG: derivative thermogravimetric; TEC: acetyl triethyl citrate; ATBC: acetyl tributyl citrate.
Differential scanning calorimetry
The results confirmed that the as-received PLA is semi-crystalline, due to the clear glass transition and melting peaks around 58°C and 149°C, respectively, as shown in Figure 2(a). It is well known that PLA has a very low crystallisation rate and that it primarily crystallises during heating. 33 This is confirmed from Table 2 and Figure 2(a). In addition to the glass transition and melting peaks, a clear cold crystallisation peak around 119°C and a very low crystallisation rate of about 1.56% calculated from equation (2) were reported. It is observed that the ΔHcc was almost equal to the melting enthalpy, confirming that PLA does not form any crystal during the controlled cooling in the DSC analysis. 34
With regard to PCL, which is also semi-crystalline, a well-defined melting peak around 57°C and high crystallisation rate of about 45.31% calculated from equation (1) were reported, whereas no crystallisation peak was noticed. Its glass transition peak is well known to be around −60°C, but it wasn’t detected in the scans. The melting enthalpy of neat PCL is much higher than that of neat PLA, indicating much higher crystallinity compared to PLA.
These findings agree with the results obtained previously. 35,36 In fact, polymers with low molecular weight have a high content of crystallinity. 37 After blending with PCL, the Tm of PLA somehow hasn’t changed significantly with the addition of PCL, showing values around 149°C range. The same applies to the melting peak of PCL, it hasn’t changed with varying PCL content, and it remains at 57°C as for neat PCL. Double peaks have been observed in the melting of the blends, which are located between 140°C and 160°C depending on the PCL content. These findings reveal the existence of two different types of PLA crystals with different perfections within the system. The first endothermic peak shows PLA crystals with less perfection probably due to the presence of a limited amount of miscible PCL chains within the formed structure, which is in agreement with previous studies. 14,38
However, with varying PCL content, some changes have occurred on the rest of the thermal properties of both PLA and PCL phases. The Tcc of PLA has barely shifted to a lower temperature, the ΔHcc, PLA and ΔHm, PLA of PLA have decreased and the ΔHcc, PCL has increased significantly, thus indicating that the crystallisation of PLA may be affected by the addition of PCL. 39 As shown in Table 2, the Xc, PLA has slightly increased by increasing the PCL content, suggesting that the presence of PCL favours the crystallisation of PLA, acting as a nucleating agent. 40,41 It should be noted that almost all the thermal transitions of PLA and PCL in rigid PLA/PCL blends appear at the same typical temperatures as those of the corresponding individual polymers, and the variation of Tg, Tcc, Tm and Xc, PLA values is usually assigned to the interactions between components, which reveals a very weak interaction between these two polymers. 32,42
Several citrate esters are commercial plasticisers for PLA, including ATBC and TEC. 43 The Tg, the melting peak or Tcc may shift due to the plasticiser’s effect on the material. 44 The effectiveness of the plasticiser on PLA is expected by the reduction of the glass transition of the amorphous phase and by the depression of the melting point of the crystalline region. 45
As shown in Figure 2(b) and Table 2, the Tg of neat PLA has decreased by increasing the plasticiser content and has increased by increasing the PCL content for plasticised PLA/PCL blends. The largest reduction of Tg when compared with neat PLA was from 58°C to 12°C and 13°C for PLA/PCL 10 (PLAPCL10) by adding up to 30 wt% ATBC and TEC, respectively. The most probable explanation may be the low molecular weight of the plasticiser allows it to occupy intermolecular spaces between the polymer chains, reducing the energy for molecular motion and the formation of hydrogen bonding between the polymer chains, which in turn increases free volume and polymer chain mobility. These findings are in accordance with previous results. 45 -47
As shown in Table 2, the Tcc of PLA has decreased from 119°C for neat PLA to 66°C and 73°C for PLA/PCL 10 (PLAPCL10) by adding up to 30 wt% of ATBC and TEC, respectively. The addition of citrate plasticisers has also decreased the Tm of PLA. From Figure 3(a) and (b), the Xc, PLA in plasticised PLA/PCL blends has increased considerably depending on the plasticiser content. For PLA/PCL 10 (PLAPCL10) plasticised with 30 wt% ATBC, greatest crystallinity has been observed for a value of 19.96%, while only 9.46% and 8.87% for 20 wt% and 30 wt% TEC, respectively. This important increase in crystallinity of PLA by the addition of both ATBC and TEC suggests that the plasticiser increased the polymer chain mobility by reducing the energy required for crystallisation. Similar results have been evidenced previously. 8
X-Ray diffraction
The XRD spectrum of PLA in Figure 3(a) has shown a broad double peak between 5° and 40° corresponding to amorphous regions confirming that the PLA is completely amorphous after initial melting and cooling. This peak positions are in accordance with those reported articles. 34,47 Although a slight Xc of about 1.56% was observed in the diffraction scanning calorimetry thermogram, no crystal structure was detected in the XRD. The Xc, PCL obtained from DSC and XRD is found to be 45.31% and 52.26%, respectively. The obtained results are in accordance with what has been stated where the crystallinity range for PCL is known to be between 42% and 60%.
The PCL spectrum has shown several peaks between 15° and 40°, with two high-intensity and other low-intensity peaks between 20° and 25° and between 35° and 40°, respectively. Moreover, the peaks were sharp and distinct, which indicated that the sample was a crystalline material. Similar findings are in accordance with those reported works. 48 -50 In addition, the spectrum of the other blends has shown the same crystalline peaks overlapping the amorphous curve of PLA. The peak intensities vary with the variation of PCL content. The DSC observation of increasing crystallinity with increasing PCL content in the blends could, therefore, be confirmed by the XRD results. Poly(lactic acid)/poly(ε-caprolactone)10-30 tributyl O-acetyl citrate (PLAPCL10-30ATBC) and poly(lactic acid)/poly(ε-caprolactone)10-30 triethyl 2-acetyl citrate (PLAPCL10-30TEC) patterns shown in Figure 3(b) have shifted towards lower angles between 15° and 20°, which probably indicates the variation in the distance between parallel planes of atoms. According to Bragg’s law, if the spacing of the crystallographic planes changes, the Bragg angle should decrease towards lower 2θ values. 51
Thermogravimetric analysis
Thermogravimetric curves obtained under nitrogen atmosphere have displayed a single-stage decomposition for PLA, PCL and their rigid blend (PLAPCL10). It can be noted that the PLA degradation had occurred at lower temperatures, at around 345°C, while the PCL degradation had taken place at around 382°C. PCL is known to have extraordinary thermal stability at high temperature compared to other polyesters due to its high Xc (45–60%) and its high hydrophobicity. 33,52 The addition of citrates has affected the thermal stability of PLA. The weight loss of the plasticised blends has occurred in two-stage decomposition. A significant shift in the onset temperature has been observed compared to the rigid PLA/PCL blend. Derivative thermogravimetric curves have displayed a clear temperature range between 291°C and 316°C corresponding to the evaporation of citrate plasticiser components, where as mentioned by the supplier, the boiling point of ATBC alone is known to be 173°C.
Conclusions
With the increase of PCL content, DSC revealed a depression in both Tg and ΔHcc of PLA. The Xc, PLA in rigid PLA/PCL blends slightly increased compared with neat PLA, suggesting the presence of PCL may favour the crystallisation of PLA and acting as a nucleating agent. The plasticising effect was highlighted through the significant decrease in Tg, Tm and ΔHcc of PLA. Depending on the plasticiser and on its content, the Xc, PLA in plasticised PLA/PCL blends increased considerably compared to neat PLA, suggesting that the plasticiser increased the polymer chain mobility by reducing the energy required for crystallisation. The changes shown by DSC indicate the partial miscibility of both rigid and plasticised PLA/PCL blends. XRD confirmed that PLA is partially amorphous. Moreover, PCL sample was also confirmed to be a highly crystalline material. TGA showed that the addition of ATBC has promoted a decrease in thermal stability of PLA/PCL blends. The rigid and plasticised PLA/PCL films show less transparency than that of the pure films.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
